Abstract
Objective
To examine the effectiveness of rivaroxaban compared to enoxaparin in patients diagnosed with cancer and venous thromboembolism.
Methods
A search of Pub Med, Scopus, and Google Scholar, from inception through April 2023 was conducted. Articles comparing rivaroxaban with enoxaparin in patients with cancer and VTE/PE/DVT were included. Review Manager Version 5.2 was utilised for the analysis of the following outcomes; VTE, PE, DVT, major bleeding, and mortality.
Results
A total of 8 articles and 2276 patients were included in the final analysis. Pooled analysis showed that rivaroxaban had a statistically insignificant reduced association with VTE occurrence (RR:0.83, 95% CI:0.58–1.18, P:0.3) as well as a statically insignificant reduction in major bleeding (RR:0.79, 95% CI:0.53–1.18, P:0.25). Analysis showcased that there was an insignificant reduction of mortality rivaroxaban as compared to enoxaparin (RR:0.74, 95% CI: 0.46–1.20, P:0.23).
Conclusion
Rivaroxaban can serve as a viable alternative to enoxaparin, with no appreciable drawbacks, for preventing and managing VTE in patients with malignancy.
Introduction
Venous thromboembolism (VTE) is a well-documented comorbidity frequently found among cancer patients. It is observed that patients diagnosed with venous thromboembolism commonly present with deep venous thrombosis and pulmonary embolism (PE).1,2 Moreover, it is recognized that around 15% to 20% of individuals who experience pulmonary embolism or venous thromboembolism have a documented history of cancer. Low-molecular-weight heparin (LMWH) is the recommended initial treatment for patients with active cancer who are diagnosed with venous thromboembolism. 3 However, patients find LMWH injections inconvenient and express a preference for oral anticoagulants as an alternative. Warfarin, the widely utilized oral vitamin K antagonist (VKA), presents certain drawbacks such as the need for regular monitoring of international normalized ratio (INR), an elevated risk of bleeding, and reduced time spent within the therapeutic range. 4 Hence, there is a requirement for more advanced anticoagulation approaches to address the needs of patients suffering from both VTE and cancer. The introduction of newer factor Xa inhibitors, such as rivaroxaban, holds promise for improved therapeutic efficacy in this context.
Clinical evidence has shown that rivaroxaban is comparable in efficacy to VKAs for the treatment of VTE in the general population. 5 The inclusion of cancer patients in earlier studies was limited in scope.6–8 Moreover, the available evidence comparing and contrasting rivaroxaban with LMWH is limited in size. In our meta-analysis study, we utilized data from multiple studies to examine the effectiveness of rivaroxaban compared to enoxaparin in patients diagnosed with cancer and venous thromboembolism.
Methodology
This systematic review and meta-analysis was carried out in compliance with Cochrane 9 and PRISMA 10 guidelines.
Data Sources and Search Method
A search of PubMed, SCOPUS and Google scholar was conducted from inception to April 2023 on 18th April 2023 independently by two researchers (AR and AA). No language filters were set and the following keywords with Boolean operators were used: ‘(embolism OR thromboembolism OR DVT OR thrombosis OR deep vein thrombosis) AND (cancer) AND (rivaroxaban OR xarelto) AND (enoxaparin OR clexane)’. Additional articles were manually identified by screening the reference lists of obtained studies and similar clinical trials to ensure all potentially relevant publications were included. A PRISMA flowchart summarising the total number of studies and included studies is shown in Figure 1.
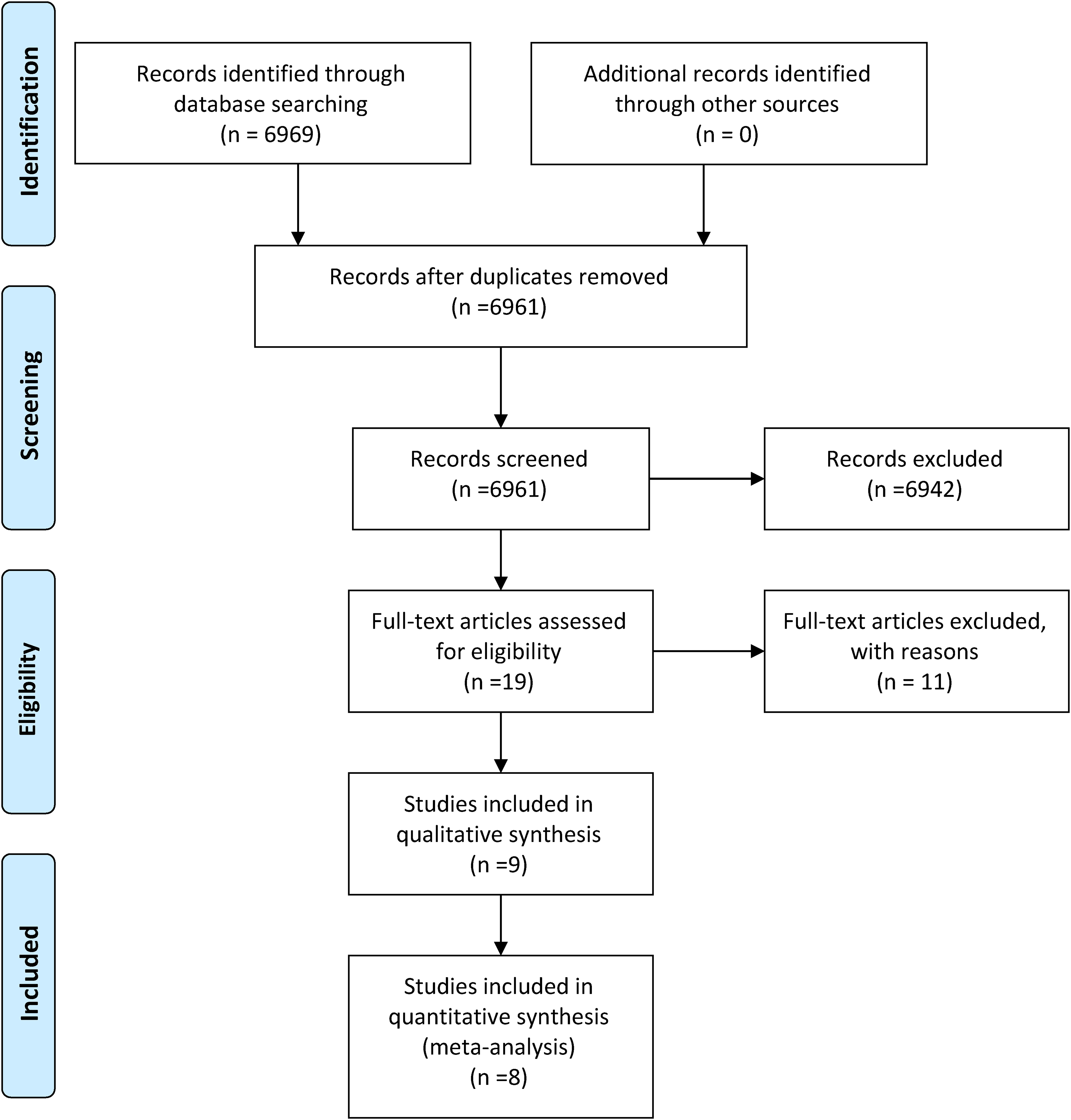
PRISMA flow diagram.
Study Selection and Eligibility Criteria
Articles that conformed to all the following criteria were selected for inclusion: (1) compared rivaroxaban with enoxaparin, (2) included cancer patients with thromboembolism/pulmonary embolism/DVT, (3) included relevant data on of one or more of the following outcomes: VTE recurrence, DVT recurrence, PE recurrence, Death, Total incidence of bleeding, Major bleeding.
In addition to the above-mentioned inclusion criteria, we also ensured that only original studies were included in our analysis. No assumptions were made regarding any unavailable data.
Data Extraction and Quality Assessment
Articles retrieved from the search were manually screened to remove duplicates. The title and abstracts of the remaining articles were then evaluated to ensure relevance and the complete text of the final articles was then reviewed to confirm inclusion. The reference lists of the included articles were also combed through to ensure inclusion of all relevant studies. 2 reviewers (ARA, AA) screened the articles and any disagreement was settled by a third independent reviewer (MD). The quality and bias assessment of the randomised controlled trials was done using the JADAD scale 11 while the Newcastle Ottawa Scale 12 was used to evaluate the risk of bias in the observational trials. The JADAD scale consists scoring of 3 items namely randomization, blinding and account of all patients. The Newcastle Ottawa Scale consists of 6 items with 3 subscales: selection, comparability, and outcome. The scoring was performed independently by two reviewers (SH and AS) and a consensus was reached between the 2 authors in cases of disagreements.
Outcome Definitions
The outcomes assessed in our meta-analysis included VTE, PE, DVT, major bleeding and mortality.
VTE was defined as a symptomatic deep vein thrombosis (DVT), pulmonary embolism (PE) or both during the study period. PE was described as either a clinically confirmed diagnosis of pulmonary embolism or confirmation on imaging, CTPA, of a symptomatic embolus. DVT was classified as a new clot or thrombus on Doppler Ultrasound. Mortality was identified as death due to either a thromboembolic event, critical haemorrhage or any other established diagnosis secondary to either anticoagulant therapy or the underlying disease itself. Major bleeding construed bleeding leading either to loss of life, bleeding in a critical location or into a major cavity or bleeding requiring transfusion of greater than 2 units of packed red blood cells.
Statistical Analysis
Papers that met our pre-set inclusion criteria had their study details, baseline demographics, and outcome data exported onto an Excel sheet by 2 reviewers. Any disagreement that arose was settled by discussion with a third independent reviewer. Statistical analysis was performed on Review Manager (RevMan Version 5.4. The Cochrane Collaboration, 2020.). 13 The number of incidents as well as the total number of patients in each study were retrieved. Using a random effect model and a confidence interval of 95%, the software calculated the risk ratio (RR, 95% CI) of each outcome. Heterogeneity was assessed using the I2 statistic 14 followed by subgroup analysis to reduce heterogeneity where necessary. A P-value of ≤0.05 was taken to be statistically significant. Publication bias could not be evaluated as only 9 studies were chosen for inclusion. In cases where multiple studies were conducted by the same author or institution within a similar timeframe and reported comparable outcomes with potential for overlap, only the most recent study was incorporated into this meta-analysis for the corresponding outcome. Therefore two studies, namely Houghtoun et al 15 and Wysokinski et al 16 reported outcomes of the same patients from the same centre over a different period. Houghton reported outcomes between March 1, 2003, and April 20, 2020, while Wysokinski reported outcomes between 1 March 2003 and 30 January 2018. Since, Houghton et al included patients reported by Wysokinski et al, we excluded outcomes from Wysokinski et al except those that were not reported by Houghton et al
Results
Baseline Characteristics
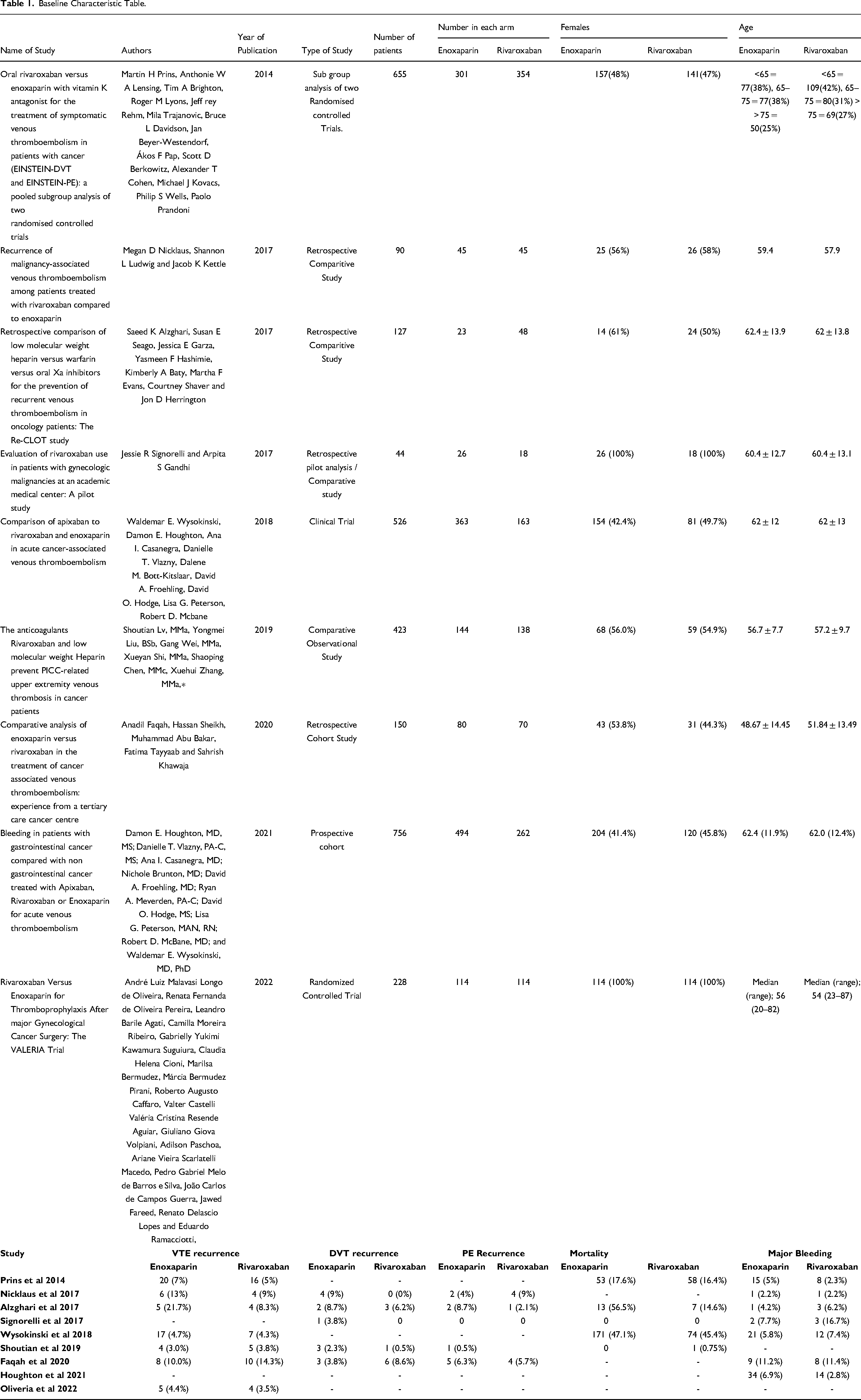
An initial search of PubMed/Medline, Scopus and Cochrane CENTRAL library databases yielded a total of 6969 articles, 8 of which were duplicates. On screening, 6942 articles were excluded. 19 of the remaining articles were then assessed by two independent reviewers (AR and AA) and 11 were excluded as they did not fit our inclusion criteria. Hence, we included 8 studies, 6 of which were retrospective cohort studies7,8,15,17–19 while the remaining 2 were randomised controlled trials.20,21 The articles we included were published between 2014 and 2022. The total number of subjects in the 8 studies were 2,276, with 1219 patients in the rivaroxaban group while the enoxaparin arm had 1057 patients. Analysis of the baseline characteristics revealed that the number of female participants were greater for the rivaroxaban group. The mean age was higher for the enoxaparin group as well as the body mass index (BMI). The incidence of lung, GI tract, haematological and breast cancer was higher in the enoxaparin group while the incidence of genitourinary and ovarian cancer was higher in the rivaroxaban group. The number of patients undergoing chemotherapy were almost similar in both groups while a greater number of patients in the enoxaparin group were suffering from metastatic disease. The incidence of pulmonary embolism and deep vein thrombosis was comparable in both groups. However, the incidence of a combined pulmonary embolism and deep venous thrombosis was higher for the rivaroxaban group. The number of patients that had undergone surgical intervention for their disease was significantly higher in the enoxaparin group. Table 1 summarises the baseline characteristics of the included studies.
Baseline Characteristic Table.
Results of Meta-Analysis
The results of this meta-analysis are presented as detailed forest plots in Figures 2–6
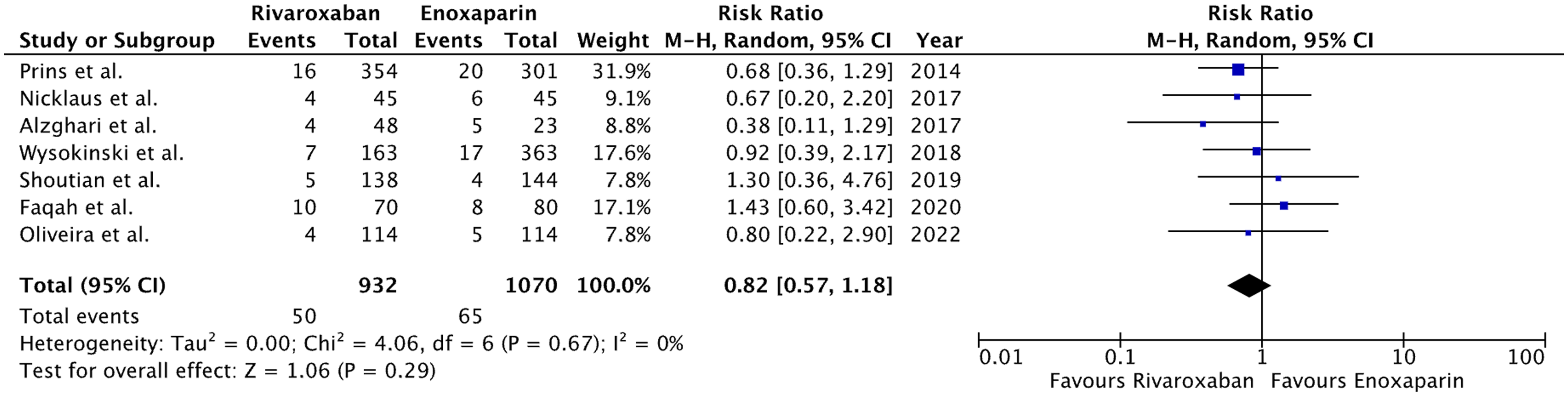
Forest plot for recurrence of VTE.
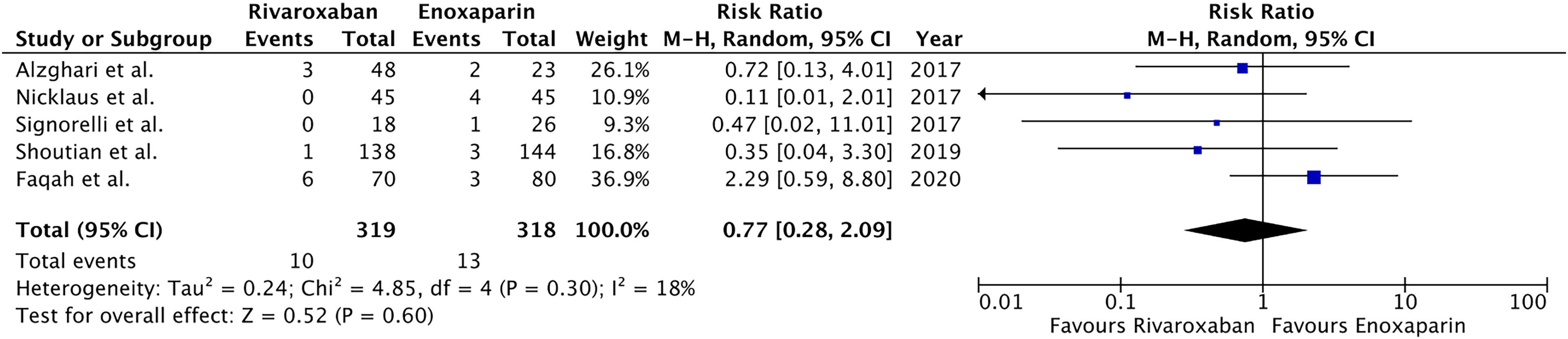
Forest plot for recurrence of DVT.
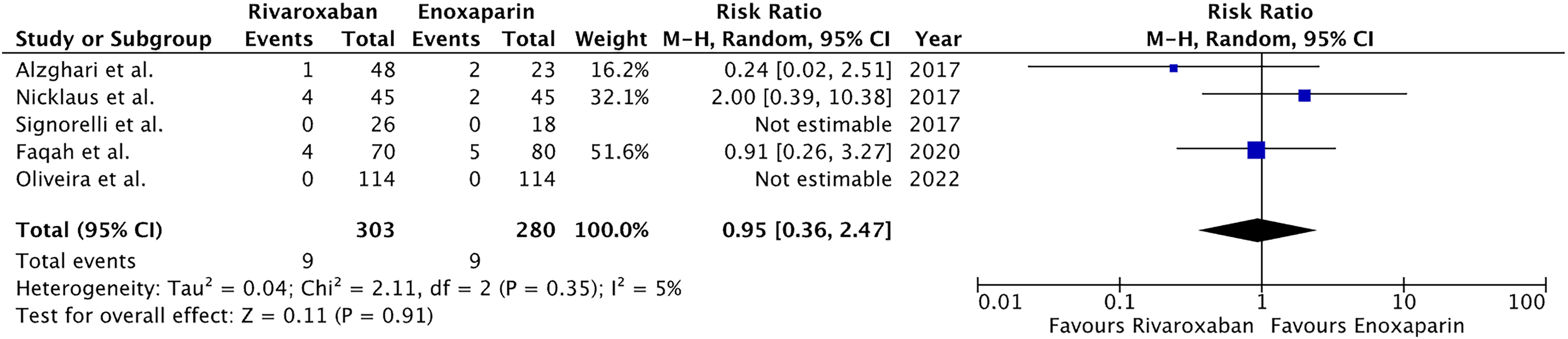
Forest plot for recurrence of PE.
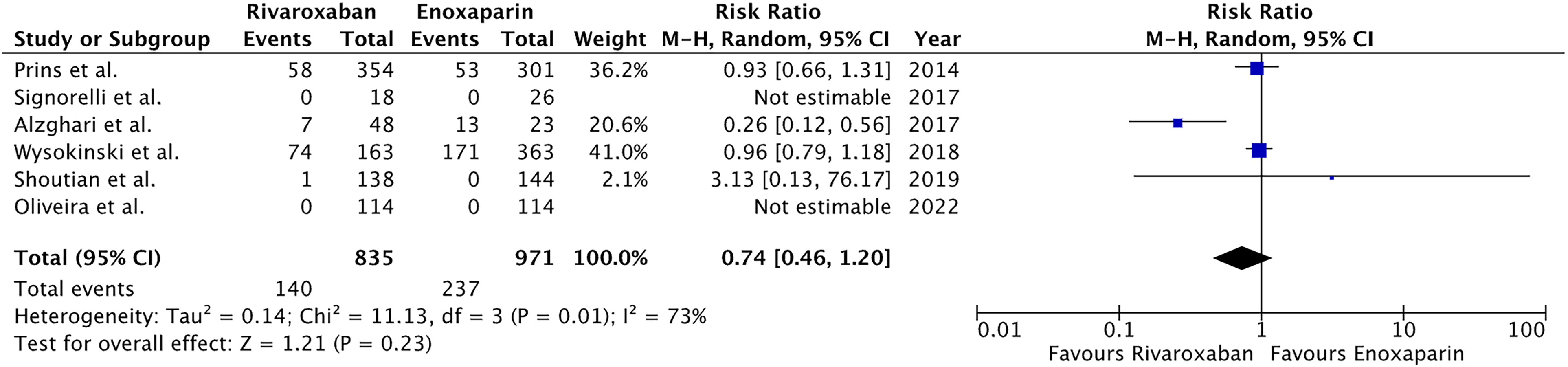
Forest plot for Mortality.
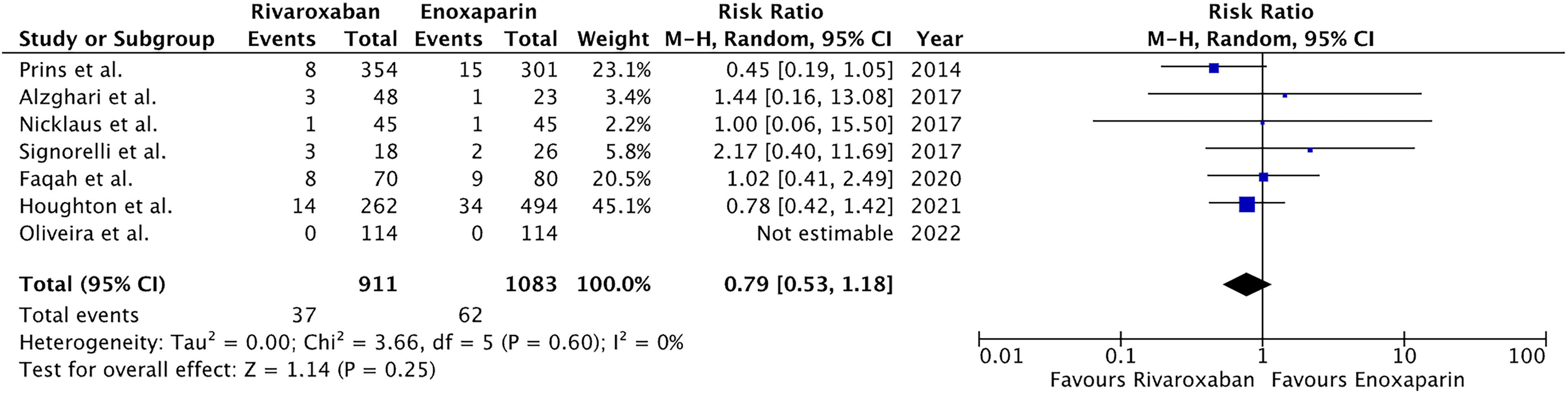
Forest plot for Major bleeding.
Discussion
Our study pools together a significant number of selected patients and expands on the previously done meta-analysis. 22 According to the authors, the study findings indicated that rivaroxaban demonstrated comparable safety and efficacy to enoxaparin in preventing recurrent venous thromboembolism (VTE) in patients with malignancy. They identified rivaroxaban as a viable alternative for individuals with cancer and VTE. In addition to a greater sample size incorporated in our study showcasing a truer representation of the previously limited population, a considerable number of emerging studies on this topic have been reported since Xing et al's study was published. Therefore, a meta-analysis based on updated data is needed to further study the link between VTE and cancer patient mortality.
To the best of the authors knowledge, the literature mostly focuses on comparing rivaroxaban with enoxaparin. While there is work that compares other OACs and LMWHs, that data can not be pooled into this analysis. Other drug members have different safety and efficacy profiles and it's is not statistically prudent to involve them in this review.
VTE commonly gives rise to morbidity and is a strong risk factor for mortality in cancer patients, with Signorelli at el. stating it as the second most leading cause of deaths, along with being an independent prognostic factor for mortality. 8 A study from Khorana et al 23 suggested that VTE rates in patients had an association with the type of cancer and the patient's demographics along with the treatment offered to them. Alzghari et al 17 highlights that receival of DOAC, enoxaparin or warfarin in patients suffering from cancer resulted in decreased VTE recurrence rates (at 3 months).
Our analysis also consolidates these findings, where two-thirds of the included studies reported VTE occurrence following the initiation of anti-coagulant therapy; with rivaroxaban showing a decreasing trend in association with VTE occurrence versus enoxaparin. The results, however, were not statistically significant. This could be due to a small sample size in the trials and overall population size (larger and well-designed studies should be done to explore this problem), concomitant anti-platelet therapy, unknown adherence issues, and low warfarin time in therapeutic range (TTR). Only 4 studies had mortality as a patient outcome, with mortality reduced in the rivaroxaban group as compared to those in enoxaparin, albeit statistically insignificant. This could be explained by a small study sample size in addition to it not being randomized. The sensitivity analysis concluded that Alzghari et al, 17 is contributing to a high heterogeneity in our results, due to a limited sample size, undergoing apixaban treatment.
Previously, low molecular weight heparin (LMWH) has remained the choice of treatment in cancer related VTE, whereas warfarin was preferred for preventing stroke in patients with atrial fibrillation. More recent studies in malignancy patients having VTE, have shown efficacy with rivaroxaban and edoxaban versus dalteparin, with variable bleeding rates when it comes to Direct Oral Anti-coagulants (DOACs). Due to the possible drug-to-drug interactions, incorporating DOAC therapy in cancer patients is limited, as co-treatment affects systems and poses a potential risk of the drug levels being outside the therapeutic range, even though theoretically. Co-administration of any DOACs in cancer therapies is also not recommended. Renal impairment, as well as thrombocytopenia, is relatively common in cancer patients, affecting the safety of DOAC therapy. Ensuring appropriate dosage adjustments in cancer patients with renal impairment is vital. It is also preferred to switch to a different anticoagulant that has lower renal clearance, rather than monitoring anti-XA levels. 24
According to the 2020 guidelines from the American Society of Clinical Oncology (ASCO), prophylaxis should be considered for acute medical oncology patients who have additional risk factors for venous thromboembolism (VTE) and do not have any contraindications or active bleeding. It suggested the use of LMWH over unfractioned heparin (UFH), with Pneumatic compression device (PCD) for patients with contraindications and advises against DOACs. ASCO states initial anticoagulation in treatment of cancer-associated VTE may involve LMWH, UFH, fondaparinux, and rivaroxaban, and for prolonged anticoagulation, prefers LMWH, edoxaban, rivaroxaban for over 6 months against Vitamin-K antagonists (VKAs). Vitamin K antagonists may be used in case of unavailability of LMWH or DOACs, albeit inferior in terms of control and therapeautic effect. In patients with ongoing cancer/metastatic cancer/chemotherapy, an extended use of anticoagulation may be used depending on the need. However, monitoring is required in these patients with periodic testing. In patients for whom anticoagulation is not a viable option, IVC filters may be used in high-risk, rapidly deteriorating settings. After calculating risk-benefit ratio, management of isolated subsegmental PE or splanchnic/visceral vein thrombi is an option that can be offered to select patients. 25
ASCO suggests the use of apixaban, rivaroxaban, and LMWH for high-risk ambulatory medical oncology patients (Khorana Risk Score (KRS) ≥ 2), provided that there are no discernible risk factors for bleeding or pharmaceutical interaction for up to 6 months.
Patients opting for cancer surgery should be given prophylaxis with UFH (Low-dose UFH three times a day) or LMWH (if CrCl ≥30 mL/min) unless contraindicated, as per ASCO's recommendations. Pharmacological prophylaxis should be continued for 7–10 days post-operatively. It is to be noted that in high-risk patients undergoing a significant surgical procedure, pharmacological prophylaxis with LMWH should be continued for up to 4 weeks. 26
Rivaroxaban is administered more conveniently and easily than enoxaparin because it is taken orally as a tablet. Enoxaparin requires subcutaneous injections. With fewer frequent injections required with oral administration, patients experience less pain and adhere to the treatment plan better. 27
Due to the fixed dosing schedule for rivaroxaban, the same dose is applied to all patients regardless of the indication. Due to the predicted anticoagulant impact of rivaroxaban, routine monitoring of variables like INR or aPTT is not necessary for consistent and reliable anticoagulation. On the other hand, enoxaparin needs to be monitored for anti-Xa levels to guarantee proper anticoagulation, as due to its mechanism of action (inactivates Factor Xa using a complex formed with anti-thrombin III) it cannot be monitored by PT and aPTT. 28 To prevent the common side effect of bleeding 29 anti-Xa is regarded as the gold standard for monitoring enoxaparin. 30
Rivaroxaban has a specific reversal agent called andexanet alfa, which can be used in emergency situations when immediate reversal of its anticoagulant effects is required. 31 Enoxaparin like all LMWHs does not have an antidote. 32 In general, rivaroxaban is more expensive than enoxaparin, but when the monitoring requirements are considered, rivaroxaban is seen as more effective and less expensive than enoxaparin. 33
Interesting results were found in this meta-analysis study on the effectiveness of rivaroxaban against enoxaparin in patients with cancer and venous thromboembolism (VTE). One of the major findings of the investigation was that there were no appreciable drawbacks to using rivaroxaban in comparison to enoxaparin. As a result of this discovery, rivaroxaban is a practical substitute for managing VTE in patients with cancer.
Particularly, in comparison to the enoxaparin group, VTE and DVT incidence in the rivaroxaban group showed a modest decrease, albeit statistically insignificant. Furthermore, no identifiable difference was found between the two therapy groups in terms of the frequency of PE recurrence, mortality or major bleeding.
All these results hint and direct us towards rivaroxaban being a viable and secure substitute for enoxaparin in the treatment of VTE in cancer patients. Given its possible advantages, such as being orally administered and easily monitored, rivaroxaban is an appealing alternative provided appropriate patient selection criteria are met.
Limitations
Although the meta-analysis study sheds light on the effectiveness of rivaroxaban in comparison to enoxaparin, it is important to recognise its limitations and advocate for restraint when extrapolating the results. The bulk of the included studies were observational in nature, which imposes some constraints, and this is a crucial factor to consider.18,21 Because observational studies rely on already-collected data, selection bias and confounding variables may have an impact on the findings. Therefore, it is important to interpret these results cautiously. Additional research, especially randomised controlled trials (RCTs), is needed to confirm the trends that have been identified.
The study also found significant variation in key outcomes, including mortality and clinically relevant non major bleeding disorder (CRNMB). The various causes of heterogeneity were determined using sensitivity analysis, and one study was shown to be a substantial contributor. To guarantee consistent and trustworthy outcomes, this emphasises the need for more robust and standardised research techniques.
It is also important to keep in mind that the meta-analysis study only included a small number of papers, which may have an impact on how generalizable the results are. The results of more research with greater sample sizes and various demographics would be more trustworthy and useful.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Informed consent for patient information to be published in this article was not obtained because this was a review article and did not include data directly from patients.
