Abstract
Enoxaparin is indicated for thromboprophylaxis in non-orthopedic surgical patients at a fixed dose of 40 mg daily. According to the US Food and Drug Administration’s enoxaparin prescribing information, this dose exposes low-weight patients (males < 57 kg, females < 45 kg) to a higher risk of bleeding. This study aimed to determine the rate of achieving a prophylactic peak anti-factor Xa (AFXa) level in low-weight surgical patients using enoxaparin 30 mg daily. Low-weight patients admitted for abdominopelvic or noncardiac thoracic surgery from May 2018 to May 2019 were prospectively studied. After receiving daily enoxaparin 30 mg, peak AFXa levels were assessed for achieving a prophylactic level (0.2-0.5 IU/mL). In 121 patients, the proportion of achieving a prophylactic peak AFXa level was 66.1%. More females (84.8%) achieved a prophylactic level compared to males (54.7%, P = .001). All out-of-range peak AFXa levels (33.9%) were sub-prophylactic. The median peak AFXa level was lower in males (0.24 [0.1-0.47] IU/mL) than females (0.31 [0.1-0.5] IU/mL; P < .001). On univariate analysis, female sex and weight were associated with achieving a prophylactic peak AFXa level. On multivariate analysis, only female sex was independently associated with an adequate prophylactic AFXa level (odds ratio 3.17, 95% CI: 1.32-11.94; P = .014). Four venous thromboembolism events (3.3%) were observed in patients with sub-prophylactic peak AFXa levels (9.7%). Two-thirds of low-weight surgical patients achieved a prophylactic peak AFXa level using daily enoxaparin 30 mg. This dose is likely to provide adequate thromboprophylaxis in low-weight females.
Introduction
Low-molecular-weight heparin (LMWH), a class of parenteral anticoagulants, is recommended by the American College of Chest Physicians guidelines for pharmacological prophylaxis of venous thromboembolism (VTE) in patients with or without cancer who are undergoing surgery. 1 A meta-analysis of 8 trials demonstrated a 70% reduction in clinical VTE in surgical patients who received LMWH prophylaxis compared to no prophylaxis. 2 This marked reduction in VTE was accompanied by an approximately 2-fold increase in the risks of major bleeding and wound hematoma after surgery.
Enoxaparin is a LMWH approved by both the US Food and Drug Administration (FDA) and the European Medication Agency for VTE prophylaxis at a daily dose of 40 mg subcutaneously for abdominal, pelvic, and lung surgery. 3,4 The effectiveness and safety of fixed-dose enoxaparin in preventing VTE in non-orthopedic surgery have been demonstrated in many randomized trials. 5,6 Due to its predictable pharmacokinetics and pharmacodynamics, the administration of fixed doses of enoxaparin for prophylaxis requires no monitoring or adjustment for weight, except in patients with morbid obesity or renal insufficiency. 7
Direct enoxaparin monitoring is inapplicable in clinical practice; therefore, anti-factor Xa (AFXa) levels are measured as a surrogate marker to reflect the adequacy of thromboprophylaxis. 8 AFXa levels are inversely related to the development of VTE, whereas the risk of bleeding increases with AFXa levels above 0.8 to 1.0 IU/mL. 9,10 AFXa peak levels ranging from 0.2 to 0.5 IU/mL at 4 hours after subcutaneous enoxaparin administration are considered adequate for VTE prophylaxis. 8,11,12
The US FDA-approved prescribing information for enoxaparin recommends careful observation for signs and symptoms of bleeding in low-weight patients receiving 40 mg for thromboprophylaxis. 3 Compared with average-weight subjects, a single dose of enoxaparin 40 mg resulted in 52% and 27% higher AFXa levels in low-weight women (<45 kg) and low-weight men (<57 kg), respectively. 3 However, no specific dose of enoxaparin was suggested for VTE prophylaxis in these low-weight individuals.
Substantial weight loss is prevalent among patients undergoing surgery for many solid tumors and inflammatory bowel diseases (IBD). Preoperatively, the rate of cachexia exceeds 19% in patients with cancer, whereas 11% of patients with Crohn’s disease are considered underweight. 13,14 Low-weight surgical patients are often extremely ill, with a high risk of VTE secondary to their underlying disease or prolonged hospitalisation. 13,15,16
To date, no prospective study has examined whether prophylactic AFXa levels are achieved in low-weight patients receiving a receiving a substandard prophylactic enoxaparin dose. This study aimed to determine the percentage of low-weight surgical patients achieving peak AFXa levels appropriate for thromboprophylaxis after subcutaneous administration of a fixed dose of enoxaparin 30 mg once per day.
Methods
A prospective cohort study was conducted at King Faisal Specialist Hospital and Research Centre, Riyadh, in accordance with the amended Declaration of Helsinki and after receiving institutional review board approval (RAC#2181086). The inclusion criteria were as follows: age > 18 years, low bodyweight (<57 kg in males and <45 kg in females), and admission for abdominal, pelvic, or noncardiac thoracic surgery from May 2018 to May 2019. The exclusion criteria were as follows: pregnancy, renal impairment (glomerular filtration rate of <60 mL/min/1.72 m2, calculated using the Modification of Diet in Renal Disease equation), known bleeding disorder or history of thrombosis, <3 consecutive doses of enoxaparin, development of heparin-induced thrombocytopenia or a platelet count of <50 000/µL, or bodyweight <30 kg.
On admission to the hospital, low-weight patients began VTE prophylaxis with subcutaneous enoxaparin (Sanofi-Aventis) 30 mg once daily, which was administered at a scheduled time of 21:00. Patients fulfilling the inclusion and exclusion criteria were consented for enrollment in the study. Patients admitted multiple times to our surgical service were enrolled for their first hospital stay only and sampled for one AFXa level.
Peak AFXa level was measured 4 hours after the third dose of enoxaparin had been administered subcutaneously; each dose was given once daily. If a peak AFXa level was sampled before surgery, it was considered “preoperatively assessed,” while if it was sampled after surgery, it was considered “postoperatively assessed.” AFXa levels obtained from patients admitted for surgical intervention but who received conservative management were considered “preoperatively assessed” during the data analysis. The daily enoxaparin dose was changed to 40 or 20 mg for sub- or supra-prophylactic peak AFXa levels, respectively. Anti-factor Xa levels were assessed using the chromogenic-based liquid STA assay for unfractionated heparin and LMWH (Diagnostica, STAGO, S.A.S).
Patient demographics and clinical characteristics including the Caprini score were recorded. 17 The surgical diagnosis (i.e, indication for surgery), underlying disease state (malignancy or IBD), time of initiating enoxaparin prophylaxis, and duration of enoxaparin prophylaxis were also recorded. Bilirubin level was obtained on the day of blood sampling for AFXa level.
Patients were followed for 4 weeks to assess whether they developed VTE or bleeding. Those who did not undergo surgery were followed for 4 weeks after hospital admission and those who underwent surgery were followed for 4 weeks postoperatively. Patients discharged from the hospital before the end of the 4-week follow-up period were evaluated at the surgical clinic.
Study Outcomes
The primary study endpoint was the achievement of a peak AFXa level adequate for VTE prophylaxis (0.2-0.5 IU/mL). 9 Secondary endpoints included the association between achieving a prophylactic peak AFXa level and various patient factors and clinical states: sex, age, weight, body mass index (BMI), low-BMI (BMI < 18.5 kg/m2) or normal BMI (≥18.5 kg/m2), time of measuring AFXa level (pre- or postoperatively), diagnosis of cancer or IBD, and bilirubin level. Secondary endpoints also included the correlation between peak AFXa level and patient weight or BMI, as well as the incidence of VTE or clinically relevant bleeding. VTE was defined as deep vein thrombosis (DVT) or pulmonary embolism (PE) or both and was classified as symptomatic or incidental. The diagnosis of VTE was based on imaging analysis (ultrasound for DVT and spiral computed tomography for PE) and was confirmed by a non-investigator treating physician. Major bleeding was defined according to the International Society on Thrombosis and Haemostasis (ISTH) definition for surgical patients. 18 Bleeding that did not meet this definition was considered clinically relevant non-major bleeding (CRNMB) and recorded as well.
Statistical Analysis
We enrolled all patients who met the inclusion and exclusion criteria over 1 year. A sample size calculation was performed to ensure enrolment of a representative number of patients. In the absence of a compatible study in the literature for sample calculation, the number of low-weight patients admitted for surgery to our institution in the preceding year was used for this purpose (165 patients). Therefore, the minimum sample size required to estimate achievement of a prophylactic AFXa level, with a margin of error of 5% and 95% confidence interval (CI), was 115 patients.
Normally distributed data were expressed as mean ± SD. Non-normally distributed continuous data were reported as medians with ranges (minimum to maximum or interquartile range). Student t test was used to compare the means of normally distributed data, while the Mann–Whitney U test was used to compare the differences between continuous and non-normally distributed data. The χ2 test or Fisher’s exact test (for cell counts < 5) was used to examine associations between categorical variables reported as counts and percentages. Spearman ρ test was used to assess correlations. Univariate analysis using logistic regression analysis with the forced entry technique was used to assess the associations for predetermined covariates. Covariates with a P < .1 during univariate analysis were considered for the multivariate logistic regression model. Statistical significance was set at P < .05. Statistical analysis was performed using SPSS, version 20.0 IBM.
Results
Patient and Treatment Characteristics
Of 158 eligible patients, 121 were enrolled in the study, with the majority of exclusions due to renal impairment (17 patients). The remaining exclusions were due to a history of hematologic or thrombotic disease (7 patients), failure to complete 3 consecutive doses of enoxaparin (10 patients), and consent rejection (3 patients). The demographic and clinical characteristics of the included patients are presented in Table 1. Males constituted 61.9% of the cohort, and 57.9% of the study patients had a malignancy. Among the 51 cancer-free patients, 22 (18.2% of the total cohort) had a diagnosis of IBD. The majority of the patients were classified into “high risk” category for VTE according to their Caprini score. Eighteen (14.8%) patients were admitted to the hospital for possible surgery but received only conservative management. Bodyweight differed according to sex: mean weight was lower in females (39.2 ± 5.8 kg) than males (48.0 ± 6.2 kg, P < .001), while the mean BMI was comparable between females (17.1 ± 3.0 kg/m2) and males (17.3 ± 2.2 kg/m2; P = .773).
Clinical Characteristics and Demographics of the Study Cohort.

Abbreviations: BMI, body mass index; HIPEC, hyperthermic intraperitoneal chemotherapy.
The median time to start enoxaparin VTE prophylaxis postoperatively was 17.7 hours (6-53 hours); 42 (41.1%) of the 102 patients who underwent surgery received enoxaparin within the first 12 hours postoperatively. The median calculated enoxaparin dose was 0.66 mg/kg and was higher in females (0.73 [0.56-1.01] mg/kg) than in males (0.61 [0.40-0.90] mg/kg, P < .01).
Outcomes
Overall, 80 (66.1%) of the 121 study patients achieved a prophylactic peak AFXa level (Table 2). The proportion of achieving a prophylactic AFXa level per sex was higher in female patients (84.8%) compared to male patients (54.7%, P = 0.001). Table 3 shows the clinical characteristics of patients who did or did not achieve a prophylactic peak AFXa level. All out-of-range peak AFXa levels were sub-prophylactic (33.8%). The median peak AFXa level in the cohort was 0.29 (0.1-0.5) IU/mL and was lower in males (0.24 [0.1-0.47] IU/mL) than females (0.31 [0.1-0.5] IU/mL; P < .001). Bodyweight was lower in patients who achieved the prophylactic level than those who did not. The median of AFXa levels obtained were 0.23 (0.1-0.5) IU/mL preoperatively and 0.21 (0.1-0.48) IU/mL postoperatively (P = .62).
Outcome Characteristics for the Study Cohort.

Abbreviations: AFXa, anti-factor Xa; VTE, venous thromboembolism.
Comparisons of Patient Characteristics With Prophylactic and Sub Prophylactic AFXa Levels.
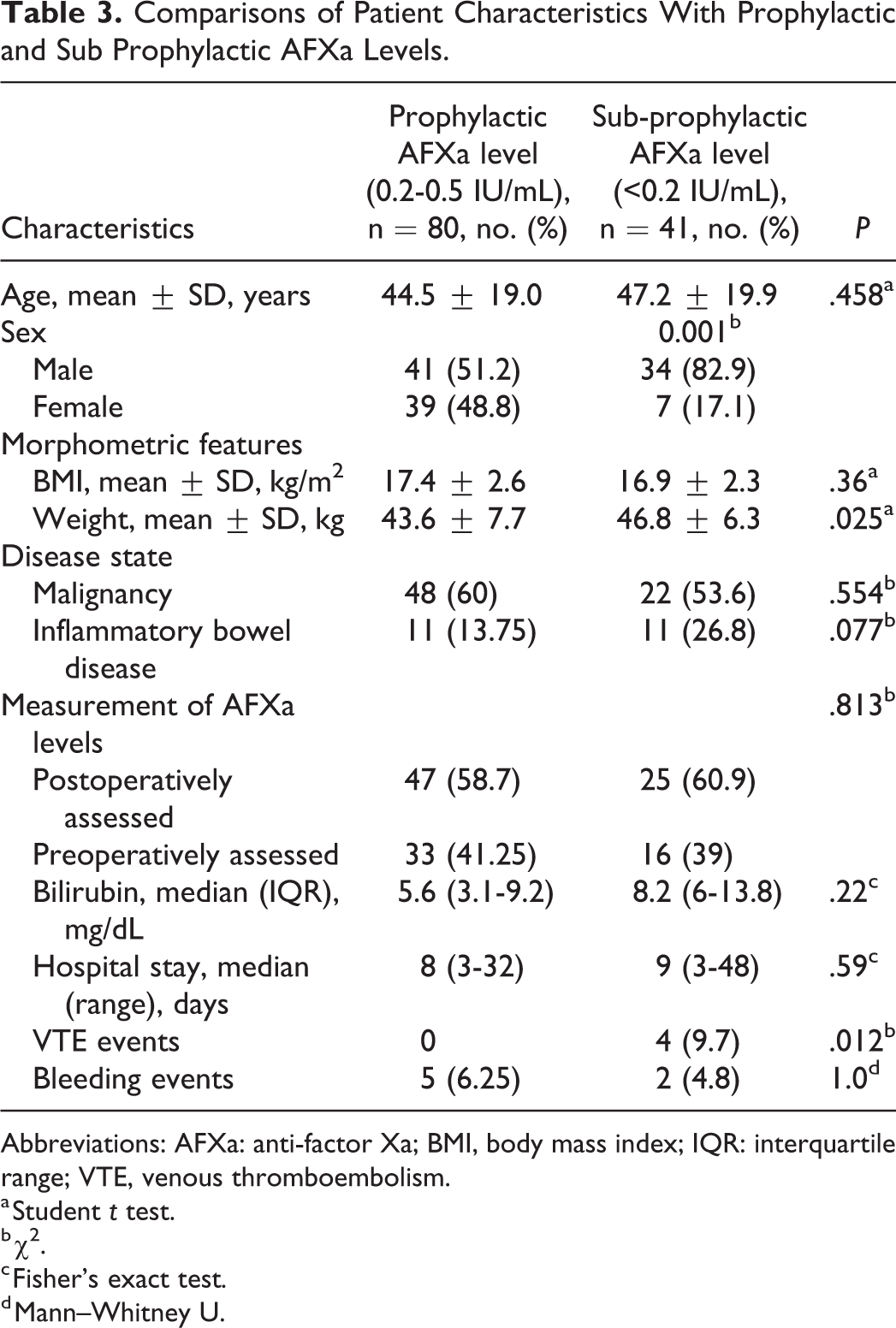
Abbreviations: AFXa: anti-factor Xa; BMI, body mass index; IQR: interquartile range; VTE, venous thromboembolism.
a Student t test.
b χ2.
c Fisher's exact test.
d Mann–Whitney U.
On univariate analysis, females were 4.6 times more likely to achieve a prophylactic AFXa level than males (odds ratio [OR]: 4.6; 95% CI: 1.83-11.64; P = .001; Table 4). A significant negative association was observed between weight and peak AFXa prophylactic level (OR: 0.93; 95% CI: 0.88-0.99; P = .028). For each 1 kg increase in weight, the log of odds of achieving a prophylactic level was decreased by 6.4% (Figure 1). Multivariate logistic regression analysis showed that sex was independently associated with achieving a prophylactic peak AFXa level (OR: 3.17; 95% CI: 1.32-11.94; P = .014) but not weight (OR: 0.98; 95% CI: 0.92-1.05; P = .63) or IBD (OR: 0.43; 95% CI: 0.16-1.18; P = .1). In patients with weights < 45 kg, the percentage of achieving a prophylactic peak AFXa level was also lower in males (52.2%) than in females (84.8%; P = .004). A weak negative correlation was observed between prophylactic peak AFXa level and weight (r = −0.22, P = .02) but not with BMI (r = 0.08 and P = .38).
Univariate and Multivariate Logistic Regression Analyses of the Relative Achievement of a Prophylactic AFXa Level With Baseline Variables.
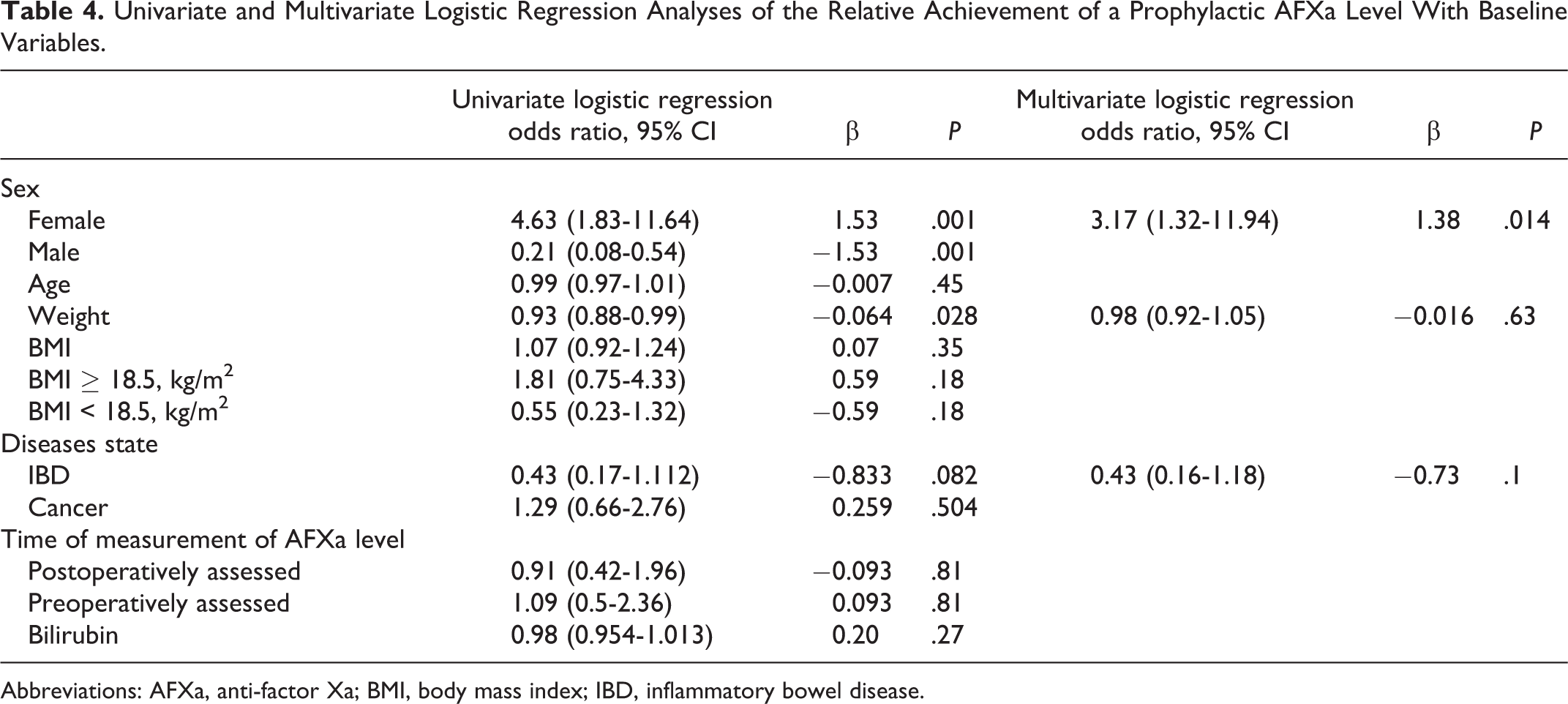
Abbreviations: AFXa, anti-factor Xa; BMI, body mass index; IBD, inflammatory bowel disease.

Patient weight versus anti-factor-Xa level in low-weight surgical patients receiving enoxaparin 30 mg.
In total, 4 VTE events (3.3%) were diagnosed in the study cohort. All events occurred in 4 hospitalized patients with sub-prophylactic peak AFXa levels (9.7%); no patient with a prophylactic peak AFXa level developed a VTE (P = .012). Two patients developed a PE event postoperatively, and two developed a central catheter associated subclavian DVT (Table 2).
One patient developed a segmental PE (on day 5 postoperatively) after enoxaparin was temporarily discontinued due to postoperative bleeding. The other patient was incidentally diagnosed with subsegmental PE 20 days postoperatively. Both DVT events were symptomatic: one was diagnosed preoperatively, and the other was diagnosed 15 days postoperatively. Nine (7.4%) patients did not return to the surgery clinic for follow-up after discharge from the hospital.
Two (1.6%) patients had major bleeding events, according to the ISTH criterion of a decrease in hemoglobin > 2 g/dL (20 g/L). Both patients received a transfusion of packed red blood cells, although no frank bleeding was observed. One patient experienced a decrease in hemoglobin level on day 3 postoperatively after sampling for peak AFXa level. The other patient, who was receiving conservative treatment for small-bowel obstruction, developed low hemoglobin after increasing enoxaparin dose to 40 mg following a sub-prophylactic peak AFXa level. Five additional patients developed CRNMB, resulting in a total of 7 (5.8%) patients who developed bleeding events. No differences were observed in the percentage of bleeding events between patients achieving a prophylactic peak AFXa level (6.25%) and those with a sub-prophylactic level (4.8%; P = 1.0). No fatalities were observed during the follow-up period.
Discussion
In this prospective study, 66% of low-weight, non-orthopedic surgical patients achieved prophylactic peak AFXa levels with a fixed dose of once daily subcutaneous enoxaparin 30 mg. At this dose, bodyweight was weakly inversely correlated with the achievement of a prophylactic level of peak AFXa in our low-weight patients (males <57 kg and females <45 kg). Dosing with subcutaneous enoxaparin 30 mg per day produced a median dose-to-weight ratio of 0.66 mg per kg in our low-weight patients.
A recent retrospective study of 35 low-weight patients (<55 kg) demonstrated a slightly higher proportion (74%) of adequate AFXa levels. However, 22% of the patients in that study developed supra-prophylactic AFXa levels, unlike our study, which had had no supra-prophylactic levels. This difference could be explained by the variability of enoxaparin doses used in the retrospective study, with majority of patients received 30 mg once daily. 19 The median dose of enoxaparin administered in the retrospective study (0.6 mg per kg) was similar to that in our study (0.66 mg per kg). Both median doses are lower than that used by Rojas et al who prospectively administered 40 mg enoxaparin to 56 patients weighing < 55 kg, for a calculated median dose of 0.8 mg per kg. 20 Rojas et al reported that 60.7% of their patients had an AFXa of ≥ 0.5 IU/mL. This high percentage of supra-prophylactic AFXa levels may increase the risk of bleeding in surgical patients, especially if enoxaparin is administered close to surgery. 2
The ENOXACAN study reported an 18% rate of bleeding in patients undergoing abdominal and pelvic surgery, all of whom received enoxaparin 40 mg as thromboprophylaxis. Patients with a BMI as low as 15.9 kg/m2 were included in that trial, and no reductions in enoxaparin dose were applied. This might explain the higher bleeding rates in the ENOXACAN study compared to our study. Both the ENOXACAN study and our study excluded renally impaired patients. We excluded patients with any stage of renal insufficiency because of the lack of evidence supporting the safety of enoxaparin thromboprophylaxis in low-weight patients who also have impaired renal function.
A prospective study of 2 prophylactic doses of enoxaparin, 40 mg daily and 30 mg twice daily administered to average weight cancer patients, demonstrated the association of female sex with adequacy of peak AFXa level. 21 Another study of obese patients who received enoxaparin VTE prophylaxis (88.5% of patients received 40 mg daily) found that female sex was associated with higher AFXa level. 22 This sex effect disparity for AFXa level was also observed in our study, where a higher proportion of females achieved the prophylactic range of AFXa. Therefore, females are more likely than males to achieve adequate prophylaxis across a range of body weights.
The higher rate of adequate prophylaxis observed in females could be explained by their lower body water content and plasma volume compared to males. 23 As a sodium salt, enoxaparin is a hydrophilic compound that distributes primarily into the vascular space. 10,24 Accordingly, enoxaparin would be more concentrated in smaller aqueous environments, such as blood. Another possible explanation is that sex-dependent differences in biotransformation have been observed for some drugs, including heparin. As a heparin derivative, enoxaparin might undergo lower clearance in females compared to males. 25 Although in our study, the mean weight of females was lower than that of males, even when we limited our analysis to only patients <45 kg in both sexes, the percentage of achieving a prophylactic peak AFXa level remained higher in females than males.
Based on the World Health Organization classification, adults with a BMI of < 18.5 kg/m2 are considered underweight. 26 In our study, the likelihood of achieving a prophylactic level of peak AFXa did not differ between underweight patients and those with a BMI ≥18.5 kg/m2. Moreover, no association was observed in our study between BMI and achievement of a prophylactic AFXa level. Therefore, for patients with an extremely low weight, lowering the dose of prophylactic enoxaparin according to weight rather than BMI is preferred. When prescribing or dispensing enoxaparin, clinicians should be cognizant that “underweight” and “low-weight” are not interchangeable terms. Of note, the definition of low bodyweight was inconsistent among studies assessing enoxaparin prophylaxis, with <55 kg set as a cutoff weight in some studies, irrespective of patient sex. 19,20 A prospective study found a significant increase in bleeding risk among patients receiving enoxaparin for VTE treatment and weighing <55 kg. 27 However, that study assessed a therapeutic dose of enoxaparin (1 mg per kg every 12 hours) for the treatment of an established VTE. Therefore, extrapolating outcomes of therapeutic doses of enoxaparin to prophylactic doses is impractical.
The effectiveness of a fixed-dose enoxaparin strategy for VTE prophylaxis has been questioned by many experts, who recommended either weight-based dosing regimens or real-time monitoring of AFXa levels as alternatives. 20,23 Although the majority of patients in our study had a peak AFXa level consistent with adequate VTE prophylaxis using a fixed-dose strategy, our patients’ weights were within a relatively narrow range: 30 to 57 kg for males and 30 to 45 kg for females.
Significant weight loss is commonly observed in patients transferred to tertiary care centers for surgical management. Guidelines recommend 7 to 10 days of preoperative parenteral nutrition (PN) in severely malnourished patients with IBD or cancer to reduce the risk of postoperative complications. 28,29 However, the need for central venous access for PN infusions predisposes to catheter-related thrombosis, PE, and vena cava syndrome. 30 In our study, two subclavian DVTs developed in patients receiving PN via a central catheter, representing 50% of all VTE events. One of these upper extremity DVTs developed in a patient receiving PN preoperatively. Therefore, the real-world effectiveness of achieving prophylactic AFXa levels in surgical patients should be assessed in the context of the entire hospital stay. Of note, we found no difference between the AFXa levels obtained pre- and postoperatively.
Our study had some limitations. Firstly, it lacked a control arm, such as patients receiving enoxaparin 40 mg. However, we chose not to expose our low-weight surgical patients, who are prone to bleeding, to 40 mg of enoxaparin, which could be contrary to the US FDA label advice. Secondly, we did not reassess the AFXa levels after increasing the enoxaparin dose in patients with sub-prophylactic peak AFXa levels, as we were unsure whether these patients would remain hospitalized long enough to remeasure their AFXa levels. Thirdly, we reported the rate of VTE development in our cohort; however, we want to stress that the study was not powered to detect a difference between patients who achieved and those who did not achieve the prophylactic AFXa range.
Although the majority of low-weight patients received adequate thromboprophylaxis with a daily dose of enoxaparin 30 mg, one-third of the patients did not achieve the prophylactic AFXa range. Therefore, we recommend that clinicians monitor peak AFXa levels when using this fixed dose of enoxaparin in this patient population, especially in males.
In conclusion, this prospective study demonstrated that two-thirds of low-weight, non-orthopedic surgical patients achieved a prophylactic peak AFXa level with a fixed-dose of enoxaparin 30 mg once daily. With this dose, low-weight females (<45 kg) were more likely than males to achieve peak AFXa levels considered adequate for thromboprophylaxis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
