Abstract
Cancer-associated venous thromboembolism (CAT) guidelines recommend direct oral anticoagulants as alternatives to low-molecular-weight heparin (LMWH) in most patients. This study compared the effectiveness and safety of rivaroxaban versus LMWH for a broad CAT cohort. The cohort study used electronic health data from January 2012 to December 2020 to evaluate patients with active cancer experiencing acute venous thromboembolism (VTE) and treated with rivaroxaban or LMWH. Propensity score-overlap weighted hazard ratios (HRs) and 95% confidence intervals (CIs) for VTE, bleeding-related hospitalization, and all-cause mortality were calculated. In total, 4935 patients were identified (27.9% on rivaroxaban and 72.1% on LMWH). The cancer types included gastrointestinal (29.4%), genitourinary (26.2%), lung (24.0%), breast (19.7%), and hematologic (14.4%). Rivaroxaban was associated with a reduction in recurrent VTE versus LMWH among all patients with cancer (HR = 0.78; 95%CI = 0.61-0.99) at 3 months. No differences in bleeding-related hospitalization or all-cause mortality were observed. Directionally similar results to those at 3 months were observed at 6 months for all outcomes. In conclusion, we observed fewer recurrent VTE cases and no increase in bleeding-related hospitalizations with rivaroxaban versus LMWH at 3 months in this patient cohort with various cancer types.
Introduction
VTE is a clotting condition that includes deep vein thrombosis (DVT) and pulmonary embolism (PE). Malignancy and chemotherapy increase patients’ risk for VTE, with the annual incidence of VTE in patients with cancer estimated at roughly 1 case per 200 persons.1–4 Aside from cancer progression, thromboembolism is one of the most prominent causes of mortality in patients with cancer. 5
Anticoagulant therapy is recommended for the treatment of CAT for a duration of 6 months.6,7 Unfortunately, there is a potential for increased harm when using anticoagulant therapy because patients with cancer also have an inherently higher risk of bleeding and major bleeding events.2,8
LMWH treatment became a standard of care for CAT following the Randomized Comparison of Low-Molecular-Weight Heparin vs Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) trial, which was published in 2003. 9 Recent trials have demonstrated that direct oral anticoagulants (DOACs) are at least as efficacious as LMWH therapy for VTE treatment in patients with cancer.10–14 Although trials suggest that DOACs are an acceptable alternative to LMWHs for lowering the incidence of VTE events, some have differed in the impact on major bleeding events.6,7 Thus, guidelines suggest that DOACs are acceptable alternatives to LMWHs for many patients, with caution to be used in patients with cancer with an increased risk of bleeding on DOACs, such as those with gastrointestinal (GI) and genitourinary cancers.6,7
We performed a secondary analysis of the Observational Study in Cancer Associated Thrombosis for Rivaroxaban-United States (OSCAR-US) program (NCT04979780) to compare the effectiveness and safety of rivaroxaban versus LMWH treatment in an unselected cohort of patients with CAT. Prior publication analyses had focused on CAT patients meeting guideline criteria and had a lower risk of bleeding.6,7,15
Methods
Data Source
This was a retrospective cohort study performed in the US Optum® deidentified electronic health records (EHR) dataset. 16 Data from January 2012 through December 2020 were utilized. In its entirety, the Optum EHR database provides longitudinal patient-level medical record data for approximately 91 + million insured and uninsured persons of all ages seen at approximately 700 hospitals and 7000 clinics in the US. The database includes diagnosis and procedure codes, laboratory results, vital signs, body measurements, other clinical observations, and data on prescribed and self-reported medications. 16
Cohort Selection
We included adults with active cancer presenting to the hospital, observation unit, or emergency department for acute VTE (DVT and/or PE) on or after January 1, 2013 (in line with the US availability of rivaroxaban for VTE, though rivaroxaban does not have a specific indication for CAT), and these patients were treated with therapeutic VTE doses of rivaroxaban or LMWH (doses of enoxaparin 30 or 40 mg once daily were excluded) per written prescription or self-report on day 7 post-acute VTE diagnosis (designated as the index date). Additionally, patients had to be active in the EHR dataset for ≥12 months before the index event and had ≥1 provider visit in the 12 months before the index VTE event (designated as the baseline period). Active cancer was defined as cancer actively treated with systemic therapy (ie, chemotherapy, radiation therapy, surgery, or immunotherapy) within 6 months prior to the index CAT, or metastatic disease, regardless of the time from cancer diagnosis or treatment. Patients were excluded if they had atrial fibrillation or valvular heart disease, used anticoagulation during the baseline period, initiated rivaroxaban at a dose other than 15 mg twice daily, used nontherapeutic doses of other DOACs or LMWHs, or were pregnant.
The outcomes of this study included the composite of recurrent VTE 17 or any bleeding-related hospitalization (per the Cunningham algorithm), 18 recurrent VTE and any bleeding-related hospitalizations separately, and all-cause mortality at 3 and 6 months from the index date. Recurrent VTE was identified by the presence of an appropriate primary inpatient diagnosis code at discharge. 17 All study outcomes were ascertained based on International Classification of Diseases–9/10–Clinical Modification diagnosis or procedure codes, Current Procedural Terminology–4 or Healthcare Common Procedure Coding System codes, or vital statistics.
Statistical Analysis
Baseline demographics and comorbidities were reported using descriptive statistics. Categorical data were expressed as percentages, while continuous data were reported as means ± standard deviations or medians and interquartile ranges as appropriate.
The presence of a comorbidity was determined through billing codes and/or supporting laboratory or observation data, whereas the absence of data supporting a comorbidity's existence was presumed to represent the absence of disease. Consequently, binary comorbidity disease states did not have any missing data and did not require imputation for missing values. Missing values for continuous laboratory and observation data (eg, estimated glomerular filtration rate and body mass index) were imputed using a fully conditional specification linear regression model multiple imputation approach utilizing all other available variables in the model. Imputation of missing outcome data was not performed.
A logistic regression model was used to calculate propensity scores (PS) based on covariates and risk factors associated with differential anticoagulant exposure identified at baseline. 19 PSs were subsequently used to assign weights to each patient using a PS overlap weighting (OLW) method. 20 This method assigns weights to individual patients proportional to their probability of belonging to the opposing treatment group (‘1 – PS’ for rivaroxaban, and ‘PS’ for LMWH patients). By design, OLW yields the exact balance in all covariates entered in the PS model. This method retains all patients in the dataset and assigns less weight to those with PSs at the extremes (thereby avoiding common concerns with PS matching or inverse probability weighting).
We fit weighted Cox regression models to evaluate HRs with accompanying 95% CIs for rivaroxaban versus LMWH, with the anticoagulant received as the only covariate included in the model. Statistical analyses were performed using IBM SPSS version 27.0 (IBM Corp., Armonk, NY) and SAS version 9.4 (SAS Institute, Cary, NC). The proportional hazard assumption was tested and shown to be valid for all analyses. Patients were censored in the models at the time of outcome occurrence, end-of-EHR activity, end of data availability, or end of follow-up (capped at 6 months), whichever came first; and they were analyzed using an intent-to-treat approach (ie, evaluated according to their index anticoagulant group). Time from index anticoagulant start to end of follow-up was considered as “time at risk.” p < 0.05 was deemed statistically significant, unless otherwise stated. A Kaplan-Meier analysis was performed for the composite outcome of recurrent VTE or bleeding-related hospitalization to generate a time-to-event curve at 6 months. An exploratory analysis limited to patients in whose time on anticoagulation could be assessed (ie, both start and stop dates for index anticoagulant were available) and analyzed using an on-treatment approach (ie, censored at outcome occurrence, index anticoagulant switch or discontinuation, end-of-EHR activity, end of data availability, or end of follow-up) was also conducted. Two additional exploratory analyses were performed: an analysis of clinically relevant non-major bleeding and an analysis of bleeding-related hospitalization subtypes in rivaroxaban versus LMWH users at 3 and 6 months. The bleeding-related hospitalization subtypes included critical organ (intracranial, intraspinal, intraocular, retroperitoneal, intra-articular, pericardial, or intramuscular with compartment syndrome), intracranial, and extracranial hemorrhage.
Subgroup and Sensitivity Analyses
Subgroup analyses were conducted in order to evaluate the effectiveness and safety of rivaroxaban versus LMWH in patients with the presence or absence of metastatic disease, undergoing or not undergoing surgical procedures within 6 months, and stratified by individual cancer subtypes with at least a 10% prevalence in both the rivaroxaban and LMWH treatment arms. For this study, GI cancers were defined as esophageal, stomach, intestine, colorectal, hepatobiliary, and pancreatic. Genitourinary cancers included bladder, kidney, urothelial, prostate, testicular, and penile. Hematologic cancers included any leukemia or lymphoma, including myeloma. p values for interaction were calculated to test for the presence of statistical interactions across these subgroups. To reduce the chances of obtaining false-positive results due to multiple hypothesis testing, we utilized a Bonferroni-corrected p value < .0125 to indicate a statistically significant subgroup interaction. Two sensitivity analyses were performed to compare rivaroxaban to LMWH. The first analysis was performed whereby a proportional hazards model for the sub-distribution of competing risk was fit. The second analysis was performed to evaluate the primary outcomes of rivaroxaban compared to LMWH in patients with a VTE index event on or after January 1, 2018. PS weights were re-calculated for each subgroup analysis, including unique cancer types.
Research Ethics and Reporting
To preserve patient anonymity and confidentiality, the Optum EHR dataset is deidentified in accordance with the Health Insurance Portability and Accountability Act (HIPAA) of 1996. The Optum EHR dataset has been determined by the New England Institutional Review Board (IRB) to not involve human subjects research and, therefore, has been deemed exempt from further IRB review. 15
This paper was authored to meet the “reporting of studies conducted using observational routinely collected health data statement for pharmacoepidemiology” (RECORD-PE) statement criteria. 21
Results
In total, we identified 4935 active cancer patients experiencing a CAT event who were treated with rivaroxaban (27.9%) or LMWH (72.1%) (Figure 1). Of the LMWH patients, 99.4% were treated with enoxaparin at a median dose per administration of approximately 1.0 mg/kg twice daily. The median duration of therapy in the rivaroxaban cohort was 180 (60, 180) days and 98 (40, 180) days in the LMWH cohort. Prior to OLW, patients in the LMWH cohort appeared to have more advanced disease (higher proportions of metastatic disease, pulmonary embolism, patients being frequently admitted to the hospital, and ongoing or recent active cancer treatment) than those receiving rivaroxaban (Table 1). Cancer types also varied between the two anticoagulant treatment groups (breast, the lungs, pancreatic, and GI) prior to OLW. Following OLW, the characteristics of the rivaroxaban and LMWH groups were similar for all variables in the multivariate logistic regression PS model. At baseline, about one-quarter of patients were ≥75 years of age, 55.9% were female, 19.0% of patients’ body mass index was ≥35 kg/m2, and 18.2% had estimated glomerular filtration rates <60 mL/min. The type of index CAT event was PE ± DVT in 52.0% of included patients. Nearly 50% of all patients were admitted to the hospital ≥2 times in the 12 months before the index VTE event. In total, 46.3% of patients’ malignancies were metastatic, and 60% had received systemic cancer treatment during the 4 weeks before or after their CAT. The most frequent cancers observed were GI (29.4%), genitourinary (26.2%), the lungs (24.0%), breast (19.7%), and hematologic (14.4%).

Flow diagram for patient inclusion and exclusion. CAT, cancer-associated venous thromboembolism; DOAC, direct oral anticoagulant; and LMWH, low-molecular-weight heparin.
Characteristics of Unweighted and Overlap-Weighted Rivaroxaban and Low-Molecular-Weight Heparin Patients with Cancer-Associated Thrombosis.

BMI, body mass index; eGFR, estimated glomerular filtration rate; LMWH, low-molecular-weight heparin; NSAID, nonsteroidal anti-inflammatory drug; OLW, overlap weighted; SD, standard deviation; PPI, proton pump inhibitor; and VTE, venous thromboembolism.
Frailty was assessed in this study using the Hospital Frailty Risk Score developed and validated by Gilbert T, et al31 The Hospital Frailty Risk Score has been shown to perform at least as well as existing frailty or risk stratification tools. It is derived from the international classification of disease codes and implemented into electronic health records and claims data sets.
Cancer types add up to greater than 100% as both patients’ primary and metastatic cancer locations were counted.
Table 2 provides weighted outcomes data at 3 and 6 months. At 3 months, there was a significant 29% relative hazard reduction in the incidence of the composite outcome of recurrent VTE or bleeding-related hospitalization associated with rivaroxaban versus LMWH use (HR 0.71; 95% CI 0.58-0.87). This hazard reduction, with rivaroxaban versus LMWH treatment, of the composite outcome was also demonstrated by the Kaplan-Meier curve (Figure 2). There was a significant 22% relative hazard reduction in recurrent VTE (HR 0.78; 95% CI 0.61-0.99) and a nonsignificant 21% relative hazard reduction in bleeding-related hospitalization (HR 0.79; 95% CI 0.59-1.07) with rivaroxaban versus LMWH use. No difference in all-cause mortality at 3 months (HR 0.94; 95% CI 0.77-1.14) was observed.

Time to the composite outcome of recurrent venous thromboembolism or bleeding-related hospitalization. Kaplan-Meier curve for the composite outcome of recurrent venous thromboembolism or bleeding-related hospitalization (rivaroxaban = solid line, low-molecular-weight heparin = dashed line).
Outcomes at 3 and 6 Months.

CI, confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin; OLW, overlap weighted; and VTE, venous thromboembolism.
At 6 months, there was a significant 21% relative hazard reduction in the incidence of the composite outcome with rivaroxaban versus LMWH use (HR 0.79; 95% CI 0.66-0.95). Although not statistically significant, rivaroxaban use was associated with numerically lower risks of recurrent VTE (HR 0.87; 95% CI 0.69-1.08) and bleeding-related hospitalization (HR 0.84; 95% CI 0.64-1.09) versus LMWH. No difference in all-cause mortality was observed at 6 months (HR 0.91; 95% CI 0.78-1.06).
Exploratory Analyses
Table 3 provides clinically relevant non-major bleeding data at 3 and 6 months. There was a nonsignificant 18% and 13% risk reduction of clinically relevant non-major bleeding at 3 and 6 months with rivaroxaban versus LMWH (HR 0.82; 95% CI 0.61-1.09 and HR 0.87; 95% CI 0.68-1.09).
Clinically Relevant Non-Major Bleed at 3 and 6 Months of Rivaroxaban Versus Low-Molecular-Weight Heparin Cohorts.
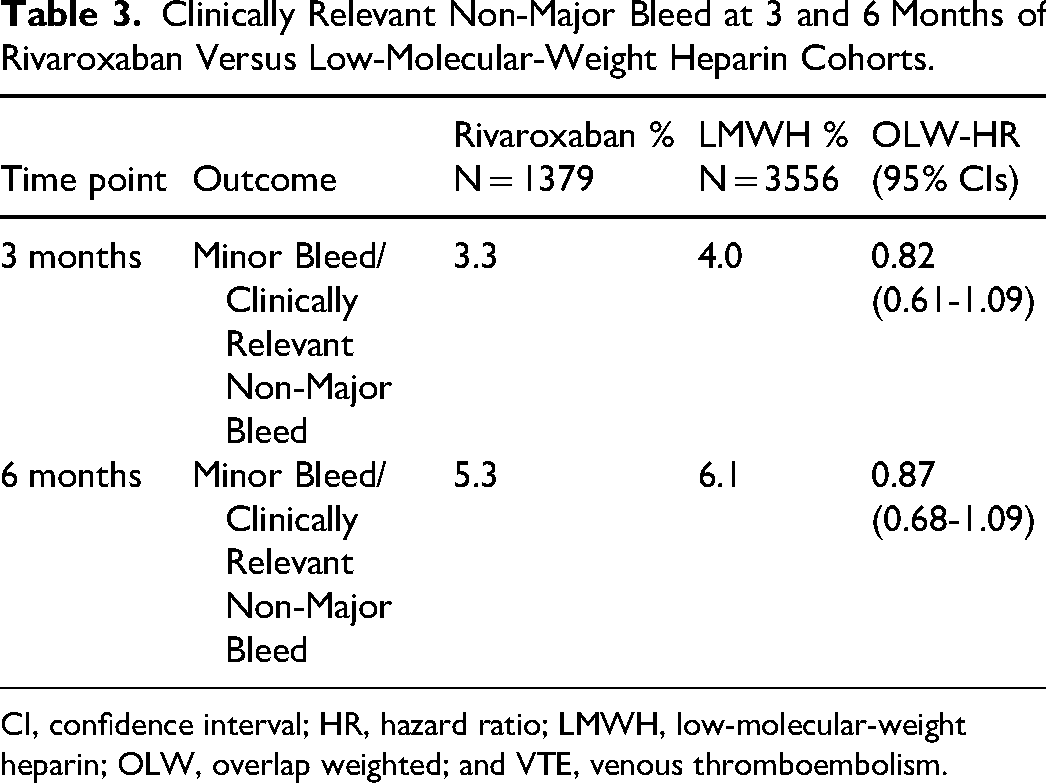
CI, confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin; OLW, overlap weighted; and VTE, venous thromboembolism.
Table 4 provides the evaluation of the outcomes of the subtypes of bleeding-related hospitalization data. The evaluation of the specific bleeding-related hospitalization outcomes suggested that rivaroxaban use was associated with significantly fewer critical organ bleeds versus LMWHs at 3 and 6 months (HR range: 0.08-0.59). These reductions appear to be driven by lower intracranial hemorrhage rates with rivaroxaban compared with LMWH. There was no difference in extracranial bleeding outcomes at 3 and 6 months between the two cohorts.
Bleeding Outcomes at 3 and 6 Months of Rivaroxaban Versus Low-Molecular-Weight Heparin Cohorts.

CI, confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin; OLW, overlap weighted; and VTE, venous thromboembolism.
Results of the on-treatment analysis were similar in direction to the main intention-to-treat analysis but showed more robust effect sizes in favor of rivaroxaban, and the net benefit was maintained from 3 months through 6 months (Supplemental Table 1).
Subgroup and Sensitivity Analyses
No statistical interaction was observed for the composite outcome of recurrent VTE or bleeding-related hospitalization, the outcomes separately, or for all-cause mortality when analyses were stratified by the presence or absence of metastatic disease (p ≥ .08 for all; Table 5) at both 3 and 6 months. There was no statistical interaction between patients with and without surgery within 180 days for the composite outcome of recurrent VTE or bleeding-related hospitalization, the outcomes separately, or for all-cause mortality at 3 and 6 months (p ≥ .32 for all; Table 6). The effectiveness and safety of rivaroxaban versus LMWH at 3 and 6 months stratified by individual cancer subtypes are depicted in Table 7. There were significant reductions in the risk of recurrent VTE in both lung and GI cancers with the use of rivaroxaban at the 3-month (HR 0.60; 95% CI 0.38-0.96 and HR 0.59; 95% CI 0.36-0.96) and 6-month (HR 0.60; 95% CI 0.39-0.93 and HR 0.59; 95% CI 0.37-0.94) time periods, respectively, but no changes in bleeding-related hospitalizations versus LMWH were seen. Conversely, there was no reduction in recurrent VTE in genitourinary cancer at 3 months and 6 months, but the risk of both bleeding-related hospitalizations (HR 0.24; 95% CI 0.09-0.67) and all-cause mortality (HR 0.31; 95% CI 0.15-0.65) were significantly reduced at 3 months.
Outcomes in Patients with or Without Metastatic Disease.

Unweighted population sizes: metastatic disease (rivaroxaban, n = 549; LMWH, n = 2074); no metastatic disease (rivaroxaban, n = 830; LMWH, n = 1482).
CI, confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin; OLW, overlap weighted; and VTE, venous thromboembolism.
Outcomes in Patients Undergoing or not Undergoing Surgery Within 180 Days.
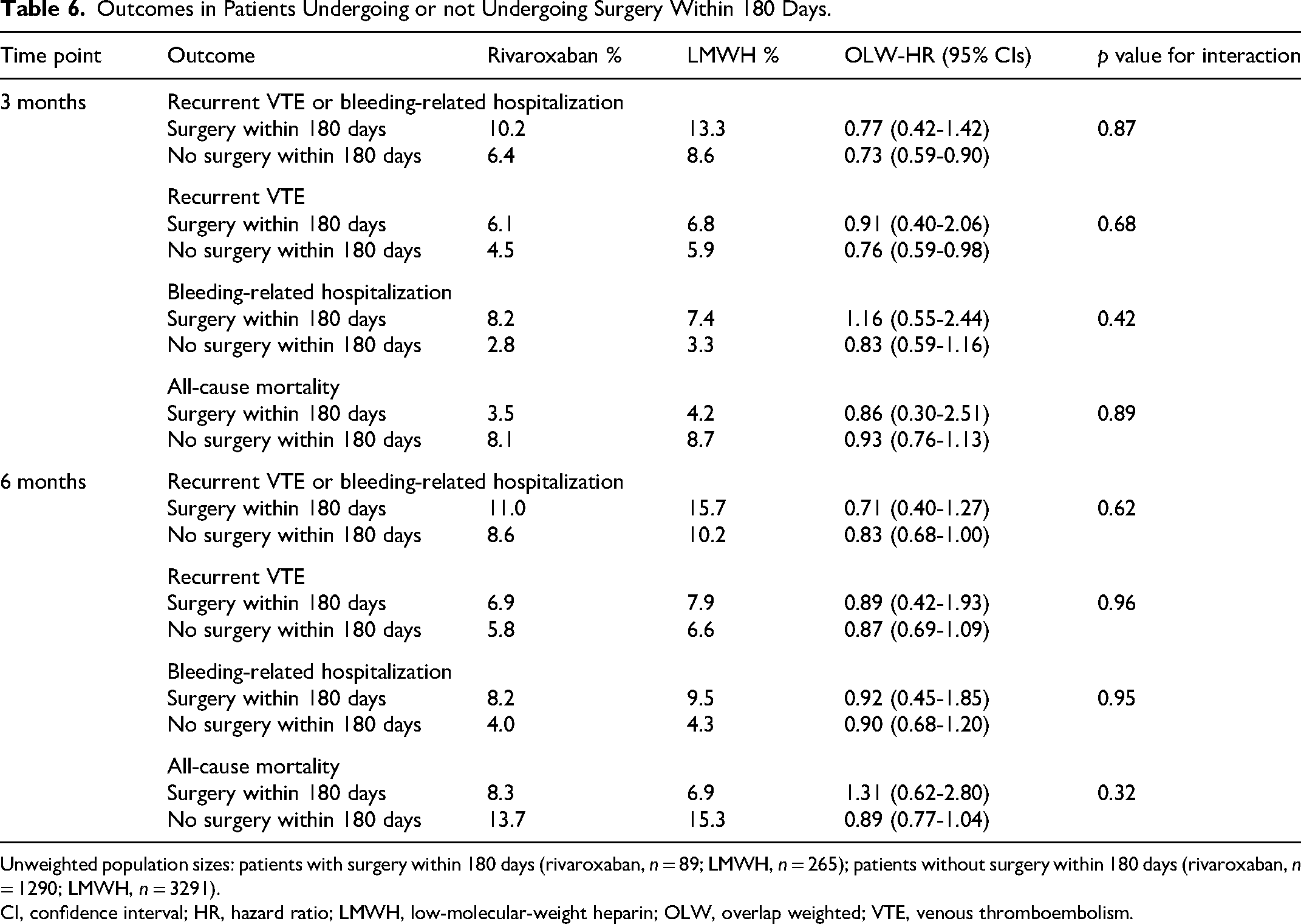
Unweighted population sizes: patients with surgery within 180 days (rivaroxaban, n = 89; LMWH, n = 265); patients without surgery within 180 days (rivaroxaban, n = 1290; LMWH, n = 3291).
CI, confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin; OLW, overlap weighted; VTE, venous thromboembolism.
Outcomes by Cancer Types Occurring in at Least 10% of Patients.
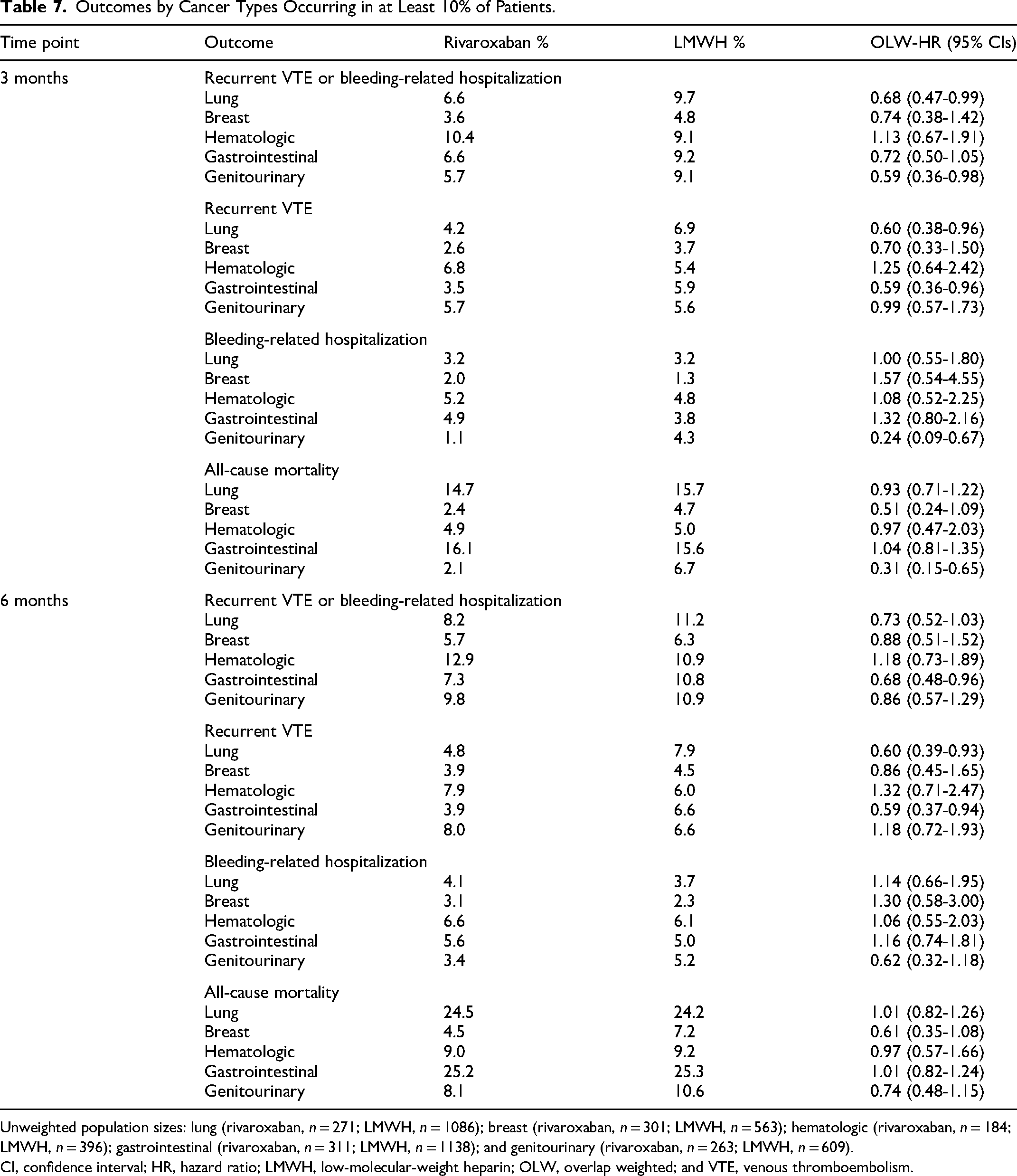
Unweighted population sizes: lung (rivaroxaban, n = 271; LMWH, n = 1086); breast (rivaroxaban, n = 301; LMWH, n = 563); hematologic (rivaroxaban, n = 184; LMWH, n = 396); gastrointestinal (rivaroxaban, n = 311; LMWH, n = 1138); and genitourinary (rivaroxaban, n = 263; LMWH, n = 609).
CI, confidence interval; HR, hazard ratio; LMWH, low-molecular-weight heparin; OLW, overlap weighted; and VTE, venous thromboembolism.
When a proportional hazards model for the sub-distribution of competing risk was applied, results for the recurrent VTE and bleeding-related hospitalization at 3 (HR 0.81; 95% CI 0.51-1.30 and HR 0.82; 95% CI 0.47-1.42) and 6 months (HR 0.89; 95% CI 0.56-1.40 and HR 0.77; 95% CI 0.47-1.28) had similar direction and magnitude of effect sizes.
In the sensitivity analysis of patients with an index event on or after January 1, 2018, rivaroxaban had directionally similar results to all the primary outcomes in the main analyses, except for a nonsignificant 28% and 25% higher risk of bleeding-related hospitalization at 3 and 6 months. (Supplemental Table 2).
Discussion
Our study of nearly 5000 patients with active cancer and experiencing a VTE showed that patients treated with rivaroxaban had a reduced hazard of the composite outcome of recurrent VTE or bleeding-related hospitalization compared with LMWH at both 3 months and 6 months. Patients treated with rivaroxaban had a reduced hazard of recurrent VTE at 3 months; however, while the direction of effect showed a persistent reduction at 6 months, it did not maintain statistical significance. Although not statistically significant, the HRs <1.0 for bleeding-related hospitalization at 3 and 6 months suggest a lower risk associated with rivaroxaban use. The use of rivaroxaban versus LMWH did not differ in all-cause mortality at 3 or 6 months. No statistical interaction was observed for the composite outcome or the individual outcomes for the presence or absence of metastatic disease in subgroup analysis. Our exploratory analyses also found reductions in VTE recurrence in patients with lung and GI cancers at 3 and 6 months and reductions in bleeding-related hospitalization events and all-cause mortality in patients with genitourinary cancer at 3 months in rivaroxaban-treated patients versus LMWH-treated patients. These results are interesting; however, further (larger) analyses are needed to evaluate DOAC and LMWH use for CAT in patients with lung, GI, and genitourinary cancers.
In the Anticoagulation Therapy in Selected Cancer Patients at Risk of Recurrence of Venous Thromboembolism (SELECT-D) trial, 14 406 patients with active cancer and a VTE were randomized to receive either rivaroxaban or dalteparin for 6 months. 14 The investigators found a significant reduction in recurrent VTE with rivaroxaban versus dalteparin (4% vs 11%; HR 0.43; 95% CI 0.19-0.99), with the two arms diverging in risk around 2–3 months after anticoagulation initiation. This result is consistent with the present study's finding of a significant 22% reduction in recurrent thrombosis risk with rivaroxaban compared to LMWH at 3 months (4.7% vs 5.9%; HR 0.78; 95% CI 0.61-0.99). One limitation of the SELECT-D trial is that the inclusion criteria were changed during the trial due to the high risk of gastrointestinal bleeding in those with luminal upper GI tumors. The fact that a minority of patients in the rivaroxaban and LMWH cohorts in our study were still receiving treatment after 3 months may explain the loss of statistical significance for the reduction in recurrent VTE with rivaroxaban at 6 months but not 3 months (5.9% vs 6.7%; HR 0.87; 95% CI 0.69-1.08). These data also raise the question of whether longer durations of anticoagulation following acute CAT might be optimal, particularly in those with a lower risk of bleeding.
The Cancer-Associated Thrombosis, A Pilot Treatment Study Using Rivaroxaban (CASTA-DIVA) trial randomized 158 patients to receive either rivaroxaban or dalteparin for a 3-month duration of treatment. 12 The results found no significant difference between treatment groups for the risk of recurrent VTE (HR 0.75; 95% CI 0.21-2.65), but the HR point estimate was very similar to our study, suggesting that their small sample size (n = 158) resulted in a type II error (underpowered analysis). As in our current study, the risk of major bleeding in CASTA-DIVA at 3 months was not significantly different between rivaroxaban and LMWH use.
In a meta-analysis of the SELECT-D, 14 Hokusai-VTE Cancer, 13 ADAM-VTE, 10 Caravaggio, 11 and CASTA-DIVA trials, 12 Planquette and colleagues found a significant reduction in the risk of VTE recurrence (HR 0.63; 95% CI 0.47-0.86) in DOAC users (rivaroxaban, edoxaban, and apixaban) with no difference in major bleeding risk (HR 1.26; 95% CI 0.84-1.90) compared with LMWH users. These meta-analysis results are similar to our current study in which the risk of recurrent VTE is significantly reduced while the risk of major bleeding is similar. In our study, almost all patients in the LMWH group received enoxaparin, whereas in the SELECT-D, Hokusai-VTE Cancer, CARAVAGGIO, ADAM-VTE, and CASTA-DIVA trials, dalteparin was the comparator LMWH.10–14 Enoxaparin has shorter average molecular length heparin strands than dalteparin, which equates to greater activated factor X selectivity. 22 The extent to which differences in activated factor X selectivity between enoxaparin and dalteparin could impact our results is not known, but a comparison between our findings and the meta-analysis of DOACs versus dalteparin randomized controlled trials suggests it is likely minimal.
Current guidelines recommend a DOAC as a reasonable alternative to LMWH for the treatment of CAT in patients with cancer types not associated with a high risk of bleeding when taking a DOAC.6,7 GI malignancies have been identified as a cancer type in which DOACs appear to result in an increased risk of bleeding.13,14,23 This was first shown in a subgroup analysis of the Hokusai-VTE Cancer trial, where patients with GI cancers were shown to have a significantly higher risk of major bleeding (13.2% vs 2.4%; p = .0169) when taking edoxaban compared with dalteparin. 13 Moreover, a meta-regression analysis performed as part of a meta-analysis by Sabatino et al 23 concluded that DOACs were associated with an increased risk of clinically relevant nonmajor bleeding compared with dalteparin (risk ratio 1.47; 95% CI 1.16-1.85), driven significantly by GI cancers (p = .03) including malignancies in both the upper (p = .03) and lower (p = .05) GI tracts.
We utilized a retrospective cohort study design, and data were obtained from routinely collected EHR databases; therefore, various biases could have impacted the results. 24 Indication bias can occur when the risk of an outcome is related to the indication for medication use but not the use of the medication itself. 24 Indication bias may at least in part explain results from our study, such as the significant reduction in all-cause mortality with rivaroxaban observed in patients with genitourinary cancers at 3 months (HR 0.31; 95% CI 0.15-0.65). Differences in mortality based upon anticoagulant received were not anticipated based on the results of prior randomized controlled trials.12,16 Misclassification bias is a common concern in retrospective cohort analyses. 18 In this study, we attempted to attenuate the risk of misclassification by using validated coding algorithms (whenever possible) to identify active cancer diagnoses, covariates, and outcomes.16,17,25,26 Furthermore, identification of recurrent VTE was limited to the presence of ≥1 of a set of VTE-associated billing codes restricted to the primary coding position of a hospital admission (previously shown to have a positive predictive value of ∼95%). 17 To identify bleeding-related hospitalizations, we used the coding algorithm published by Cunningham et al 18 Both these VTE and bleeding-related hospitalization outcome detection algorithms have been validated and used in prior real-world studies evaluating the comparative effectiveness of anticoagulants to prevent CAT.27,28,29 In nonrandomized studies, unmeasured or residual confounding bias remains a concern and cannot be completely ruled out.20,24 To address this risk, we used PS-based OLW to balance many important baseline covariates between rivaroxaban- and LMWH-treated patients. 20 This method has the advantage of retaining all patients in the analysis dataset (unlike PS matching) and gives less weight to patients with PSs close to 0.0 or 1.0 (a concern with inverse probability weighting). 20 Our use of an intent-to-treat methodology may have resulted in bias toward a null effect, especially with longer follow-up durations. To further investigate the effect of patients discontinuing anticoagulation therapy, we provided an on-treatment analysis and rivaroxaban showed a similar net benefit over LMWH, and this benefit was maintained through 6 months. Future analyses with larger population sizes with prescription claims data should be performed to further assess the safety and effectiveness of rivaroxaban compared to LMWH. The choice of utilizing day 7 as the index date was selected to prevent early therapy switching from biasing our findings. However, the index date at day 7 could have created an immortal time bias and a potential underestimation of the reporting of recurrent VTE or major bleeding events. This limitation cannot be resolved in a retrospective claims-based study because identifying events such as recurrent VTE or major bleeding occurring during the index date time period is not possible given the common claim-based event methodology used. Other limitations to our study include our inability to reliably identify the original date of cancer diagnosis, which resulted in our active cancer inclusion criteria varying slightly from randomized trials. Selecting only participants with actively treated cancer or metastatic disease may have resulted in a sicker CaVTE population. Furthermore, we used the broad definition of GI cancer in our analysis. Further breaking down of this population would result in subgroups with population sizes too small to provide an assessment of the outcomes. Finally, this study evaluated US patients receiving rivaroxaban and (predominantly) enoxaparin. Consequently, our results and conclusions are most generalizable to this patient population. Focusing on rivaroxaban versus LMWHs instead of all DOACs reduced potential heterogeneity associated with broader DOAC inclusion. This would be important if the bleeding risk does, in fact, differ between DOACs.
Conclusion
Our study examined a larger, unselected cohort of patients with acute cancer, reflecting real-world administration of rivaroxaban and LMWHs for acute VTE events. Our data support the use of rivaroxaban as an alternative to LMWH for CAT treatment, with rivaroxaban associated with significant reductions in the composite of recurrent VTE or bleeding-related hospitalization versus LMWH. These data are generally consistent with those from the SELECT-D trial and lend support to current CAT treatment guidelines.
Disclosures
KS Caroti reports no conflicts of interest. AA Khorana reports consulting fees, honoraria, and travel support from Bayer AG; consulting fees, honoraria, and travel support from Janssen; consulting fees and honoraria from Bristol Myers Squibb; consulting fees and honoraria from Anthos; consulting fees and honoraria from Pfizer; consulting fees and honoraria from Sanofi; and honoraria from WebMD. C Becattini reports consulting fees and honoraria from Bayer AG; consulting fees and honoraria from Bristol Myers Squibb; consulting fees and honoraria from Daiichi Sankyo; and consulting fees from Pfizer. AYY Lee reports consulting fees and honoraria from Bayer AG; consulting fees and honoraria from LEO Pharma; consulting fees and honoraria from Pfizer; consulting fees from Servier; and honoraria from Bristol Myers Squibb. A Ekbom reports no conflicts of interest. M Carrier reports grants and consulting fees from Pfizer; grants and consulting fees from LEO Pharma; grants and consulting fees from Bristol Myers Squibb; consulting fees from Bayer AG; consulting fees from Sanofi; and consulting fees from Servier. AT Cohen reports grants, consulting fees, and honoraria from Alexion/AstraZeneca; grants, consulting fees, and honoraria from Bristol Myers Squibb/Pfizer; and consulting fees and honoraria from Bayer AG. C Brescia reports no conflicts of interest. K Abdelgawwad and G Psaroudakis are employees of Bayer AG. M Rivera was an employee of Bayer AG at the time of conduct, and her current affiliation is with Janssen Research and Development, Barcelona, Spain. B Schaefer is an employee of Bayer AG. G Brobert is a consultant for Bayer AG. CI Coleman reports grants, consulting fees, and travel support from Bayer AG; grants and consulting fees from Janssen Pharmaceuticals; grants and consulting fees from Alexion Pharmaceutical; and honoraria from Medscape.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231189282 - Supplemental material for Rivaroxaban Versus Low-Molecular-Weight Heparins in a Broad Cohort of Patients With Cancer-Associated Venous Thromboembolism: An Analysis of the OSCAR-US Program
Supplemental material, sj-docx-1-cat-10.1177_10760296231189282 for Rivaroxaban Versus Low-Molecular-Weight Heparins in a Broad Cohort of Patients With Cancer-Associated Venous Thromboembolism: An Analysis of the OSCAR-US Program by Kimberly Snow Caroti, Alok A. Khorana, Cecilia Becattini, Agnes Y.Y. Lee, Anders Ekbom, Marc Carrier, Alexander T. Cohen, Christopher Brescia, Khaled Abdelgawwad, George Psaroudakis, Marcela Rivera, Bernhard Schaefer, Gunnar Brobert and Craig I. Coleman in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors would like to acknowledge Tom Rowles from Chameleon Communications International, who provided editorial services with funding from Bayer AG.
Authors’ Note
Marcela Rivera is currently affiliated to Janssen Research and Development, Barcelona, Spain.
Author Contributions
KS Caroti contributed to the design and conduct of the study; analysis, management, and interpretation of the data; preparation, review, and approval of the manuscript; and decision to submit the manuscript for publication. AA Khorana, C Becattini, AYY Lee, A Ekbom, M Carrier, AT Cohen, K Abdelgawwad, G Psaroudakis, M Rivera, B Schaefer, and G Brobert contributed to the design and conduct of the study; management and interpretation of the data; preparation, review, and approval of the manuscript; and the decision to submit the manuscript for publication. C Brescia contributed to the management and interpretation of the data; and preparation, review, and approval of the manuscript. CI Coleman prepared the initial draft of the manuscript and contributed to the design and conduct of the study; management and interpretation of the data; review and approval of the manuscript; and decision to support the manuscript for publication.
All authors have read the manuscript and approve of its submission to Research and Practice in Thrombosis and Haemostasis.
Guidelines and results from recent trials support the use of DOACs for the treatment of CAT The real-world effectiveness and safety of rivaroxaban were evaluated in a broad cancer cohort Rivaroxaban showed reduced recurrent VTE events and a similar safety profile versus LMWH These data complement clinical trial results supporting the use of rivaroxaban for the treatment of CAT
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bayer (grant number NA).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
