Abstract
Anticoagulation therapy is the standard treatment of patients with symptomatic venous thromboembolism (VTE), including deep vein thrombosis and pulmonary embolism. Until recently, treatment of VTE was based on parenteral or low-molecular-weight heparin for initial therapy (5-10 days) and oral vitamin K antagonists for long-term therapy. Those treatments have some limitations, including parenteral administration (heparins), the need for frequent monitoring and dose adjustments, interactions with several medications, and dietary restrictions (vitamin K antagonists). Rivaroxaban is a new oral direct factor Xa inhibitor with a wide therapeutic window, predictable anticoagulant effect, no food interactions, and few drug interactions. Consequently, no periodic monitoring of anticoagulation is needed, and fixed doses can be prescribed. EINSTEIN program demonstrated that rivaroxaban was as effective as and significantly safer than standard therapy for treatment of VTE. Rivaroxaban was recently authorized so doubts exist about how to use it in daily clinical practice. This document aims to clarify common questions formulated by clinicians regarding the use of this new drug.
Keywords
Introduction
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE), is a major health problem. It has been estimated that 370 000 people die from VTE-related conditions in the European Union every year. Remarkably, about 75% of these deaths are due to hospital-acquired VTE. 1
Anticoagulation therapy is recommended for both primary prevention and treatment of VTE. 2 –6 Until recently, it was restricted to parenteral anticoagulants for short-term therapy and oral vitamin K antagonists (VKAs) for long-term therapy. 4 The limitations of low-molecular-weight heparin (LMWH) include the route of administration, the lack of a specific antidote, and the need for dose adjustment according to renal function. Unfractionated heparin has a specific antidote (protamine) and does not need to be adjusted for renal function. However, the intravenous route as well as the need for period`ic monitoring are relevant disadvantages. Vitamin K antagonists also have a number of limitations, including a narrow therapeutic window, multiple food and drug interactions, and variability in metabolism, all of which translate into the need for frequent monitoring of coagulation, dose adjustments, surveillance of interactions with other medications, and dietary restrictions. 7 –9
Rivaroxaban is an oral, highly selective direct inhibitor of factor Xa. It has a wide therapeutic window and a predictable anticoagulant effect. Moreover, it does not interact with food and rarely interacts with other drugs. Consequently, no routine periodic monitoring of coagulation is needed, and fixed doses can be prescribed. 3,5,10,11 Several clinical trials have analyzed the efficacy and safety of rivaroxaban in the prevention and treatment of thromboembolic complications in different clinical settings. 12 –21 In fact, of the newly approved oral anticoagulants, rivaroxaban has the most clinical indications (Table 1). 22,23 However, there is no specific antidote for rivaroxaban at this moment, and dose adjustments according to renal function are needed. 22,23
Current Indications of Rivaroxaban in Clinical Practice.a
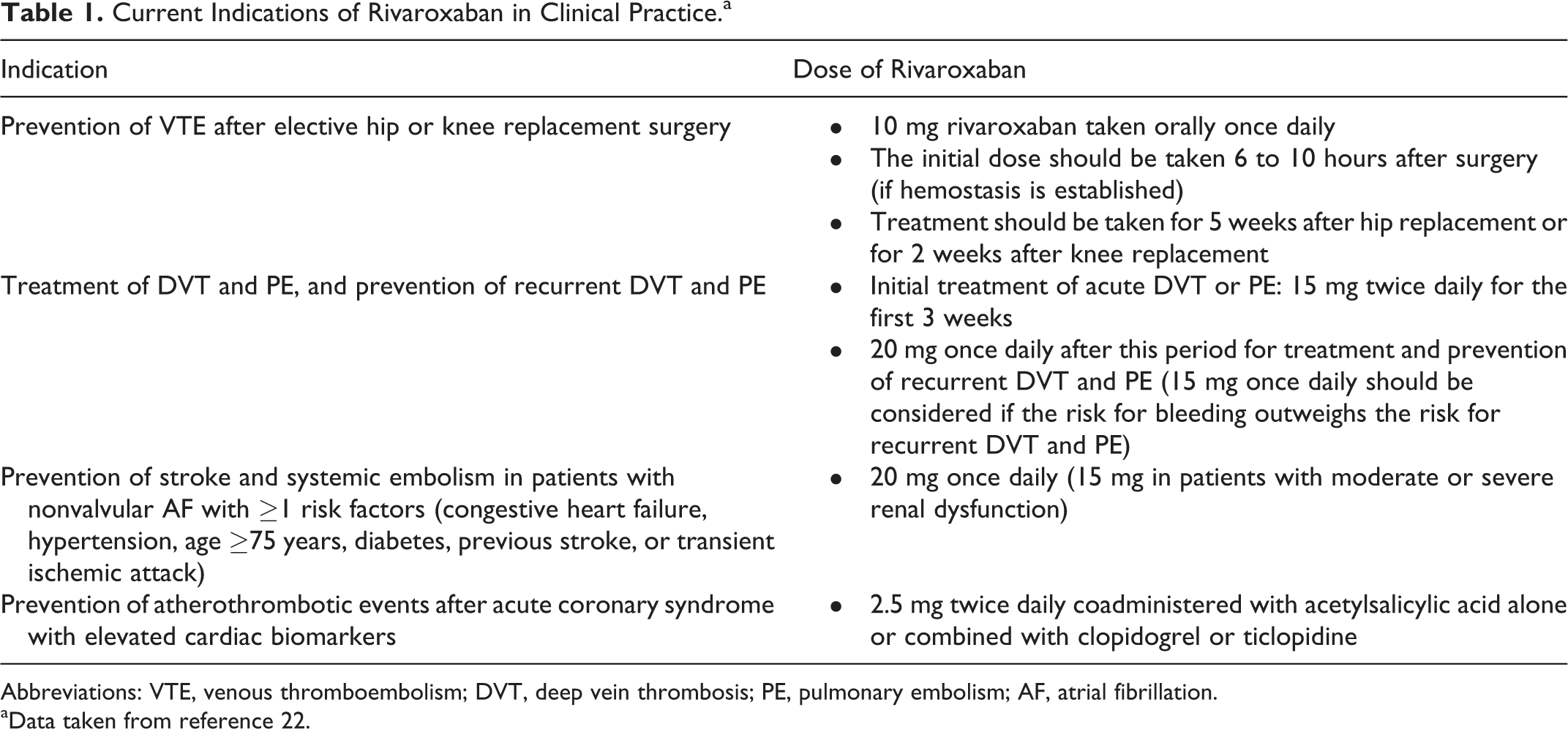
Abbreviations: VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism; AF, atrial fibrillation.
aData taken from reference 22.
The EINSTEIN program included 3 clinical trials that analyzed the efficacy and safety of rivaroxaban: EINSTEIN-DVT, which analyzed treatment of acute DVT, EINSTEIN-PE, which analyzed the treatment of acute PE, and EINSTEIN-EXTENSION, which was an extension phase study after treatment for VTE. In summary, the EINSTEIN program showed that rivaroxaban was noninferior to standard therapy (enoxaparin followed by VKA) in the prevention of recurrent VTE in patients with acute symptomatic DVT or PE. Remarkably, although rates of the combined end point of major bleeding or clinically relevant nonmajor bleeding were similar, the risk of major bleeding events was less frequent with rivaroxaban in patients with PE (Table 2). 16,17,24
Efficacy and Safety of Rivaroxaban in the EINSTEIN Program.a

Abbreviations: VTE, venous thromboembolism; DVT, deep vein thrombosis; PE, pulmonary embolism; VKA, vitamin K antagonists; CI, confidence interval; HR, hazards ratio.
aData taken from references 16 and 17.
An additional advantage of rivaroxaban over standard therapy is the option to simplify treatment and thus reduce complications and improve adherence. 11 Rivaroxaban can be administered as monotherapy in all phases of VTE, including the acute phase as well as for secondary prevention in patients with DVT and in patients with PE with or without DVT (Table 1). 16,17,22 –25 Dabigatran has recently been approved by Food and Drug Administration for treating DVT and PE in patients who have been treated with a parenteral anticoagulant for 5 to 10 days. As a result, although dabigatran has been developed to replace warfarin, rivaroxaban may replace both, LMWH and warfarin. Finally, apixaban also has EMEA approval for VTE.
The aim of this document was to provide a practical approach for the management of patients having VTE with rivaroxaban by posing and answering a series of questions according to the available evidence and the opinion of different experts in the management of patients with VTE.
Rivaroxaban and Treatment of VTE
1. How should rivaroxaban be taken?
To ensure an optimal efficacy and safety profile for rivaroxaban in the treatment of VTE, it is essential to follow the dosage and timing used in the EINSTEIN DVT and EINSTEIN PE trials, namely, 15 mg twice daily for the first 3 weeks, followed by 20 mg once daily for 3, 6, or 12 months. As the bioavailability of rivaroxaban 20 mg is 66% without food, but the area under the curve increases by 39% when the drug is taken with food, rivaroxaban 15 mg and 20 mg should be taken simultaneously with food. 22,26,27
In patients with moderate or severe renal insufficiency (creatinine clearance [CrCl] 15-49 mL/min), the above-recommended doses could be maintained. Of note, patients with CrCl below 30 mL/min were excluded in the EINSTEIN trials and the experience in this group of patients is limited. A reduction to 15 mg once daily is recommended in those individuals in whom the risk of bleeding outweighs the risk of recurrent DVT and PE. 16,17,22
In the EINSTEIN DVT and EINSTEIN PE trials, patients could not be included if they had received therapeutic doses of LMWH, fondaparinux, or unfractionated heparin for more than 48 hours or if they had received ≥1 dose of a VKA before randomization. 16,17 As a result, rivaroxaban can be used as previously indicated in patients who have taken unfractionated heparin, LMWH, or fondaparinux during the first 48 hours. In fact, 73% of patients included in EINSTEIN DVT received treatment with these drugs, in most cases for 1 day. In this group, rivaroxaban should be administered at discontinuation of unfractionated heparin, 0 to 2 hours before the next scheduled dose of LMWH or 12 to 24 hours after the last dose of fondaparinux. 16,17,22
The duration of treatment for secondary prevention should be tailored, considering the risk of recurrent VTE and the risk of bleeding. Thus, a short treatment period (3 months) should be reserved for those patients with transient risk factors (ie, recent surgery, traumatic injuries, or immobilization). Longer periods of treatment should be based on the presence of permanent risk factors or for cases of idiopathic VTE. Experience with rivaroxaban for periods longer than 12 months is limited.
3,11
2. When is rivaroxaban indicated/contraindicated in the treatment of patients with VTE?
Rivaroxaban has been approved for the treatment of acute DVT and PE. This indication is based on the favorable data obtained in EINSTEIN DVT and EINSTEIN PE. The former included patients with acute, symptomatic, and objectively confirmed proximal DVT and no symptomatic PE; the latter included patients with acute, symptomatic, and objectively confirmed PE, with or without symptomatic DVT. 16,17 In both trials, the results for efficacy and safety were consistent across different subgroups of patients in terms of age, weight, sex, renal function, and history of cancer. This finding is particularly important in patients who are more fragile, including those aged >75 years, weighing <50 kg, and/or with CrCl <50 mL/min. 16,17 A recent pooled analysis of both trials showed that in those patients with a previous venous thromboembolic event, rivaroxaban reduced the risk of recurrent VTE by 55% compared to standard therapy. 28
In EINSTEIN DVT and EINSTEIN PE, patients with CrCl <30 mL/min, clinically significant liver disease (including transaminase levels >3 times upper limit of normal [ULN]), bacterial endocarditis, active bleeding or high risk of bleeding, contraindication for anticoagulation therapy, blood pressure levels >180/110 mm Hg, or concomitant use of strong cytochrome P-450 3A4 inhibitors or inducers were excluded as were pregnant and breast-feeding women. 16,17 Consequently, data on the efficacy and safety of rivaroxaban in these patients are lacking.
According to the summary of product characteristics, rivaroxaban is contraindicated in patients with hypersensitivity to this drug or to any of its excipients, clinically significant active bleeding, lesions or conditions that increase the risk of major bleeding, concomitant treatment with any other anticoagulant drug (except when switching therapy to or from rivaroxaban), liver disease associated with coagulopathy, and clinically relevant bleeding risk (ie, Child-Pugh B and C cirrhosis). It is also contraindicated in pregnant and breast-feeding women.
22
3. How should rivaroxaban be used in patients with renal insufficiency?
About two-thirds of the dose of rivaroxaban is metabolized by the liver before being eliminated by the kidneys and in feces in equal proportions. The remaining third is eliminated directly by the kidneys through active renal secretion. 22 For this reason, patients with severe renal insufficiency (CrCl < 30 mL/min) were excluded from phase III clinical trials. Moreover, it has been demonstrated that as renal function decreases, exposure to rivaroxaban increases. In fact, the area under the curve of rivaroxaban increased by 1.4 in patients with a CrCl 50 of 80 mL/min, by 1.5 in those with CrCl 30 of 49 mL/min, and by 1.6 in those with CrCl 15 of 29 mL/min. Unfortunately, no data are available on the use of rivaroxaban 15 mg twice daily in patients with CrCl <30 mL/min or the use of rivaroxaban in patients with CrCl <15 mL/min. 22,29
Therefore, before initiating treatment with rivaroxaban, it is mandatory to determine renal function, the risk of bleeding, and the possible benefit of rivaroxaban over standard therapy. 3,11,22,30
No dose adjustment is required in patients with CrCl ≥50 mL/min during the initial period of treatment. Patients with CrCl 30 to 49 mL/min should be also treated with 15 mg twice daily for the first 3 weeks. Although the summary of product characteristics also recommends this dosage regimen for patients with CrCl 15 to 29 mL/min, 22 it should be noted that clinical experience with rivaroxaban in this population is very limited. Consequently, the risk of bleeding should be carefully weighed against the benefits over standard therapy. Finally, rivaroxaban should not be used in patients with CrCl <15 mL/min. 3,22
After this period, the recommended dose is 20 mg once daily. However, this dose may be reduced to 15 mg once daily when the perceived risk of bleeding exceeds the risk of recurrent DVT and PE.
3,22
During treatment with rivaroxaban, renal function and the risk of bleeding should be periodically reassessed, particularly in patients at higher risk, such as those with chronic kidney disease or elderly patients, as well as in specific situations (ie, infections and hypovolemia).
22
4. How should rivaroxaban be used in patients with liver insufficiency?
About two-thirds of the dose of rivaroxaban is metabolized in the liver. Moreover, patients with liver insufficiency are at increased risk of bleeding. Patients with clinically significant liver disease (including acute hepatitis, chronic active hepatitis, and cirrhosis) and patients with alanine aminotransferase levels >3 times ULN were excluded from EINSTEIN DVT and EINSTEIN PE. 16,17
Data from the pooled analysis of the RECORD trials and pharmacokinetic/pharmacodynamic studies have shown that in patients with cirrhosis and Child-Pugh stage B, the area under the curve of rivaroxaban is increased by 2.3 and the inhibition of factor Xa activity by 2.6. By contrast, no significant changes were observed in patients with Child-Pugh stage A cirrhosis. 12 –15,22,31 No clinical trials have included patients with Child-Pugh C cirrhosis. 22
Consequently, rivaroxaban is contraindicated in patients with liver disease associated with coagulopathy and clinically relevant bleeding risk, including patients having cirrhosis with Child-Pugh stages B and C. By contrast, it can be given without dose adjustment in patients with Child-Pugh stage A cirrhosis or other liver diseases without coagulopathy or a clinically relevant risk of bleeding.
22
5. How to switch between rivaroxaban and other anticoagulants?
As enoxaparin does not interfere with the pharmacokinetic properties of rivaroxaban, and their half-lives are very similar, both drugs can be used sequentially or as bridging therapy. 10,22,32 For patients taking LMWH, rivaroxaban should be started 0 to 2 hours before the next scheduled dose of the LMWH that is discontinued. However, for patients receiving intravenous unfractionated heparin, rivaroxaban should be started at discontinuation of parenteral heparin (Table 3). 3,11,22
Switching From Other Anticoagulants to Rivaroxaban and Vice Versa.a

Abbreviations: LMWH, low-molecular-weight heparin; VTE, venous thromboembolism; VKA, vitamin K antagonists; NVAF, nonvalvular atrial fibrillation; INR, international normalized ratio.
aData taken from reference 22.
By contrast, in patients taking rivaroxaban requiring a switch to heparin (subcutaneous or intravenous), heparin should be started at the time of the next scheduled dose of rivaroxaban that is discontinued (Table 3). 3,11,22
Vitamin K antagonists (ie, warfarin and acenocoumarol) have a slow onset and duration of action. Thus, switching from or to VKAs is not as rapid as with parenteral anticoagulants and should be based on international normalized ratio (INR) values. If the patient is taking VKAs for VTE, VKAs should be discontinued, and rivaroxaban will be started when INR is ≤2.5 (≤3.0 in case of atrial fibrillation; Table 3). 3,11,22 International normalized ratio is not a valid tool for measuring the anticoagulant activity of rivaroxaban, and therefore should not be used.
On the other hand, when rivaroxaban is switched to VKAs, both drugs should be taken simultaneously until the INR reaches 2.0, and then only VKAs should be used and rivaroxaban discontinued. As rivaroxaban can increase INR levels, INR should be determined before the next scheduled dose of rivaroxaban. When rivaroxaban is discontinued, the next determination of INR should be performed at least 24 hours after the last dose (Table 3).
3,11,22
6. How does rivaroxaban interact with food and other drugs?
Unlike VKAs, rivaroxaban rarely has interactions with other drugs. Although no food restrictions are required with rivaroxaban, food increases absorption and area under the curve. 3,10,11,22
Although infrequent, interactions between rivaroxaban and other drugs can be relevant. Rivaroxaban is metabolized via CYP3A4, CYP2J2, and CYP-independent mechanisms and is a substrate of the transporter protein P-glycoprotein (P-gp). Thus, coadministration of rivaroxaban with potent inhibitors of both CYP3A4 and P-gp could increase plasma rivaroxaban concentrations and, secondarily, the risk of bleeding. However, coadministration of rivaroxaban with drugs that moderately inhibit CYP3A4 or P-gp promotes a lesser increase in rivaroxaban plasma concentrations. Caution should be exercised, when these drugs are used in combination, particularly in patients with a high risk of bleeding (ie, elderly patients or those with renal dysfunction; Table 4). 3,22
Interactions Between Rivaroxaban and Other Drugs.a
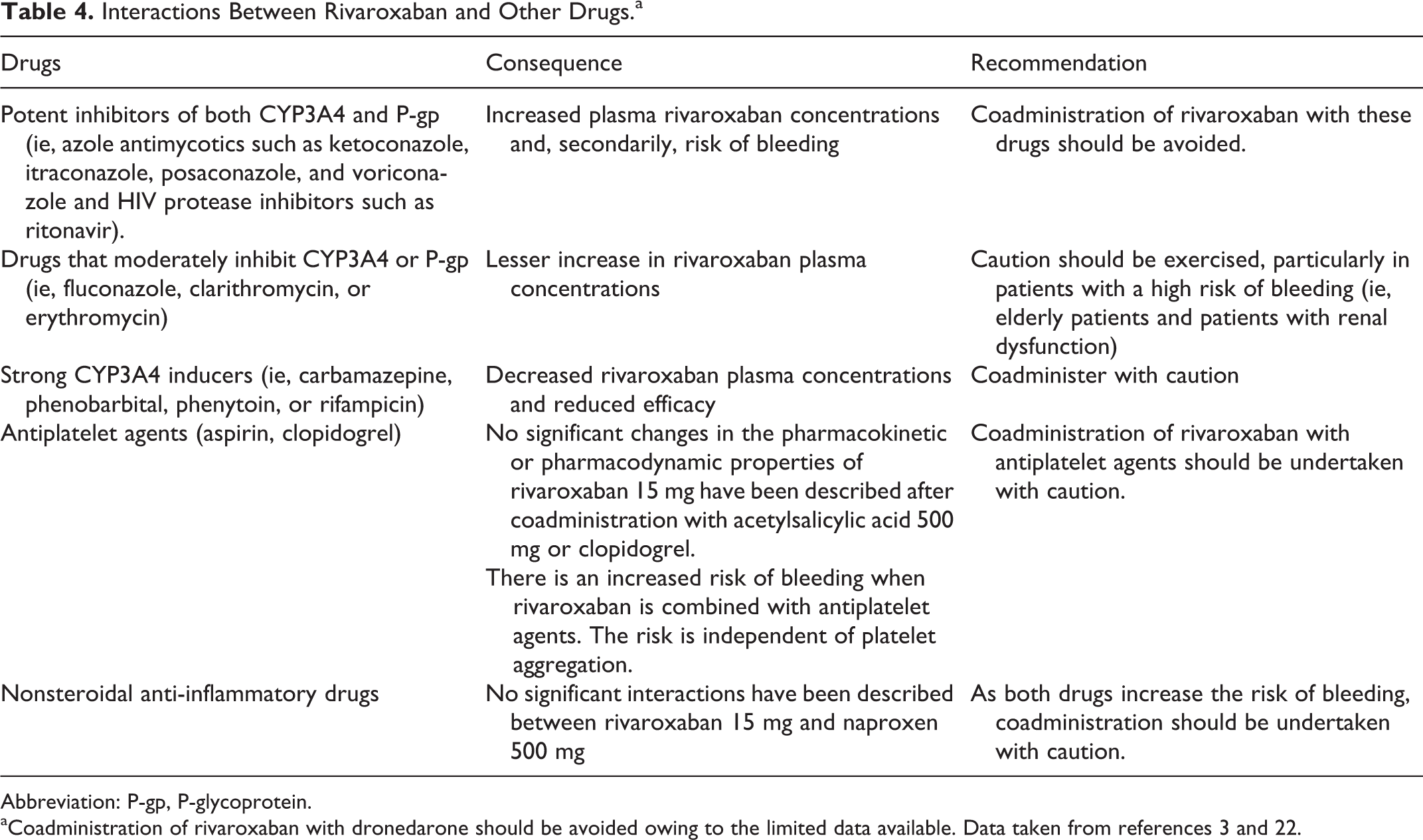
Abbreviation: P-gp, P-glycoprotein.
aCoadministration of rivaroxaban with dronedarone should be avoided owing to the limited data available. Data taken from references 3 and 22.
By contrast, strong CYP3A4 inducers can decrease rivaroxaban plasma concentrations, thus reducing its efficacy. Therefore, these drugs should be coadministered with caution, particularly in patients with high risk of recurrent disease (Table 4). 3,22
Finally, given the limited data available, coadministration of rivaroxaban with dronedarone should be avoided (Table 4). 22 In clinical practice, it is not unusual for a patient taking an anticoagulant to need concomitant antiplatelet therapy and vice versa. 3,33 Although no direct effects of rivaroxaban on platelets have been described, and no significant changes in the pharmacokinetic or pharmacodynamic properties of rivaroxaban 15 mg have been described for coadministration with acetylsalicylic acid 500 mg or clopidogrel (300 mg loading dose followed by 75 mg maintenance dose), 22,34 the fact is that the concomitant use with a nonsteroidal anti-inflammatory drugs or aspirin is associated with an increased risk of clinically relevant and major bleeding. 35
Therefore, concomitant use of rivaroxaban with antiplatelet agents or nonsteroidal anti-inflammatory drugs should be addressed with caution, particularly in patients with a known increased risk of bleeding. Furthermore, as the efficacy and safety of rivaroxaban have only been investigated in combination with the antiplatelet agents aspirin and clopidogrel/ticlopidine, concomitant use with prasugrel or ticagrelor is not recommended.
22
7. What is the approach when a patient receiving rivaroxaban has to undergo elective surgery?
Few data are available on the optimal approach for patients undergoing surgery while taking rivaroxaban. The recommendations given are based on the opinion of experts and on the pharmacokinetic properties of rivaroxaban. 3,22,36,37
When a patient treated with rivaroxaban for VTE (15 mg twice daily or 20 mg once daily) requires a surgical intervention, in order to establish how long should the drug be discontinued, it is necessary to determine the risk of bleeding related to the surgery (Table 5). If the risk of bleeding is low, surgery should be delayed until at least 24 hours after the last dose of rivaroxaban. However, some authors recommend that in patients with renal insufficiency (CrCl < 50 mL/min), surgery should be delayed at least 48 hours. 3,36,37
Risk of Bleeding According to Type of Surgery.a

Abbreviation: ENT: ear, nose and throat.
aData taken from references 3, 36, and 37.
If the risk of bleeding is moderate or high, 2 options have been suggested.
3,22,36,37
: Option 1 (proposed by North American and British guidelines): The moment of rivaroxaban discontinuation before surgery should be adjusted in accordance to the renal function; surgery should be delayed between 24 and 48 hours when CrCl is ≥50 mL/min and at least 48 hours when CrCl is <50 mL/min, even 3 to 4 days in those with low or moderate thrombotic risk. Option 2 (proposed by Spanish and French anesthetists): Rivaroxaban should be stopped 5 days before surgery and LMWH used as bridging therapy (starting LMWH 12 to 24 hours after the last dose of rivaroxaban, and finishing 24 hours before surgery). According to Llau et al,
36
this is the preferred option in patients with high risk of bleeding or thrombosis or CrCl <50 mL/min. Despite current limitations in the monitoring of coagulation in patients receiving new oral anticoagulants, coagulation tests (prothrombin time [PT], preferably with highly sensitive thromboplastin, activated partial thromboplastin time [aPTT], and anti-factor Xa chromogenic assay) may be considered before surgery in some cases (ie, high risk of bleeding). Although prolongation of PT can be associated with high plasma concentrations of rivaroxaban, it is not sensitive at lower plasma concentrations. Nevertheless, the normality of these parameters seems to imply that hemostasis is acceptable.
3,36,37
Restarting anticoagulation therapy after surgery in a patient previously treated with rivaroxaban depends on the possibility of oral intake and the risk of bleeding. If oral intake is possible, rivaroxaban can be restarted 12 to 24 hours after surgery when the risk of bleeding is low and 48 to 72 hours when the risk of bleeding is high. By contrast, if oral intake is not possible, heparin should be used, preferably LMWH, starting when hemostasis is established, usually 24 hours after surgery. When oral intake is possible again, rivaroxaban can be taken 12 hours after the last dose of LMWH.
In case of minor procedures, the general recommendations should be the same as those proposed for patients undergoing elective surgery with low risk of bleeding, namely, delaying surgery until 24 hours after the last dose of rivaroxaban. In the case of tooth extraction, most guidelines recommend maintaining treatment with rivaroxaban, although the extraction should be performed at least 8 hours after the last dose of rivaroxaban.
36,37
8. What is the approach when a patient treated with rivaroxaban has to undergo emergency surgery?
In cases of emergency surgery, it is important to know when the last dose of rivaroxaban was taken. When possible, it would be preferable to delay surgery at least 2 times the half-life of rivaroxaban or until the absence of significant antifactor Xa activity is confirmed. If this is not possible, the decision to operate or not should be made on an individual basis according to the increase in the risk of bleeding and the need for surgery. In this setting, in which complex laboratory tests are not always available, normal PT and aPTT values could indicate adequate hemostasis. 36,37
There is no specific antidote for rivaroxaban to reverse anticoagulation. When active bleeding is diagnosed and surgery cannot be delayed, providing hemostatic agents such as prothrombin complex concentrate (PCC) or alternatively, recombinant factor VIIa (rFVIIa) could be used. Experimental studies performed in animals and healthy human volunteers treated with rivaroxaban showed that these agents improve some coagulation tests. However, in an experimental rabbit model none of them reversed rivaroxaban-induced bleeding, despite partially improving laboratory parameters.
38
Therefore, these agents should be administered on individual basis and an expert in hemostasis should be consulted. Prophylactic administration of PCC or rFVIIa is not recommended before surgery in the absence of bleeding.
36
–39
9. How should bleeding be managed in patients taking rivaroxaban?
When a clinically significant bleeding event occurs, rivaroxaban should be discontinued in order to recover the hemostasis and to reevaluate the indication of anticoagulation therapy. It is mandatory to establish the severity and location of the bleeding and to ensure the hemodynamic stability. In patients with normal renal function, it is expected that hemostasis can be restored 16 to 24 hours after the last dose of rivaroxaban. If the bleeding episode is mild in intensity, in many cases it is sufficient to apply local compression and delay or stop the next dose of rivaroxaban. 3,11,22,40
When it is necessary to urgently reverse anticoagulation therapy (ie, because of the risk of fatal bleeding or intracranial, pericardial, or intraspinal hemorrhage), the options include PCC, activated PCC (aPCC), and rFVIIa, given that no specific treatment is currently available. 3,11,22,38 –42 Recent studies have shown that these agents can normalize coagulation test results in animals and healthy volunteers. 38,39 In some cases, more than 1 dose of factor concentrates may be necessary to restore coagulation. However, normalization of coagulation test results does not necessarily translate into cessation of bleeding, and data on the effects of these concentrates in patients with active bleeding are lacking. Fresh frozen plasma contains factor X and may reduce the anticoagulant effect of rivaroxaban, although the duration of the volume infusion may delay the procedure. If the patient is taking antiplatelet therapy, transfusion of platelets can accelerate the normalization of hemostasis. Studies performed in animals have shown that the infusion of recombinant activated factor X completely restores the effect of rivaroxaban, but its efficacy in humans is unknown. 43
If the patient receives high doses of rivaroxaban (whether by mistake or intentionally), gastric lavage and administration of activated charcoal may be useful in the first 2 hours after intake. Owing to high plasma protein binding (>90%), rivaroxaban is not expected to be dialyzable. Small increases in PT or aPTT may indicate exposure to high doses of rivaroxaban, although these markers are not useful for evaluating the intensity of anticoagulation. 44
It is also important to determine whether the patient with bleeding requires hospitalization, surgery, or endoscopic procedures to stop the bleeding; consequently, knowledge of the exact time of intake is essential. The pharmacokinetics of rivaroxaban are predictable. In patients with normal renal function, Cmax is achieved 2 to 3 hours after intake, and half-life is 7 to 11 hours. Therefore, the intensity of anticoagulation can be determined at the moment of bleeding. It has been suggested that the time to safely perform an invasive procedure in anticoagulated patients is 2 terminal half-lives after Cmax is reached. 45 If possible, it is advisable to wait this length of time. Hemodynamic and renal perfusion must be verified in order to ensure renal elimination. 22
When bleeding has ceased, the risk of thrombotic complications and bleeding events and the need for concomitant medication (ie, antiplatelet agents) should be evaluated. If the bleeding is mild in intensity and controlled, the next dose of rivaroxaban can be delayed 6 to 8 hours after hemostasis is established or omitted, as appropriate. If the cause of bleeding has been resolved (ie, polypectomy), rivaroxaban can be given safely 24 to 72 hours after the procedure. In case of severe bleeding and high risk of thromboembolism, the decision should be individualized, but in many cases, anticoagulation therapy can be restarted 7 to 10 days after bleeding has resolved. Other options, such as inferior vena cava filter to prevent recurrent PE, should be considered.
3,11,22,38
–42,46
10. Should rivaroxaban be used in patients with an increased risk of bleeding?
According to the summary of product characteristics, rivaroxaban is contraindicated in patients at high risk of major bleeding (eg, current or recent gastrointestinal ulcer and arteriovenous malformations). 22 Moreover, since these patients have not been included in clinical trials, 16,17 no evidence-based strong recommendations can be established for these settings. Therefore, the decision to use rivaroxaban in these patients should be made on individual basis.
In some patients, these risk conditions can be resolved (eg, by surgery or endoscopic procedures) and anticoagulation therapy could be started. However, if this is not possible, the alternative in patients with recent PE is vena cava filter insertion either alone or associated with low-dose anticoagulants to prevent new VTE episodes. In patients without a vena cava filter but who are at high risk of bleeding, the administration of low doses of anticoagulants, such as rivaroxaban 10 mg/d or low-dose LMWH, may be considered after the first weeks of treatment, although this approach has not been tested in clinical trials. 11. How should recurrence of VTE be managed in patients treated with rivaroxaban?
This question has not been specifically analyzed in clinical trials. Therefore, until more evidence is available, we recommend an approach similar to the patients taking VKAs,
4,47,48
as follows: Assess adherence and whether patient is taking rivaroxaban correctly (with food) as well as the presence of concomitant medication that may reduce the efficacy of rivaroxaban. If the patient is taking rivaroxaban, the effects of the drug on laboratory parameters should be determined (see subsequently). However, even if insufficient activity is observed, there is no clinical experience with the use of higher doses of rivaroxaban. In cases of recurrent PE despite appropriate anticoagulation therapy, imaging the lower limbs for acute DVT and administering a therapeutic dose of LMWH/unfractionated heparin should be first considered and the vena cava filter reserved for those with residual acute DVT or poor cardiopulmonary reserve.
49
Switch anticoagulation therapy: the results of EINSTEIN DVT and EINSTEIN PE showed that this approach was not more effective in reducing thrombotic recurrences than rivaroxaban.
16,17
In summary: Recurrence in initial phase: start treatment with LMWH or unfractionated heparin. In cases of PE and limited cardiopulmonary function, consider insertion of a vena cava filter. Recurrence during long-term treatment: use VKAs.
12. How should patients be managed when oral intake is not possible?
When oral intake is not possible, rivaroxaban should be discontinued and replaced by a parenteral anticoagulant, preferably LMWH or fondaparinux. The first dose of LMWH should be administered at the time of the next rivaroxaban dose. Low-molecular-weight heparin should be continued until oral intake is restored.
22
Rivaroxaban should then be administered 12 hours after the last dose of LMWH or when intravenous unfractionated heparin is discontinued.
22
13. How should a missed dose of rivaroxaban be managed?
Rivaroxaban has a high bioavailability and is absorbed rapidly, reaching maximum concentrations 2 to 4 hours after oral intake. Thus, its onset of action is fast. Moreover, terminal half-life ranges from 5 to 9 hours in young patients (11-13 hours in the elderly patients). 22,49
If the patient is taking rivaroxaban 15 mg twice daily in the initial treatment phase and misses 1 dose, the drug should be taken as soon as possible to ensure the daily dose of 30 mg (2 tablets of 15 mg can be taken together) and then to continue as previously prescribed.
If the patient is in the once-daily treatment phase (day 22 and onwards) and misses 1 dose, then the missed dose should be taken as soon as possible and the following dose continued as prescribed.
Although some studies have shown that higher doses of rivaroxaban than those prescribed for the treatment of VTE are safe, taking a double dose of rivaroxaban to make up for the missed dose is not recommended.
22,25,50
14. How should an overdose of rivaroxaban (without bleeding) be managed?
Despite the ceiling effect in the absorption of rivaroxaban, overdose can increase the risk of bleeding. In a dose-escalation study, doses of up to 80 mg were shown to be safe. In phase II studies in patients with proximal DVT, doses of 60 mg daily taken in 2 doses of 30 mg and up to 40 mg once daily, a slight increase was observed in the risk of bleeding compared with enoxaparin followed by VKA or lower doses of rivaroxaban. In fact, it has been reported that doses higher than 50 mg once daily are not associated with significant further increases in average plasma exposure. Moreover, factor Xa activity is dose-dependently inhibited and maintained for approximately 12 hours after dosing. 51,52 Terminal half-life ranges from 5 to 13 hours (5 to 9 hours in young patients and 11 to 13 hours in patients >75 years). 22,25,49 –54
Thus, in cases of overdose, patients should be monitored for 12 to 24 hours, and rivaroxaban could be restarted after 24 hours (or later if required). The use of activated charcoal, preferably in the 2 first hours after intake, may reduce the absorption of rivaroxaban. As rivaroxaban is highly bound to plasma protein, it is not dialyzable. Moreover, no specific antidote for rivaroxaban exists.
22
In the case of bleeding, see question 9. 15. What is the role of laboratory tests in patients taking rivaroxaban?
Rivaroxaban has a wide therapeutic window and a predictable anticoagulant effect. Consequently, monitoring of anticoagulant activity is not necessary. 22 In fact, phase III clinical trials on the benefits and risks of rivaroxaban were carried out without monitoring anticoagulant activity (Table 6). 12 –21
Rivaroxaban and Laboratory Tests.a

Abbreviations: P-gp, P-glycoprotein; INR, international normalized ratio.
aData taken from references 56-59.
However, there are situations in which it would be interesting to assess the exposure to rivaroxaban. These include emergency surgery, severe bleeding, overdose, doubts about adherence, thrombotic events in treated patients, and high risk of bleeding (i.e. those with renal insufficiency, hepatic insufficiency, older age, low weight, and concomitant medications that interfere with rivaroxaban; Table 6). 3,10,11
The recommended tests for assessing exposure to rivaroxaban are specific anti-factor Xa chromogenic assay and PT. The anti-factor Xa chromogenic assay calibrated with rivaroxaban is specific and sensitive for measuring plasma rivaroxaban concentrations, although these concentrations should be correlated with the timing of drug administration. PT, measured in seconds, correlates with rivaroxaban concentrations when highly sensitive thromboplastin is used for the assay. With respect to other laboratory tests, conventional INR should not be used in patients taking rivaroxaban. Activated partial thromboplastin time is prolonged depending on the dose administered, but it is not recommended to assess the pharmacodynamic effect of rivaroxaban (Table 6).
3,22,44,55
–58
In the case of lupus anticoagulants, false-positive results have been observed depending on the assay system used.
59
16. Can rivaroxaban be taken during pregnancy?
Rivaroxaban belongs to pregnancy category C of the United States Food and Drug Administration. The safety and efficacy of rivaroxaban have not been analyzed in pregnant women. Nonetheless, studies performed in animals have shown that rivaroxaban crosses the placenta and that is associated with reproductive toxicity and with an increased risk of bleeding. 60,61 Consequently, rivaroxaban is contraindicated during pregnancy. Similarly, as it is secreted into breast milk, it is contraindicated during breast-feeding. 22
Women should be fully informed about the potential risks of rivaroxaban during pregnancy, the need to avoid it during the treatment period, and, when pregnancy is suspected, the importance of informing their physician. If a patient becomes pregnant or is trying to become pregnant, rivaroxaban should be discontinued and switched to LMWH 24 hours after the last dose of rivaroxaban. The patient should be followed up in specific obstetric units. 22,62
Conclusions
The EINSTEIN program showed that rivaroxaban is as efficacious as and safer than standard therapy in the treatment of VTE. However, since this is a novel agent, many areas of use in clinical practice remain unclear, as this document has tried to clarify.
More research is needed to provide evidence-based specific answers to many questions that emerge in clinical practice. Meanwhile, this document show how to address common problems related to rivaroxaban use based on the available evidence and expert opinion. The indications, precautions, and contraindications for rivaroxaban in the treatment of VTE are summarized in Table 7.
Indications, Precautions, and Contraindications of Rivaroxaban in the Treatment of Patients With Venous Thromboembolism.

Abbreviations: VTE, venous thromboembolism; VKA, vitamin K antagonist; CrCl, creatinine clearance; PE, pulmonary embolism; DVT, deep vein thrombosis.
Footnotes
Acknowledgments
The authors thank Content Ed Net for providing editorial assistance in the preparation of this manuscript; editorial assistance was funded by Bayer.
Declaration of Conflicting Interests
J.I.A. received honoraria as advisor from Bayer HealthCare, Daiichi Sankyo, Janssen, and Rovi; and lecture fees from Pfizer, Rovi, Aspen, and Sanofi. P.D. has been consultant during advisory board meetings of Bayer. C.F.C. received compensation for advisory-board membership from Boehringer-Ingelheim, Bayer Health Care, Daiichi Sankyo, Rovi, Sanofi-Aventis and Leo Pharma and lectures fees from Bayer Health Care, Boehringer-Ingelheim, Menarini, Rovi, and Sanofi-Aventis. R.G. received honoraria as advisor from Bayer HealthCare, Boehringer-Ingelheim and lecture fees from Bayer HealthCare, Pfizer, Sanofi-Aventis and Leo Pharma. D.J. received compensation for advisory-board membership from Boehringer-Ingelheim, Bayer HealthCare, Bristol-Myers Squibb, Daiichi Sankyo, Leo Pharma, Pfizer, and Rovi; and lecture fees from Boehringer-Ingelheim, Bayer HealthCare, Bristol-Myers Squibb, Daiichi Sankyo, Leo Pharma, Pfizer, Rovi, and Sanofi-Aventis. S.J. received compensation for advisory-board membership from Bayer HealthCare and Rovi; and lecture fees from Boehringer-Ingelheim, Bayer HealthCare, Rovi and Sanofi-Aventis. F.S.L. received compensation for advisory-board membership from Boehringer-Ingelheim, Bayer Health Care, Daiichi Sankyo, Rovi and Sanofi-Aventis; and lectures fees from Bayer Health Care, Daiichi Sankyo, Glaxo Smith Kline, Leo Pharma, Menarini, Rovi, and Sanofi-Aventis. M. M. has received funding for research from Sanofi and Bayer HealthCare, compensation for advisory-board membership from Bayer HealthCare and Daiichi Sankyo, and lecture fees from Bayer HealthCare, Daiichi Sankyo, Leo Pharma and Sanofi. J.A.N. received advisory fees from Boehringer, Bristol-Mayer and Bayer, lecture fees from Sanofi-Aventis and Bayer and participated in multicentre clinical trials sponsored by Boehringer and Bayer. J.A.P declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. .
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
