Abstract
Annually, 10% of patients with atrial fibrillation (AF) or venous thromboembolism (VTE) treated with non-vitamin K oral anticoagulants undergo diagnostic or therapeutic procedures. This subanalysis of the multicenter, prospective, observational Edoxaban Management in Diagnostic and Therapeutic Procedures real-world registry included patients in Europe and Asia with AF or VTE who underwent transcatheter cardiovascular (CV) procedures. Edoxaban interruption and clinical outcomes were assessed for all arterial or venous access procedures and stratified by bleeding risk. Overall, 2695 procedures were reported; 755 (28.0%) were transcatheter CV procedures, of which 373 (49.4%) were arterial access and 382 (50.6%) were venous access procedures. Patients with arterial versus venous access procedures had significantly higher bleeding and stroke and thromboembolism risk scores (P < 0.0001 for both) and underwent procedures that were more frequently classified as having a higher European Heart Rhythm Association bleeding risk. Edoxaban was interrupted in 59.5% (222) arterial versus 42.4% (162) venous access procedures, mostly either only preprocedurally or both pre- and postprocedurally. The combined incidence of clinically relevant ischemic or bleeding event rates and deaths was low (0.8 events/100 procedures). This subanalysis showed that while edoxaban was interrupted in approximately half of all interventions, ischemic events and major bleeding were low, suggesting transcatheter CV procedures can be performed safely in high-risk patients with AF or VTE. Patient and procedural factors should be considered to personalize the decision of edoxaban management around the time of a transcatheter CV procedure.
Keywords
Introduction
Atrial fibrillation (AF) is an increasingly common condition with rising incidence and prevalence in advanced age, affecting more than 20% of octogenarians.1–4 The majority of patients with AF require long-term treatment with antithrombotic agents to reduce the risk of stroke. Non
Among the most frequently performed procedures are those requiring arterial and/or vascular access, such as coronary angiographies, percutaneous coronary interventions, valvular procedures, and ablations.10,14,15 No clear evidence exists for the optimal time to interrupt or reinstate oral anticoagulation, balancing the reduction of MB against the potential higher risk of ischemic events, notably, thromboembolic stroke. It is also unclear which criteria physicians use to determine their periprocedural oral anticoagulation strategy and whether they would decide to use other antithrombotic agents during that interruption.
Therefore, the above questions were addressed by investigating the real-world clinical practice of edoxaban management in a subgroup of the Edoxaban Management in Diagnostic and Therapeutic Procedures real-world registry in patients with AF or VTE (EMIT-AF/VTE) 14 with the focus on effectiveness and safety parameters. The objective of the present work was to analyze the periprocedural management of edoxaban and clinical outcomes in patients with AF or VTE undergoing unselected diagnostic or therapeutic transcatheter cardiovascular (CV) procedures in daily practice.
Methods
Study Design
EMIT-AF/VTE (NCT02950168, NCT02951039) is a multicenter, prospective, observational program conducted in Europe and Asia in accordance with the Declaration of Helsinki and with the approval of local Institutional Review Boards. All participants provided written informed consent prior to enrollment. Periprocedural management of edoxaban was at the discretion of the investigators. The protocol design and overall results of the Global EMIT-AF/VTE program were previously published.14,16
Patient Recruitment
Enrollment commenced in December 2016 and was completed in April 2020 for the countries included in the Global EMIT-AF/VTE program. Patients were recruited from Belgium, Germany, Italy, the Netherlands, Portugal, South Korea, Spain, Taiwan, Thailand, and the United Kingdom. Eligible patients were adults with AF or VTE who were treated with edoxaban according to the local labels and were not enrolled concurrently in any interventional study. This subanalysis of the Global EMIT-AF/VTE program included patients who underwent transcatheter CV procedures.
Outcome Parameters
Time and duration of edoxaban interruption relative to the procedure, as reported by the physician, and dosing of edoxaban were recorded. Data on edoxaban interruption were collected from 5 days prior to the procedure to 30 days after; ischemic and bleeding events were collected from the day of the procedure to 30 days thereafter. Events were defined in accordance with the International Society of Thrombosis and Haemostasis. 17 Primary outcome parameter was the composite of ischemic stroke, transient ischemic attack (TIA), acute coronary syndrome (ACS), any systemic embolic event (SEE), and the rate of MB.
Secondary outcomes included ACS, ischemic stroke, TIA, SEE, deep vein thrombosis, pulmonary embolism, as well as CV and all-cause mortality. Other outcome parameters included the incidences of clinically relevant nonmajor bleeding (CRNMB), hemorrhagic stroke, and all strokes combined. All incidences of MB, CRNMB, ACS, and acute thromboembolic events were reviewed and unanimously adjudicated by the Steering Committee. Bleeding had to commence during or after the procedure to be classified as a procedural complication.
Observations
The observation period of the study started 5 days before and ended 30 days after the procedure. Details of edoxaban treatment and clinical outcomes were documented for the 30-day period after each procedure. “No interruption” of edoxaban therapy was defined as edoxaban administration on each day of the observation period. Any interruption of edoxaban treatment was recorded as the number of days without administration of edoxaban. Any dose skipped before or on the day of the procedure was categorized as preprocedural. Heparin bridging was defined as any heparin use during the time period ranging from the day before the procedure until the day after the procedure. Details of edoxaban treatment, use of concurrent antiplatelet therapy (APT), type of diagnostic/therapeutic procedures, periprocedural EHRA bleeding risk, 13 modified HAS-BLED (hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, elderly, drugs/alcohol concomitantly), 18 and CHA2DS2-VASc scores were recorded at the baseline. 12
Statistical Analysis
Data quality checks were performed on a regular basis to ensure that reported data were accurate and complete and that the conduct of the study complied with the observational plan and regulatory requirements. 16 Binary, categorical, and ordinal parameters were summarized as absolute and percentage numbers. Numerical data were described by descriptive statistics. Comparisons of baseline demographics and clinical characteristics between patients with arterial versus venous procedures were performed using Fisher's exact test. Edoxaban interruption, clinical outcomes, and APT were summarized for all vascular access procedures, by access route (arterial or venous) and by EHRA procedural bleeding risk. The frequency and duration of edoxaban interruption were assessed at the following time points: preprocedure only, postprocedure only, and both pre- and postprocedure. Clinical event rates are presented as the number of events per 100 procedures. Statistical analyses were performed using SAS® version 9.3 or higher (SAS Institute, Cary, NC, USA).
Results
To date in the Global EMIT-AF/VTE program, 2695 procedures in 1952 patients were reported, of which 755 were transcatheter CV procedures, 373 (49.4%) through arterial and 382 (50.6%) via venous access (Supplemental Figure S1). Most arterial access procedures were coronary angiographies (66.4%); venous access procedures were primarily electrophysiologic interventions or studies (72.5%; Figure 1).
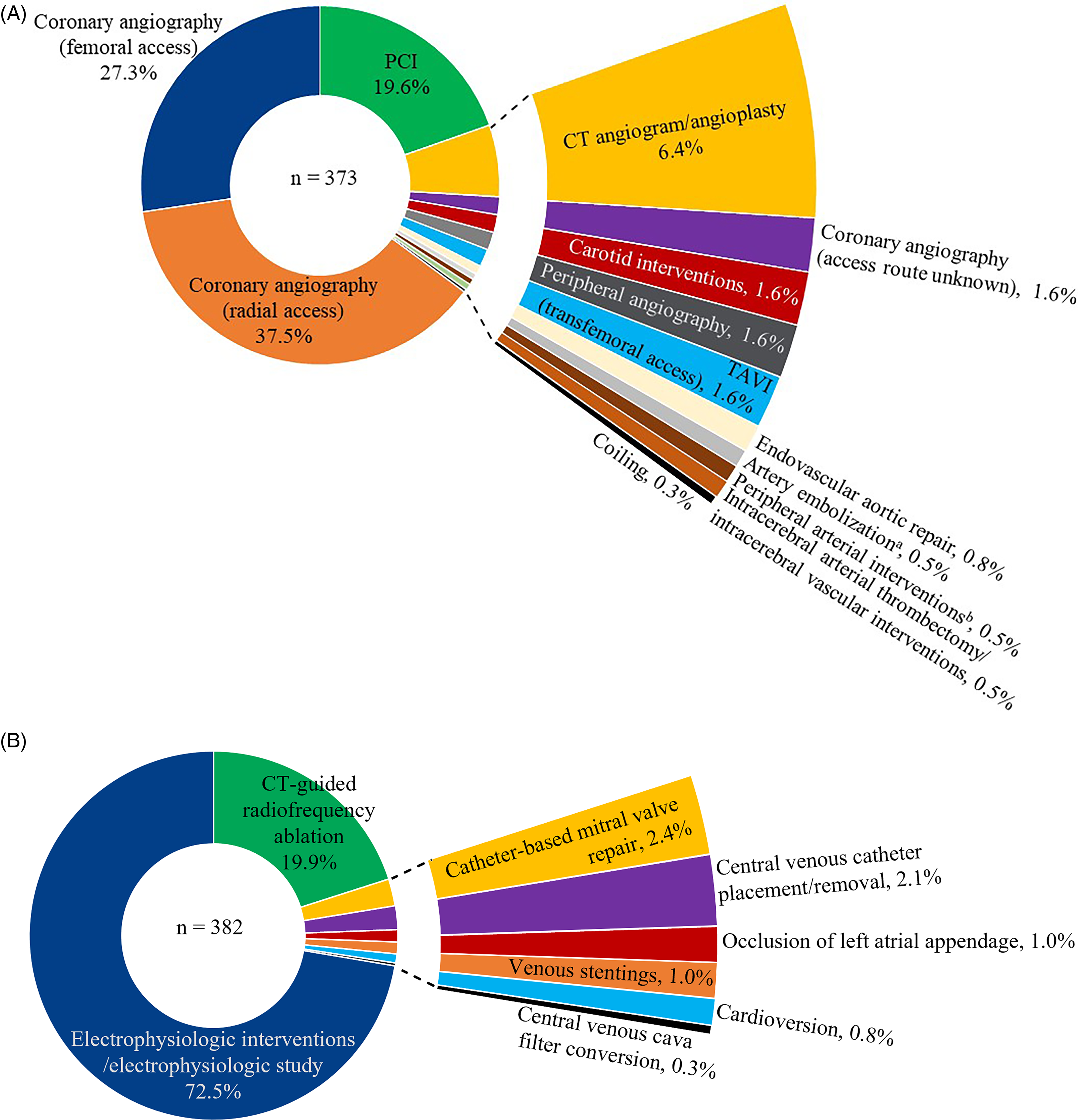
Arterial (A) and venous (B) access procedure subtypes. aNon-bleeding indications. bExcluding carotids. CT, computed tomography; PCI, percutaneous coronary intervention; TAVI, transcatheter aortic valve implantation.
Most patients (657 [86.9%]) underwent a single CV procedure; 43 patients (5.6%) underwent 2 procedures, whereas 3 patients (0.4%) underwent 3 or more. At the baseline, almost all CV risk factors (ie, arterial hypertension, coronary heart disease, diabetes, or heart failure) were considerably more frequent or more pronounced in patients with procedures through arterial versus venous access routes (P ≤ 0.0001 for all). When combined, this led to significantly higher mean ± standard deviation CHA2DS2-VASc (3.4 ± 1.5 vs 2.2 ± 1.5) and modified HAS-BLED scores (1.9 ± 1.0 vs 1.3 ± 0.9; P < 0.0001 for both) in patients who underwent arterial compared with venous access procedures (Table 1). Arterial (53.6%) versus venous (7.4%) access procedures were more frequently classified as a low or high bleeding risk category. Patients who underwent procedures with a higher EHRA bleeding risk level had higher mean CHA2DS2-VASc and HAS-BLED scores compared with patients who underwent procedures with a lower bleeding risk level (low or minor risk). For the overall population, CHA2DS2-VASc and HAS-BLED scores showed a significant (P < 0.0001), though moderate, correlation (r = 0.46). More patients with arterial than venous access procedures fulfilled at least one edoxaban dose reduction criterion and, subsequently, received edoxaban 30 mg once daily (P = 0.0003).
Baseline Patient Demographics and Clinical Characteristics.
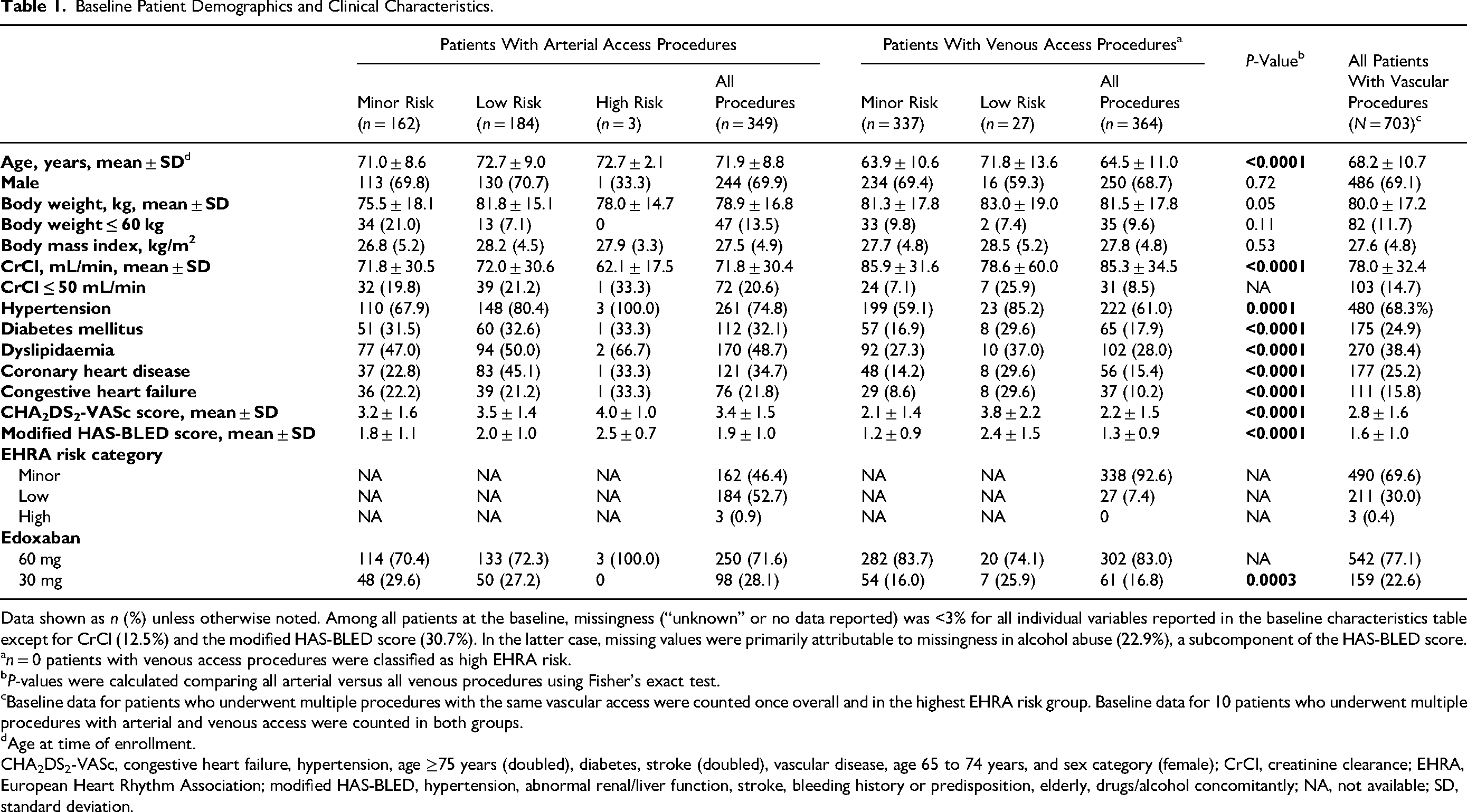
Data shown as n (%) unless otherwise noted. Among all patients at the baseline, missingness (“unknown” or no data reported) was <3% for all individual variables reported in the baseline characteristics table except for CrCl (12.5%) and the modified HAS-BLED score (30.7%). In the latter case, missing values were primarily attributable to missingness in alcohol abuse (22.9%), a subcomponent of the HAS-BLED score.
n = 0 patients with venous access procedures were classified as high EHRA risk.
P-values were calculated comparing all arterial versus all venous procedures using Fisher's exact test.
Baseline data for patients who underwent multiple procedures with the same vascular access were counted once overall and in the highest EHRA risk group. Baseline data for 10 patients who underwent multiple procedures with arterial and venous access were counted in both groups.
Age at time of enrollment.
CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75 years (doubled), diabetes, stroke (doubled), vascular disease, age 65 to 74 years, and sex category (female); CrCl, creatinine clearance; EHRA, European Heart Rhythm Association; modified HAS-BLED, hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, elderly, drugs/alcohol concomitantly; NA, not available; SD, standard deviation.
Edoxaban was interrupted in approximately half of all procedures, with a somewhat higher percentage of interruptions occurring for arterial (222/373 [59.5%]) versus venous access procedures (162/382 [42.4%]). In patients with a high bleeding and/or stroke risk, as reflected by patient characteristics (ie, CHA2DS2-VASc and HAS-BLED scores) and EHRA procedural bleeding risk classification (ie, more arterial procedures were classified as high-risk procedures), edoxaban was interrupted more frequently and for a longer period (Figures 2 and 3). As a consistent pattern between arterial and venous access procedures, edoxaban was interrupted primarily only preprocedurally (253/754 [33.6%]) or both pre- and postprocedurally (98/754 [13.0%]), whereas postprocedural-only interruption was rare (32/754 [4.2%]; Supplemental Table S1). One patient had 1 arterial access procedure and 1 venous access procedure on the same day. These procedures were counted once each in arterial and venous procedures; however, they are counted only once in all vascular procedures.
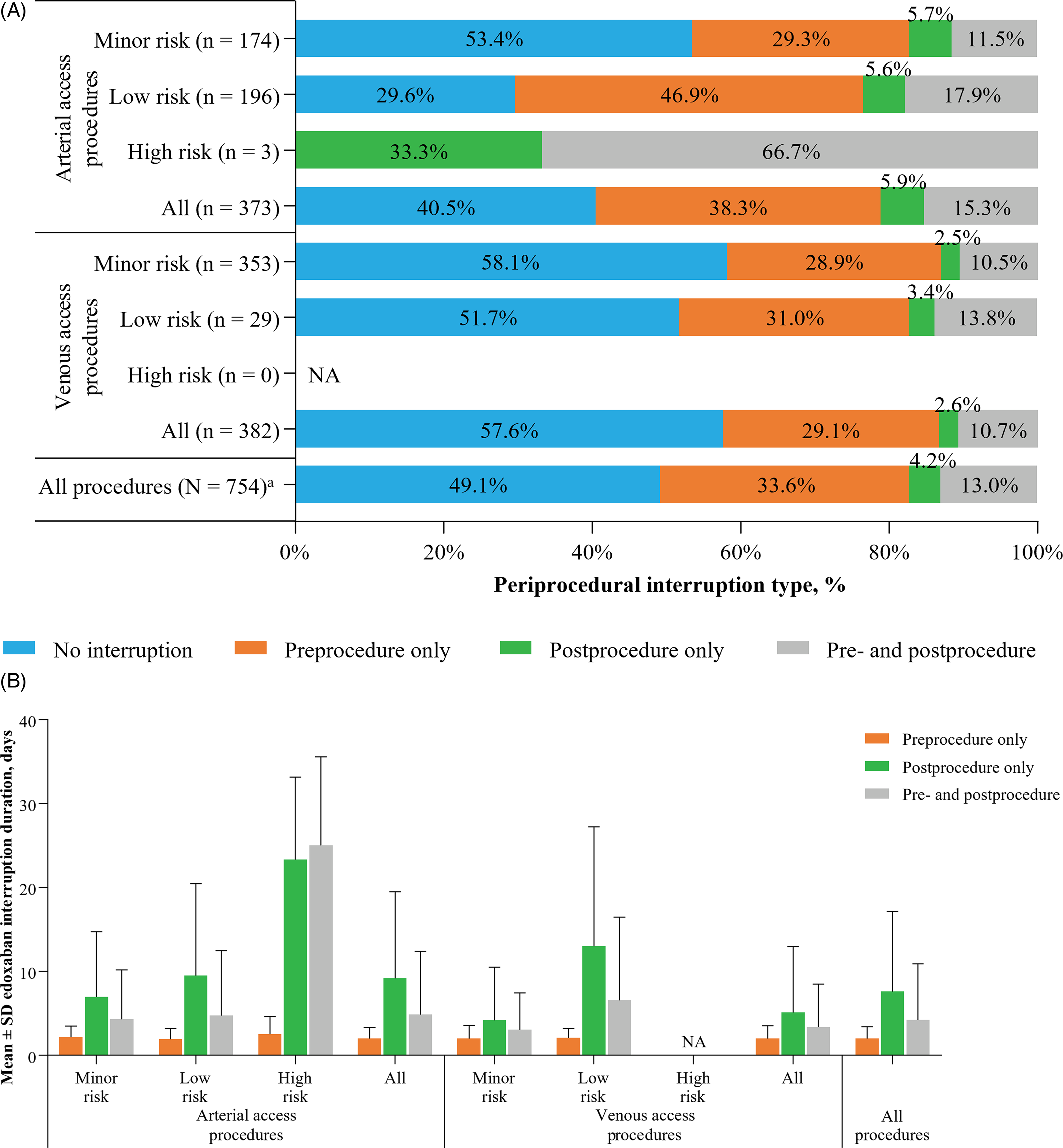
Type (A) and mean ± SD duration (B) of edoxaban interruption by EHRA risk score. aOne patient had 1 arterial and 1 venous access procedure on the same day. These procedures were counted once each in arterial and venous procedures, respectively; however, they are counted only once in all vascular procedures. Mean ± SD interruption duration excludes procedures without edoxaban interruption. EHRA, European Heart Rhythm Association; NA, not applicable; SD, standard deviation.
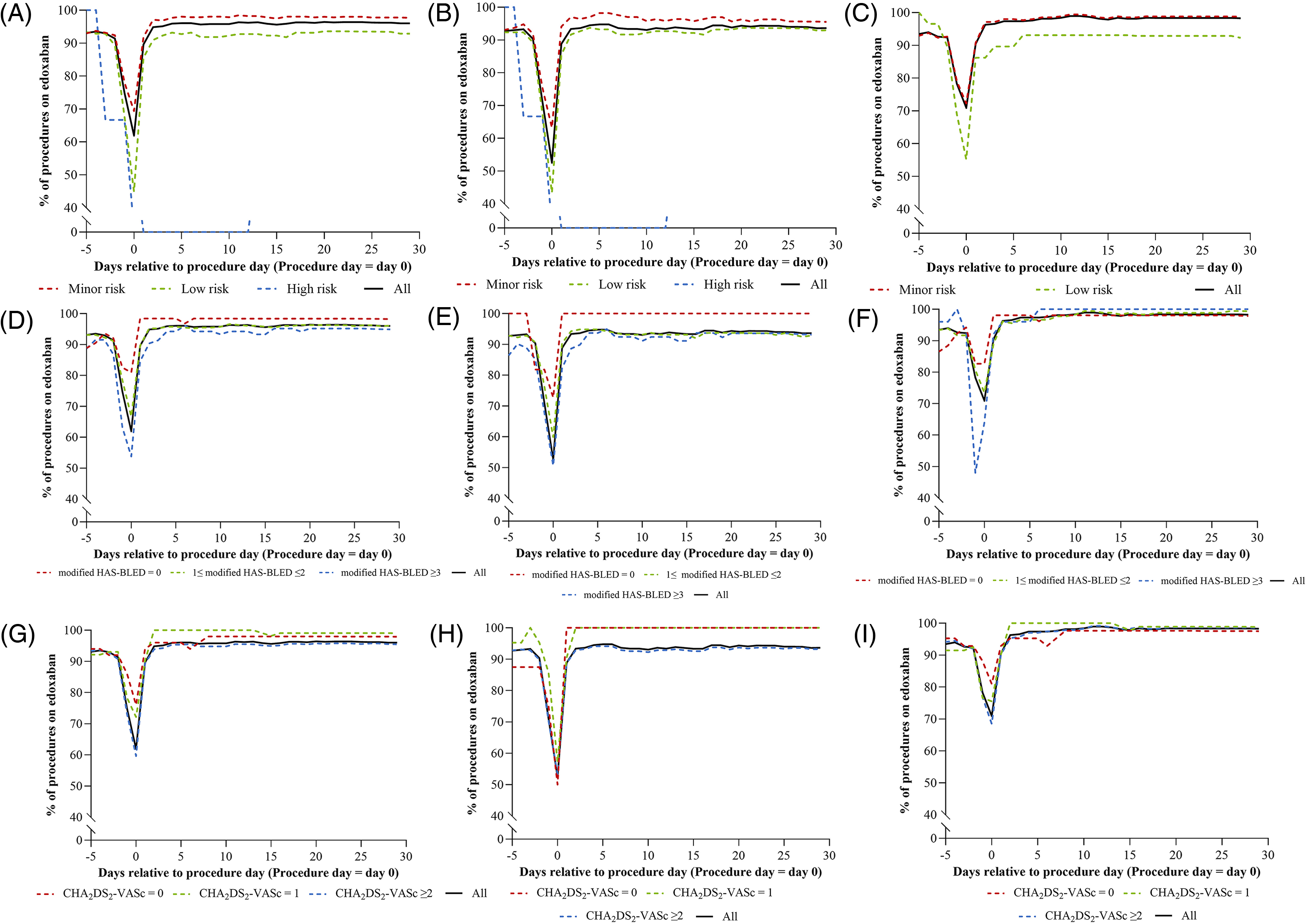
Periprocedural edoxaban use over time for all procedures, arterial access procedures, and venous access procedures by EHRA risk score (A–C), modified HAS-BLED score (D–F), and CHA2DS2-VASc score (G–I). CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75 years (doubled), diabetes, stroke (doubled), vascular disease, age 65 to 74 years, and sex category (female); EHRA, European Heart Rhythm Association; modified HAS-BLED, hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, elderly, drugs/alcohol concomitantly.
APT was more frequent for arterial versus venous procedures on the day of the procedure and during follow-up (24% vs <3%). During the observation period, APT spiked around the day of the procedure and was higher post- than preprocedurally. APT was more common in arterial and venous procedures for low (41.8% and 24.1%) versus minor (30.5% and 5.1%) bleeding risk (Supplemental Figure S2). For both arterial and venous access procedures, single APT was more common than dual APT at all time points assessed (Supplemental Figure S3).
Clinically relevant ischemic or bleeding events occurred in 0.8 of 100 procedures (Table 2). Ischemic and bleeding events occurred more frequently in venous access procedures compared with arterial access procedures.
Clinical Outcomes for Arterial and Venous Access Procedures.
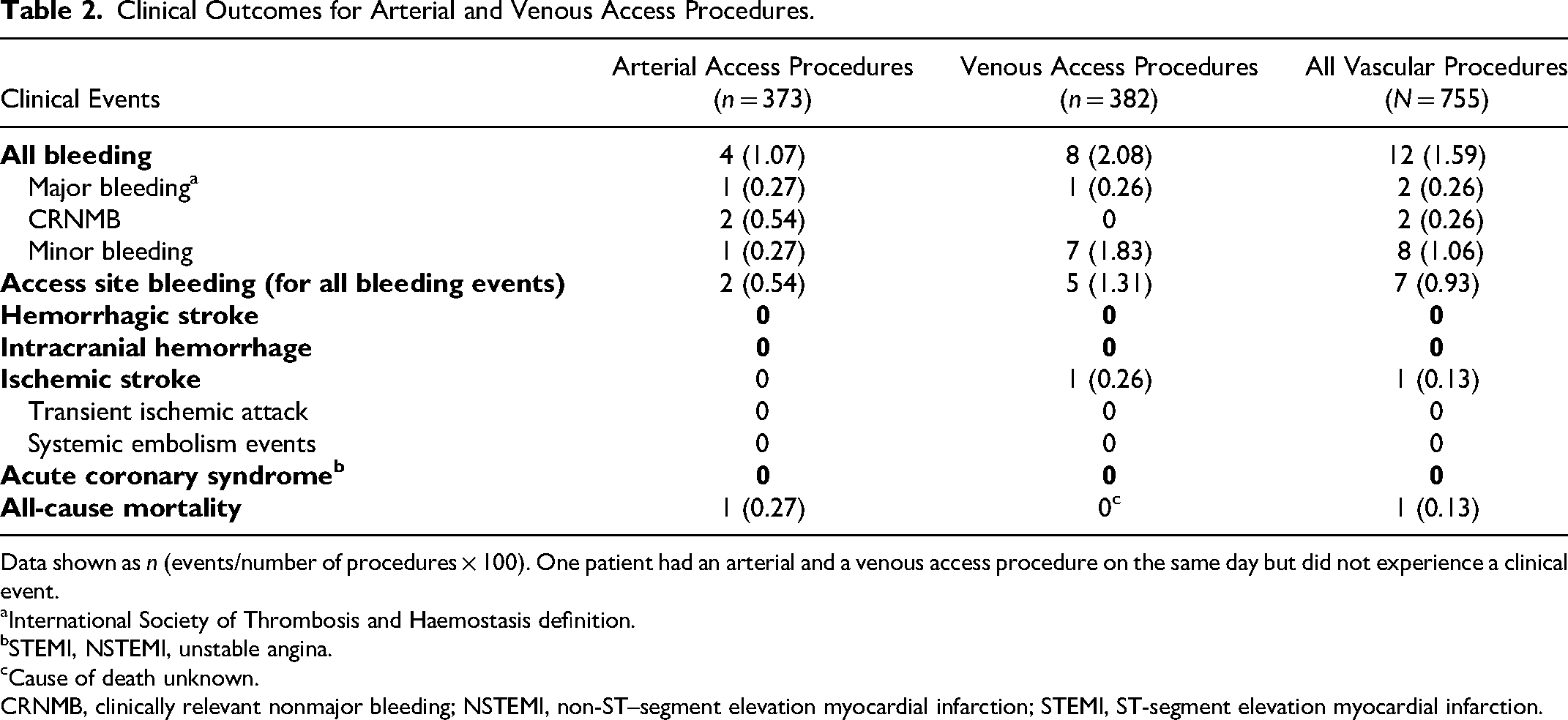
Data shown as n (events/number of procedures
International Society of Thrombosis and Haemostasis definition.
STEMI, NSTEMI, unstable angina.
Cause of death unknown.
CRNMB, clinically relevant nonmajor bleeding; NSTEMI, non-ST–segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction.
In aggregate, while patients were on edoxaban (<72 h since the last dose), 2 MB and 1 ischemic stroke events occurred. One of the MB events occurred during the observation period and was related to a polypectomy done during that time frame. No MB or ischemic events were reported in patients off edoxaban. Six clinically relevant events (ie, 2 MB, 2 CRNMB, and 1 ischemic stroke, and 1 all-cause death) occurred on Days 0 to 8 of the procedure (Table 3). The only death occurred on Day 19 after the procedure, with a very unlikely causal relation to the index angiography procedure.
Clinically Relevant Ischemic or Bleeding Events.
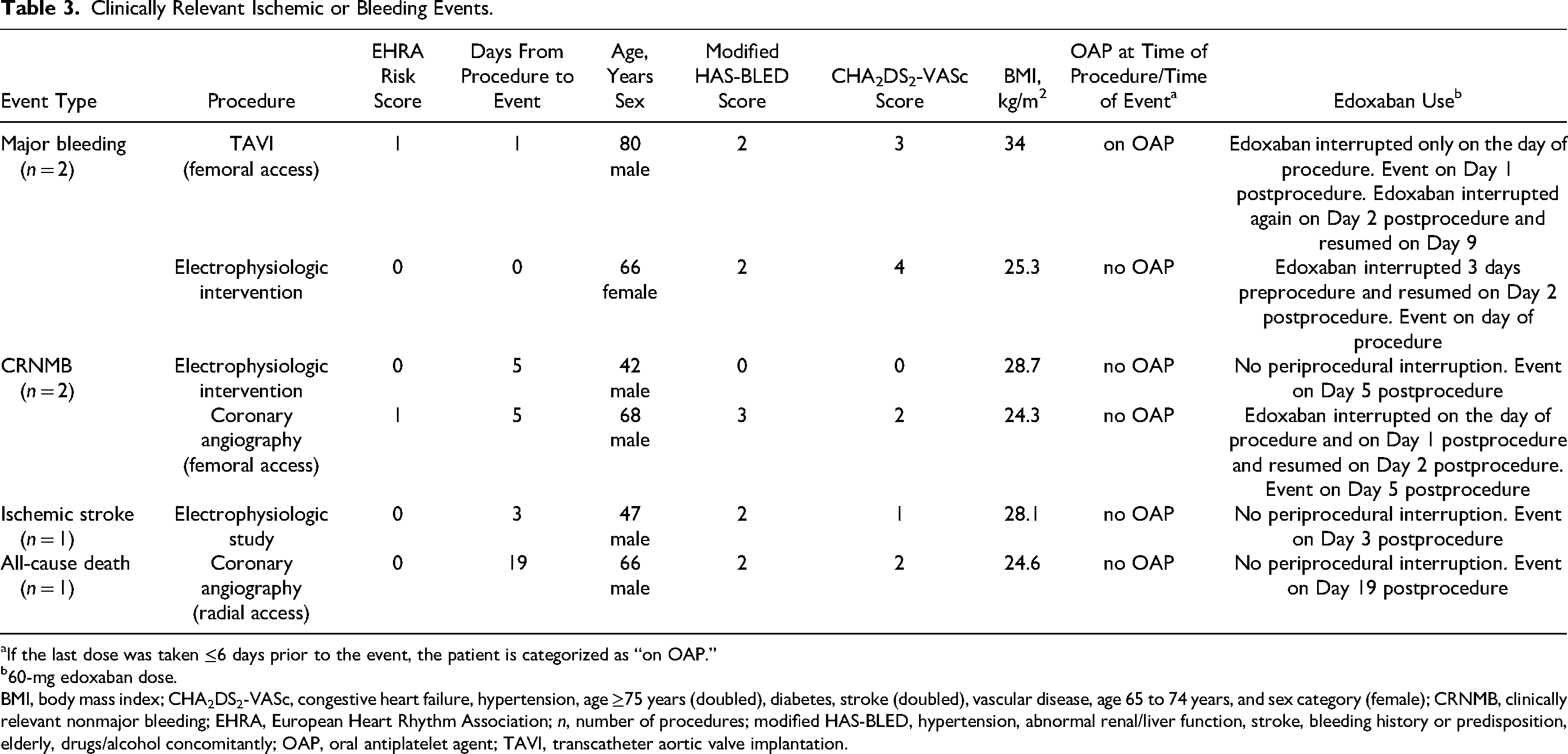
If the last dose was taken ≤6 days prior to the event, the patient is categorized as “on OAP.”
60-mg edoxaban dose.
BMI, body mass index; CHA2DS2-VASc, congestive heart failure, hypertension, age ≥75 years (doubled), diabetes, stroke (doubled), vascular disease, age 65 to 74 years, and sex category (female); CRNMB, clinically relevant nonmajor bleeding; EHRA, European Heart Rhythm Association; n, number of procedures; modified HAS-BLED, hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, elderly, drugs/alcohol concomitantly; OAP, oral antiplatelet agent; TAVI, transcatheter aortic valve implantation.
Discussion
In this large, international, prospective, real-world subanalysis of the Global EMIT-AF/VTE program in patients undergoing transcatheter CV procedures, physicians interrupted edoxaban in about half of all interventions. Interruption was more frequent in higher bleeding risk procedures and in patients with a higher bleeding risk score. APT was more common in arterial versus venous procedures and was most often used on the day of procedures or postprocedurally. Despite relatively high ischemic and bleeding risks, patients undergoing transcatheter CV procedures had a very low risk of ischemic and MB events or deaths for an observation period of 30 days, indicating adequate periprocedural management of edoxaban in these patients in routine clinical practice. Understanding which factors were involved in the choice and timing of interruption can further help to improve periprocedural management.
Factors associated with a higher likelihood of interruption were higher risks for stroke and bleeding based on patient characteristics (CHA2DS2-VASc and HAS-BLED scores) and procedural risk (EHRA classification). 13 Arterial versus venous access procedures were more likely interrupted. This finding may have been due partly to a greater risk of ischemia and MB in patients undergoing arterial access procedures. Arterial access procedures, such as coronary angiography or percutaneous coronary interventions, may have an increased bleeding risk in patients on chronic oral anticoagulation. 19 Procedures in patients with higher risks for ischemic events, as described by the CHA2DS2-VASc score, were more frequently interrupted in this registry compared with procedures in patients with lower ischemic risks. At first glance, this seems counterintuitive. However, as the CHA2DS2-VASc and HAS-BLED scores show a significant correlation in this registry, it appears reasonable to hypothesize that ischemic risk was unlikely a reason for edoxaban interruption rather than a reflection of a somewhat higher risk for MB. This is in line with other previous studies relating bleeding risk with CHA2DS2-VASc score, HAS-BLED score, and EHRA classification.20–23
When physicians interrupted anticoagulant treatment, they did so before or on the day of the procedure in the majority (92%) of cases, whereas postprocedural-only interruptions were rare (8%). These cases may have been more complex procedures in which physicians deemed the risk of subsequent bleeding higher.
Major bleeding was very rare. The 2 MB events (0.26% for the entire cohort) occurred in 1 patient on the day following a transcatheter aortic valve implantation (TAVI) but before a scheduled colonoscopy with polypectomy and in 1 patient on the day of an electrophysiologic study. There were no clinical sequelae from these 2 MB or the 2 CRNMB events.
Although the number of adverse events was low and despite a higher frequency of anticoagulation interruption, patients that underwent arterial access procedures had a higher rate of MB or CRNMB events compared with those who underwent venous access route procedures (0.8 vs 0.3 events per 100 procedures). More patients with arterial (24%) than venous (<3%) access procedures were administered oral APT during the observation period to a large extent driven by percutaneous coronary interventions. However, excluding the patient who underwent TAVI, no patient with an ischemic, MB, or CRNMB event was on oral APT. One death of unknown cause occurred on Day 19 after an uneventful coronary angiography and was very unlikely considered as a direct consequence of the procedure.
To the authors’ knowledge, this is the first analysis assessing periprocedural management and outcomes for patients with AF or VTE receiving NOACs who underwent transcatheter CV procedures in real-world clinical practice. Though tempting, the juxtaposition of the current study with a subanalysis of the Effective Anticoagulation With Factor Xa Next generation in Atrial Fibrillation-Thrombolysis in Myocardial Infarction 48 (ENGAGE AF-TIMI 48) randomized, controlled trial that included a wide variety of surgical procedures warrants caution. 24 These patients had a higher number of and more severe CV and bleeding risk factors on the one hand but longer (up to 10 days) interruptions of edoxaban on the other. 24 Ischemic and MB event rates were higher in the ENGAGE AF-TIMI 48 trial than in the current EMIT subanalysis. However, only 10% were percutaneous coronary interventions, and the EHRA classification was not reported. In the Global EMIT registry, which included all types of diagnostic procedures, the overall rates of thromboembolic, CRNMB, and MB events were low (0.6%, 0.7%, and 0.4%, respectively). 14 The Perioperative Anticoagulation Use for Surgery Evaluation (PAUSE) study assessed perioperative management of apixaban, dabigatran, and rivaroxaban in patients with AF undergoing 13 different types of elective surgery or procedures and found the overall 30-day perioperative rates of MB were 1.35% in the apixaban cohort, 0.90% in the dabigatran cohort, and 1.85% in the rivaroxaban cohort. 11 The overall rates for CRNMB and minor bleeding were 1.67% and 4.3% in apixaban, 1.95% and 5.69% in dabigatran, and 2.4% and 5.73% in rivaroxaban. 11 In comparison, the rate of MB, CRNMB, and minor bleeding in patients receiving edoxaban in the current study was 0.26%, 0.26%, and 1.06%, respectively. A lower MB rate in the current study may be due to the inclusion of a more specific population, in which only patients undergoing transcatheter CV procedures were included, whereas the PAUSE study included primarily cardiothoracic and gastrointestinal procedures. 11 Additionally, 33.5% of patients in the PAUSE study underwent high bleeding risk procedures compared with 0.4% of patients in the current analysis. Notably, the PAUSE study used a predefined interruption protocol for surgeries with different bleeding risks, while in the current analysis, periprocedural management of edoxaban was at the discretion of the physicians. 11
Two main limitations of this study are the lack of a comparator arm and the investigation of only one NOAC. Given the low event rates, either a larger registry or a randomized trial would be desirable, along with the attempt to differentiate between different oral anticoagulants. However, these approaches require a much greater sample size and longer duration, which may not be feasible from a timing and economic perspective. 25 For the same reasons, no attempt was undertaken to evaluate the impact of geographical location or ethnicity. Furthermore, edoxaban management was not standardized, as it was at the discretion of the investigator. To account for the challenges of data collection, patients were provided with memory aids to support data recall. Data elements were reviewed at the patient level, and all incidents of MB, CRNMB, ACS, and acute thromboembolic events were centrally adjudicated.
Conclusions
In patients on chronic oral anticoagulation with edoxaban, transcatheter CV procedures can be performed safely. Periprocedurally, a brief interruption of edoxaban in high-risk patients was not associated with high risks of ischemic events. Additionally, continuing edoxaban in selected patients was also not associated with a high risk of bleeding events. Patient and procedural factors should be considered to personalize the decision of edoxaban management around the time of a transcatheter CV procedure.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241260728 - Supplemental material for Periprocedural Edoxaban Management and Clinical Outcomes in Patients Undergoing Transcatheter Cardiovascular Procedures in the EMIT-AF/VTE Program
Supplemental material, sj-docx-1-cat-10.1177_10760296241260728 for Periprocedural Edoxaban Management and Clinical Outcomes in Patients Undergoing Transcatheter Cardiovascular Procedures in the EMIT-AF/VTE Program by Martin Unverdorben, Paolo Colonna, James Jin, Sabine Köhler, Amparo Santamaria, Manish Saxena, Amanda Borrow, Cathy Chen, Christian von Heymann and Thomas Vanassche in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors thank Petra Laeis, PhD, for operating the EMIT program. Medical writing and editorial assistance for this manuscript were provided by Kimberly Dent-Ferguson, MBS, MPH, of AlphaBioCom, a Red Nucleus company, and funded by Daiichi Sankyo, Inc.
Data Sharing Statement
The data underlying this article cannot be shared publicly, as the Global EMIT-AF/VTE study is currently ongoing.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AB, CC, JJ, and MU are employees of Daiichi Sankyo. PC reports grants and personal fees from Daiichi Sankyo; personal fees from Bayer, Boehringer Ingelheim, Daiichi Sankyo, and Pfizer/BMS; and nonfinancial support from the European Society of Cardiology (ESC) and the Italian Cardiology Association (ANMCO). CvH reports grants and personal fees from Daiichi Sankyo; personal fees from Artcline GmbH, CSL Behring, HICC GbR, Mitsubishi Pharma, Novo Nordisk Pharma, Shionogi Pharma, and Sobi Pharma; receipt of a mandate from the German Society of Anaesthesiology and Intensive Care Medicine (DGAI) to write the German Guidelines on Preoperative Anemia; receipt of a mandate from the Deutsche Interdisziplinäre Vereinigung für Intensiv- und Notfallmedizin (DIVI) to take part in the writing group of the guidelines on the Prevention of Venous Thromboembolism of the Gesellschaft für Thrombose- und Hämostaseforschung (GTH); participation in the writing group of the Patient Blood Management Guideline in cardiac surgery on behalf of the European Society of Cardiothoracic Anaesthesiologists (EACTA) in conjunction with the European Society of Cardiothoracic Surgery (EACTS); and receipt of a mandate to take part in the writing group of the guidelines on the Diagnostics and Treatment of Peripartum Haemorrhage of the Deutsche Gesellschaft für Gynäkologie und Geburtshilfe (DGGG). SK reports consulting fees from Daiichi Sankyo. AS reports nothing to disclose. MS reports grants and personal fees from Daiichi Sankyo and personal fees from Daiichi Sankyo; TV report grants and personal fees from Daiichi Sankyo; and personal fees from Bayer, Boehringer Ingelheim, Daiichi Sankyo, and LEO Pharma.
Ethics Approval
EMIT-AF/VTE (NCT02950168, NCT02951039) is a large multicenter program conducted in Europe and Asia in accordance with the Declaration of Helsinki and with the approval of local Institutional Review Boards.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was funded by Daiichi Sankyo, Inc.
Informed Consent
All participants provided written informed consent prior to enrollment.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
