Abstract
Vitamin K antagonists (VKAs) remain the standard therapy for anticoagulation in prevention and treatment of venous thromboembolism (VTE) and for the prevention of stroke in atrial fibrillation (AF). Due to numerous limitations of VKAs, target-specific oral anticoagulants have been developed. Edoxaban is a direct activated factor X inhibitor with attractive features among which are once daily dosing, no need for routine monitoring, and minimal drug–drug interactions. In patients undergoing orthopedic surgery, edoxaban was superior to enoxaparin in preventing VTE. Furthermore, a recent large-scale phase III trial in patients with symptomatic VTE demonstrated that edoxaban was noninferior to warfarin in preventing recurrent VTE and reduced bleeding. In the largest trial of anticoagulation in patients with AF to date, edoxaban was noninferior to warfarin in the prevention of stroke or systemic embolism and reduced bleeding and cardiovascular mortality. This review provides an overview of the pharmacology, clinical trial results, and potential indications for edoxaban.
Introduction
Venous Thromboembolism
Venous thromboembolism (VTE) is the third most common cardiovascular disorder with an estimated mortality in the United States between 100 000 and 180 000 deaths annually. 1,2 The most common manifestations of VTE are deep vein thrombosis of lower extremities and pulmonary embolism (PE). Each year in the United States, more than 4 000 000 surgical patients and 8 000 000 medical patients are hospitalized and are at risk of VTE. Among hospitalized medical patients, the 4 most common risks factors are prior VTE, bed rest, peripherally inserted central venous catheter, and cancer. 2
In addition, clinical trials in patients undergoing orthopedic surgery have demonstrated the importance of VTE prophylaxis. The prevalence of deep venous thrombosis (DVT) after total hip replacement (THR), total knee replacement (TKR), surgery, or surgery for hip fracture is 45% to 57%, 40% to 84%, and 36% to 60%, and prevalence of PE in these patients is 0.7% to 30%, 1.8% to 7%, and 4.3% to 24% respectively. 3
Atrial Fibrillation
Atrial fibrillation is the most prevalent cardiac arrhythmia, affecting more than 2.2 million people in the United States. 4 It is associated with excess mortality and increasing risk of ischemic stroke by a factor of 4 to 5. 5,6 Heparin followed by vitamin K antagonists (VKAs) has been the standard of care for prevention and treatment of VTE 7 and atrial fibrillation (AF). 6
Limitations of VKAs
Although VKAs are effective in preventing VTEs and stroke in AF, they have many limitations, such as a narrow therapeutic window, the need for frequent monitoring and dose adjustments, a slow onset and offset of action, interpatient variability in metabolism, and many drug–drug and food–drug interactions. 5 As a result, adherence is low and many patients remain inadequately anticoagulated. 8 Furthermore, since efficacy of VKAs depends on the time in the therapeutic range, poor monitoring leads to increasing hemorrhagic and thrombotic events. 9
Target-Specific Oral Anticoagulants
These limitations have prompted the development of target-specific oral anticoagulants (TSOACs), which appear to be an effective and safer alternative. The first 3 TSOACs introduced into clinical practice were dabigatran, rivaroxaban, and apixaban. In the United States, all 3 agents are currently approved for stroke prevention in patients with AF, while rivaroxaban is the only TSOAC approved for VTE prophylaxis and treatment in patients with TKR and THR. Dabigatran is a direct thrombin inhibitor (DTI), and rivaroxaban and apixaban are activated factor X (FXa) inhibitors.
By inhibiting thrombin, direct thrombin inhibitors (DTIs) prevent thrombin formation, reduce thrombin production, and decrease platelet activation. Rivaroxaban and apixaban are the only oral FXa inhibitors approved in the United States and Europe. 10,11 Since FX is at the junction of intrinsic and extrinsic pathways, inhibition at this stage blocks the downstream thrombin activation from either pathway. 11
Several phase III trials with TSOACs, in patients with recent orthopedic surgery, acute VTE, and AF, demonstrate that TSOACs are at least as good as warfarin in preventing VTE and reducing bleeding. 8,12 As these target-specific anticoagulation agents namely, apixaban, rivaroxaban, and dabigatran have been extensively reviewed, 13 -16 the focus of this article will be on edoxaban, the newest FXa inhibitor to complete phase III testing.
Edoxaban/Preclinical Studies
Edoxaban (DU-176b; Figure 1) was developed by Daiichi-Sakyo (Tokyo, Japan). It is an oral, reversible direct FXa inhibitor. 5,19 In preclinical studies, it was found to be >10 000 more potent than other serine proteases at inhibiting FXa, demonstrating high specificity for FXa with maximum anti-FXa activity of 2.1 and 3.8 IU/mL at 30 and 60 mg doses of edoxaban, respectively. 18 In animal studies, edoxaban at peak plasma concentration caused 93% reduction in FXa activity. 20 A high therapeutic index of 133 was demonstrated in a rat model of intracerebral hemorrhage, indicating a high degree of separation between toxic and safe doses.
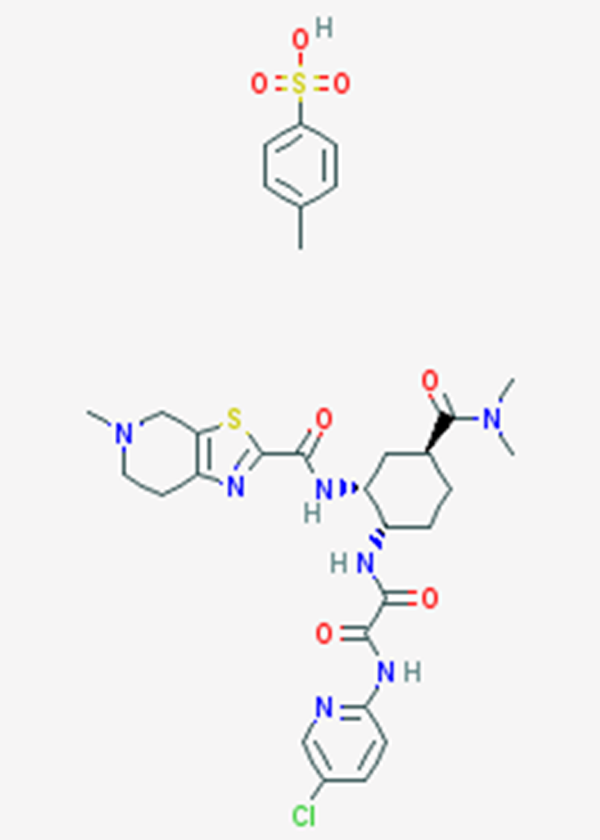
Chemical structure of edoxaban. 17 In comparison to the first small molecule inhibitor direct factor Xa inhibitor developed in 1994, DX-9065a, the chemical structure of edoxaban lacks the amide group. The increased oral bioavailability and >100 000-fold more potent inhibition of FXa are attributed to this chemical change. 18
Phase I Trials of Edoxaban
Edoxaban was shown to be safe and well tolerated at single doses up to 150 mg and multiple doses up to 120 mg up to 10 days in healthy volunteers. Edoxaban has dose-dependent anti-FXa activity 18 with predictable pharmacokinetic (PK) and pharmacodynamic (PD) profiles. 19 Edoxaban also exhibits dose-dependent prothrombin time (PT) and partial thromboplastin time (PTT) prolongation. At 30 mg and 60 mg of edoxaban daily, the PT was prolonged by 49% and 99%, respectively, with time to reach maximum PT at 1 hour. The time to maximum plasma concentration is 1 to 2 hours. 19 The renal elimination of edoxaban ranges from 35% to 39% in healthy volunteers 21 and more recent studies show renal elimination of 50%. 22 Edoxaban is a substrate for p-glycoprotein (P-gp), a transporter located in the intestine that prevents drug absorption into the systemic circulation. Thus, when coadministered with other strong P-gp inhibitors (ie, quinidine, verapamil, and dronedarone), a dose reduction of 50% of edoxaban is recommended. 23 Table 1 compares the key PK and PD parameters of edoxaban and other TSOACs.
Abbreviations: ACS, acute coronary syndrome; TSOAC, target-specific oral anticoagulant; FXa, activated factor X; PE, pulmonary embolism; Cmax, maximum plasma concentration.
a An additional 33% is excreted in the urine unchanged.
b Dose reduction recommended. Additional drugs may require dose adjustment based on age, weight, renal clearance, and other conditions that increase bleeding risk (see 10 for details).
c Azoles: ketoconazole, itraconazole, voriconazole, posaconazole (ketoconazole and itraconazole contraindicated with edoxaban).
d Contraindicated.
e No data.
f Anticonvulsants: carbamazepine, phenytoin, phenobarbital.
g Macrolides: erythromycin, azithromycin, clarithromycin.
Phase II
Several phase II trials were conducted which compared edoxaban to placebo or heparin in the prevention of VTE after TKR and THRs. 28,29 Edoxaban, at doses 15, 30, 60, or 90 mg once daily, was shown to be superior to heparin in decreasing the incidence of VTE following THR (N = 903) with no significant difference in major bleeding between edoxaban and heparin. 29 In 523 patients following total knee arthroplasty, edoxaban at doses of 5, 15, 30, 60, or 90 mg once daily was associated with significant reductions in the development of VTE versus placebo. With respect to safety analysis, there was no difference in major or clinically relevant bleeding across all edoxaban doses and in comparison to placebo. 28
Currently, a phase II randomized study is underway comparing the efficacy and safety of edoxaban monotherapy versus heparin/warfarin combination in patients with symptomatic DVT (NCT #01662908). The primary outcome being measured is the change in the thrombus volume at 14 to 21 days. 30
The major phase II study that lay groundwork for study of edoxaban in AF was conducted by Weitz et al. 31 In this randomized, multinational, parallel-group, dose-finding trial, edoxaban was compared with warfarin in 1146 patients with AF for 12 weeks. The study was double blind to the dose of edoxaban but open label to randomization between edoxaban and warfarin. Four regimens of edoxaban were utilized (30 mg once daily, 30 mg twice daily, 60 mg once daily, and 60 mg twice daily) versus warfarin titrated to international normalized ratio (INR) of 2.0 to 3.0. The study showed that the 2 once-daily regimens of edoxaban (30 mg once daily and 60 mg once daily) had a safety profile similar to warfarin and were associated with less bleeding than the twice-daily regimens. 23,31 It was particularly noteworthy that even with the same total daily dose of 60 mg, less bleeding was seen with once daily 60 mg than twice daily 30 mg. The pharmacokinetic analysis showed that the total plasma exposure of edoxaban was the same with 30 mg twice daily versus 60 mg once daily. However, with the once-daily regimen of 60 mg, the maximum plasma concentration was higher and trough levels were lower as compared to 30 mg twice-daily regimen. 31,32 Furthermore, the risk of bleeding correlated best with trough levels (which are lower with once-daily regimen) than with either peak levels of area under the curve. Based on these results, 30 mg once-daily and 60 mg once-daily dose of edoxaban were selected for testing in the phase III Effective aNticoaGulation with factor xA next GEneration in Atrial Fibrillation trial: Thrombolysis In Myocardial Infarction study 48 (ENGAGE AF-TIMI 48) trial. 31
Phase III Trials of Edoxaban
Edoxaban in prevention of VTE
Three phase III trials were conducted with edoxaban in a total of 1418 patients in the prevention of thromboembolism following orthopedic surgery. STARS E-3, STARS J-4, and STARS J-V were randomized double-blind trials that enrolled patients following unilateral total knee arthroplasty, trochanteric/subtrochanteric fracture repair, and unilateral total hip arthroplasty, respectively. 33 -35 All 3 trials compared 30 mg once-daily dose of edoxaban to enoxaparin with primary and secondary outcomes measured being VTE events and bleeding, respectively. The STARS J-4 trial showed similar rates of VTE between edoxaban and placebo (6.5% vs 3.7%; Table 2). 35 The STARS E-3 trial showed that edoxaban was associated with significantly fewer VTE events. 34 The STARS J-V trial showed that edoxaban was superior to enoxaparin in preventing VTE. 33 In all 3 trials, there was no significant difference in major bleeding between edoxaban and enoxaparin
Abbreviations: ENGAGE AF-TIMI 48, Effective aNticoaGulation with factor xA next GEneration in Atrial Fibrillation trial: Thrombolysis in Myocardial Infarction study 48; VTE, venous thromboembolism; SEE, systemic embolic event.
a Dose reduced by 50% in patients with creatinine clearance 30 to 50 mL per minute, body weight < 60 kg or concomitant use of potent P-glycoprotein inhibitors.
Edoxaban in VTE treatment
The Hokusai-VTE study was a double-blind, noninferiority trial of patients with acute symptomatic VTE who received either enoxaparin or unfractionated heparin for at least 5 days and were then randomized to receive either warfarin or edoxaban. A total of 8292 patients were enrolled across 37 countries. The dose of edoxaban tested was 60 mg once daily for most patients with a dose reduction to 30 mg once daily in patients with any one or more of the following: creatinine clearance 30 to 50 mL per minute, body weight ≤60 kg, or concomitant use of potent P-gp inhibitors. The control group received warfarin with an INR goal of 2.0 to 3.0. Blinding was maintained by the use of an encrypted point-of-care device to measure the INR with the use of sham INRs in patients randomized to edoxaban. The treatment was continued for a minimum of 3 months to a maximum of 12 months, with the duration determined by the treating physician. The primary efficacy outcome was the recurrence of VTE and the primary safety outcome measured was the incidence of major or clinically relevant nonmajor bleeding. 36 The noninferiority margin for the upper bound of the 95% confidence interval (CI) for the primary efficacy end point was 1.5. 37
Among all patients randomized, the rate of recurrent thromboembolism was 3.2% in the edoxaban group versus 3.5% in the warfarin group (P < .001 for noninferiority; Figure 2). Furthermore, the risk of clinically relevant bleeding was 8.5% in the edoxaban group versus 10.3% in the warfarin group (P = .004 for superiority). In the subgroup of patients with PE (n = 3319), the rate of recurrent thromboembolism was 2.8% in the edoxaban group versus 3.9% in the warfarin group (hazard ratio [HR] 0.73, 95% CI 0.50-1.06). Among high-risk patients with PE and right ventricular dysfunction (defined as an elevated N-terminal probrain natriuretic peptide levels, n= 938), the rate of recurrent thromboembolism was 3.3% in the edoxaban group versus 6.2% in the warfarin group (HR 0.52, 95% CI 0.28-0.98). 37

Efficacy and safety data from Hokusai-VTE trial. 37 The primary efficacy outcome was the recurrence of VTE and the P value reflects the noninferiority analysis. The primary safety outcome measured was the incidence of relevant bleeding and the P value reflects the superiority analysis. HR indicates hazard ratio; VTE, venous thromboembolism.
The rates of major bleeding were also significantly lower in the edoxaban group. The number of deaths and other serious adverse events were similar in the 2 groups. Thus, the Hokusai-VTE trial established that in patients with broad spectrum of VTE burden, edoxaban once daily was noninferior to warfarin with respect to efficacy and superior with respect to bleeding. 37 Also, edoxaban was significantly more effective that warfarin in patients with a large PE with right ventricular dysfunction.
Edoxaban in AF
The ENGAGE AF-TIMI 48 was a Phase III randomized, double-blind, double-dummy noninferiority trial that compared 2 doses of edoxaban to warfarin. The study population consisted of 21 105 patients with AF at moderate or high risk of stroke (CHADS2 score ≥ 2). The key exclusion criteria included creatinine clearance < 30 mL/min, high risk of bleeding, use of dual antiplatelet therapy, preplanned invasive procedures, acute coronary syndromes, and stroke within 30 days of randomization. 23 The patients were randomized to receive high-dose edoxaban (60 mg once daily), low dose edoxaban (30 mg once daily), and warfarin titrated to INR 2.0 to 3.0. Blinding of the INR was maintained as in the Hokusai trial. The primary efficacy end point of the trial was to determine whether edoxaban was noninferior to warfarin in preventing stroke or systemic embolism (SEE) and the primary safety end point was major bleeding. The noninferiority margin of 1.38 was derived to maintain 50% of the benefit of warfarin based on prior meta-analyses of placebo-controlled trials of warfarin in patients with AF. 23,38
Both edoxaban dose regimens were shown to be noninferior to warfarin in prevention of stroke or SEE. The rates of stroke or SEE were 1.50% with warfarin and 1.18% with high-dose edoxaban (HR 0.79, 97.5% CI [0.63-0.99]); P < .001 for noninferiority) and 1.61% with low-dose edoxaban (HR 1.07; 97.5% CI, 0.87-1.31; P = .005 for noninferiority; Figure 3).
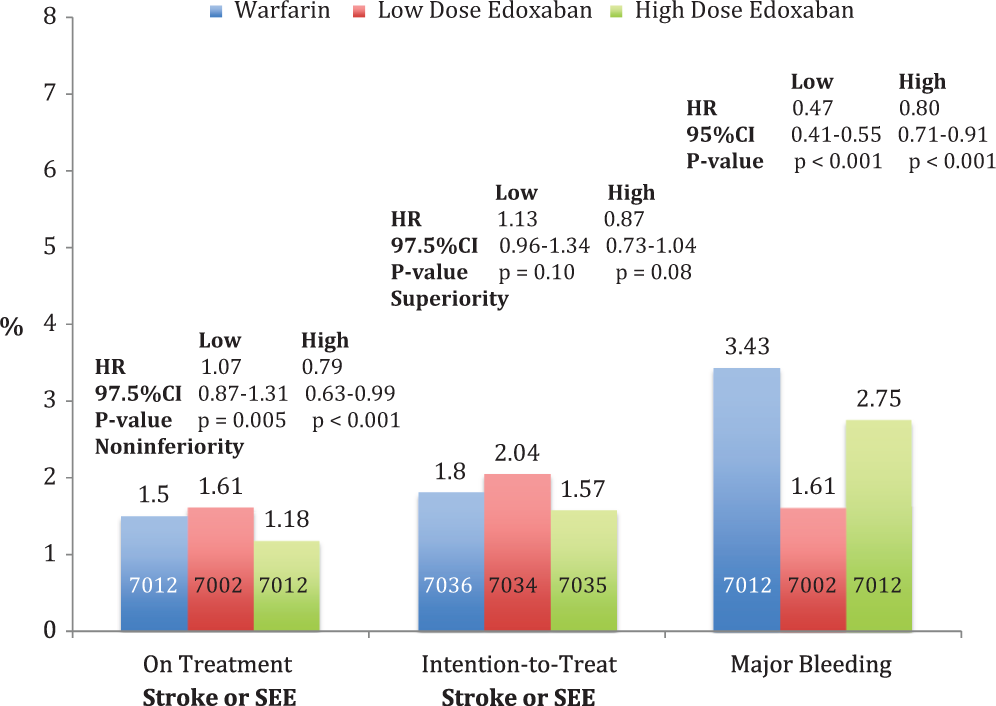
Efficacy and safety data from ENGAGE AF-TIMI 48 Trial. 38 The primary efficacy end point was stroke or systemic embolism and principal safety end point was major bleeding. The P value for the “on-treatment” group reflects the noninferiority analysis. The P value for the “intention-to-treat” group reflects the superiority analysis. ENGAGE AF TIMI-48 indicates Effective aNticoaGulation with factor xA next GEneration in Atrial Fibrillation trial: Thrombolysis In Myocardial Infarction study 48.
The rates of major bleeding were 3.43% with warfarin, 2.75% with high-dose edoxaban (HR 0.80, 95%CI 0.71-0.91; P < .001), and 1.61% (HR 0.47; 95%CI 0.41-0.55; P < .001) with low-dose edoxaban. The rates of cardiovascular death were 3.17%, 2.74% (HR 0.86, P = .013), and 2.71% (HR 0.85, P = .008). Thus, the ENGAGE-AF TIMI 48 trial established that in patients with AF, both high and low doses of edoxaban were noninferior to warfarin with respect to efficacy, and both doses were superior to warfarin with respect to bleeding and cardiovascular death. 38 The higher dose regimen of edoxaban appears to be the best option for the majority of patients, given the lower rates of bleeding and superior stroke prevention as compared to warfarin. However, in patients who are at very high risk of bleeding, the low-dose edoxaban regimen represents a reasonable safer option that further reduces the risk of bleeding and lowers mortality as compared to warfarin. Although it is difficult to compare results with TSOACs across trials, several features of ENGAGE AF-TIMI 48 trial may lead to preferential selection of edoxaban over other agents. Edoxaban is administered once daily and studies show improved compliance with once daily therapies compared to multiple daily dosing. 32,39 The availability of 2 dosing regimens, with dynamic dose adjustments during and after the trial, provides multiple dosing options spanning a 4-fold range that can be customized to patients according to their specific risk profile. An additional unique feature from the ENGAGE AF-TIMI 48 trial was the successful development of an edoxaban-specific transition plan that permits the safe and effective conversion between oral anticoagulants in patients with AF.
Conclusion
Target-specific oral anticoagulants have emerged as an attractive alternative to the standard therapy with warfarin in VTE prevention, acute VTE treatment, and prevention of stroke in patients with AF. Edoxaban is a once-daily novel FXa inhibitor with dose-dependent anti-FXa activity, no need for routine monitoring, and minimal drug–drug and food–drug interactions. Phase III trials established the superiority of edoxaban over enoxaparin in prevention of VTE. The Hokusai-VTE trial further demonstrated that edoxaban was at least as good as warfarin in preventing recurrent VTE and reduced bleeding in patients with acute symptomatic VTE. The ENGAGE-AF TIMI 48 trial showed that in patients with AF, both high and low doses of edoxaban were as good as warfarin in preventing stroke or SEE while reducing bleeding and cardiovascular death.
Future Perspective
Assay/Monitoring
Given the many advantages of TSOACs, we anticipate that they will be widely used in situations requiring long-term anticoagulation; however, several outstanding issues still remain. Two of the most important open questions are how can we measure the anticoagulant effect of TSOACs and how can these newer agents be reversed in case of urgent clinical situations?
One of the advantages of TSOACs is the lack of need for routine monitoring. However, in urgent clinical situations, such as emergent surgery or life-threatening bleeding, the ability to easily assay the anticoagulant effect of a TSOAC would be desirable. 40
Since the standard coagulation tests (PT and PTT) measure time to initiation of clot formation, they are not ideal to assess the anticoagulant effect of edoxaban. Edoxaban transiently increases the PT and activated partial thromboplastin time (aPTT) in a concentration-dependent manner; however, therapeutic doses of edoxaban result in PT measurements at the lower limit of discrimination. These coagulation tests should be elevated 1 to 4 hours after dosing and thus might be helpful to confirm compliance with edoxaban. However, we do not advise using the PT or aPTT for clinical decision making. Although there are no approved assays for monitoring the activity of edoxaban, 2 methods that have been studied include the prothrombinase-induced clotting time (PiCT) 41 and HepTest. 41 The PiCT is an assay that can be used to monitor anti-FXa and anti-FIIa activity. The assay involves adding FXa and Russell viper venom factor V activator (RVV-V), phospholipids, and calcium chloride to plasma. The RVV-V activates factor V, which causes downstream formation of prothrombin complexes, which convert prothrombin to thrombin. The HepTest is another clotting time test that combines plasma and FXa and quantifies the time it takes for clot formation. 41
Reversal Agents
There are currently no approved reversal agents for TSOACs. In patients with major bleeding, general measures such as cessation of all antithrombotic drugs, tamponade of the bleeding site or surgery (if applicable), and transfusion of red blood cells should be initiated. 42 In addition to these supportive measures, more specific reversal strategies can be considered.
Prothrombin complex concentrate (PCC; PPSB-HT), recombinant activated factor VIIa (rFVIIa, NovoSeven), and an activated PCC (aPCC, Feiba) were shown to significantly reverse anticoagulant activity of edoxaban in vitro. 43 Prothrombin complex concentrate and aPCC are purified from human plasma. Prothrombin complex concentrate contains nonactivated vitamin K-dependent coagulation factors and heparin. Feiba contains factor VII, mainly in the activated state, and factors II, VII, IX, and X, mainly in a nonactivated state. In rat models, rFVIIa and Feiba were effective at significantly reversing the anticoagulant effects of edoxaban. Furthermore, plasma thrombin–antithrombin complex (TAT) levels may be a better indicator of anticoagulant activity than PT since TAT levels and bleeding time are negatively correlated. 43
One of the universal FXa reversal agents being studied is PRT4445 developed by Portola. 42 This novel recombinant protein binds to FXa inhibitors in the blood, preventing their interaction with FXa. Following promising preclinical and safety studies, the first phase II trials (NCT 01758432) 44 are expected to conclude in 2013.
Another universal reversal agent is PER977, a synthetic molecule developed by Perosphere 45 that directly binds TSOACs including edoxaban. In the rat-tail transection bleeding model, PER977 was shown to reverse the anticoagulant effect of TSOACs. Phase I safety and tolerability study is currently underway studying single dose of PER977 as monotherapy following a single dose of edoxaban (NCT01826266). 46
Future Landscape
Edoxaban is 1 of the 4 TSOACs (dabigatran, rivaroxaban, and apixaban) shown to be as effective and safer than the standard warfarin therapy in preventing and treating VTE and stroke prevention in patients with AF.
The TSOACs are similar in that there is no need to routinely monitor the anticoagulant effect, there are few drug interactions, and there are no food interactions. The differences exist among the TSOACs primarily in the availability of different doses, regimens, and dose-adjustment in patients with reduced renal clearance. Future research should focus on developing assays for measuring and reversing the anticoagulant effect of TSOACs since this may be important in cases of overdose, questions of compliance, and urgent surgical interventions. Also, assessment of the efficacy and safety in special populations such as patients with renal insufficiency, extreme body weights, and children should be undertaken. Further studies of TSOACs in other indications for anticoagulation (eg, acute coronary syndrome, percutaneous coronary intervention, and mechanical heart valve replacement) are underway and may expand the use of these agents in clinical practice.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Giugliano reports receiving consulting fees from Daiichi Sankyo, Janssen Pharmaceuticals, Merck, and Pfizer; lecture fees from Bristol-Myers Squibb, Daiichi Sankyoo, Merck, and Sanofi; and grant support through his institution from Daiichi Sankyoo, Merck, Johnson & Johnson, Sanofi, and AstraZeneca.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


