Abstract
Patients with renal impairment require dose adjustments for direct oral anticoagulants (DOACs), though there is uncertainty regarding their use in severe chronic kidney disease. Inappropriately dosed DOACs may increase risk of ischemic events when under-dosed, or risk of bleeding when over-dosed. The purpose of this study was to describe DOAC selection, dosing strategies, and associated clinical outcomes in patients with moderate to severe renal impairment at our institution. This was a single-center retrospective analysis of adult outpatients with moderate to severe renal impairment (estimated creatinine clearance <50 mL/min, including need for hemodialysis) who were prescribed a DOAC by a cardiologist between June 1, 2015 and December 1, 2018. Outcomes evaluated included the percentage of patients who received appropriate and inappropriate DOAC dosing, prescriber reasons for inappropriate DOAC dosing if documented, and incidence of thrombotic and bleeding events. A total of 207 patients were included. Overall, 61 (29.5%) patients received inappropriate dosing, with 43 (70.5%) being under-dosed and 18 (29.5%) being over-dosed as compared to FDA-labeled dosing recommendations for atrial fibrillation or venous thromboembolism (VTE). By a median follow-up duration of 20 months, stroke occurred in 6 (3.3%) patients receiving DOACs for atrial fibrillation, and VTE occurred in 1 (4.3%) patient receiving a DOAC for VTE. International Society on Thrombosis and Haemostasis major or clinically relevant nonmajor bleeding occurred in 25 (12.1%) patients. Direct oral anticoagulants were frequently prescribed at off-label doses in patients with moderate to severe renal impairment, with a tendency toward under-dosing.
Keywords
Introduction
Current guidelines recommend direct oral anticoagulants (DOACs), including apixaban, rivaroxaban, dabigatran, and edoxaban, over warfarin for stroke prevention in atrial fibrillation (AF) and treatment of venous thromboembolism (VTE) due to similar efficacy and improved safety. 1 –3 Recently, DOAC prescriptions have surpassed that of warfarin across a broad range of patients and indications. 4 –6 All 4 DOACs with FDA approval for treatment of AF and VTE have dose adjustments and/or contraindications based on renal function (Supplementary Table 1). 7 –10 Apixaban has additional dosing considerations for age and weight for its AF indication. As DOAC dosing is nuanced and different with each agent and indication, prescribers may not be as familiar with these agents as they are with warfarin, causing dosing errors to frequently occur in the general population. 11
Patients with renal impairment may be at heightened risk for dosing errors, which is of particular concern. 11,12 Over-dosing of DOACs in patients with renal impairment has been associated with worse safety and no benefit in efficacy, whereas under-dosing has been associated with decreased efficacy and no benefit in safety. 12
The purpose of this study was to describe DOAC agent selection, dosing strategies, and potential reasons for off-label dosing in patients with moderate to severe renal impairment at our institution. Associated outcomes including stroke, recurrent VTE, and bleeding events were also analyzed.
Methods
This was a single-center retrospective analysis conducted at a tertiary academic medical center. All adults with a new outpatient prescription for dabigatran, rivaroxaban, apixaban, or edoxaban within the Division of Cardiovascular Medicine between June 1, 2015 and December 1, 2018 were considered for inclusion. Moderate to severe renal impairment was defined as an estimated creatinine clearance (eCrCl) <50 mL/min, including need for hemodialysis. Due to the study’s retrospective design, patients with prior DOAC use were excluded to capture the precise point of clinical decision-making and the prescriber’s rationale for dose selection. Patients were also excluded if they had an eCrCl ≥50 mL/min, or if a baseline serum creatinine within 3 months prior to initiation was not available to calculate eCrCl. This study protocol was approved by our health system’s Institutional Review Board.
Patients were identified from computer-generated reports of outpatient medication orders. Manual chart review of the electronic health record (EHR) was performed to collect patient demographics, medical conditions, and baseline labs. Serum creatinine within 3 months of DOAC prescription entry was used to calculate the eCrCl using the Cockroft-Gault equation. Actual body weight was used when calculating eCrCl if a patient weighed less than 120% of their ideal body weight, and adjusted body weight was used if the patient weighed greater than 120% of their ideal body weight.
Appropriate DOAC dosing was defined as dosing consistent with FDA-labeled dosing for treatment of AF and VTE as of December 2018. For patients prescribed apixaban for AF, serum creatinine and meeting additional criteria for low body weight and older age, rather than eCrCl, was used to define appropriate dosing. 9 For non-FDA-labeled indications, appropriate dosing was defined as consistent with the FDA-labeled dosing for the most similar indication (i.e. left ventricular thrombus dosing was adjudicated for dosing appropriateness as consistent with dosing for stroke prevention in atrial fibrillation). Inappropriate DOAC dosing was defined as either under-dosing (no renal indication for dose reduction but received reduced dose) or over-dosing (renal indication for dose reduction but received standard dose). 12
Endpoints of this study included the percentage of patients who received appropriate and inappropriate DOAC dosing, prescriber reasons for inappropriate DOAC dosing if documented, and incidence of thrombotic and bleeding events. Bleeding events were adjudicated as major, clinically relevant non-major, and minor bleeding using the International Society on Thrombosis and Haemostasis (ISTH) definitions. 13 Incidence of DOAC dose changes or discontinuations were collected along with the corresponding reasons determined by chart review. Descriptive statistics including mean ± standard deviation or median (IQR) were used to analyze data.
Results
A total of 3,537 patients were evaluated for inclusion. After exclusion, a total of 207 patients were included in the analysis (Figure 1). Overall, 61 (29.5%) patients received inappropriate dosing, with 43 (70.5%) being under-dosed and 18 (29.5%) being over-dosed.
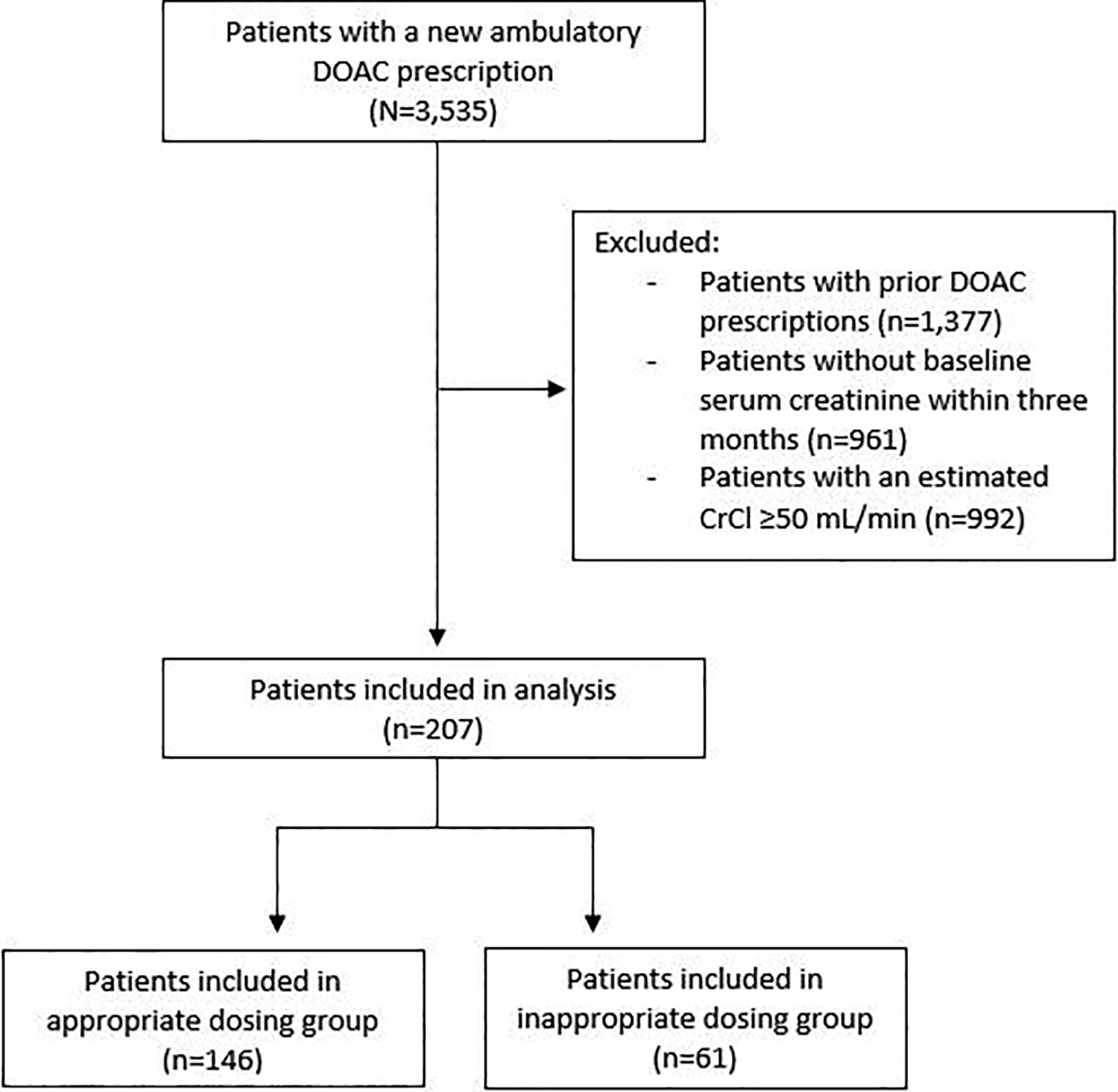
Patient inclusion.
Baseline characteristics are described in Table 1. The median age was 81 years (75-87). Most patients were prescribed a DOAC for stroke prevention in AF. The median serum creatinine and eCrCl were 1.25 mg/dL (1.05-1.55) and was 39.5 mL/min (33.0-43.2) in the appropriate dosing group and 1.23 mg/dL (1.08-1.53) and 40.3 mL/min (34.2-45.6) in the inappropriate dosing group. Only 3 patients had end-stage renal disease dependent on hemodialysis. Besides a greater percentage of overweight patients in the inappropriate dosing group compared to the appropriate dosing group, there were no statistically significant differences in baseline characteristics. There was a trend toward greater aspirin use at baseline in the inappropriate dosing group. Only 3 patients were on triple antithrombotic therapy, 2 in the appropriate dosing group and 1 in the inappropriate dosing group.
Baseline Characteristics.

BMI, body mass index; TIA, transient ischemic attack; VTE, venous thromboembolism; VKA, vitamin K antagonist; SCr, serum creatinine; CrCl, creatinine clearance.
* Values signify means ± SD.
† Values signify median (IQR).
‡ Five patients had dual indication so total percentage >100%.
§”Other” indications for anticoagulation included left ventricular (LV) non-compaction, LV thrombus, stroke of unknown etiology, and renal infarct.
|Major interacting medications included amiodarone, diltiazem, ranolazine, and fluconazole. None required dose adjustment per package insert recommendations.
Creatinine clearance was estimated using the Cockroft-Gault equation. Actual body weight was used if a patient weighed less than 120% of their ideal body weight, and adjusted body weight was used if the patient weighed greater than 120% of their ideal body weight.
Direct oral anticoagulant choice and dosing appropriateness are described in Table 2. Most patients were treated with apixaban or rivaroxaban, with very few patients receiving dabigatran or edoxaban. Prescriber reasons for under-dosing DOAC therapy are listed in Table 3. Rationale for under-dosing was not clearly documented in a third of patients. A considerable number of under-dosed patients had more than 1 reason documented in their medical record. The most commonly documented reason was physician-described renal dysfunction, although these patients did not meet the manufacturer-defined eCrCl cutoff for renal dose adjustment. The majority of under-dosing was in patients with atrial fibrillation and an eCrCl above—or serum creatinine below, for apixaban—the FDA-labeled criteria. There were only 2 patients being treated for the venous thromboembolism indication who were under-dosed: 1 patient who was indicated for a reduced apixaban dose for their atrial fibrillation but who had not completed 6 months of full-dose anticoagulation for their pulmonary embolism, and 1 patient where the prescriber intentionally opted for an off-label reduced dose, citing severe renal dysfunction bordering on the need for dialysis, frailty, and unknown acuity versus chronicity of the deep vein thrombosis.
Medication and Dosing Regimen.

Prescriber Reasons for Under-Dosing Direct Oral Anticoagulant Therapy.

* Fourteen patients had more than 1 reason documented in the medical record making total percentage >100%.
In the 9% of patients who were over-dosed, rationale for over-dosing was not clearly discernible from the medical chart. As the majority of the over-dosed patients were prescribed rivaroxaban for atrial fibrillation, it is possible that providers were unaware that a reduced dose was indicated, given rivaroxaban’s less stringent criteria compared to dabigatran’s (where renal dose adjustment starts at CrCl of 30 mL/min) or apixaban’s (where patients must meet an a second criteria in addition to impaired renal function) (Supplementary Table 1).
Clinical thrombotic and bleeding events are described in Table 4. Within this limited study population, patients with inappropriate dosing did not seem to experience an excess of adverse thrombotic or bleeding events. The median follow-up duration was 20 months (12.8-35.1). A quarter of patients in each dosing group discontinued DOAC therapy within the follow-up period for a variety of reasons (Table 4).
Treatment Outcomes by Medication Dosing Strategy.

Stroke was assessed as the efficacy outcome for patients prescribed DOACs for AF, and recurrent VTE was assessed as the efficacy outcome for patients prescribed DOACs for VTE. Bleeding was assessed as a safety outcome in all indications.
* Patients with an “other” indication for anticoagulation were not included in efficacy outcomes.
† Values signify median (IQR).
‡“Other adverse events” included nausea, diarrhea, lightheadedness, rash, pruritis, and dyspepsia.
Discussion
In this retrospective study assessing dosing strategies of DOACs in patients with moderate to severe renal impairment, we found that over a quarter of patients received initial doses that were inappropriate based on their baseline kidney function. Most of the inappropriate dosing was under-dosing, where the patient did not have a renal indication for dose reduction but received a reduced dose.
Overall, prescribing patterns at our institution revealed a heavy preference for apixaban in patients with renal impairment, comprising three-quarters of DOAC prescriptions. This trend may be due to apixaban being the least renally cleared DOAC, its retained safety in elderly patients, and its administration without consideration for meals. 14 Of note, the majority of DOAC under-dosing occurred in patients who were prescribed apixaban for AF, where the criteria for renal dose adjustment is more complex.
It is important to note that our study population is unique compared to those of randomized clinical trials or studies of DOACs in chronic kidney disease. The median age of patients in our study was 81 years, and most patients had a serum creatinine less than 1.5 mg/dL despite having a reduced eCrCl. Whereas elderly age generally reduces creatinine clearance estimation, there may also be a component of CrCl overestimation in the setting of lower muscle mass and frailty. A fraction of elderly patients was intentionally under-dosed due to age or age-related risk factors such as increased bleeding risk, frailty, or fall risk. This pattern toward under-dosing in elderly patients has been documented in another recent cohort. 15
Another important distinction is that for overweight patients weighing >120% of their ideal body weight, we used adjusted body weight in the Cockroft-Gault equation to estimate CrCl. The randomized clinical trials assessing efficacy and safety of the DOACs used actual body weight for CrCl estimation. 16 –19 The guidance to use actual body weight has not been readily available on DOAC labeling, although it was recently added to prescribing information for rivaroxaban. 8 In routine clinical practice, adjusted body weight is commonly chosen in overweight patients as CrCl may be overestimated when actual body weight is used. Thus, we included patients with this reduced CrCl using adjusted body weight to more accurately reflect real world practice. Additionally, there are several alternative methods to Cockroft-Gault CrCl for describing renal function, such as glomerular filtration rate (GFR) which is employed in chronic kidney disease staging. 20 In our study, we used Cockroft-Gault CrCl as opposed to GFR as this was the methodology chosen in the pivotal DOAC clinical trials. 16 –19
Our thrombotic and bleeding event rates were similar to those observed in the pivotal DOAC clinical trials, although our study was not powered to compare incidence rates between dosing groups and was descriptive in nature. 16 –19 Of note, 2 patients who experienced ischemic stroke (1 in each dosing group) were not fully anticoagulated at the time of the event; 1 patient had been holding apixaban for 1 week prior to a surgery and the other was only taking dabigatran once daily instead of twice daily as prescribed.
In a previous study done at our institution, authors found that of the 224 patients who received dose-reduced DOAC therapy, only 43.4% of patients fit criteria for a dose adjustment according to manufacturer recommendations. 11 Many patients in this study had renal dysfunction, drug-drug interactions, and/or a past medical history of bleeding events, although 15% of patients had no apparent clinical rationale from chart review to explain a dose reduction. Several other studies have highlighted the prevalence of dosing errors, usually in the direction of lower than recommended dosing strategies. 15,21,22
Our study has several limitations. First, due to the retrospective design, there were no prespecified selection criteria for DOAC initiation, and dosing strategies reflect individual provider discretion. Reasons for inappropriate dosing and incidence of clinical events were contingent upon accurate EHR documentation. We assumed that patients were adherent to their DOAC unless otherwise stated. Second, due to the small sample size, this study was not powered to detect differences in clinical thrombotic and bleeding outcomes. Finally, the end of the study period was set at December 2018 to ensure at least 1 year of follow-up in assessing thrombotic and bleeding events. Several studies which support the safety of DOACs in severe renal impairment and hemodialysis were published around this time. 23 –25 Additionally, for patients with ESRD, defined as CrCl <15 mL/min or on dialysis, national guidelines on atrial fibrillation were recently updated to add apixaban as an alternative to warfarin for oral anticoagulation. 1 Thus, DOAC prescribing in patients with moderate to severe renal impairment including hemodialysis has likely seen an uptick in more recent months.
To maximize the duration and quality of patient follow-up in our study, we included prescriptions written by outpatient cardiologists, who do not benefit from prospective medication order review by pharmacists. Some prescribers may not be well-versed in FDA-labeled CrCl cutoffs for DOAC dose adjustments based on indication, or they may intentionally under-dose patients with additional risk factors for bleeding.
Our study highlights the need for improved prescriber education and/or computerized decision support with DOAC prescribing, as incorrect dosing in patients with renal impairment has been associated with worse clinical outcomes. 12 Select patient characteristics and risk enhancers may help identify patient populations that either require up-front dose adjustment or close supervision (i.e., enrollment into a long-term anticoagulation clinic) (Figure 2). Implementation of pharmacist-managed anticoagulation clinics has been shown to identify and resolve dosing errors and offer additional benefits such as medication access, patient counseling, adverse effect monitoring and peri-procedural planning. 26 –28

Considerations for patient enrollment into anticoagulation clinics for direct oral anticoagulant supervision.
Conclusion
Although our study was done at a large academic medical center with expert physician staff, we observed frequent DOAC prescribing at off-label doses, with a tendency toward under-dosing and often for unknown reasons. Apixaban was the most commonly prescribed DOAC in patients with moderate to severe renal impairment. Implementation of computerized decision support surrounding DOAC prescribing may help optimize patient safety.
Supplemental Material
Supplemental Material, sj-docx-1-cat-10.1177_1076029620987900 - Evaluation of Direct Oral Anticoagulant Prescribing in Patients With Moderate to Severe Renal Impairment
Supplemental Material, sj-docx-1-cat-10.1177_1076029620987900 for Evaluation of Direct Oral Anticoagulant Prescribing in Patients With Moderate to Severe Renal Impairment by Clara Ting, Megan Rhoten, Jillian Dempsey, Hunter Nichols, John Fanikos and Christian T. Ruff in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
All authors contributed to the conception and design of the study. C. Ting collected the data. All authors analyzed the results. C. Ting wrote the manuscript. M. Rhoten, J. Dempsey, H. Nichols, J. Fanikos, and C. Ruff critically revised the manuscript. All authors approved the final version of the manuscript for publication. Ethical approval to report this study was obtained from Brigham and Women’s Hospital (Protocol # 2019P002287). Informed consent for patient information to be published in this article was not obtained because a waiver of consent was approved by the Brigham and Women’s Hospital Institutional Review Board.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John Fanikos reports grants and personal fees from Boehringer-Ingelheim, personal fees from Portola, grants from Astra-Zeneca, personal fees from Ipsen, outside the submitted work. Christian Ruff reports grants and personal fees from Anthos, grants and personal fees from Boehringer Ingelheim, grants and personal fees from Daiichi Sankyo, grants from AstraZeneca, grants from National Institutes of Health, personal fees from Bayer, personal fees from Bristol Myers Squibb, personal fees from Janssen, personal fees from Pfizer, personal fees from Portola, outside the submitted work; he is also a member of the TIMI Study Group which has received institutional research grant support through Brigham and Women’s Hospital from: Abbott, Amgen, Anthos Therapeutics, Aralez, AstraZeneca, Bayer HealthCare Pharmaceuticals, Inc., Daiichi-Sankyo, Eisai, Intarcia, MedImmune, Merck, Novartis, Pfizer, Quark Pharmaceuticals, Regeneron Pharmaceuticals, Inc., Roche, Siemens Healthcare Diagnostics, Inc., Takeda, The Medicines Company, Zora Biosciences. The rest of the authors have nothing to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
