Abstract
The aim of our study was to assess hospital budget implications of substituting dabigatran for warfarin in patients enrolled in a large anticoagulation service. The study population was identified using criteria from randomized controlled trials of dabigatran. We obtained labor costs ($483 per patient) from the hospital’s anticoagulation service budget, laboratory costs of international normalized ratio (INR) tests ($267 per patient), and wholesale costs of warfarin 5 mg tablets ($31 per patient) and dabigatran 150 mg capsules ($2464 per patient). A total of 1774 (93.5%) of 1898 patients were eligible to substitute dabigatran for warfarin. The annual projected hospital expense for anticoagulation with dabigatran was $4 371 136, attributable to drug cost alone. The annual projected cost of warfarin management was $1 385 494. This was comprised of $856 842 for labor, $473 658 for INR testing, and $54 994 for the drug cost of warfarin. Substitution will result in increased expense due to drug cost.
Anticoagulation therapy with warfarin is hampered by a narrow therapeutic index, dosing that requires adjustment according to international normalized ratio (INR) testing, and multiple interactions with drugs and food. Novel oral anticoagulants offer several improvements over warfarin.1–3 Dabigatran is an oral direct thrombin inhibitor administered in fixed doses without the need for laboratory coagulation monitoring. Dabigatran has the promise of simplifying anticoagulation management for stroke prevention in atrial fibrillation and for treating acute venous thromboembolism.4–6 In patients with atrial fibrillation, dabigatran decreased the rate of stroke and systemic embolism by one third when compared with warfarin.4,7 Major bleeding did not differ between the agents, but dabigatran reduced the frequency of intracranial hemorrhage by 60%. 4 For venous thromboembolism, a fixed dose of dabigatran was as effective as warfarin in preventing recurrent events and decreased any bleeding episodes by 29%. 5 While the cost of warfarin is low, the inconvenience and costs associated with labor and laboratory monitoring are problematic. 8 The aim of our study was to assess the hospital budget implications of substituting dabigatran for warfarin in patients with atrial fibrillation and venous thromboembolism at a single, large anticoagulation service.
Methods
The study population was derived from inclusion and exclusion criteria of the RE-LY and RE-COVER trials.4,5 Patients were considered eligible if they were receiving warfarin therapy for stroke prevention for atrial fibrillation or for the treatment of acute venous thromboembolism. Patients were excluded if they had a stroke or transient ischemic attack within the past 6 months, an estimated glomerular filtration rate ≤30 mL/min, or aspartate aminotransferase and alanine aminotransferase that exceeded 2 times the upper limit of normal. To evaluate the hospital budget implications of the potential switch, we obtained annual costs for labor ($483 per patient) from the 2010 fiscal year anticoagulation service budget, INR blood tests ($267 per patient) from the hospital laboratory, and wholesale acquisition of warfarin 5 mg daily ($31 per patient) and dabigatran 150 mg capsules twice daily ($2464 per patient) through our pharmacy.
Results
The anticoagulation service managed 2493 patients, of whom 1329 (53%) were treated for atrial fibrillation and 569 (23%) for venous thromboembolism. We found that 1256 (94.5%) of 1329 patients with atrial fibrillation and 518 (91%) of 569 patients with venous thromboembolism were eligible to substitute dabigatran for warfarin (Figure 1 ). Hence, 1774 (93.5%) of 1898 patients with atrial fibrillation or venous thromboembolism were eligible for dabigatran substitution. Among the 1898 patients with atrial fibrillation or venous thromboembolism, reasons for dabigatran ineligibility were renal dysfunction (n = 89, 4.7%), liver dysfunction (n = 24, 1.3%), and recent stroke or transient ischemic attack (n = 11, 0.6%).
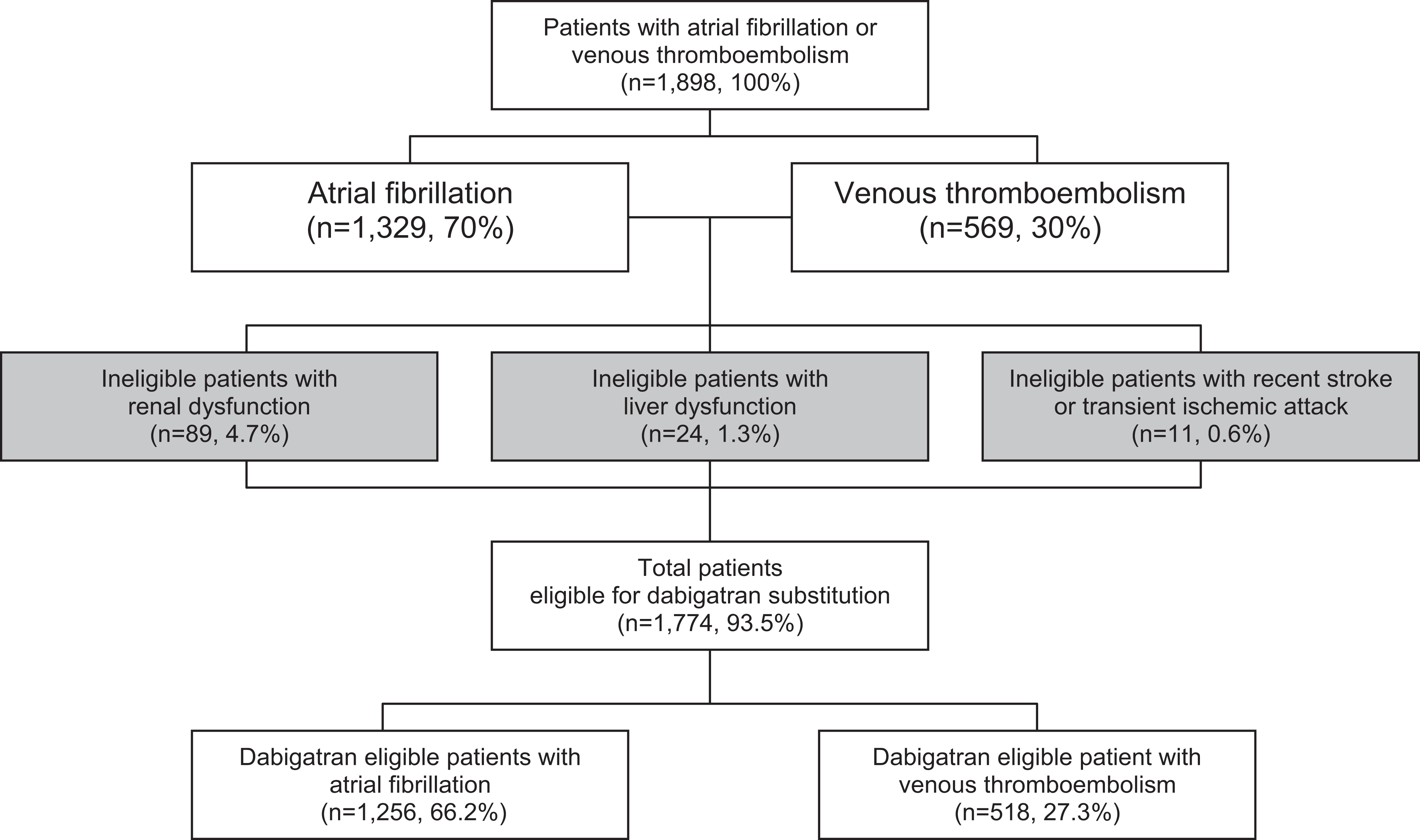
Anticoagulation service patient population. Of 1898 patients with atrial fibrillation or venous thromboembolism, 124 were excluded for renal dysfunction, liver dysfunction, or recent stroke.
For 1774 dabigatran-eligible atrial fibrillation and venous thromboembolism patients, we estimated the annual cost of anticoagulation management to be $4 371 136 for dabigatran, compared with $1 385 494 for warfarin, yielding a difference of $2 985 642. The annual drug cost for dabigatran was $4 371 136, compared with $54 994 for warfarin (Figure 2 ). The laboratory and labor costs for dabigatran were $0 since these costs were eliminated with dabigatran use. The annual laboratory cost for INR monitoring for the 1774 dabigatran-eligible patients who had been prescribed warfarin was $473 658, and the labor cost was $856 842.
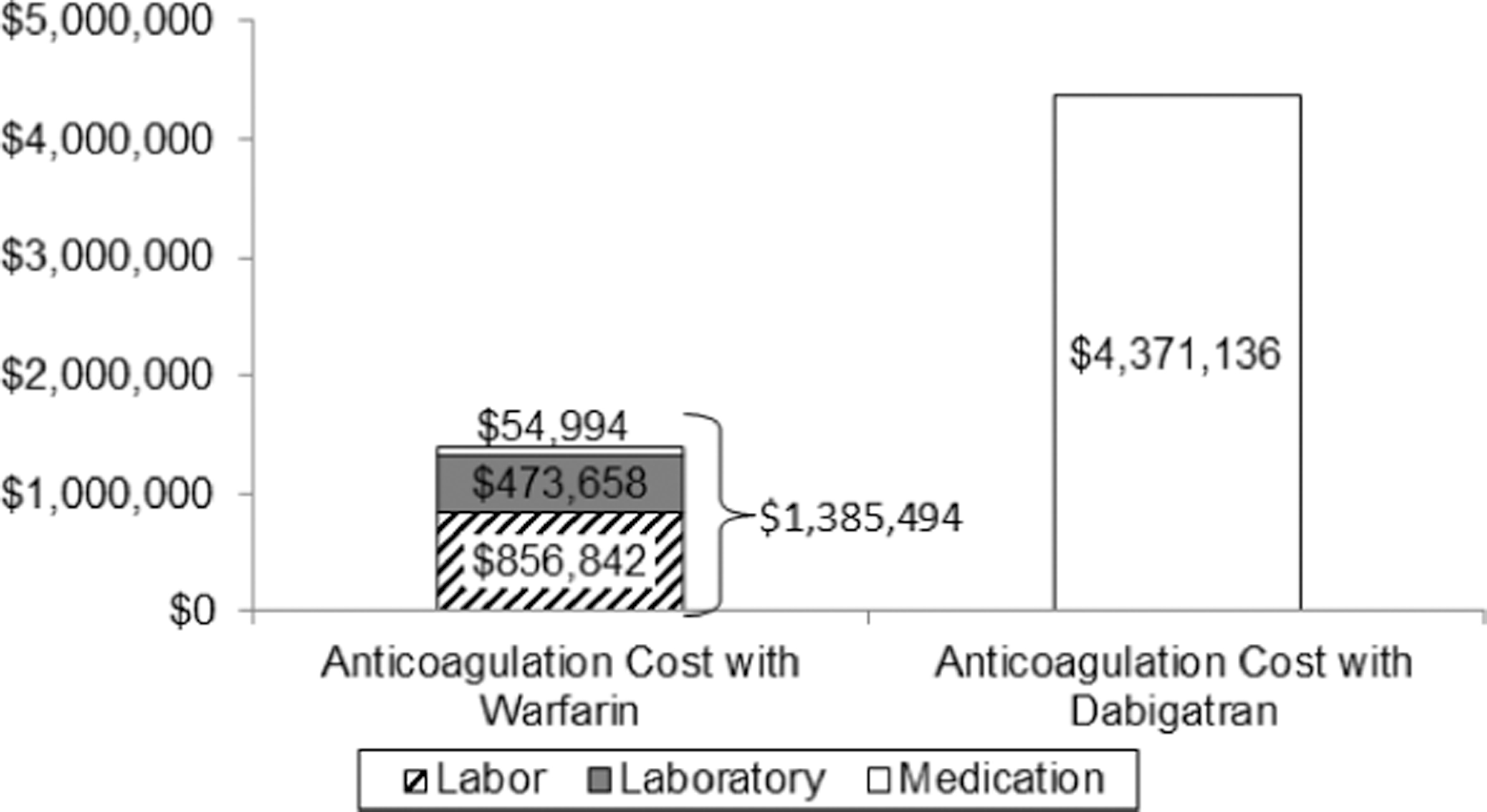
Annual costs of anticoagulation management. Currently, the drug cost and monitoring of warfarin for 1774 patients is $1 385 494. If all the 1774 patients who qualify for substitution switched to dabigatran, the cost of anticoagulation would increase to $4 371 136.
Discussion
We found that the majority of patients with atrial fibrillation and venous thromboembolism will be eligible for substitution of dabigatran for warfarin. Substitution of dabigatran for warfarin in a large anticoagulation service for patients with atrial fibrillation and venous thromboembolism will increase hospital expenses due to higher drug purchase cost. This increase in medication cost far exceeds the budgetary savings from eliminating all costs for labor and laboratory coagulation monitoring. Consequently, clinicians and patients can expect imposition of external cost-control measures that may include a preferred formulary choice, prescription preauthorization, increasing patient copayments, or an initial trial of generic warfarin.
A recent cost-effectiveness analysis of 10 000 patients with atrial fibrillation found that dabigatran 150 mg twice daily yields an additional 0.56 quality-adjusted life years compared with warfarin. 9 The incremental cost effectiveness ratio was $45 372 per quality-adjusted life year with dabigatran when compared with warfarin. This satisfies the threshold of $50 000 per quality-adjusted life year that most policy makers use as a reasonable measure of the value of an intervention. 10
Patients with atrial fibrillation who are well managed on warfarin with a high percentage of time in therapeutic range, >72%, may not derive incremental stroke prevention benefit from dabigatran. 7 The mean time in therapeutic range for the patients who received warfarin in the RE-LY trial was 64%. 4 For most patients receiving warfarin therapy, dabigatran is likely to prevent more strokes than warfarin.
Our analysis did not consider the costs associated with adverse outcomes from anticoagulation, including the costs of stroke or bleeding, postthrombotic syndrome, and long-term care for debilitation from strokes.11–13 The incremental dabigatran expense for atrial fibrillation patients will eventually be offset by decreasing long-term costs of caring for debilitated stroke patients and eliminating INR monitoring. 14 The average cost of an acute stroke to the community has been estimated at $19 018. 15 The lifetime cost per patient of a first stroke has been estimated at $103 576 averaged across all stroke subtypes. 16 In addition to decreasing stroke rates, dabigatran also decreases bleeding, which is reported to cost $14 975 per event. 17 We did not consider patient-associated costs, which include travel, parking, time, and inconvenience. However, quality of life issues and the “hassle factor” of warfarin for patients may not provide sufficient reasons for hospital administrators and insurers to approve the use of dabigatran in lieu of warfarin.
We present results from a single-center anticoagulation service with experience in the management of complex anticoagulation patients. Because the population of eligible patients in our service, and therefore, the cost burden, may differ from others, our results may not be generalizable to all anticoagulation services. In addition, we did not address the budget implications of using a 50% dose reduction of dabigatran in patients with an estimated glomerular filtration rate of 15 to 30 mL/min. 18
Our report does represent the “real-world” setting of a large anticoagulation service, and actual costs were used to analyze labor, laboratory, and drug costs. Study criteria from the RE-COVER and RE-LY trials were used to determine eligibility and consequently, our population is representative of the patients who will actually be considered for dabigatran substitution. Our analysis suggests that substituting dabigatran for warfarin will increase pharmacy budget costs associated with anticoagulation due to the increased drug costs.
Footnotes
This article was presented at ACC.11 60th Annual Scientific Session & Expo, April 2-5, 2011; New Orleans, Louisiana.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Piazza is supported by a Research Development Award (K12 HL083786) from the National Heart, Lung, and Blood Institute (NHLBI).
