Abstract
To investigate the possible risk factors for proximal pulmonary embolism (PE) in patients with lower extremity deep vein thrombosis (LEDVT). A single-center, retrospective cohort study was conducted. Consecutive patients diagnosed with unilateral LEDVT and confirmed PE by computed tomography (CT) pulmonary angiography and indirect CT venography between September 2015 and January 2022 were included. Patient demographics, presentation of LEDVT, laboratory examination, thrombus characteristics, comorbidities and risk factors for LEDVT, and imaging findings were analyzed to determine the univariable and multivariable associations. The associations between thrombus location, stenosis, and proximal PE were assessed using a stratified analysis. A total of 115 eligible patients (60.08 ± 15.18 years, 59.1% male) were included. Of these patients, 40 (34.8%) were in the proximal PE group (proximal thrombus involved main or lobar pulmonary artery), and 75 (65.2%) were in the distal PE group (without main and labor pulmonary artery involvement). The univariable analysis showed that onset time of LEDVT symptoms ≤ 7 days (P = .041), inferior vena cava (IVC) involvement (P = .035), and severe iliac vein stenosis (IVS) (stenosis ratio > 61.3%) (P = .010) were associated with the occurrence of proximal PE. Further multivariant analysis showed that IVC involvement was associated with an increased odds of proximal PE (OR: 4.45, 95% CI: 1.083 − 18.248, P = .038), whereas severe IVS (stenosis ratio > 61.3%) was associated with a decreased odds of proximal PE (OR: 0.294, 95% CI: 0.110-0.783, P = .014). Among patients with unilateral LEDVT, IVC involvement increased the risk of proximal PE; patients with severe IVS (stenosis ratio > 61.3%) had a lower risk of developing proximal PE compared with distal PE; and severe IVS seemed to be a protective factor for proximal PE.
Introduction
Venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and its complications, is a significant public health concern as well as an important source of morbidity and mortality.1,2 Pulmonary embolism (PE), which is known as one of the most serious manifestations along the disease spectrum of VTE and a potentially life-threatening condition,3,4 can generally be divided into 4 major disease processes, consisting of vein thrombosis, thrombus in transit or PE, acute PE, and pulmonary circulation reconstruction.2,3 As previously reported, the prevalence of isolated PE is lower, indicating that PE is less common; it most frequently arises from lower extremity DVT (LEDVT) and triggers 900,000 hospitalizations and 200,000 deaths each year.3,5 However, the true incidence of PE is uncertain and likely to be underestimated because many episodes are undetected and left untreated.
The pulmonary arterial vascular tree becomes progressively tapered from proximal to distal branches, 4 and PE locations are correspondingly classified as main, lobar, segmental, or subsegmental pulmonary arteries based on the position of the most proximal luminal filling defect.5,6 Of these, Torbicki et al 6 defined proximal PE as thrombosis that could be unequivocally visualized in the main or lobar pulmonary arteries. Of note, compared with distal PE, proximal PE may be more prone to be associated with increased right ventricular afterload and to be followed potentially life-threatening conditions,6,7 whereas patients with distal PE showed minimal symptoms or no symptoms or even were serendipitously detected among LEDVT patients. 8 Why were some individuals prone to a risk of proximal PE but others were not? It would be clinically useful to have measurable factors to predict which patient is at a higher risk.
We hypothesized that proximal PE may also be closely related to those with severe IVS based on the observation that inferior vena cava (IVC) thrombosis is less common in patients with left severe iliac vein stenosis (IVS). IVS commonly arises from May‒Thurner syndrome, in approximately 22% of the population, and IVS is an anatomic variant in which the right common iliac artery compresses the left common iliac vein against the lumbar vertebrae. 8 This single-center retrospective study was conducted in patients with both LEDVT and PE. The aim was to investigate the risk factors for PE patients with proximal pulmonary artery involvement compared with patients with distal pulmonary artery involvement, as well as to evaluate the relationship between IVC-involved thrombosis, IVS, and proximal PE in LEDVT patients.
Methods
Patients and Study Design
This was a retrospective cohort study involving consecutive patients older than 18 years old at a single academic center from September 06, 2015, to January 18, 2022. The data collection protocol and informed consent were both approved by our Institutional Review Board. The inclusion criteria were patients who simultaneously suffered from unilateral LEDVT and PE, had complete clinical data, and underwent combined computed tomography (CT) pulmonary angiography (CTPA) and indirect CT venography (CTV) within 48 h (h) after their admission. The exclusion criteria were bilateral LEDVT, previously indwelled IVC filter, unhealed previous LEDVT or incomplete data. The data were retrospectively derived from the medical database system and/or paper records to identify eligible patients.
The baseline demographics, PE-related clinical symptoms (including dyspnea, chest pain, cough, hemoptysis, syncope, tachycardia, hypoxia, and decreasing oxygen saturation), onset time (from onset of possible LEDVT symptoms to admission, hematological examination within the first 24 h, limb location of unilateral LEDVT, LEDVT segments (proximal LEDVT or isolated distal deep vein thrombosis (IDDVT)), IVS, comorbidities and thrombotic risk factors for LEDVT were analyzed.
Diagnosis of LEDVT and Measurement of IVS
The initial diagnosis of LEDVT was verified by Doppler ultrasonography and D-dimer value. A combined CTPA and CTV (128-slice dual-source CT, SOMATOM Definition Flash, Siemens, Germany) were objectively performed within 48 h after the initial diagnosis to confirm the distribution and extent of thrombus in pulmonary arteries and deep veins. Proximal pulmonary artery thrombus was defined as confirmation of an intraluminal filling defect within the main and lobar pulmonary arteries (proximal PE) on CTPA imaging. The diagnosis of PE was initially performed by radiologists via CT angiography and was ultimately confirmed by 2 independent reviewers (JK and BXZ). The 5-mm or 1-mm reconstructed contiguous axial images were used to determine the stenosis level of the iliac vein, and measurements of the iliac vein diameters were independently performed by the reviewer (XH), who was blinded to the PE location of each subject.
The appraisal phase of IVS was comprised of an assessment of any degree of compression along the iliocaval vein segments according to previous reports.9,10 The minimum diameter of the left IVS was measured at the point of maximum compression (if any), where the right common iliac artery crossed anterior to the left common iliac vein, whereas in the single case of right IVS stenosis, the minimum diameter of the right IVS was measured 10 mm below the IVC bifurcation. Since all patients had unilateral LEDVT, the patent contralateral iliac vein diameter was used as an internal control. 9 We calculated the level of IVS as follows: (1-D1/D2) × 100%, where D1 is the minimal diameter ipsilateral to the LEDVT, and D2 is the contralateral minimum diameter. The ratio ranged from 0% (diameter equal to or greater than the contralateral leg) to 100% (complete loss of lumen).
Results
Baseline Demographics and Characteristics of Proximal and Distal PE Patients with LEDVT
The demographics, presentation, hematological examination, thrombus characteristics, comorbidities, and risk factors for all patients are summarized in Table 1. A total of 316 potentially eligible patients with identified LEDVT as well as PE were collected, and 201 were subsequently excluded because of bilateral LEDVT (n = 71), indwelled IVC filter (n = 56), unhealed VTE (n = 24), or lack of complete data (n = 50). Of the remaining 115 included patients, based on whether proximal pulmonary artery thrombus was observed, 34.8% (40/115) of the patients were divided into the proximal PE group, and 65.2% (75/115) were divided into the distal PE group. The mean age of these included patients was 60.08 ± 15.18 years, and 59.1% (68/115) were male. The mean onset time of LEDVT symptoms at presentation was 9.64 ± 11.30 days, of which 67.8% (78/115) of the patients had an onset time ≤ 7 days. Hematological examination primarily focused on the D-dimer test, and the mean value was 9.80 ± 11.20 μg/ml. On the CTPA imaging findings, thrombus involvement in the main, lobar, segmental, and subsegmental pulmonary arteries was detected in 10.4% (12/115), 34.8% (40/115), 64.3% (74/115), and 100% (115/115) of these patients, respectively. In the CTV examination session, 60% (69/115) of these patients suffered from left-side LEDVT, and 87.0% (100/115) of them underwent proximal LEDVT. With regard to comorbidities and risk factors for LEDVT, the leading comorbidities of these patients were hypertension, accounting for 38.3% (44/115), and the major risk factors for LEDVT were immobilization, presenting in 28.7% (33/115) of these patients.
Demographics, Presentation, Hematological Examination, Lesion Characteristics, Comorbidities and Risk Factors for LEDVT in Proximal or Distal PE Patients.

Continuous data are presented as the means ± standard deviations; categorical data are given as the counts (percentage). PE, pulmonary embolism; LEDVT, lower extremity deep vein thrombosis; SD, standard deviation; IVC, inferior vena cava; IDDVT, isolated distal deep vein thrombosis; IVS, iliac vein stenosis; CAD, coronary artery disease; CVD, cerebral vascular disease; PAD, peripheral artery disease.
Predictive Factor of Severe IVS for Proximal PE
The mean IVS level in the included patients with proximal PE was lower than that in the patients with distal PE (40.81 ± 22.55% vs 50.32 ± 25.21%, P = .048). After univariable analysis, the predictive values of severe IVS level were analyzed by a receiver operating characteristic (ROC) curve to identify the optimal cutoff value. A stenosis ratio > 61.3% was discriminant (area under ROC curve = 0.625, 95% CI: 0.519-0.731, P < .05) for predicting proximal PE. This indicator had a sensitivity of 40.0% and a specificity of 85.0%. The positive and negative predictive values were 83.3% and 43.0%, respectively. Therefore, the unilateral LEDVT patients with severe IVS > 61.3% were less likely to develop proximal PE than the patients with IVS < 61.3%.
Relationship Between Proximal PE and Thrombus Distribution of LEDVT
Age and gender were not significantly different between the 2 groups (both P > .05). The mean onset time of LEDVT symptoms at presentation in the proximal PE patients was shorter than that in the distal PE patients (6.71 ± 6.98 vs 11.20 ± 12.80 days, P < .05). The incidence of PE symptoms in the patients with proximal PE was significantly higher than that in the patients with distal PE (57.5% vs 14.6%, P < .001). Of note, the patients with proximal PE experienced more life-threatening conditions (hypotension and circulatory arrest) (30.0% vs 2.6%, P < .001). The main PE locations in the proximal PE patients were the bilateral pulmonary arteries and on unilateral pulmonary arteries in the distal PE patients (62.5% vs 40%, P = .021). The proximal PE patients encountered similar proximal LEDVT and IDDVT incidences compared with those in the distal PE patients (both P > .05). In addition, the severe IVS (stenosis ratio > 61.3%) incidence was higher in the distal PE patients than in the proximal PE patients (41.3% vs 17.5%, P = .010), which was higher among patients with left-side LEDVT than in the patients with right-side LEDVT. For the comorbidities and risk factors for LEDVT, the differences were not significant (all P > .05).
Univariable associations with demographics, presentation, hematological examination, thrombus characteristics, comorbidities and risk factors for LEDVT were all analyzed between the 2 groups, which showed that compared to the patients with distal PE, the factors that predicted the probability of proximal PE were as follows: onset time of LEDVT symptoms at presentation ≤ 7 days (P = .041), IVC involvement (P = .035), and severe IVS (stenosis ratio > 61.3%) (P = .010). Then, multivariate logistic regression was performed, the results of which are visualized in Figure 1. It was shown that the onset time of LEDVT symptoms at presentation ≤ 7 days (OR: 2.31, 95% CI: 0.903–5.930, P = .080) was not significant. IVC thrombosis was associated with increased odds of proximal PE (OR: 4.45, 95% CI: 1.083−18.248, P = .038), and severe IVS (stenosis ratio > 61.3%) was associated with a decreased odds of proximal PE (OR: 0.294, 95% CI: 0.110-0.783, P = .014).
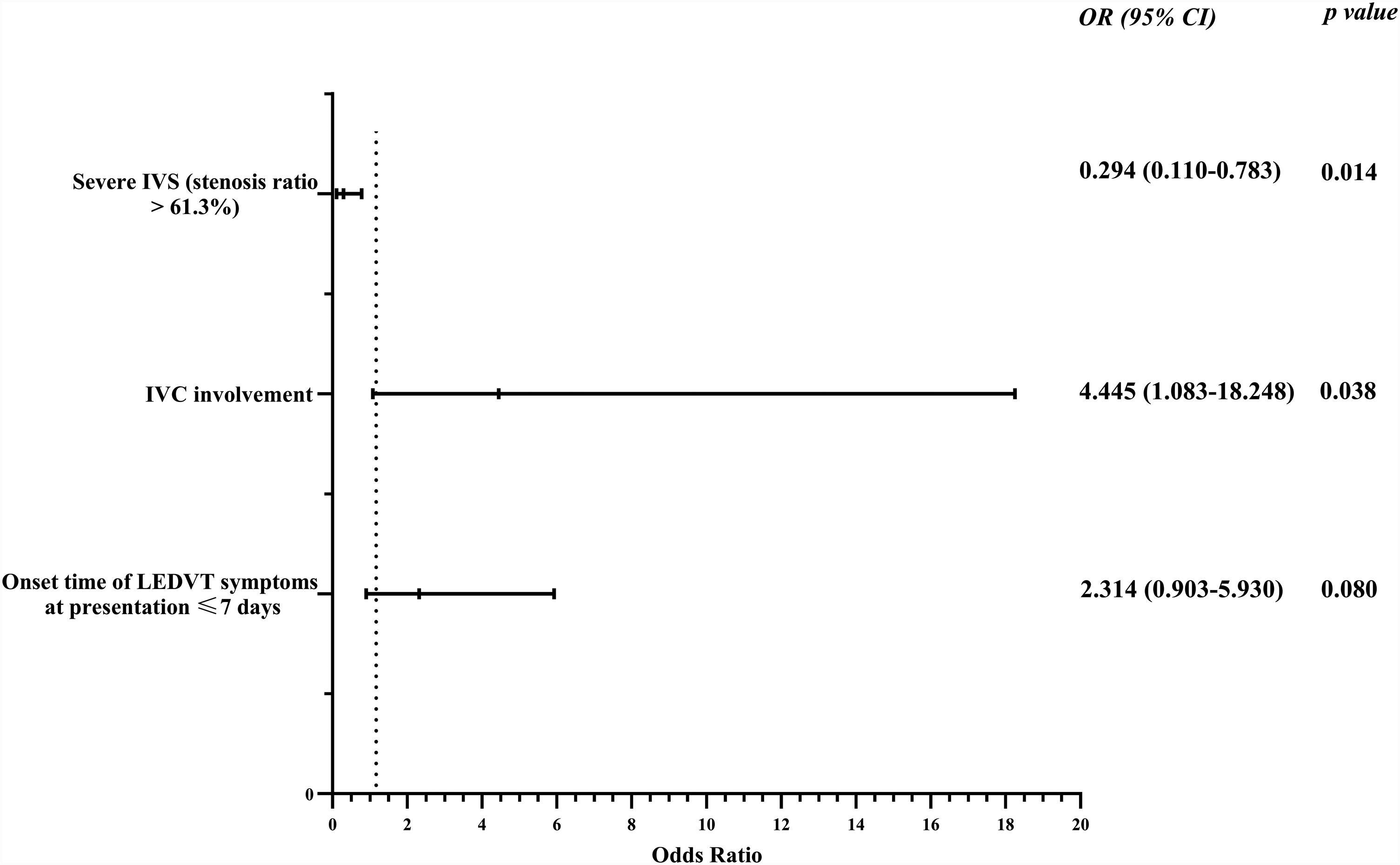
A forest plot was used to visualize the multivariate regression analysis of risk factors for proximal pe in LEDVT patients. It showed severe IVS (stenosis ratio > 61. 3%) was a protective risk factor, and IVC involvement was a risk factor for proximal PE. PE , pulmonary embolism; LEDVT, lower extremity deep vein thrombosis; IVS, iliac vein stenosis; IVC, inferior vena cava.
The subgroup analysis is summarized in Table 2. In the left-side LEDVT patients, the difference in the mean onset time of LEDVT symptoms ≤ 7 days between the proximal and distal PE patients was not significant (68.0% vs 65.9%, P = .859). Severe IVS (stenosis ratio > 61.3%) was more prevalent in the distal PE patients (43.2% vs 16.0%, P = .021). In right-side LEDVT patients, the mean time from the onset of symptoms consistent with LEDVT to hospital admission (≤ 7 days) was associated with increased odds of proximal PE than in the distal PE patients (100% vs 54.8%, P = .001). The incidence of severe IVS (stenosis ratio > 61.3%) was not significant between the 2 groups (P = .204).
The Characteristic of IVS in Subgroup Patients with Proximal or Distal PE.
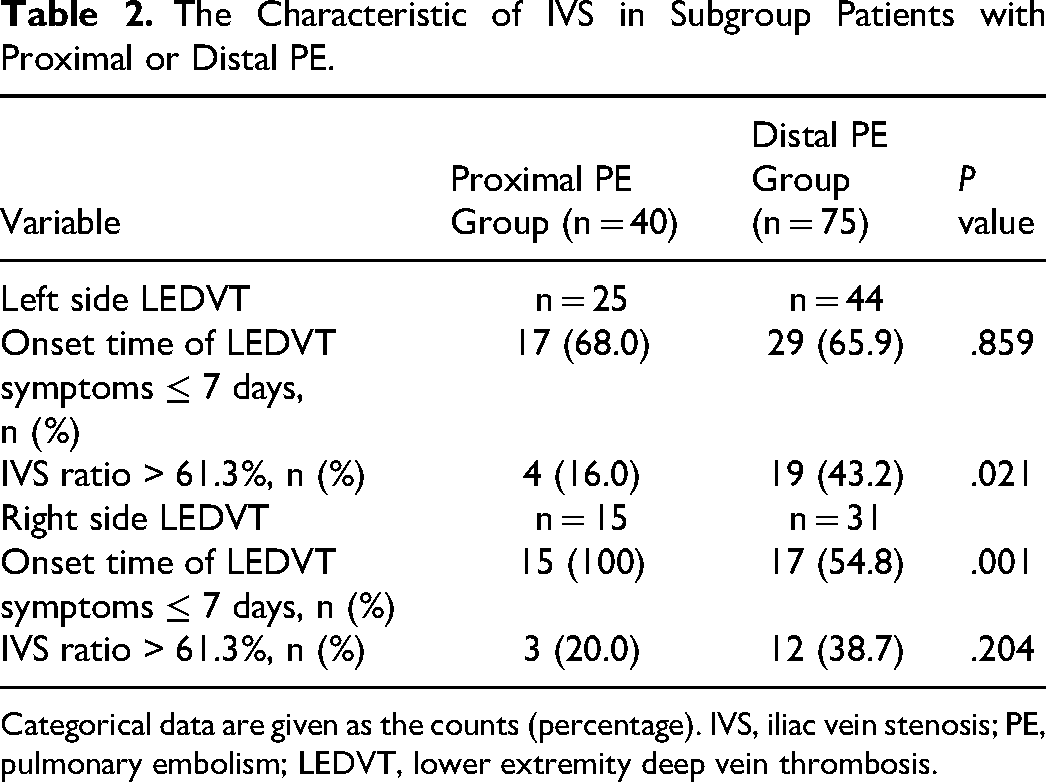
Categorical data are given as the counts (percentage). IVS, iliac vein stenosis; PE, pulmonary embolism; LEDVT, lower extremity deep vein thrombosis.
Discussion
PE has been taken as a sequela of LEDVT that detaches with thrombus centrally into the pulmonary circulation, and it is estimated that up to a quarter of patients with PE have suffered from sudden death. 9 However, these studies were mostly limited to PE that was not well stratified into proximal or distal PE.3,9 Intuitively, proximal PE is more likely to develop into a severe condition; however, the risk factors for its formation remain poorly understood or identified. Previously, many fundamental biological factors were identified in the thrombogenesis of LEDVT. Indeed, PE is considered to have a similar pathological and clinical spectrum as LEDVT.2,10 Of these, IVS was initially addressed as a major factor for the significant incidence of LEDVT. 8 Subsequently, Chan et al 9 found that this naturally anatomic compression of the common iliac vein played a significant role in reducing the risk of symptomatic PE. However, determining whether an IVS is present, what the degree or severity it is, and the nature of that stricture from this underlying condition is still a clinical dilemma. 11 Hence, carrying out this retrospective study among patients with proximal and distal PE and seeking the relationship between severe IVS and proximal PE may yield an important clinical implication, which has rushed into the forefront of clinicians. In this retrospective cohort study, based on demographics, presentation of LEDVT, hematological examination, thrombus characteristics, comorbidities, and risk factors for LEDVT, we found that onset time of LEDVT symptoms at presentation ≤ 7 days (OR: 2.31, 95% CI: 0.903−5.930, P = .080) did not demonstrate a significant relationship between proximal PE and distal PE. IVC-involved thrombosis was associated with increased odds of proximal PE (OR: 4.45, 95% CI: 1.083–18.248, P = .038), whereas severe IVS (stenosis ratio > 61.3%) was associated with a decreased odds of proximal PE (OR: 0.294, 95% CI: 0.110-0.783, P = .014).
In this study, patients afflicted with proximal PE were more likely to experience clinical symptoms than those with distal PE, which was consistent with a previous study concerning symptomatic PE. 11 Of note, 30% of these proximal PE patients had suffered from life-threatening conditions, which was significantly higher than that of patients with distal PE, indirectly implicating that more attention should be given to patients with proximal PE. Additionally, as shown in prior studies,6,9,12 the larger the thrombus burden the patients experienced, the more adverse clinical events they might suffer from. With the proliferation of smooth muscle and endothelial cells, 13 patients with unhealed proximal PE are more likely to develop chronic thromboembolic pulmonary hypertension (CTEPH). In our study, 34.8% of the included patients were found to have proximal PE, which means that a non-negligible number of patients would encounter a potential risk of prolonged CTEPH without appropriate treatment. Whether these patients would experience it remains an unanswered question, and further studies are required to elucidate this issue.
In this study, it was shown that gender was not significant between the proximal and distal PE patients, which was similar to the conclusion of previous studies that there was no significant difference in the incidence of symptomatic and silent PE between sexes.2,3,14 Additionally, the mean age of the patients with proximal PE was slightly older than that of the patients with distal PE but was not statistically significant. In addition, as the data showed in our cohort, we found that the left side was the predominant thrombus limb for proximal and distal PE, which may be related to the fact that a higher LEDVT incidence presented in the left side limb as well as LEDVT being a dominant factor for PE. 4 Proximal LEDVT was closely related to the occurrence of PE. 15 The same was true in our study as well, whereas the distributions of proximal LEDVT and IDDVT did not demonstrate a significant relationship between the proximal and distal PE patients. In the subgroup analysis, the onset time of LEDVT symptoms and severe IVS were considered to be associated with proximal and distal PE. For left-side LEDVT patients, the mean onset time of symptoms at presentation was not statistically significant, but the incidence of severe IVS was lower in the proximal patients, which suggested that severe IVS may be a protective factor for proximal PE in left-side LEDVT patients. In regard to right-side LEDVT, the incidence of severe IVS was similar, whereas the onset time of LEDVT symptoms in the proximal PE patients was shorter than that in the distal PE patients. It seemed that a short duration of onset time was associated with a greater risk of migration, which was consistent with the conclusion of previous reports.1,3,7
IVS occurs in approximately 33.0% of the general population and has been found to be a strong independent risk factor for LEDVT on the left side.14,15 The thrombus burden is strongly associated with the stenosis level of the iliac vein. 9 Intuitively, patients with IVS should have an increased incidence of proximal PE due to the large thrombus burden. In our study, however, the patients with massive proximal LEDVT and severe IVS presented with lower proximal PE. It is possible that although stenosis-induced flow stasis contributes to an increased incidence of LEDVT, the same stenosis may also determine whether embolization of LEDVT will occur and cause proximal PE. Unless the thrombus extends beyond the stenosis, this stricture may limit the embolic fragments to small, clinically insignificant pieces that occlude only small pulmonary arteries. In our study, thrombus extension beyond the stenosis into the IVC was a risk factor for the incidence of proximal PE, and LEDVT patients with IVC involvement were considered to have a 4.45-fold increased risk of proximal PE compared with those without IVC involvement. Moreover, the data showed that proximal PE was also associated with the degree or severity of IVS. However, the severe IVS level was subjectively partitioned into 50% or 70%,9,10 and the recommendation of severe IVS has not been elucidated. In this study, we found that a severe IVS level of 61.3% (sensitivity 40.0%, specificity 85.0%) was discriminative of proximal PE occurrence. Multivariate regression showed that a stenosis ratio > 61.3% was a protective factor for proximal PE in LEDVT patients, and patients with severe IVS had a lower odds of developing proximal PE, although more proximal LEDVTs were identified in these patients with severe IVS. 2
The present study has several limitations that merit discussion. First, this is a single-center retrospective cohort study, and biases were possibly introduced due to the inherent nature of our study. Second, prognostic classification, which is related to severity, is more important in the clinic. 16 However, the limited numbers of patients with massive PE in our center limited the subgroup assignments in our study. Third, 50 patients who were confirmed to undergo both PE and LEDVT were not included because of missing and incomplete data. Therefore, selection bias may be an introduction. Nevertheless, our finding of IVS as a protective factor was not different compared with a previous study concerning symptomatic PE. 9 We presume that the effect of the selection of only the patients with complete information may have not had a substantial impact on the overall results. Fourth, the sample sizes included in this study were relatively small for assessing the risk factors, and subsequently establishing a multivariate predictive model was not achieved. Finally, some patients underwent anticoagulation treatment prior to blood sample collection, which may have somewhat influenced the D-dimer results. Some thrombophilia conditions, including a lack of protein C and protein S, were not investigated. In the future, a study including more factors as well as excluded confounding factors to overcome these limitations can be designed for further research.
Conclusion
This study showed that IVC involvement was a risk factor for the occurrence of proximal PE, which was associated with increased odds of proximal PE. Severe IVS decreases the incidence of proximal PE in unilateral LEDVT patients compared with distal PE by limiting the migration of large embolic fragments from the lower extremity to the proximal pulmonary artery, and severe IVS seems to be a protective factor.
Footnotes
Authors’ Contributions
MFG contributed to this project development, manuscript writing/editing. YDS and ZLL contributed to data collection, manuscript associated editing. JK contributed to data analysis. BXZ and XH contributed to manuscript editing. JPG contributed to project development, manuscript editing. All authors contributed to the article and approved the submitted version.
Availability of Data and Materials
The datasets generated and analyzed during the current study are not publicly available, as the experimental data are related to other experiments that are progressing but are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (8187 1463) and Xinghuo Talent Program of Nanjing First Hospital.
Ethical Approval
The study protocol was reviewed and approved by the institutional review board (IRB) of the Nanjing First Hospital, Nanjing Medical University (Nanjing, China). In addition, the study was performed in accordance with the Declaration of Helsinki and the Ethical Guidelines for Clinical Studies.
