Abstract
Direct oral anticoagulants (DOACs) are becoming increasingly popular clinically, but their safety and effectiveness profile in patients with chronic thromboembolic pulmonary hypertension (CTEPH) is not well-established. Literature from the PubMed and EMBASE databases was systematically screened up to February 2024 to identify relevant studies on the use of DOACs in CTEPH patients. The bias risk of RCTs was assessed using the Cochrane Risk of Bias Tool 2.0. The quality of observational prospective cohorts was assessed using the Newcastle-Ottawa Scale tool. Data pooled from different studies were analyzed. Results from 4 studies were gathered, including 2 randomized controlled trials and 2 prospective cohorts, with a total of 2038 patients, of which 751 were on DOACs and 1287 were on vitamin K antagonists (VKAs). Similar rates of all-cause mortality (3.33% vs 3.33%, RD = −0.01%, 95% CI [−0.02%, 0.00%], P = .17), VTE recurrence (1.46% vs 2.12%, RD = −0.00%, 95% CI [−0.01%, 0.01%], P = .92) were observed. DOACs were associated with a nonsignificant reduction in bleeding events including major bleeding (2.22% vs 3.71%, RD = −0.01%, 95% CI [−0.04%, 0.01%], P = .30), any bleeding (5.33% vs 9.94%, RD = −0.03%, 95% CI [−0.07%, 0.01%], P = .10), and minor bleeding (4.17% vs 13.3%, RD = −0.06%, 95% CI [−0.23%, 0.10%], P = .45). Data pooled from existing perspective trials suggests the use of DOACs in CTEPH patients as an effective and safe alternative to VKAs.
Keywords
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) is a condition that arises as a result of a delayed complication following an acute pulmonary embolism. Acute pulmonary embolism occurs when a blood clot develops in the pulmonary arteries, leading to restricted blood flow and increased pressure in the lungs. In order to reduce the risk of recurrence of venous thromboembolism (VTE), which includes the formation of blood clots in the veins, current medical guidelines recommend the use of anticoagulation therapy in patients diagnosed with CTEPH..1,2
Vitamin K antagonists (VKAs) such as warfarin have been clinically proven to be effective in reducing the occurrence of blood clots in patients with CTEPH. However, the use of VKAs has certain drawbacks such as the need for regular dose monitoring and adjustments, and interactions with other medications and certain foods. In contrast, direct oral anticoagulants (DOACs) including dabigatran, rivaroxaban, apixaban, and edoxaban overcome these disadvantages associated with VKAs. These DOACs have been recommended for use in stroke prevention among adult patients with atrial fibrillation3,4 and for acute treatment and secondary prevention of VTE including pulmonary embolism and deep vein thrombosis. 5 However, their effectiveness and safety in patients with CTEPH are still not well-established. A comprehensive evaluation of the benefits and potential risks associated with DOACs in treating CTEPH patients should be performed. To address this, researchers have previously conducted metaanalyses, which are studies that combine and analyze data from multiple related studies. However, these previous metaanalyses have certain limitations as they include various retrospective data. Therefore, by analyzing the available data from the prospective studies, the objective of our current meta-analysis was to evaluate and compare the effectiveness and safety outcomes of DOACs with VKAs in patients with CTEPH.
Methods
Literature Retrieval
Our current metaanalysis was conducted based on the Cochrane Handbook for Systematic Reviews of Interventions Version 6.2. 6 In order to conduct a thorough literature retrieval, we systematically searched the PubMed and Embase databases up to February 2024 to identify relevant studies on the effect of DOACs in patients with CTEPH. To conduct a comprehensive search, we expanded our search terms to key phrases related to CTEPH, DOACs, and VKAs (Supplemental Table 1). In addition, we also thoroughly examined the reference lists of previous reviews on the subject for additional studies that may have been overlooked during the initial database search.
Eligibility Criteria
Among the available studies, RCTs and prospective cohorts were included if they assessed the effectiveness and safety outcomes of DOACs (dabigatran, rivaroxaban, apixaban, or edoxaban) compared with VKAs in patients with CTEPH. The outcomes of interest mainly included all-cause mortality, VTE recurrence, major bleeding, any bleeding, and minor bleeding. We excluded studies that only involved a single group or arm of participants and did not have any comparison or control group.
Study Selection and Data Extraction
We followed our established eligibility criteria to assess studies for inclusion in our present analysis. Initially, we reviewed the titles and abstracts of the studies to identify those that appeared to meet our criteria. We then proceeded to obtain and review the full texts of these potentially eligible studies in a sequential manner. Once we identified the studies that met our inclusion criteria, we meticulously extracted the baseline characteristics of these included studies, such as author name, year of publication, study type, age, gender, sample size, demographics, and any relevant baseline measurements.
Risk of Bias Assessment
We used the Cochrane Risk of Bias Tool 2.0 (RoB 2) 7 to assess the bias risk of RCTs. This comprehensive tool consisted of 5 key domains that were assessed to determine the risk of bias. These domains included evaluating bias arising from the randomization process, bias resulting from deviations in the intended interventions, bias due to incomplete outcome data, bias in the measurement of the outcomes, and bias related to selectively reported results. Each domain was scored as either “low risk,” “some concerns,” or “high risk,” depending on the extent of bias identified.
The quality of observational prospective cohorts was assessed using the Newcastle-Ottawa Scale (NOS) tool. 8 The NOS tool consisted of 3 domains, each contributing to a total of 9 points. These domains included the selection of cohorts, comparability of cohorts, and assessment of the outcome. A NOS score of 6 or above was categorized as moderate to high quality. On the other hand, a NOS score below 6 suggested low quality.9,10
Statistical analysis
We performed a heterogeneity test using statistical methods, including the P-value of the Cochrane Q test and the I² value. In the pooled analysis, a Mantel–Haenszel random effects model was applied due to the potential heterogeneity across the included studies (the Review Manager software, Version 5.4 [the Cochrane Collaboration 2014, Nordic Cochrane Centre Copenhagen, Denmark]). The effect measures were expressed as the absolute risk differences (RDs) and 95% confidence intervals (CIs). In addition, we conducted the subgroup analysis based on the study type. To further evaluate the potential publication bias, the funnel plots were employed to examine whether there was a bias in the publication of studies concerning the outcomes being investigated.
Results
Study Selection
In this study, the literature retrieval process was carefully conducted and the steps followed are displayed in Figure 1. A total of 402 records were retrieved, with 72 records found in PubMed and 330 records in Embase. To ensure a comprehensive analysis, the reference lists of prior reviews were also checked, but no additional studies were identified. Initially, the titles and abstracts of the retrieved records were assessed according to predefined inclusion and exclusion criteria. As a result, 392 studies that did not meet the requirements were excluded from further consideration. After this initial screening phase, the remaining 10 studies underwent full-text screenings. During this phase, a more detailed evaluation was performed to determine their relevance and suitability for the present study. Consequently, 6 additional studies11,12,13,14,15,16 were excluded because they were retrospective studies. Ultimately, a final set of 4 studies (2 RCTs and 2 prospective cohorts)17,18,19,20 were included in this metaanalysis.
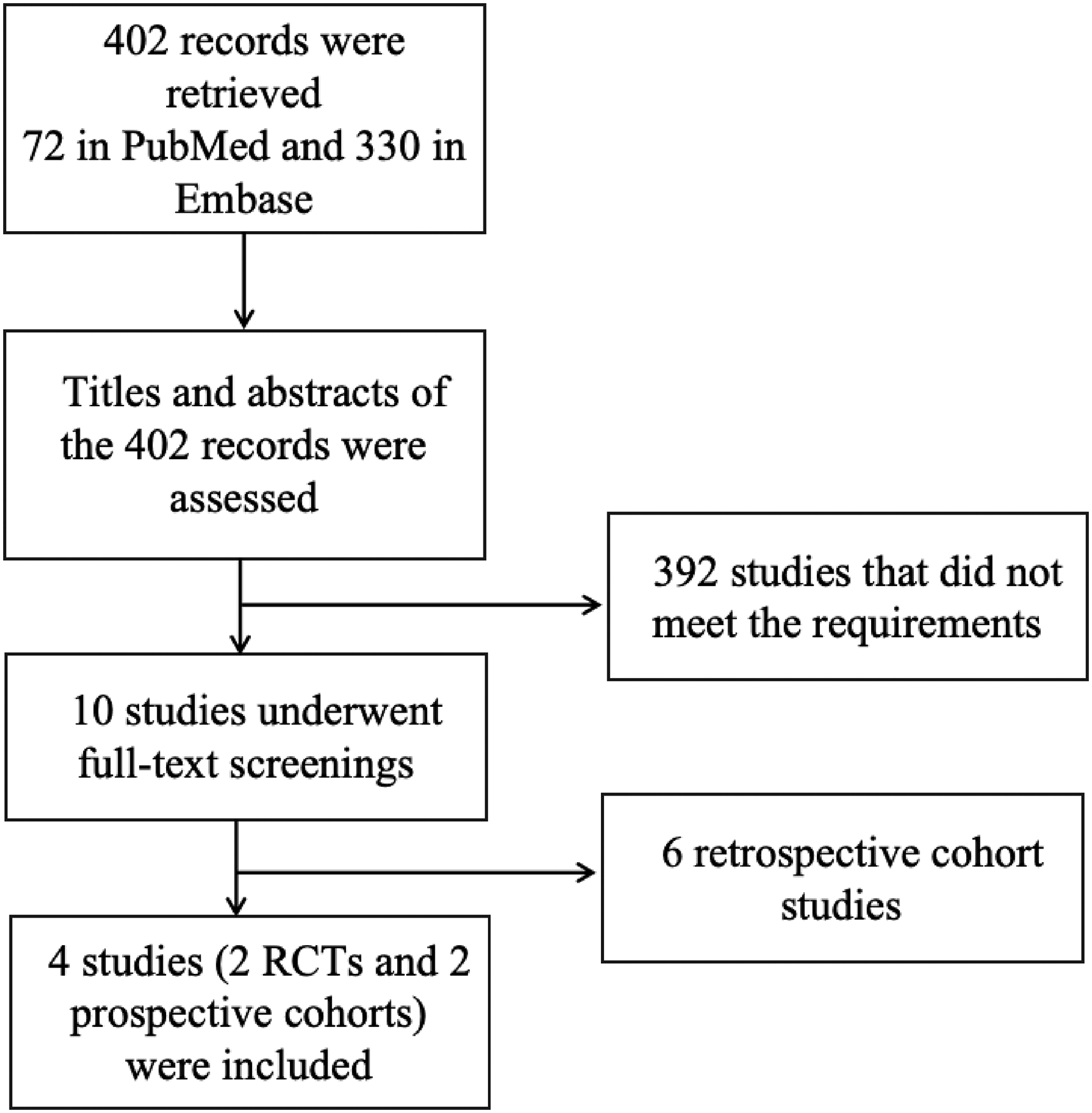
The literature retrieval process of this metaanalysis.
Baseline characteristics
The baseline characteristics of the included studies are shown in Table 1. A total of 2038 participants were enrolled, including 751 DOAC users (dabigatran, rivaroxaban, apixaban, and edoxaban) and 1287 VKA users (warfarin, phenprocoumon, and acenocoumarol). The follow-up duration ranged from 0.5 to 4 years. The 2 RCTs had a low risk of bias assessed according to the RoB 2 tool, and an NOS score of ⩾ 6 points for 2 prospective cohorts. The Japanese RCT and prospective cohort17,20 by Hosokawa et al included CTEPH patients, whereas the other RCT by Barati et al and prospective cohort by Humbert et al18,19 specifically selected CTEPH patients undergoing endarterectomy and CTEPH patients on riociguat, respectively. Riociguat is a soluble guanylate cyclase used as a first line treatment for inoperable CTEPH patients or patients with persistent pulmonary hypertension after pulmonary endarterectomy. It shows both tolerability and efficacy across various age groups, irrespective of comorbidities. Longer disease duration in the VKA group compared to the DOACs group was noted in 2 studies, suggesting that DOACs could be a more popular option in newly diagnosed CTEPH, potentially due to the short time that DOACs have been clinically available. The prospective cohort by Hosokawa et al 20 reported a higher proportion of patients with a history of acute VTE and patients with no history of previous reperfusion treatment in the DOACs arm. Inoperable, persistent pulmonary hypertension following reperfusion therapy was more common, whereas riociguat monotherapy was less common in the VKA group in the population included by Humbert et al. 19 A higher percentage of patients with previous thrombosis in the DOAC group was also observed by Barati et al. 18
The Baseline Data of the Included Studies in this Review.

DOAC, direct oral anticoagulant; VKA, vitamin K antagonist; HAS-BLED, hypertension, abnormal renal and liver function, stroke, bleeding, labile international normalized ratio, elderly, drugs or alcohol; CTEPH, chronic thromboembolic pulmonary hypertension; BNP, brain natriuretic peptide; VTE, venous thromboembolism; PVR, pulmonary vascular resistance; PAP, pulmonary arterial pressure; APS, antiphospholipid syndrome;
Outcomes Between DOACs and VKAs in Patients With CTEPH
In the pooled analysis shown in Figure 2, compared with VKAs, DOACs showed similar rates of effectiveness outcomes including all-cause mortality (3.33% [9/270] vs 3.33% [26/781]; RD = −0.01%, 95%CI [−0.02%, 0.00%, P = .17), VTE recurrence (1.46% [11/751] vs 2.12% [26/1227]; RD = −0.00%, 95%CI [−0.01%, 0.01%], P = .92) in patients with CTEPH. For the safety events, compared with VKAs, the use of DOACs had nonsignificant reductions in the occurrence rates of major bleeding (2.22% [6/270] vs 3.71% [29/781]; RD = −0.01%, 95%CI [−0.04%, 0.01%], P = .30), any bleeding (5.33% [40/751] vs 9.94% [122/1227]; RD = −0.03%, 95%CI [−0.07%, 0.01%, P = .10), and minor bleeding (4.17% [3/72] vs 13.3% [13/98]; RD = −0.06%, 95%CI [−0.23%, 0.10%], P = .45).
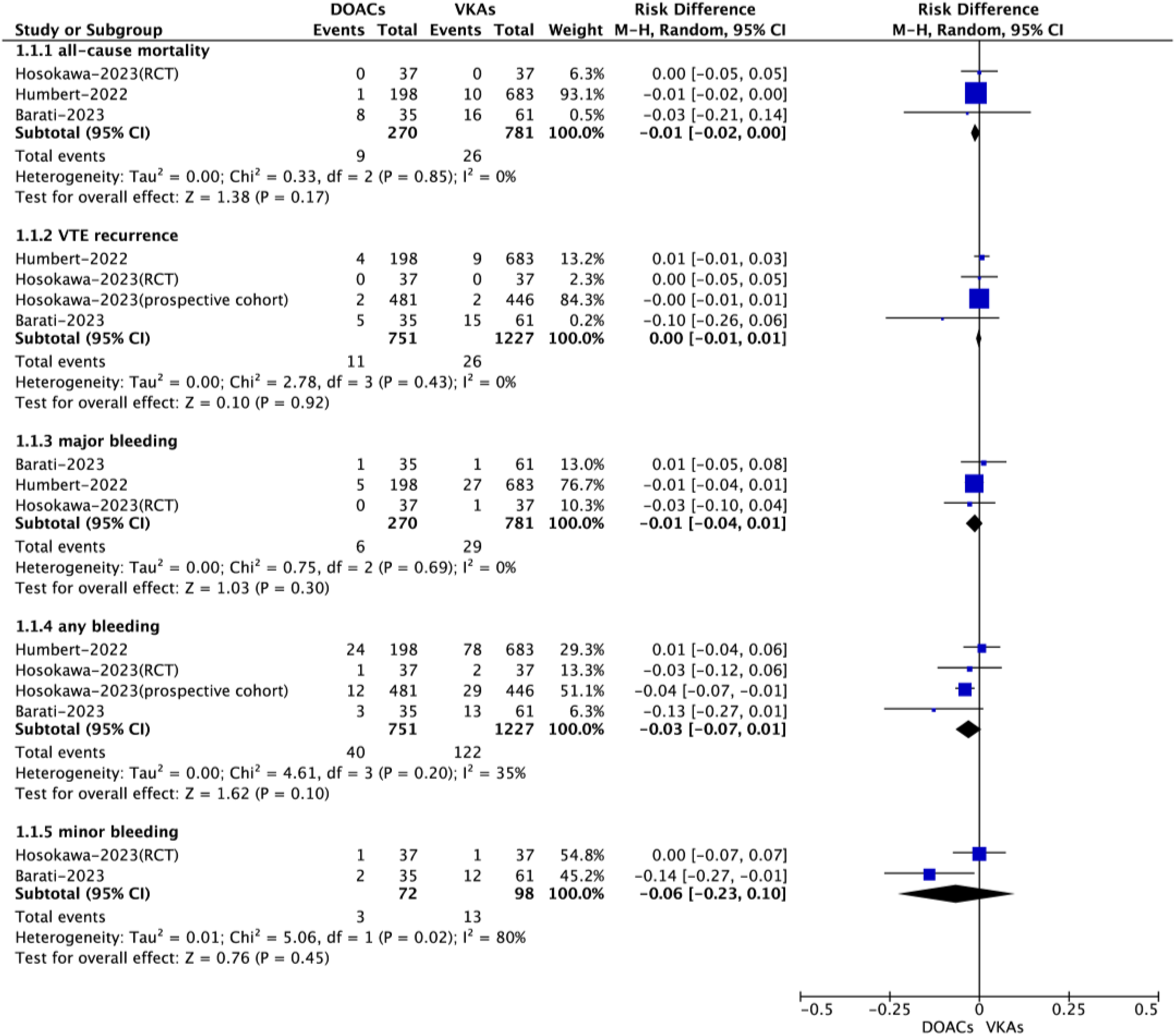
Effectiveness and safety outcomes between direct oral anticoagulants and vitamin K antagonists in patients with chronic thromboembolic pulmonary hypertension.
Subgroup Analysis and Publication Bias
Based on the subgroup analysis focusing on the type of study (RCT vs prospective cohorts), the results suggested that there were no significant interactions between the different study types. This suggested that the observed outcomes between DOACs and VKAs did not significantly differ between RCTs and prospective cohort studies. The findings from the funnel plots indicated that there was no clear evidence of publication bias for the studied outcomes.
Discussion
In our comprehensive analysis, researchers examined data from multiple prospective cohorts and RCTs to compare the outcomes of patients using DOACs versus VKAs. The results indicated that DOACs showed a comparable rate of all-cause mortality and VTE recurrence when compared to VKAs. Additionally, the analysis also revealed a nonsignificant reduction in bleeding events among patients taking DOACs. This finding suggests that DOACs may have a slight advantage over VKAs in terms of lower bleeding risks, although the difference was not statistically significant. It is worth noting that DOACs are likely to offer several advantages over VKAs, including increased convenience and ease of use for both healthcare providers and patients. Thus, these findings suggest that DOACs could become a more reliable and user-friendly option in the management of CTEPH, improving patient outcomes and overall treatment experience.
Prior systematic reviews and metaanalyses21,22,23 have been conducted on the use of DOACs in CTEPH patients. VKA participants outnumbered DOAC participants in all 3 studies. The systematic review by Sedhom et al 21 published in 2021 included retrospective studies and case series. The authors concluded that DOACs are possibly associated with an increased risk of recurrent VTE as compared to VKAs, as well as a lower or similar risk for major bleeding events. The systematic review was conducted without employing a quantitative statistical method such as metaanalysis. The results solely relied on a qualitative synthesis of existing data. Therefore, the conclusion drawn from this review may be less conclusive due to the lack of statistical power and an inability to assess heterogeneity among the included studies. A newer metaanalysis published in 2023 by Burmeister et al 22 included 3 retrospective studies and 2 prospective trials, but with no RCTs. All-cause mortality appeared to be significantly lower in the DOACs arm in comparison to VKAs. No significant difference was observed in VTE recurrence, total bleeding events, and major and minor bleeding events. The most recently published metaanalysis by Salazar et al 23 incorporated a total of 10 studies, consisting of the 2 prospective studies and 2 RCTs included in our metaanalysis, in addition to 6 retrospective observational studies, resulting in the highest number of participants. The pooled analysis showed no significant difference in all-cause mortality between the 2 groups and similar rates of VTE recurrence and major bleeding events.
The previous metaanalysis reports on this topic have provided inconsistent and contradictory conclusions. This may be attributed to certain limitations associated with those reviews, such as the inclusion of both prospective and retrospective trials. By exclusively including prospective trials in our study, we aim to address the limitations of the previous reviews. Prospective trials are more reliable as they avoid selection bias and incorporate randomization, which enhances the quality of the data. We believe that focusing on prospective trials will enable us to generate more valid and reliable results, offering a stronger understanding of the topic at hand. Overall, our study aims to provide a comprehensive and robust elucidation of the subject matter. Importantly, it is worth noting that our metaanalysis stands out as the first of its kind to evaluate the prospective use of DOACs specifically in patients with CTEPH. This demonstrates the novelty and significance of our study, providing valuable insights into the effectiveness and safety of DOACs in this particular patient population.
We hope our findings can provide clinicians with more definitive evidence as to the choice of long-term anticoagulation therapy when it comes to managing patients with CTEPH. By utilizing data from prospective clinical studies on CTEPH, it may guide clinicians in making more informed decisions that will significantly improve the quality of care provided to CTEPH patients. Furthermore, the study emphasizes the necessity of conducting future clinical trials specifically focused on the use of DOACs, an area that requires further exploration. We believe that conducting well-designed RCTs with larger sample sizes will play a crucial role in confirming their findings and shedding more light on the optimal dosing regimen, long-term outcomes, and management strategies for CTEPH patients receiving DOAC therapy.
Limitations
Our study had several limitations. Firstly, since our study involved pooling data from various studies, it was inevitable that there would be heterogeneity present. This heterogeneity stemmed from differences in the study designs, target populations, follow-up duration, intervention, endpoints, and baseline characteristics among the included studies. Secondly, it is worth noting that the number of studies included in our meta-analysis and the overall sample size were relatively small. This limited sample size might restrict the generalizability of our findings to the larger population. Lastly, an important limitation of our study was the significant imbalance in the number of participants between the DOAC and VKA arms. Specifically, there were more patients included in the VKAs arm compared to the DOACs arm. This imbalance may have introduced bias and affected the accuracy of the observed treatment effects, particularly when comparing the efficacy or safety outcomes between the 2 treatment groups. Therefore, caution should be exercised when interpreting the results related to the comparison between DOACs and VKAs in our study.
Conclusions
After conducting a comprehensive analysis of the data obtained from 4 different prospective studies, we have reached the conclusion that DOACs and VKAs demonstrate similar levels of effectiveness and safety in the treatment of CTEPH. This suggests that DOACs have the potential to serve as a viable alternative to VKAs in the administration of thromboprophylaxis for CTEPH patients in actual clinical settings. Further prospective clinical studies should be undertaken to confirm our findings.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241257931 - Supplemental material for Direct Oral Anticoagulants in Chronic Thromboembolic Pulmonary Hypertension: First Meta-Analysis of Prospective Studies
Supplemental material, sj-docx-1-cat-10.1177_10760296241257931 for Direct Oral Anticoagulants in Chronic Thromboembolic Pulmonary Hypertension: First Meta-Analysis of Prospective Studies by Tang Zhang, Linjuan Guo, Shucheng Liang and Hao Liu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We thank Dr Wengen Zhu for his help in this study.
Author contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
