Abstract
The purpose of the study was to investigate baseline inflammatory, hemostatic indicators and new-onset deep vein thrombosis (DVT) with the risk of mortality in COVID-19 inpatients. In this single-center study, a total of 401 COVID-19 patients hospitalized in Sir Run Run Shaw Hospital, Zhejiang University School of Medicine were enrolled from December 1, 2022 to January 31, 2023. The basic information, first laboratory examination results, imaging examination, and outcome-related indicators were compared between patients in the moderate and severe subgroups. We found that baseline D-dimer and baseline absolute neutrophil count (ANC) levels were associated with new-onset DVT and death in severe hospitalized patients with COVID-19. The odds ratio (OR) of baseline D-dimer and baseline ANC with mortality was 1.18 (95% confidence interval [CI], 1.08–1.28; P < .001) and 1.13 (95% CI, 1.06–1.21; P < .001). Baseline ANC was associated with the risk of death in severe hospitalized COVID-19 patients, irrespective of the DVT status. In addition, a significantly higher serum neutrophil activity was observed in severe COVID-19 inpatients with DVT or those deceased during hospital stay. New-onset DVT partially mediated the association between baseline D-dimer (indirect effect: 0.011, estimated mediating proportion: 67.0%), baseline ANC (indirect effect: 0.006, estimated mediating proportion: 48.7%), and mortality in severe hospitalized patients with COVID-19. In summary, baseline D-dimer and baseline absolute neutrophil count (ANC) levels were associated with the mortality in severe hospitalized patients with COVID-19, especially DVT inpatients. New-onset DVT partially mediated the association between baseline D-dimer, baseline ANC, and mortality in severe hospitalized patients with COVID-19.
Keywords
Introduction
The coronavirus disease 2019 (COVID-19) is a novel coronavirus caused by the severe acute respiratory syndrome, Coronavirus 2 (SARS-CoV-2), a rapidly spreading virus that causes pneumonia. In addition to respiratory involvement, SARS-CoV-2 infection can cause severe changes in coagulation function, inducing hypercoagulable states.1,2 Since the outbreak of COVID-19, several studies have reported the association between laboratory hemostatic parameters and clinical outcomes of hospitalized patients with COVID-19, with data suggesting that venous thromboembolism (VTE) may lead to acute respiratory failure in patients with COVID-19. Despite the use of low-molecular weight heparin (LMWH) for thromboprophylaxis, a high incidence of VTE is observed in hospitalized patients with COVID-19, especially those admitted to the intensive care unit (ICU). One of the first papers published in China revealed that elevated baseline D-dimer (DDI) levels were closely related to the risk of mortality in COVID-19 patients. 3 Other studies showed that patients infected with SARS-CoV-2 (COVID-19 patients), especially those treated in the ICU, are at high risk for VTE. Elevated D-dimer levels are associated with an increased risk of VTE and/or death in patients with COVID-19.4,5
COVID-19 infection usually starts with flu-like symptoms and can be asymptomatic or may have a mild to severe course.6,7 The infection is characterized with a significant burden of inflammation; hence, the association between hematological parameters and COVID-19 infection has been studied, with the absolute neutrophil count/absolute lymphocyte count (NLR)8,9 and the platelet-to-lymphocyte ratio (PLR) 10 found to be related with the infection. Theb NLR,11,12 PLR, 13 and high-sensitivity C-reactive protein (CRP)14,15 are in association with the inflammatory burden. It is also due to the strong systemic inflammatory response that patients with COVID-19 are susceptible to VTE and arterial and microvascular thrombosis. Abnormal coagulation parameters and high concentrations of proinflammatory cytokines are also associated with disease severity, poor prognosis, and incidence of VTE in patients with COVID-19.16,17 As the first line of defense against pathogen invasion, the increase in the absolute value of peripheral blood neutrophils is associated with disease severity and the risk of death in hospitalized patients with COVID-19.17,18 Neutrophil extracellular traps (NETs) are a specific form of programmed neutrophil cell death characterized by the extrusion of DNA, histones, and antimicrobial proteins in a meshwork of structures, called NETs. NETs can limit infection, whereas NET disruption may cause negative outcomes, such as immune-related inflammation and tissue damage. Moreover, activated NETs also induce hypercoagulability and thrombosis. NETs have been found in plasma and serum samples of COVID-19 patients, as well as in autopsy lung specimens. Therefore, hematological parameters and NET biomarkers may help predict disease severity, clinical outcomes, and VTE in COVID-19 patients, as well as guide anticoagulant therapy.18–21
In December 2022, the adjustment of national policies resulted in the first nationwide outbreak of COVID-19 in China. During the outbreak period, the fever outpatient service, emergency department, and ICU were overloaded, among which the peak in the ICU workload was the most challenging. Identifying the early predictors of disease progression in COVID-19 patients, as well as the risk factors of new-onset DVT and mortality in hospitalized severe COVID-19 patients, is crucial for the timely adjustment of treatment, which reduces the risk of death in severe patients. In this study, a series of laboratory markers were evaluated on admission from 401 hospitalized COVID-19 patients in Sir Run Run Shaw Hospital. The markers related to neutrophil activation were detected in the peripheral blood of early-stage patients. Subsequently, the test results and clinical outcomes of patients were compared to comprehensively determine the differences in laboratory indicators between moderate and severe inpatients. Furthermore, the risk of in-hospital mortality and the influencing factors of new-onset DVT in severe COVID-19 inpatients were evaluated, providing a reference for the assessment of outcomes of COVID-19 inpatients.
Material and Methods
A total of 401 COVID-19 patients aged ≥18 years who were hospitalized between December 1, 2022 and January 31, 2023 were enrolled in this study.
Inclusion criteria:
at least 18 years of age; positive reverse transcriptase-polymerase chain reaction (RT-PCR) for SARS-CoV-2 of nasal or oropharyngeal swabs. patients with known active cancer, pregnancy, or lactation at the time of DVT diagnosis; patients with incomplete clinical data; patients with a history of VTE.
Exclusion criteria:
The COVID-19 severity was defined according to the clinical classification of “Diagnosis and Treatment Plan for COVID-19 (Trial 10th edition)” formulated by the National Health Commission of China in 2022. The specific classification criteria are presented below.
In adults, the presence of any of the following with no other possible cause than COVID-19 infection:
shortness of breath, RR ≥ 30 beats/min; oxygen saturation ≤93% on air in resting state; [arterial partial pressure of oxygen (PaO2)/inspired oxygen concentration (FiO2)] ≤ 300mm Hg (1 mm Hg = 0.133 kPa), high altitude (above 1000 m) areas should be corrected according to the following formula: PaO2/FiO2[760/ atmospheric pressure (mm Hg)]; progressively worsening clinical symptoms, with lung imaging displaying more than 50% progression in the lesion within 24 to 48 h.
This study was approved by the ethical committee of Sir Run Run Shaw Hospital (Study No. 0264). A total of 24 patient serum samples used in this study were retained during routine laboratory testing. The activity of NET markers, such as citrullinated histone H3 (CitH3) and myeloperoxidase DNA (MPO-DNA), was detected with ELISA (CitH3: Cayman, USA; MPO-DNA:4A Biotech, China). The data of admitted patients were collected from the hospital medical record system. The baseline characteristics (age, gender, etc), comorbidities, admission to the ICU, length of hospital stay, treatment outcome (improvement or death), bleeding and thrombotic events, and baseline routine blood tests were collected, including the total white blood cell count (WBC), absolute neutrophil count (ANC), absolute lymphocyte count (LYM), NLR, hemoglobin, red blood cell distribution width (RDW), CRP, interleukin-6 (IL-6), N-terminal pro-brain natriuretic peptide (NT-proBNP), blood gas P/F ratio, and coagulation indices (platelet count [PLT], activated partial thromboplastin [APTT], international standard ratio [INR], D-dimer [DDI], fibrinogen). Blood counts were analyzed on Mindray BC6800/6900 (China). Analysis of CRP was performed on a BC5390 CR blood cell analyzer from Mindray (China). The IL-6 measurements were performed on an eCL8000 automatic chemical analyzer from Shenzhen Lifotronic Technology Corporation, Ltd. (China). The PT (INR), APTT, DDI, fibrinogen, and DDI were analyzed with STA-Rmax automatic blood coagulation analyzer from France. NT-proBNP was analyzed with a Pylon automatic fluorescence immunoassay analyzer from ET Health (USA). The analysis of arterial blood gas was performed on Roche Cobasb123 (Sweden).
DVT was diagnosed according to the “Chinese Guidelines for the Prevention and Treatment of Thrombotic Diseases” issued by the Expert Committee for the Prevention and Treatment of Thrombotic Diseases in November 2022. The thrombosis prevention and antithrombotic treatment of COVID-19 patients were mainly based on the 2022 guidelines for the diagnosis and treatment of COVID-19 of the National Health Commission of China (Trial 10th edition). Bleeding events during hospitalization were classified according to the International Society on Thrombosis and Haemostasis criteria.
Patients enrolled in this study were divided into two groups according to their SARS-Cov-2 infection severity (Severe vs Moderate), diagnosis with new-onset DVT (DVT vs Non-DVT), or in-hospital outcome (Deceased vs Survived) based on different analysis purposes. The severe COVID-19 inpatients were further divided into two subgroups considering the diagnosis with new-onset DVT (DVT vs Non-DVT), ICU-treatments (ICU vs Non-ICU), baseline ANC levels (High-ANC vs Low-ANC), or in-hospital outcome (Non-survivors vs Survivors) for various analysis purposes. The mean ± standard deviation was used to describe the variables following a normal distribution, while variables not following normal distribution were presented as median (interquartile range [IQR]). The number of cases (%) was used to represent the count data, whereas the t-test and nonparametric rank-sum test were used for the univariate analysis of continuous variables. In addition, the Fisher's exact test was used for the univariate analysis of categorical variables, and binary logistic regression analysis was used for the regression analysis of binary dependent variables.
Subsequently, a causal mediation model was constructed to analyze the mediating effect of new-onset DVT between baseline D-dimer levels and the risk of death in severe hospitalized patients with COVID-19. Baseline DDI/ANC was the predictor variable (X). DVT was the mediator (M). Mortality was the outcome variable (Y). Overall, the mediation analysis involved four steps: (1) demonstrating that the predictor variable is associated with the outcome (Model Y = βTot X) (βTot = total effect); (2) demonstrating that the predictor variable is associated with the mediator (Model M = β1 X) (β1 = indirect effect 1); (3) demonstrating which part of the outcome is explained by controlling for the predictor (Model Y = β2 M+βdir X) (β2 = indirect effect, βdir = direct effect); and (4) calculating the proportion of mediation: mediation effect (%) = (β1*β2/βTot) × 100%. Logistic regression analysis was used to measure the direct, indirect, and total effects. 22
SPSS 26.0 statistical software was used for data processing and analysis.
Results
Baseline Characteristics
The demographic and baseline characteristics of the severe and moderate patients are shown in Table 1. The age of patients in the severe group (median 80 years, IQR 71–87) was significantly higher than those in the moderate group (median 73 years, IQR 66–84) (P < .001). A larger proportion of severe COVID-19 patients suffered from hypertension (65.7%) and heart failure (3.5%) compared to moderate patients (hypertension 52.5%, P = .008; heart failure 0%, P = .015). In addition, the risk of mortality (34.3% vs 3.5%, P < .001), the incidence of DVT (40.8% vs 12.2%, P < .001), and the proportion of patients requiring ICU admission (39.3% vs 1.5%, P < .001) were higher in the severe group compared to the moderate group. No significant difference in the prevalence of diabetes, atrial fibrillation, previous history of arterial thromboembolism, chronic kidney disease, and liver cirrhosis was observed between the two groups (all P values above .05). Moreover, the median length of the hospital stay was longer in severe patients (median 16 days, IQR 9–23) than in moderate patients (median 6 days, IQR 4–9) (P < .001).
Baseline Clinical Characteristics.

The units of the variables in the table are the number of patients, except for the variables with specific labeling units.
Venous and Arterial Embolism
DVT
According to the 2022 guidelines for the diagnosis and treatment of COVID-19 pneumonia (trial, 10th edition) of the National Health Commission of China, LMWH was used to prevent thrombosis in COVID-19 patients without anticoagulant contraindication. However, DVT of the lower extremities still occurred in 105 patients (26.2%), with a significantly lower incidence in the moderate group (12.2%) compared to the severe group (40.8%) (Table 1). The median time of new-onset DVT was 2 days after admission, showing no significant difference between the severe and moderate groups (P = .816).
Subsequently, the clinical characteristics, laboratory test results, and imaging results of severe hospitalized COVID-19 patients were analyzed, revealing that requiring admission to ICU, the duration of hospitalization, baseline ANC, LYM, NLR, D-dimer, blood gas P/F ratio, and lesions covering more than 50% of the lung field on the first chest imaging were associated with new-onset DVT. Binary logistic regression analysis of the abovementioned indicators, except for NLR, showed that the duration of hospitalization (OR 1.03, 95% confidence interval: 1.01–1.06, P = .012), baseline LYM (OR 0.45, 95% CI: 0.21–0.95, P = .037), and baseline D-dimer (OR 1.12, 95% confidence interval: 1.02–1.23, P = 0.013) were independent risk factors for new-onset DVT in severe hospitalized patients with COVID-19 (Tables S1 and S2, Table 2).
Influencing Factors of New-Onset DVT in Severe Inpatients.
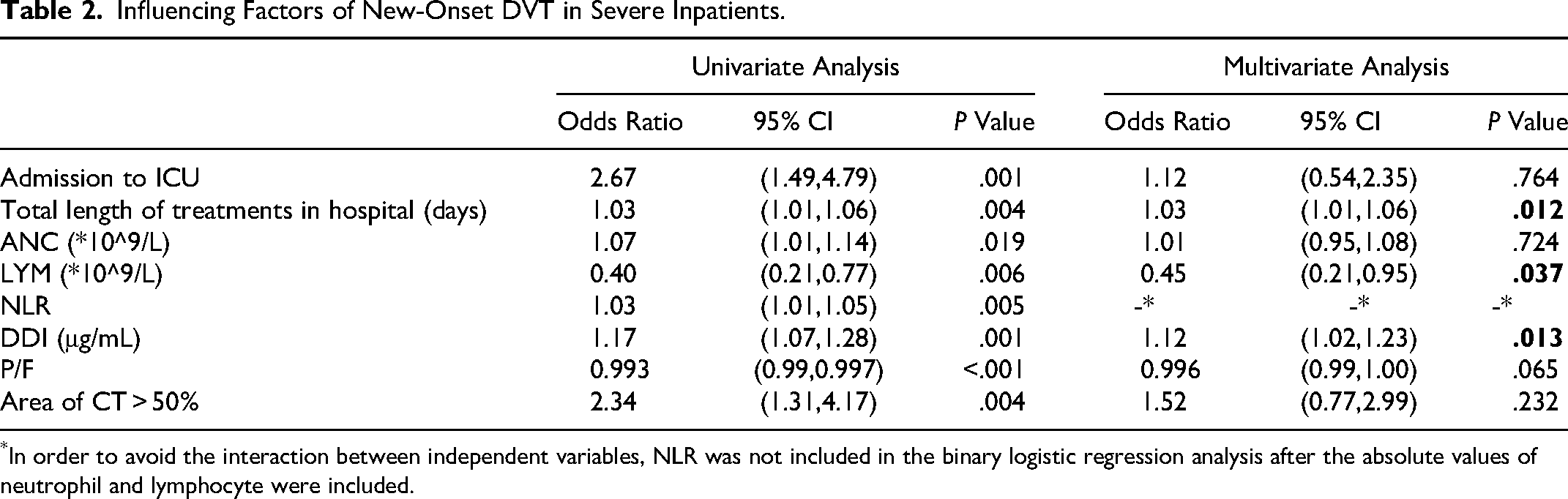
In order to avoid the interaction between independent variables, NLR was not included in the binary logistic regression analysis after the absolute values of neutrophil and lymphocyte were included.
Arterial Embolism Events
New cerebral arterial thromboembolism events were found in two patients from the severe group during hospitalization, both with deep vein thrombosis of the right lower extremity.
Bleeding
Bleeding occurred in 15 patients during the current hospitalization, with an overall incidence of 3.7%, including 10 in the severe group and 5 in the moderate group. The characteristics of these 15 cases are shown in Table S3.
Mortality
In total, 397 patients (99%) who were hospitalized with severe COVID-19 had a length of stay of less than 60 days, of which 34.3% of severe patients and 3.5% of moderate patients died (P < .001) (Table 1).
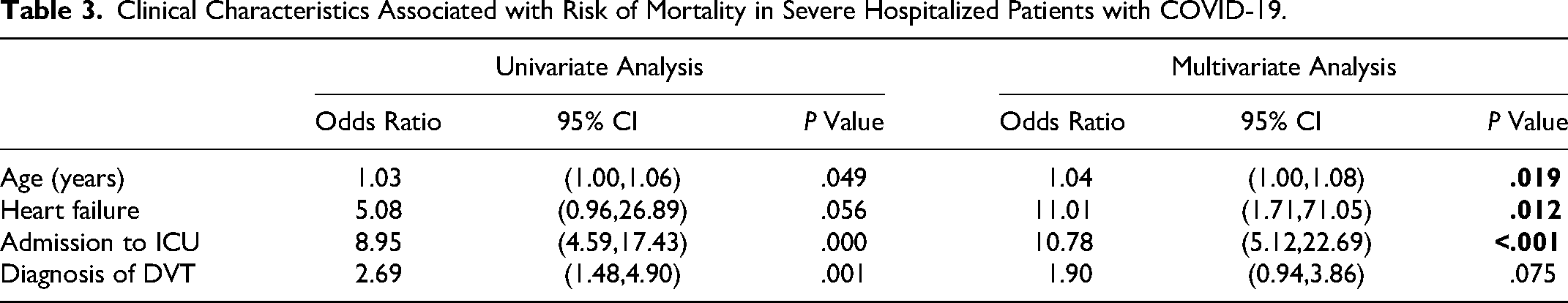
The mean age of the patients who died (80.04 ± 9.72 years) was significantly higher than that of patients who survived (76.73 ± 11.84 years) (P = .047). In this study, age, underlying heart failure, requiring admission to ICU, and new-onset DVT during hospitalization were all associated with a higher risk of death in severely hospitalized patients with COVID-19 (Tables S4 and 3). Logistic regression analysis, including the abovementioned clinical characteristics showed that age (OR 1.04, 95% CI: 1.00–1.08, P = .019), underlying heart failure (OR 11.01, 95% CI: 1.71–71.05, P = .012), and requiring admission to ICU (OR 10.78, 95% confidence interval: 5.12–22.69, P < .001) were independent risk factors for death in hospitalized severe COVID-19 patients (Table 3).
Clinical Characteristics Associated with Risk of Mortality in Severe Hospitalized Patients with COVID-19.
Other Routine Laboratory and Imaging Tests
As shown in Figure 1 and Table S5, patients in the severe group had a higher baseline RDW (median 13.4%, IQR 12.9–14) than those in the moderate group (median 12.9%, IQR 12.5–13.5) (P < .001). Severe patients also had a higher median baseline NT-proBNP level than moderate patients: 1270 pg/ml (IQR 479.5–3278.75) versus 254 pg/ml (IQR 116–739) (P < .001). In addition, the median baseline blood gas P/F ratio in the severe group (median 174, IQR 114–259.5) was significantly lower than that in the moderate group (median 372, IQR 334–413.8) (P < .001). Nevertheless, no significant difference in the baseline hemoglobin level was observed between the severe and moderate groups (all P values above .05). Moreover, the proportion of patients with lesions larger than 50% of the lung fields on first chest imaging was significantly higher in the severe group (43.9%) than in the moderate group (4.1%) (P < .001).
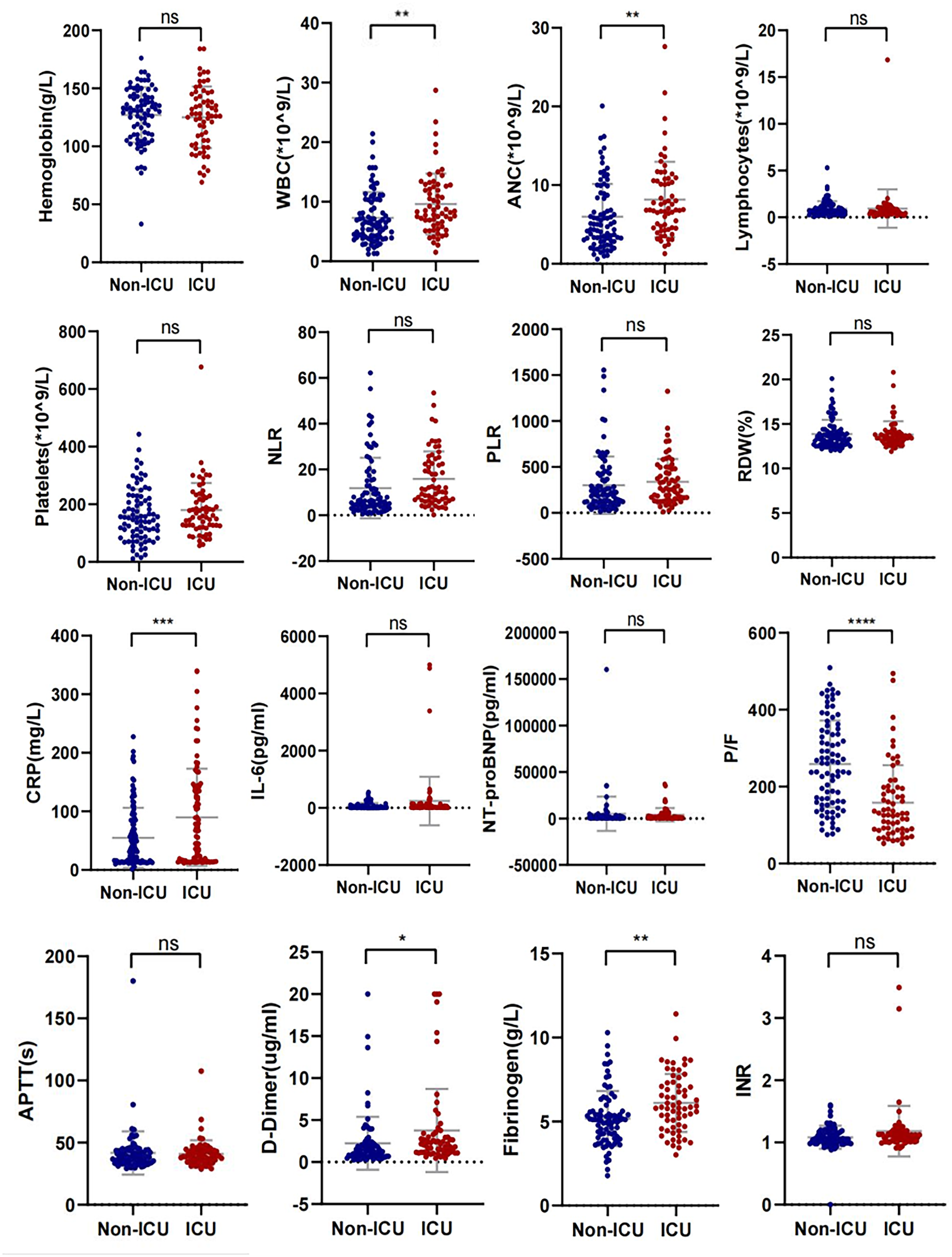
Distribution of the laboratory test results in moderate and severe patients on admission. WBC: white blood cell; ANC: absolute neutrophil count; NLR: neutrophil to lymphocyte ratio; PLR: platelet to lymphocyte ratio; RDW: red blood cell volume distribution width; CRP: C-reactive protein; P/F: PaO2/FiO2; APTT: activated partial thromboplastin time; INR: international normalized ratio. (*P < .05; **P < .001; ***P < .001; not significant n.s.)
Markers of Coagulation, Fibrinolysis, and Inflammation
As shown in Figure 1 and Table S5, patients in the severe group had higher median baseline D-dimer and fibrinogen levels than patients in the moderate group: 1.67 ug/ml (IQR 0.98–2.73) versus 0.82 ug/ml (IQR 0.46–1.46) (P < .001), 5.38 g/L (IQR 4.13–6.68) versus 4.9 g/L (IQR 4.1–5.8) (P = .006), respectively. Meanwhile, the median baseline APTT and the INR of severe patients were higher than those of moderate patients: 38.8 s (IQR 34.9–43.9) versus 38 s (IQR 34.5–41.6) (P = .043) and 1.07 (IQR 1.01–1.17) versus 1.03 (IQR 0.98–1.08) (P < .001), respectively. A lower median baseline platelet count was observed in severe patients (median 152 × 109/L, IQR 111.5–222 × 109/L) compared to moderate patients (median 169 × 109/L, IQR 127.3–221 × 109/L) (P = .017). These indicators suggest that compared with moderate patients, patients with severe COVID-19 have weakened coagulation function and increased fibrinolysis products on admission.
In severe and moderate patients, the median baseline WBC was 7.5 × 109/L (IQR 4.9–11.3) and 5.5 × 109/L (IQR 4.3–7.9) (P < .001), respectively. Patients in the severe group also exhibited significantly higher median baseline ANC 6.4 × 109/L (IQR 3.5–10.2) versus 3.9 × 109/L (IQR 2.9–5.9) (P < .001), baseline NLR 9.9 (IQR 4.63–21.84) versus 4.2 (IQR 2.7–6.6) (P < .001), and baseline PLR 240 (IQR 134.35–442.35) versus 190.7 (IQR 119.3–263.7) (P < .001). The median baseline LYM in severe patients was lower than that in moderate patients: 0.6 × 109/L (IQR 0.4–0.98) versus 0.93 × 109/L (IQR 0.65–1.28), P = .024. Other inflammatory markers, including CRP and IL-6, were significantly higher in severe patients compared to moderate patients: 78 mg/L (IQR 36.6–141.3) versus 40.4 mg/L (IQR 19.4–77.8) (P < .001), 34.5 pg/ml (IQR 9.9–143.1) versus 14.3 pg/ml (IQR 4.5–51.1) (P < .001), respectively. These inflammatory markers suggest significantly enhanced systemic inflammatory response in severe patients compared with moderate patients.
Baseline D-Dimer and Baseline ANC Associated with the Risk of New DVT and Mortality in Hospitalized Patients with Severe COVID-19
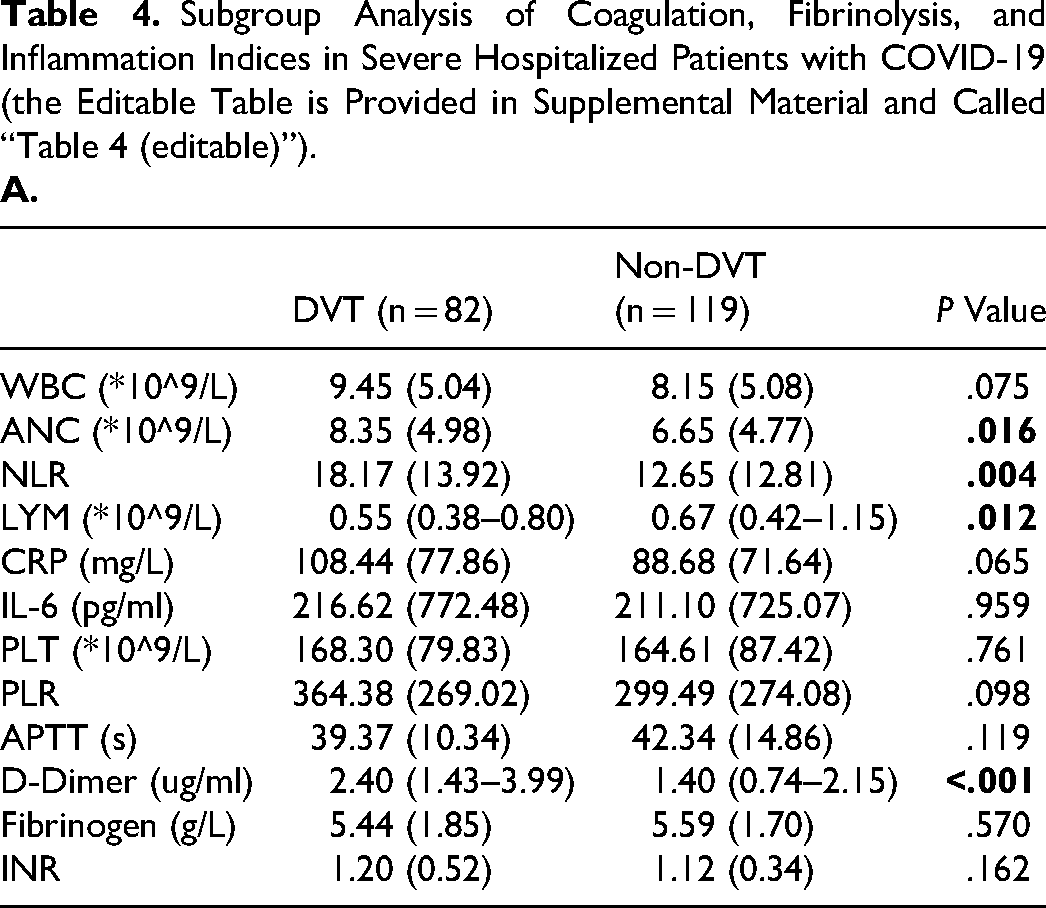
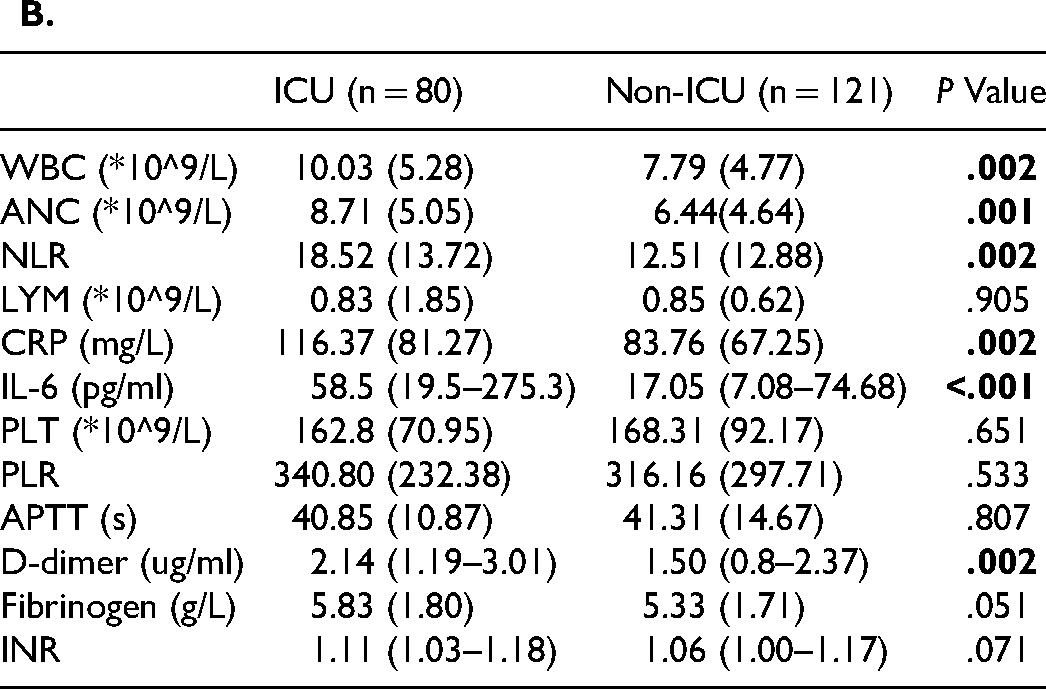
In the subgroup analysis of patients who were hospitalized with severe COVID-19, significant differences in the distribution of some test results were observed (Table 4). The baseline D-dimer levels and ANC were associated with new-onset DVT, requiring admission to the ICU and mortality in severe hospitalized patients with COVID-19 (Table 4A–C). Patients who required admission to the ICU or died had a higher baseline WBC than the corresponding controls: (10.03 ± 5.28) × 109/L versus (7.79 ± 4.77) × 109/L (P = .002) and (10.53 ± 5.44) × 109/L versus (7.72 ± 4.63) × 109/L (P < .001), respectively (Table 4B–C). In addition, patients with new-onset DVT had a lower median baseline LYM compared to patients without new-onset DVT [0.55 × 10^9/L (IQR 0.38–0.8) versus 0.67 × 10^9/L (IQR 0.42–1.15), P = .012] (Table 4A). Severe COVID-19 patients who required admission to the ICU had higher baseline CRP and median baseline IL-6 levels than severe COVID-19 patients in general wards: 116.37 ± 81.27 mg/L versus 83.76 ± 67.25 mg/L (P = .002), 58.5 pg/ml (IQR 19.5–275.3) versus 17.05 pg/ml (IQR 7.08–74.68) (P < .001), respectively (Table 4B). The median baseline INR of severe COVID-19 patients who died (median 1.12, IQR 1.01–1.23) was higher than that of severe COVID-19 patients who survived (median 1.06, IQR 1.01–1.16) (P = .048) (Table 4C).
Subgroup Analysis of Coagulation, Fibrinolysis, and Inflammation Indices in Severe Hospitalized Patients with COVID-19 (the Editable Table is Provided in Supplemental Material and Called “Table 4 (editable)”).

High Baseline D-Dimer and High Baseline ANC had an Additive Effect on the Risk of Mortality in Severe Hospitalized Patients with DVT
The baseline absolute neutrophil count and the baseline D-dimer levels were compared between severe COVID-19 patients with new DVT and those without in order to determine whether there was an additive effect on the mortality risk. The results suggested that high D-dimer levels were associated with mortality risk in severe hospitalized COVID-19 patients with DVT, but not in those without DVT. Among severe COVID-19 patients who had both DVT and high baseline d-dimer levels, d-dimer levels were associated with a risk of death (Figure 2A). However, a high baseline absolute neutrophil count was associated with mortality in severely hospitalized patients with COVID-19, regardless of the presence or absence of DVT (Figure 2B). Further analysis suggested that higher baseline D-dimer levels were associated with a higher risk of death in severe hospitalized COVID-19 patients with an absolute baseline neutrophil count above the normal value (6.3 × 109/L) and new-onset DVT, but this additive risk was not present in severely hospitalized COVID-19 patients without DVT (Figure 2C).
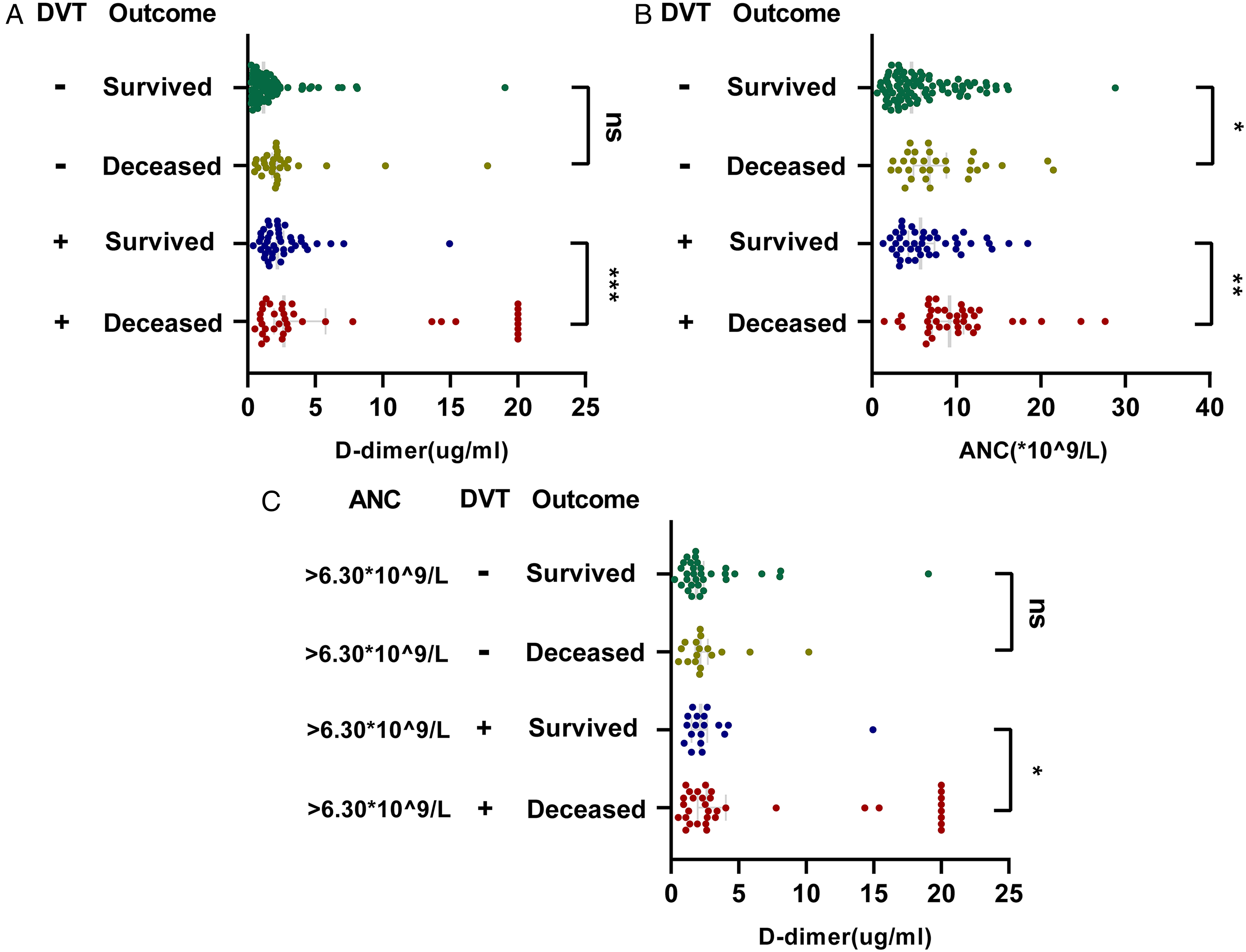
Analysis of baseline D-dimer and baseline ANC in deceased and survived severe COVID-19 inpatients.
Baseline ANC, Peripheral Blood Serum NET Activity and Risk of Mortality in Severe COVID-19 Patients
Previous results demonstrated that baseline ANC was associated with the risk of mortality in severe COVID-19 hospitalized patients, regardless of DVT. The predictive value of baseline ANC for the risk of death in hospitalized severe COVID-19 patients was explored by using the actual clinical outcomes. The ROC curve analysis of the baseline ANC was performed in 201 hospitalized severe COVID-19 patients and those with DVT (Figure 3). The area under the curve of baseline ANC for predicting the mortality of severe hospitalized patients with COVID-19 was 0.69, and the cut-off value of baseline ANC was 6.13 × 109/L with a sensitivity of 76.8% and a specificity of 61.4% (P < .001). The area under the curve of baseline absolute neutrophil count for predicting mortality in COVID-19 patients with DVT was 0.73, and the cutoff value of baseline ANC was 6.25 × 109/L with a sensitivity of 89.7% and a specificity of 58.1% (P < .001).
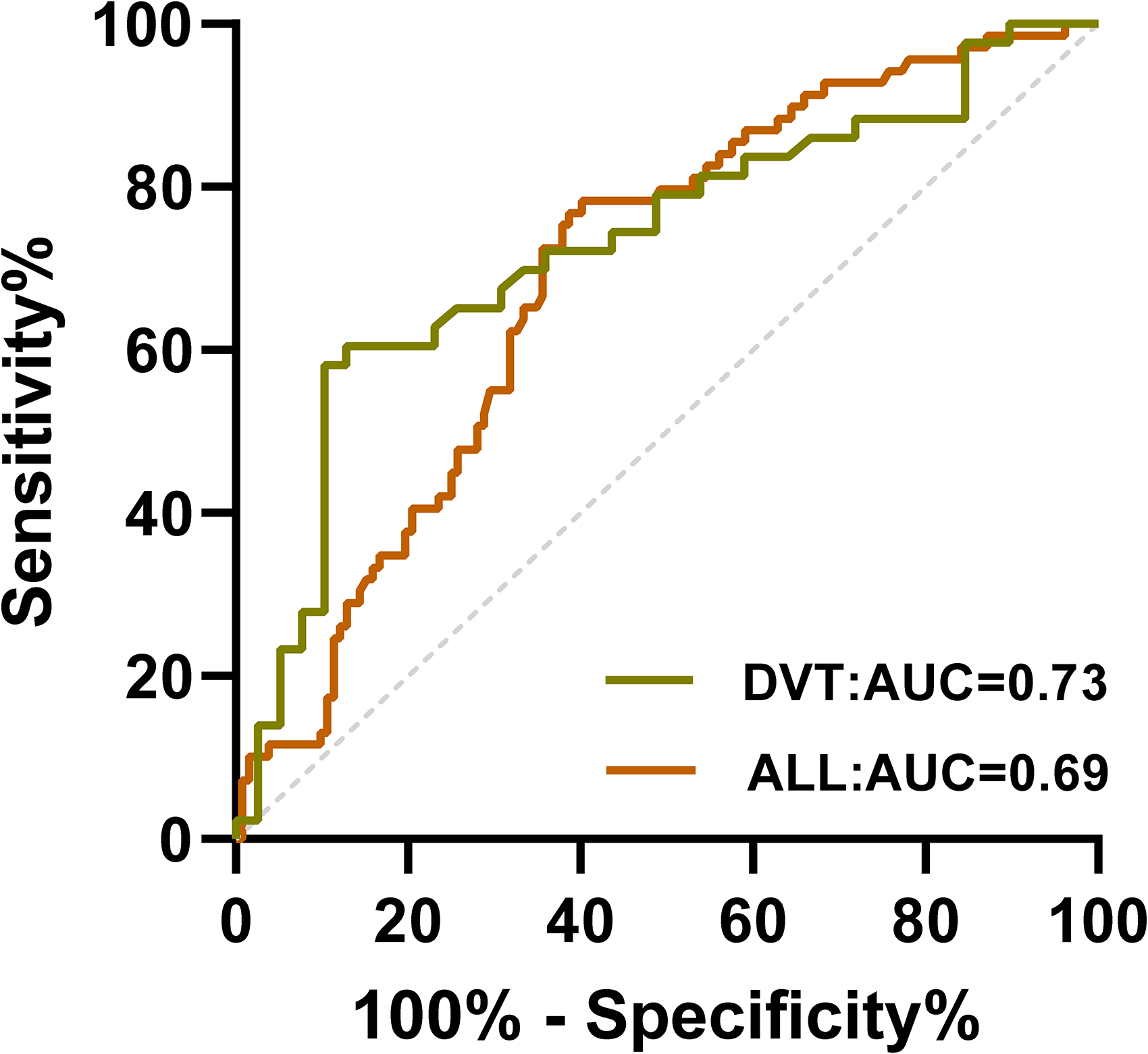
ROC curve of baseline ANC predicting the risk of mortality in severe hospitalized patients with COVID-19.
The serum levels of NET markers citrullinated histone H3 and MPO-DNA were also assessed in relation to ANC, DVT, and mortality by analyzing patients exhibiting the highest and lowest values for each factor (n = 4 for each group). The results suggested that severe COVID-19 patients with a higher baseline ANC also had a higher serum NET activity (Figure 4A). The serum NET activity was associated with the risk of mortality in severe COVID-19 hospitalized patients with or without DVT (Figure 4B).
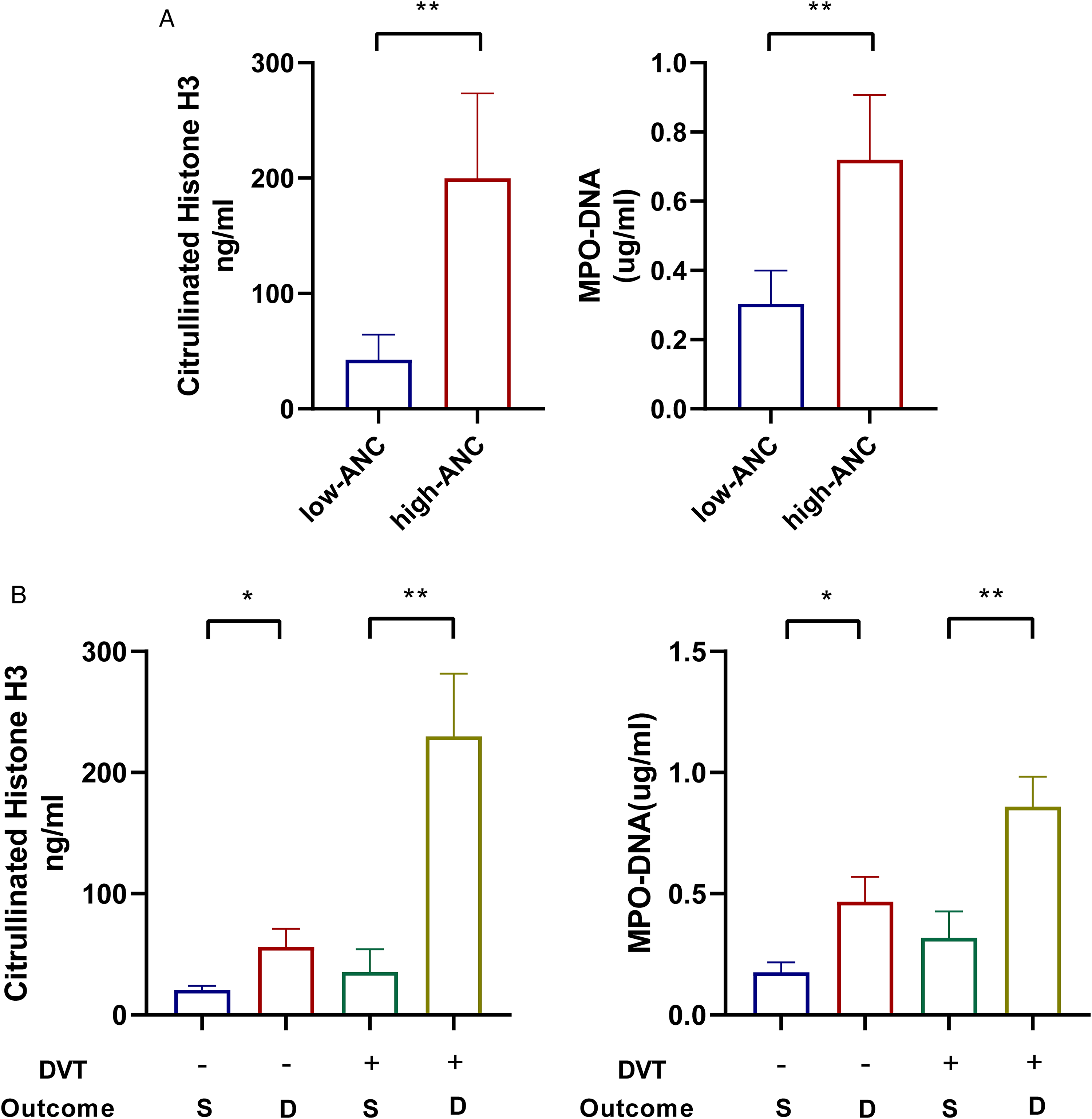
Results of NET activity detection in hospitalized patients with severe COVID-19 (Abbreviation: S = survived; D = deceased.).
Newly Diagnosed DVT Partially Mediates the Association Between Baseline D-Dimer, Baseline ANC, and Risk of Mortality in Hospitalized Patients with Severe COVID-19
The potential mediating role of new-onset DVT in the association between baseline D-dimer/baseline ANC and mortality was further explored in severe hospitalized patients with COVID-19 (Figure 5A). The mediation analysis results are shown in Table S6 and Figure 2. The overall effect of baseline DDI on mortality was 1.18 (95% CI, 1.08–1.28; P < .001), and new-onset DVT (indirect effect 0.011, estimated mediation proportion of approximately 67.0%) partially mediated the association between baseline D-dimer and mortality in severe hospitalized patients with COVID-19 (Figure 5B). The overall effect of baseline ANC on mortality was 1.13 (95% CI, 1.06–1.21; P < .001), and new-onset DVT (indirect effect 0.006, estimated mediation proportion of approximately 48.7%) partially mediated the association between baseline ANC and mortality in severely hospitalized patients with COVID-19 (Figure 5C).
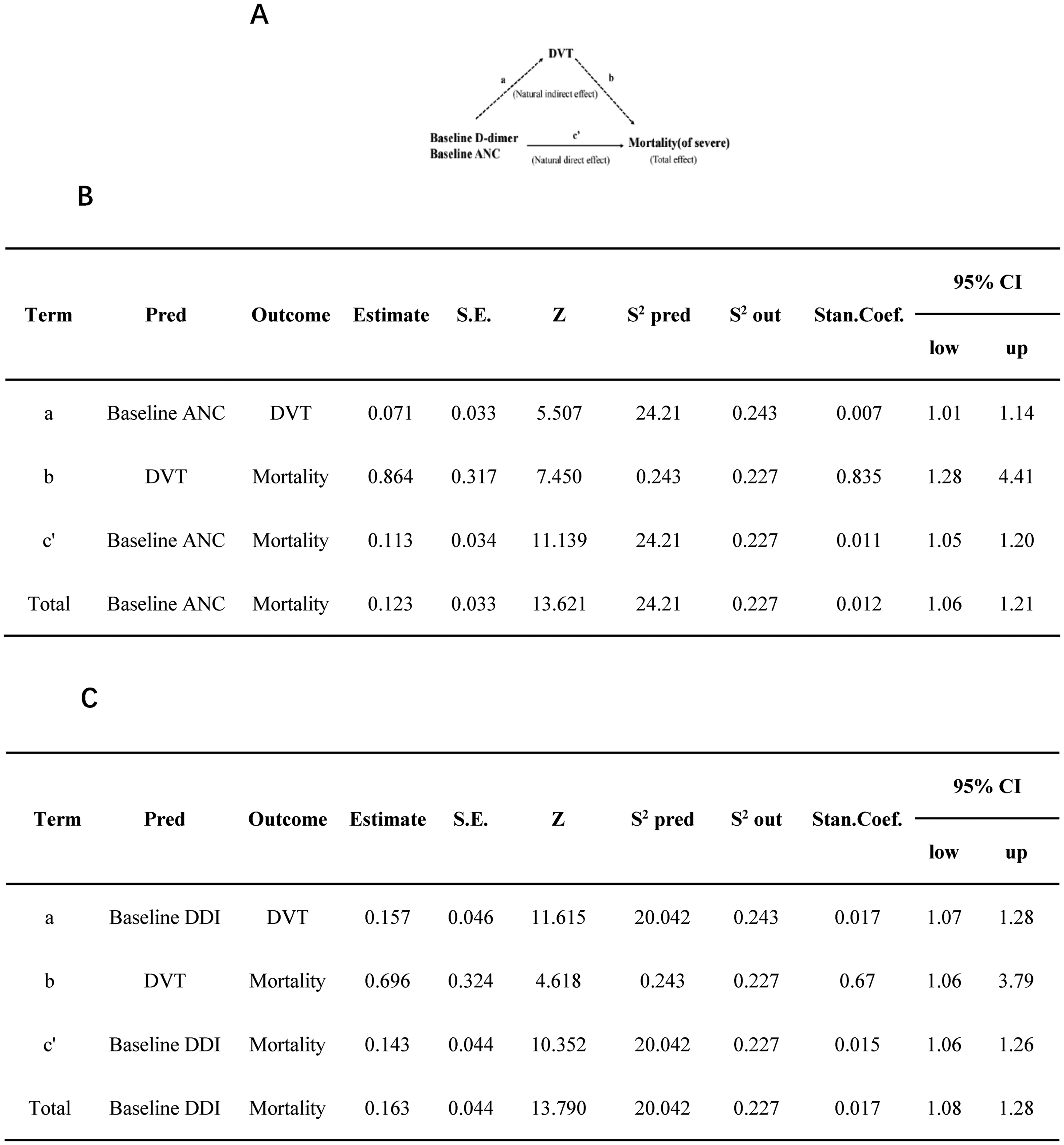
Mediating role of newly diagnosed DVT in correlation with baseline D-dimer, baseline neutrophil absolute value, and risk of death in hospitalized patients with severe COVID-19. (A) Schematic diagram of intermediary analysis; (B) analysis of the mediating effects of newly diagnosed DVT on the association between baseline D-dimer and risk of mortality in hospitalized patients with severe COVID-19; (C) analysis of the mediating effects of new-onset DVT on the association between baseline ANC and the risk of mortality in hospitalized patients with severe COVID-19. Pred: predictors; Out: outcomes; Estimate: estimate regression coefficient; S.E.: standard error; Z: (Wald Chi-squared statistics) Z-statistics; s2 Pred: variance of predictor; s2 Out: variance of outcome; Stan.Coef: standard regression coefficient.
Discussion
In an article published in 2020 by the Tongji Medical College of Huazhong University of Science and Technology in China, a high DVT prevalence was found in COVID-19 inpatients and was associated with poor outcomes. 23 COVID-19 infection is a risk factor for DVT in hospitalized patients, and even prophylactic anticoagulation therapy cannot offset the associated risk of VTE.23,24,25 Patients with severe COVID-19 are at a higher risk of DVT and may develop new and recurrent symptomatic VTE events despite anticoagulant therapy.4,26,27 Considering the severe condition of the patients in this study and the relatively limited medical resources after lifting epidemic control measures, bilateral lower extremity deep vein B ultrasound was selected instead of pulmonary CTPA as a routine screening test for vein thrombosis in hospitalized COVID-19 patients. In our study, 12.2% of moderate patients and 40.8% of severe patients developed new-onset DVT during hospitalization, similar to the incidence reported in previously published studies.2,4 In addition, patients requiring ICU treatment were more likely to develop new-onset DVT. The cumulative incidence of DVT was 54.2% in 83 patients admitted to the ICU within 60 days of admission and 18.9% in 318 patients admitted to general wards (P < .001).
A study published in JAMA in 2020 suggested that the case fatality rate of COVID-19 patients admitted to intensive care units was as high as 40%. 28 In a 2022 research from Saudi Arabia focusing on bleeding and embolism in COVID-19 patients, the risk of mortality in the ICU group was significantly higher than that in the non-ICU group (35.8% vs 2%, P < .001). In the present study, some patients were discharged against medical advice because they were too ill to be cured, dying at home or on the way home. The proportion of patients who died in the ICU group was 62.7%, which was much higher than 7.5% in the non-ICU group. The multivariate analysis confirmed that requiring ICU admission was an independent risk factor for poor outcomes in hospitalized COVID-19 patients. Furthermore, other studies have confirmed that factors related to the risk of mortality and outcome of hospitalized patients with COVID-19 include age, D-dimer, PAP, confirmed VTE, neutrophil, and platelet activation-related indicators, presence of underlying diseases, and concurrent bleeding and arterial embolism.29,30,31,32
Age is an independent risk factor for new and recurrent VTE and is an independent predictor of reduced early survival after VTE.33,34,35 In addition to being related to the incidence of embolic events, age is also associated with bleeding events in medical inpatients.36,37 At present, age has been included as an independent risk factor in the risk assessment of VTE and bleeding in hospitalized patients with acute diseases. In this study, the average age of hospitalized severe COVID-19 patients with DVT (79.73 ± 9.55 years) was higher than that of hospitalized severe COVID-19 patients without DVT (76.58 ± 12.15 years), but the difference was not statistically significant, which may be related to the older age of all admitted severe COVID-19 patients (Table S1). The total length of hospital stay, as one of the outcome indicators of hospitalized patients, is affected by new-onset DVT during hospitalization. Several studies have suggested that patients with new-onset DVT during hospitalization for other diseases have a longer length of hospital stay.38,39 In our analysis, the total length of hospital stay was found to be an independent risk factor for new-onset DVT, which may be related to more serious systemic inflammatory response and longer bed rest time in severe COVID-19 inpatients.
Heart failure (HF) is a common condition in the elderly and can be encountered at all stages of COVID-19. Heart failure itself is an independent predictor of in-hospital mortality, and patients with heart failure have a significantly increased risk of infection due to decreased immunity and decreased hemodynamic regulation ability to cope with more severe infections. 40 In HF patients, monocytes appear to produce more TNF-α than healthy subjects. Moreover, the extensive systemic inflammatory response associated with severe COVID-19 infection requires a higher cardiac output. 41 Acute COVID-19 infection leads to the release of pro-inflammatory cytokines and the recruitment of pro-inflammatory immune cells that mediate the inflammatory storm and increase metabolic demand and coagulation dysfunction, thereby inhibiting cardiac function.2,42 In this study, the underlying hypertension was related to the severity of COVID-19 infection, which was consistent with the results of a previous domestic article. 43 This may be related to the relationship between the COVID-19 virus and the renin–angiotensin–aldosterone system (RAAS). In patients infected with COVID-19, the virus enters the host cells by binding to the catalytic site of ACE2 on the cell membrane, resulting in the shedding of ACE2 from the cell membrane. Therefore, elevated ACE2 levels are observed in plasma, and ACE2 receptors are down-regulated in various organs. Most hypertensive patients have RAAS system disorders, and the use of ACE inhibitors and angiotensin receptor inhibitors increases the vulnerability of the regulation of the RAAS system ordinary infected patients. 44
D-dimer is a soluble fibrin degradation product produced by the orderly breakdown of thrombi by the fibrinolytic system. As a marker of coagulation and fibrinolytic activation, D-dimer testing has an important negative role in the diagnosis of VTE and can reduce the need for imaging in most patients with suspected DVT or PE. 45 In addition, a number of studies have shown that D-dimer has a predictive value for mortality, ICU requirement, and thromboembolic disease in patients with COVID-19.3,24,46,47,48 In the current study, baseline D-dimer was an independent risk factor for new-onset DVT in severe COVID-19 hospitalized patients, and the baseline D-dimer level of COVID-19 severe hospitalized patients with DVT was nearly twice that of patients without DVT (Table S2). Univariate analysis suggested that patients hospitalized with severe COVID-19 who required ICU admission or died (Table 4) had higher baseline D-dimer levels on admission. However, subsequent subgroup analysis showed that baseline D-dimer was only associated with the risk of mortality in severe hospitalized patients with DVT, but not in severe hospitalized patients without DVT. These findings suggested that baseline D-dimer should be comprehensively determined in combination with DVT when predicting the risk of mortality in severe hospitalized patients with COVID-19.
COVID-19 is often complicated by VTE and arterial and microvascular thrombosis considered to be caused by a strong systemic immune inflammatory response and related coagulation abnormalities.5,16 An important mechanism after COVID-19 infection is the recruitment and activation of neutrophils at the infection site, which also predict disease severity and poor outcome. Similar to the activation of NETs following a bacterial infection, various viral infections can also trigger the formation of NETs. Studies have shown that the serum of COVID-19 patients can trigger the release of NETs from the neutrophils of healthy controls in vitro, and surviving SARS-CoV-2 can directly stimulate neutrophils to release NETs in a dose-dependent manner.21,49 Activated neutrophils release NETs and neutrophil microparticles (PMN-MPs), which induce endothelial injury and platelet activation, leading to adverse coagulation dysfunction and immune thrombosis. These changes mediate multi-organ damage in severe hospitalized COVID-19 patients requiring ICU support treatment, affecting the clinical outcome and prognosis.19,50,51 Previous studies have also found no significant difference in PMN-MPs between COVID-19 patients with thrombosis and those without thrombosis, suggesting that COVID-19-related thrombosis involves NETs rather than only neutrophil activation. 52 In this study, the baseline absolute neutrophil count was not only significantly different between moderate and severe patients, but also related various clinical adverse events and outcomes, such as new DVT, ICU admission, and death in severe hospitalized patients with COVID-19. Patients with baseline ANC higher than 6.13 × 109/L and those with new-onset DVT and baseline ANC higher than 6.25 × 109/L have a higher risk of death. The subsequent detection results of NET markers were consistent with the above conclusions, confirming the relationship between the formation of NETs after neutrophil activation and the adverse clinical events/outcomes in hospitalized patients with COVID-19.
Previous literature has shown that the absolute value of lymphocytes in moderate COVID-19 patients is significantly higher than that in asymptomatic patients, but critical COVID-19 patients show an overall decrease in immune cells and lymphocyte subsets.53,54 The lymphocytes, including CD4+ T cells, CD8+ T cells, total T cells, B cells, and NK cells, in critical patients were lower than those in mild and severe patients. However, slightly higher levels of some lymphocyte subsets, such as CD21low CD38low B cells, effector T4 cells, and PD1+ depleted T8 cells, were observed in critical patients compared to mild patients. Recent studies suggested that T lymphocytes can promote the dissolution of vein thrombosis by secreting cytokines to up-regulate the number and activity of macrophages. 55 Meta-analysis results showed that COVID-19 patients with low lymphocyte absolute value had a higher risk of VTE, and patients with elevated NLR (absolute neutrophil/absolute lymphocyte) had a higher median length of hospital stay and higher in-hospital mortality.56,57 Similar to the results of previous studies, the baseline lymphocyte absolute value was an independent protective factor against new-onset DVT in severe COVID-19 patients, which further confirmed the important regulatory role of baseline lymphocyte absolute value on DVT in severe COVID-19 patients.
In addition to antiviral therapy against the virus itself, anti-cytokine therapy is also essential in preventing disease progression and decreasing the risk of mortality in hospitalized COVID-19 patients. Interleukin-6 (IL-6) inhibitors, especially tocilizumab, are the most used and studied anti-cytokine therapy at present. In clinical practice, it is generally used for severe and critical COVID-19 patients with significantly elevated IL-6 levels detected by laboratory tests. In the present study, the baseline IL-6 levels of severe patients were significantly higher than those of moderate patients, suggesting that the inflammatory response in severe patients was more intense than in moderate patients. However, in severe hospitalized patients with COVID-19, the baseline IL-6 level was not significantly related to the risk of 60-day mortality/new-onset DVT during hospitalization, which might be attributed to patients receiving IL-6 inhibitor treatment, which counterbalances the IL-6 mediated systemic inflammatory response and the risk of disease progression. Our team also published research on IL-6 inhibitors. Babaodan (BBD), a proprietary Chinese medicine, can inhibit the excessive immune response of macrophages in vitro. A pneumonia mouse model was established by inducing bacterial infection after viral infection, revealing that BBD can significantly inhibit the release of IL-6, thereby improving the survival rate of mice. 58 Therefore, the anti-hyperimmunity effect of BBD can be considered as a cytokine-targeting drug for the treatment of COVID-19.
Our study proved the relationship between the baseline ANC, baseline D-dimer, and COVID-19 inpatients’ mortality, especially severe inpatients, which could be intermediated by new-onset DVT, the baseline ANC, D-dimer levels, and new diagnosis of DVT should be considered as the key indicators to evaluate the mortality of severe COVID-19 inpatients. Correspondingly, when inflammatory cells and inflammatory molecules are significantly elevated or hypercoagulable and embolic signs are detected in the early stage of admission, relevant anti-excessive inflammation and anticoagulant therapy should be actively given without contraindications.
Nevertheless, the limitations of the present study should be acknowledged. The main viral strains isolated from COVID-19 patients in this epidemic were BA.5.2 and BF.7, and the extent to which different strains affect the outcome and risk of DVT in COVID-19 patients varies. 59 Therefore, the results of this study need to be considered as a reference for the management of patients infected with other subtypes of viruses. Due to the retrospective nature of this study, some indicators may have missing values.
Conclusions
The baseline D-dimer and baseline absolute neutrophil count (ANC) levels were associated with mortality in severe hospitalized patients with COVID-19, especially DVT inpatients. New-onset DVT partially mediated the association between baseline D-dimer, baseline ANC, and mortality in severe hospitalized patients with COVID-19.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241255959 - Supplemental material for What Is the Impact of Baseline Inflammatory and Hemostatic Indicators with the Risk of Mortality in Severe Inpatients with COVID-19: A Retrospective Study
Supplemental material, sj-docx-1-cat-10.1177_10760296241255959 for What Is the Impact of Baseline Inflammatory and Hemostatic Indicators with the Risk of Mortality in Severe Inpatients with COVID-19: A Retrospective Study by Huiqi Zhu, Yeping Li, Hua Su, Chao Yan, Wanghao Xin and Kejing Ying in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations:
ANC: Absolute neutrophil count; APTT: Activated partial thromboplastin; CI: Confidence interval; CitH3: Citrullinated Histone H3; COVID-19: Coronavirus disease 2019; CRP: High-sensitivity C-reactive protein; DDI: D-dimer; DVT: Deep vein thrombosis; ICU: Intensive care unit; IL-6: Interleukin-6; INR: International standard ratio; IQR: Interquartile range; LMWH: Low-molecular weight heparin; LYM: Absolute lymphocyte count; MPO-DNA: Myeloperoxidase DNA; NETs: Neutrophil extracellular traps; NLR: Absolute neutrophil count/Absolute lymphocyte count; NT-proBNP: N-terminal pro-brain natriuretic peptide; OR: Odds ratio; PLT: Platelet count; P/F: Arterial partial pressure of oxygen (PaO2)/ inspired oxygen concentration (FiO2); RDW: Red blood cell distribution width; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; VTE: Venous thromboembolism; WBC: White blood cell count.
Acknowledgements
Authors’ Contributions
HQZ, YPL, and KJY conceived and designed the study. HQZ, YPL, HS, CY and WHX collected information, analyzed the data and prepared figures. HQZ and YPL drafted the manuscript. KJY revised the final manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
The content of this manuscript has not been previously published and is not under consideration for publication elsewhere.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
All human serum samples were retained during routine laboratory testing (residual valid samples). This study was approved by the ethical committee of Sir Run Run Shaw Hospital (Study No. 0264). Written informed consent was obtained from each patient in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Zhejiang Province (Y21H010014) and the Medical Science and Technology Project of Zhejiang Province, China (2022KY841).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
