Abstract
Coronavirus disease 2019 (COVID-19) affects the respiratory system of patients and is characterized by pneumonia with hypoxemia. Hospitalized patients and particularly those admitted to intensive care unit (ICU) may encounter a cascade of coagulopathies, which may lead to macrovessel thrombotic events such as pulmonary embolism (PE), deep vein thrombosis (DVT), or arterial thromboembolism (ATE). These events can result in serious life-threatening diseases including cerebrovascular stroke and myocardial infarction. Despite all available information about the incidence, prevention, and treatment of venous thromboembolism (VTE) among hospitalized patients, few data are available on the incidence of both symptomatic and subclinical VTE after discharge. Therefore, there is no precise suggestion or guideline for prophylaxis against VTE in post-discharge period, and some controversies exist over the current guidelines. In the present study, we aimed to review and summarize available literature upon incidence, prevention, diagnosis, and therapeutic approaches for VTE in COVID-19 patients. Also, the pathogenic mechanisms of VTE in infected individuals with COVID-19 were discussed.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) affects the respiratory system leading to pneumonia with hypoxemia in nearly 20% of coronavirus disease 2019 (COVID-19) patients. 1 COVID-19 is accompanied by an increased risk of symptomatic venous thromboembolism (VTE).2,3 COVID-19 patients admitted to intensive care unit (ICU) are prone to encounter some coagulopathies with hypercoagulable state, such as enhanced fibrinogen concentration, D-dimer levels, and factor VIII activity.4,5 Endothelial injury and subsequent activation of thrombin generation is the main mechanism behind this immunothrombotic phenomenon. 6 This may lead to macrovessel thrombotic events like pulmonary embolism (PE), deep vein thrombosis (DVT), and arterial thromboembolism (ATE).7–13 In addition, pneumopathy-induced local coagulopathies and microvascular thrombosis may occur in patients.14,15 Although the exact incidence of VTE is unknown, large studies have reported an incidence of 6.2% for PE and 9.4% for DVT in COVID-19 ICU-admitted patients. 10 Previous studies have shown that prescription of low-molecular-weight heparin (LMWH), in either prophylactic or therapeutic doses, improves clinical outcomes of hospitalized patients; however, subclinical VTE may occur in these patients.14,16–19 Systematic evaluation of asymptomatic DVT using leg ultrasound has revealed an incidence of 11.9% to 22.5% in non-ICU and 65% to 69% in ICU-admitted patients.20–23
There is little data on the incidence of subclinical and symptomatic VTE after discharge. 2 On the other hand, there is no prophylactic strategy for VTE in post-discharge period. 24 Some studies suggest no prophylactic role; however, others believe in personalized approach according to thrombotic as well as bleeding risk factors.25,26 Moreover, previous epidemiologic studies have shown that up to 80% of all hospital-related VTEs occur after discharge. 27 In the present study, we aimed to review available data on incidence, prevention, diagnosis and treatment of post-discharge VTE in COVID-19 patients.
COVID-19 and VTE: Involved Mechanisms and Processes
Although the pathophysiology of VTE in COVID-19 infection has not been completely uncovered yet, some mechanisms and processes may have a role in this condition. For example, increased levels of hypercoagulability indices, such as D-dimer, fibrinogen, and factor VIII, have been reported in all cases with COVID-19. Also, a decrease in the levels of protein S, protein C, and antithrombin has been implicated. 28 Besides, there are records for the existence of coagulopathy in COVID-19 subjects with higher levels of lactate dehydrogenase, D-dimer, and total bilirubin. 29 Coagulation disorders are linked with VTE, and in this line, several studies have been documented. For instance, an investigation on 115 COVID-19 patients approved SARS-CoV-2 RNA presence in platelets and high cytokine levels related to platelets. 30 Histopathologic results from 38 subjects infected with this novel virus manifested neutrophil extracellular traps (DNA networks originated from extracellular neutrophils), neutrophil-platelet aggregates, and microvascular thrombi. 31 Thromboembolism pathogenesis in COVID-19 than non-COVID-19 ailments may be more dependent on platelets, endothelial inflammation, acquired antiphospholipid antibodies, hypercoagulability related to elevated coagulation factor level, and reduced levels of endogenous anticoagulant proteins.28,32 It is thought that the increased risk of VTE in COVID-19 can be in light of hyperinflammation, disseminated intravascular coagulation (DIC), and immobilization. 8 In the early stages of this viral infection, hypoxia and inflammation along with acute lung injury give rise to a profound inflammatory condition because of cytokine storm, excess, and uncontrolled release of pro-inflammatory cytokines, like interferon gamma (IFN-γ), IL-1, tumor necrosis factor alpha (TNF-α), and interleukin 6 (IL-6) indicating that there is evidence for thrombotic occurrences.33,34 Similarly, this increase in the levels of pro-inflammatory cytokines and chemokines is also observed in Middle East respiratory syndrome coronavirus (MERS-CoV) and severe acute respiratory syndrome coronavirus (SARS-CoV). 35 In other words, when SARS-COV-2 arrives at the alveolar epithelium through the angiotensin-converting enzyme 2 (ACE2) receptor, the secretion of inflammatory cytokines and chemokines occurs ,36,37 which in turn results in the stimulation of epithelial cells, neutrophils, and monocytes (Figure 1). Indeed, the infection of endothelial cells can be occurred by the ACE2 receptor, causing endothelial stimulation and dysfunction, and subsequently coagulation cascade activation that forms fibrin clot and thrombin.38,39 In the lung tissue of infected subjects with COVID-19, there is a report implicating catastrophic microvascular damage along with complement activation components, like C4d and C5b-9, and serine protease related to mannose-binding lectin. 40 Also, macrophage activation syndrome (MAS) can take part in the hypercoagulable situation and cytokine storm observed in COVID-19. This syndrome occurs while the activated antigen-presenting cells have no capability to be lysis through natural killer (NK) cells or CD8 T cells. 41

Some of the involved mechanisms in venous thromboembolism (VTE) related to COVID-19.
Incidence of post-Discharge VTE
Table 1 summarizes the findings of studies which have assessed post-discharge thrombotic events in COVID-19 patients. In a study by London's King College researchers, they evaluated 1877 patients after discharge for a COVID-19-related admission and reported 9 episodes of hospital-associated VTE (HA-VTE) within 42 days which is equal to an incidence rate of 4.8 per 1000 discharges. They compared this rate with the data from 2019 in which 18,159 cases who were discharged following any medical conditions (except for COVID-19) were assessed and found an incidence of 3.1 per 1000 for HA-VTE. As a result, they concluded that hospitalization following COVID-19 does not raise the risk for HA-VTE in comparison with other medical conditions. 42 They have also mentioned that their local protocol provides thromboprophylaxis to COVID-19 patients only during admission and it is withdrawn upon discharge.
Summary of Studies Comparing low-Dose and High-Dose Anticoagulation in the Outcomes of Coronavirus Disease 2019 (COVID-19) Patients.
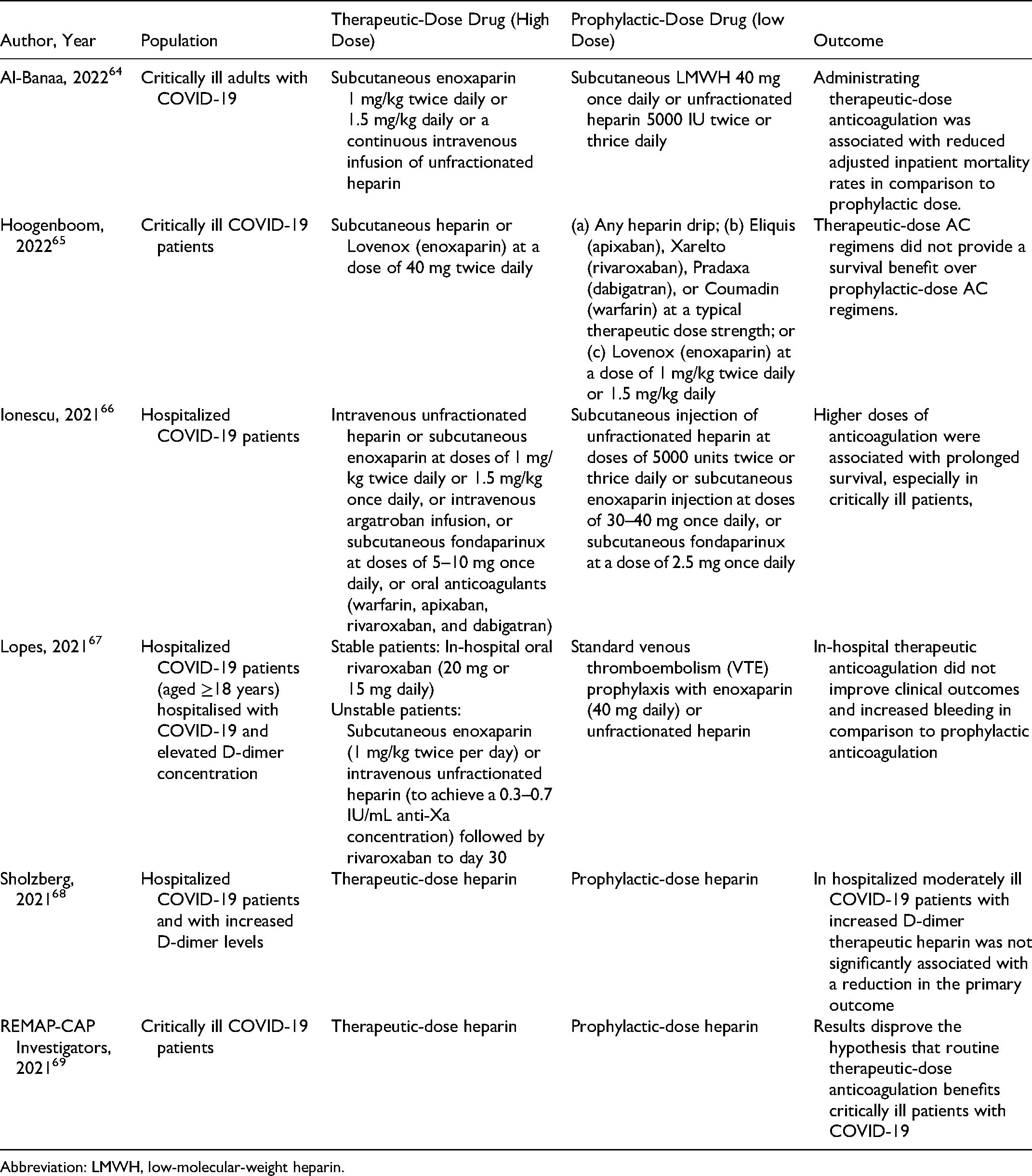
Abbreviation: LMWH, low-molecular-weight heparin.
In a retrospective study, 163 patients with COVID-19 who were discharged without thromboprophylaxis were evaluated. 43 The incidence rate of thrombotic events was 2.5% (4 out of 163) consisting of ischemic stroke, intracardiac thrombus, segmental PE, and thrombosed arteriovenous fistula. The researchers have also reported a median duration of 23 days from discharge to thrombotic event. It is worth mentioning that none of the 13 patients who were discharged with thromboprophylaxis experienced thrombotic or hemorrhagic events.
In a cohort of 146 patients, from Leuven University hospitals (Belgium), who were followed for 6 weeks after discharge from a COVID-19 hospitalization, only 1 case of asymptomatic DVT and 1 case of bilateral PE were reported. 2 They were a 65-year-old female with history of non-active malignancy and a 68-year-old male with severe myopathy and 47 days of ICU hospitalization, respectively. Some patients (except the 2 with VTEs) in this cohort were receiving a post-discharge thromboprophylaxis with low-dose enoxaparin for 2 to 6 weeks after discharge, based on individualized risk assessment. Researchers have concluded that VTE after discharge is infrequent and thromboprophylaxis can be withheld in a majority of patients who have no major risk factors.
In a comprehensive cohort of 1529 COVID-19 patients from major Iranian cites (Tabriz, Zanjan, and Qom), they were followed for at least 45 days after discharge. Researchers reported a cumulative rate of symptomatic VTE of 0.2%. 44 No risk factors have been suggested for post-discharge VTE according to the low incidence rate; however, older age, history of cancer and diabetes were among the early all-cause death. Klok et al. also securitized the incidence of arterial thrombotic complications and VTE in 184 consecutive patients with COVID-19 who had been referred to the ICU. For all patients, the minimum standard-dose thromboprophylaxis was prescribed, and diagnostic assessments were carried out only in suspected patients of VTE clinically. This published report indicated that the incidence of arterial thrombotic events (ATE) and VTE are 3.7% and 27%, respectively. 8
Giannis et al. followed 4906 COVID-19 patients for a mean duration of 90 days. 45 Post-discharge thromboprophylaxis was prescribed in 12.7% of the patients and VTE was diagnosed in 76 patients (1.55%) post-discharge. In addition, ATE was diagnosed in 84 (1.71%) patients.
Diagnosis of COVID-19-Related VTE
Reviewing the literature, we realized that there are no united guidelines upon diagnosis of VTE in COVID-19 patients, neither hospitalized nor following discharge.
In King's college hospitals, physicians do not perform any routine screening of asymptomatic patients. In case of clinical suspicion, D-dimer and pretest probability are conducted prior to VTE imaging. 42 Any VTE which is diagnosed after a surgical operation or following a 2-day hospitalization, and occurs within 90 days of discharge, is considered as a HA-VTE.
Regarding imaging options, venous duplex ultrasound was used to systematically screen DVT in all the patients in Leuven study. 2 Also, in patients suspected of PE, other imaging explorations, such as echocardiography and computed tomography pulmonary angiography (CTPA), are performed. 46 PE and DVT diagnosis are challenging in subjects with COVID-19. PE symptoms overlap with COVID-19 symptoms, and mild symptoms may fail to observe in a case that already had suffered breath shortness. In parallel, the detection of clinical manifestation of DVT may be more difficult in COVID-19, and while curing clinicians mainly focus on the respiratory situation and do not evaluate lower extremities systematically for DVT signs. 47 Serum biological and inflammatory markers can be considered as other complementary options for predicting post-discharge VTE, beside clinical manifestations. Association of the higher levels of D-dimer with poor clinical outcomes, and also its correlation with thrombotic complications, resulted in early suggestions about the role of D-dimer levels on decision-making for clinical management of patients with COVID-19.48–50 A prospective cohort have shown that patients with SARS-Cov-2 infection have sustained elevations in D-dimer levels and this occurs more commonly in older patients and those with severe acute disease. 51 The same study has suggested that the elevated D-dimer levels cannot explain the systemic coagulation activation or the low-grade DIC, as there are no links between D-dimer and other markers of coagulation or inflammation (eg, CRP). In Engelen et al. study, patients in ICU had significantly higher D-dimer levels than those on ward, both during hospitalization and at the time of discharge. 2 However, the difference between D-dimer level was no longer significant for 6 weeks after discharge. Interestingly, the trajectories of D-dimer level from discharge to follow-up point were not significantly different between patients with and without post-discharge thromboprophylaxis. 2 In sum, it seems that the predictive role of D-dimer for COVID-19 coagulopathies in post-discharge setting has remained unknown. 52 In case of a D-dimer level higher than 2000 ng/mL or clinical suspicion, CT pulmonary angiogram or ventilation/perfusion (V/Q) scan can also be performed. In the viewpoint of laboratory tests, coagulopathy assessment, particularly hypercoagulopathy, is hard to achieve by common coagulation tests, which comprise evaluation of platelet count, prothrombin time (PT), fibrinogen levels, and activated partial thromboplastin time (APTT). These landmarks can be prognosis factors for VTE associated with COVID-19. Viscoelastic hemolytic assessments, like rotational thromboelastometry (ROTEM) and thromboelastography (TEG), have more validity for coagulopathy detection compared with common coagulation tests.53,54
Addition of tissue-type plasminogen activator (tPA) to standard ROTEM is a new diagnostic entity which has been used by a small study with 22 COVID-19 patients. 55 This is a viscoelastic assay method which measures changes in viscoelastic parameters during clot formation and fibrinolysis. 56 Researchers conducted routine hemostasis tests as well as tPA ROTEM during ICU admission and 6 months after ICU discharge. They reported that tPA ROTEM measures were returned to normal values at the sixth month of discharge; however, D-dimer and fibrinogen levels were remained elevated. Gibson et al. and Spyropoulos et al. studies have assessed the risk of VTE using the International Medical Prevention Registry on Venous Thromboembolism (IMPROVE)-DD VTE score which has been validated for patients both with and without COVID-19.57,58 Figure 2 summarizes the approaches of diagnosing VTE related to COVID-19.

Flowchart of diagnostic methods for venous thromboembolism (VTE) related to COVID-19.
Prevention of COVID-19-Associated VTE (Thromboprophylaxis)
King's college hospitals have a protocol of intermediate-dose enoxaparin for patients who are admitted to critical care with COVID-19 from April 22, 2020; however, this is stopped at the time of discharge. 42 In addition, patients receive recommendations upon symptoms of VTE, at their discharge summary. Researchers have also mentioned that a short course of extended thromboprophylaxis could have decreased the incidence of post-discharge VTE by about 40% to 50%, as all the post-discharge events were occurred at early phase. 59 However, this may increase the risk of bleeding about 2 to 3 times and this is the reason that it has not been considered as a standard protocol so far. 60
Belgian society of Thrombosis and Hemostasis and the local guideline of the Leuven University hospitals, consider prophylaxis with 0.5mg/kg daily enoxaparin in the ward and 0.5mg/kg twice daily in the ICU setting.61,62 As for the other healthcare facilities, the guidelines for post-discharge prophylaxis are lacking in Leuven. So, it was prescribed for patients with known risk factors, such as atrial fibrillation, obesity, VTE, heart failure, aged over 70 years, and mechanical heart valve, with 0.5mg/kg daily enoxaparin for 2 to 6 weeks. No major bleeding events were reported in patients who received post-discharge thromboprophylaxis. In the Iranian study, it was also concluded that the routine prescription of extended thromboprophylaxis may not lead to an evident clinical benefit. 44 American guidelines have already restricted the routine extended thromboprophylaxis for medically ill patients, and it seems that these recommendations can be also applied in post-discharge period of COVID-19 patients. 63
Giannis et al. attached to their local guideline on the extended thromboprophylaxis with direct oral anticoagulant (DOAC) (rivaroxaban 10 mg orally) or LMWH (enoxaparin 40 mg subcutaneous daily) in COVID-19 discharged patients. This regimen was prescribed for 30 days following discharge if the patient had an IMPROVE VTE score of higher than 3 or a D-dimer level of twofold as upper limit of normal. 45 Several studies have also compared the effects of thrombophylactic anticoagulation (low dose) and therapeutic anticoagulation (high dose). Generally, therapeutic doses of anticoagulation were seemed to decrease mortality. However, in some studies, there were no significant differences found. Furthermore, some studies that had found statistically significant differences in mortality rates between the 2 groups were not able to associate that mortality rate to thrombotic events.64–69 Table 1 summarizes the studies comparing low-dose and high-dose anticoagulation in hospitalized COVID-19 patients.
Ramacciotti et al. conducted a study on hospitalized COVID-19 patients who were more at risk for post-discharge thromboembolism. All patients had received thromboprophylaxis with standard doses of heparin while they were hospitalized. Following discharge, patients randomly received rivaroxaban for 35 days or no anticoagulation following discharge. Patients who received extended thromboprophylaxis had better clinical outcomes in comparison to patients who didn’t receive extended prophylaxis. 70 Moreover, in the severe stage of COVID-19, the utilization of intermittent pneumatic compression (IPC) has been proposed in order to prevent VTE (Figure 3). 46 IPC is described as a mechanical device that diminishes VTE by the elevation of blood flow speed and reduction of hypercoagulability by the IPC performance inducing fibrinolytic function of vessel walls 71 (Figure 3).
Flowchart concerning methods of prevention of venous thromboembolism (VTE) related to coronavirus disease 2019 (COVID-19).
Therapeutic Options for COVID-19-Associated VTE
Several therapeutic approaches have been recommended for VTE related to COVID-19. Anticoagulation treatment is known as the main therapy for patients with VTE. The current guidelines suggested that anticoagulation agents should be administrated in suspected patients with intermediate or high clinical possibility when awaiting the findings of diagnostic tests, particularly in PE cases. 72 In the case of the parenteral way having priority for treatment, weight-regulated therapeutic-dose LMWH can be the first selection, whereas unfractionated heparin (UFH) infusion or subcutaneous fondaparinux can be utilized as an alternative approach. 37 UFH, as an anticoagulant agent, hamper the production or extension of blood clots. UFH can bind to the enzyme inhibitor antithrombin, and thus, neutralizes the impact of thrombin and other involved coagulation factors in blood clotting, namely X, IX, XI, and XII. 73 This anticoagulant needs to be infused continuously or frequently because of its short half-life, around 1 h. 74 But, LMWH has more half-life, 4 to 5 h, which can be dosed once daily. Heparin can create serious complications, such as heparin-induced thrombocytopenia (HIT), a potentially life-threatening circumstance. 75 Another factor binding to antithrombin is fondaparinux which does not suppress thrombin, unlike heparin. In fact, fondaparinux binding to antithrombin inhibits factor Xa, which in turn leads to coagulation suppression. 76 It also has more half-life compared with heparin and can be prescribed once daily by subcutaneous injection. 77 Using fondaparinux is not accompanied by HIT because it does not interplay with platelets. 78 However, this factor Xa inhibitor is more expensive in comparison with the heparin so its use may be limited. 75 Some documents stated that fibrinolytic therapy can be used as a potential tool in the treatment of systemic thrombotic events associated with COVID-19. However, it is expressed that this type of therapy in severe COVID-19 may create an increased risk of hemorrhagic occurrences, consisting of diffuse alveolar hemorrhage and intracranial hemorrhage. 79 Recently, harnessing some immunotherapeutic agents, like monoclonal antibodies, have been offered in COVID-19-associated VTE. 80 Monoclonal antibodies are formed by white blood cells exposed to a specific viral protein, which subsequently is cloned to create antibodies targeting viruses. In addition to COVID-19, these antibodies were previously used to treat some viral infections, for example, rabies and Ebola. 81 Monoclonal antibodies have a potential for inhibition of several coagulation factors, such as tissue factor, factor VIII, and factor IX. 82 Another therapeutic choice for COVID-19 cases with coagulopathy disorders is using P2Y12 inhibitors. 83 The P2Y12 receptor, which is classified in the subgroup of Gi-coupled adenosine diphosphate receptors, has a substantial role in the aggregation and activation of platelets. Thus, the suppression of the receptor can have a positive role in thrombotic event treatment. 84 Despite this, the risk of bleeding in this therapeutic candidate is present. 83
Conclusion
In conclusion, we found that there is no united reliable guideline for post-discharge thromboprophylaxis in patients with COVID-19-related hospitalization. In addition, it seems that extended thromboprophylaxis for patients with COVID-19 is not of vital necessity due to the low incidence rate of post-discharge VTE syndromes which are comparable with other medical conditions than COVID-19. Furthermore, we realized that D-dimer and inflammatory markers does not seem to play an important role as predictive indicators of post-discharge thrombosis. However, all the related studies believe that there is a need for more structured clinical trials and prospective cohorts to make physicians able for more precise decision making.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
