Abstract
Purpose
Ticagrelor is an antiplatelet drug, and its use increases the risk of bleeding. Coronary artery disease is significantly influenced by the widespread occurrence of diabetes mellitus. In order to decrease the incidence of clinical adverse events, a novel bleeding and thrombosis score is developed in this research.
Methods
We conducted a retrospective analysis of patient data from two medical centers who were diagnosed with diabetes mellitus and treated with ticagrelor. We gathered information on every patient from the electronic database of the hospital and follow-up. The collected data were statistically analyzed to obtain risk factors for bleeding and ischemic events.
Results
A total of 851 patients with diabetes mellitus who have been administered ticagrelor are included in our investigation. A total of 76 patients have bleeding events and 80 patients have ischemic events. The analysis of multiple variables indicates that characteristics like the age of >65, having a previous occurrence of bleeding, experiencing anemia, using aspirin, and taking atorvastatin are linked to a higher likelihood of bleeding. Additionally, the age of >65, smoking, having a history of blood clots, and having a BMI ≥ 30 are found to increase the risk of ischemia.
Conclusion
The A4B score established in this study was better than the HAS-BLED score,and the same is true for the ABST score to the CHA2DS-VASc score. This new risk assessment model can potentially detect patients who are at high risk for bleeding and ischemic events. For high-risk patients, the dose of ticagrelor can be adjusted appropriately or the medication can be adjusted.(2023-09-11, ChiCTR2300075627)
Introduction
There is an increasing incidence of coronary artery disease associated with diabetes mellitus (DM). The presence of DM in individuals with pre-existing atherosclerosis significantly amplifies the likelihood of ischemic events occurring in a synergistic fashion. 1 Patients with stable coronary artery disease, as well as those who have experienced ischemic events, face an elevated cardiovascular (CV) risk due to diabetes. Consequently, a significant proportion of diabetic individuals necessitate the placement of stents through percutaneous coronary intervention (PCI). In the context of secondary prevention, a combination of aspirin and one of the more recent and powerful ADP P2Y12 receptor inhibitors are employed alongside dual antiplatelet therapy (DAPT). 2
A potent and reversible platelet P2Y12 receptor antagonist, ticagrelor inhibits an amplification pathway directly and indirectly through active metabolites in platelets. Following the administration of ticagrelor twice a day, there is a rapid initiation and cessation of antiplatelet effects that remain constant in the bloodstream and can potentially be reversed. Ticagrelor, possessing inverse agonist characteristics and diverse impacts, plays a role in promoting antithrombotic, anti-inflammatory, and vasodilatory effects. The favorable clinical results in acute individuals have been attributed to the benefits of ticagrelor. 3 Numerous studies have been conducted both domestically and internationally on dual antiplatelet therapy using ticagrelor for atherosclerotic diseases,4–9 as well as ticagrelor monotherapy.10–14 Diabetes, however, may impair antiplatelet therapy's effectiveness. In people with diabetes, platelet turnover appears to be accelerated, so young platelets that don’t respond to suppressive drugs may appear more frequently in circulation. Meanwhile, the enteral absorption of any drug in diabetics is affected. Due to this, the drug may not reach the concentration required to inhibit platelets in vivo, resulting in ischemic events. Using ticagrelor may cause adverse reactions like bleeding, and the most frequently reported adverse reactions in PLATO study are dyspnea, contusion and nosebleed. Following the administration of ticagrelor, the annual incidence of significant bleeding is 11.6%, while the annual incidence of severe fatal or life-threatening bleeding is 5.8%. Moreover, the combined occurrence of major and minor bleeding is greater in the ticagrelor group compared to the clopidogrel group (16.1% vs 14.6%, P = .0084). Bergmeijer TO's study indicates that 30.2% of patients have bleeding events within one year of using ticagrelor. 15 In diabetic patients, angiogenesis function is weakened, with elasticity decreased and brittleness increased, while rupture risk is increased, leading to bleeding events.
This study aims to investigate the risk factors associated with bleeding and ischemic events in diabetic individuals following the administration of ticagrelor. Additionally, it aims to develop a predictive scoring system for assessing the likelihood of bleeding and ischemic risks in diabetic patients. The study will differentiate patients based on their respective scores for bleeding and ischemic events. Customized management can be implemented to decrease the occurrence of bleeding and ischemic occurrences. According to individual characteristics, the duration of treatment will be shortened to ensure that ticagrelor could prevent thrombus and reduce the incidence of cardiovascular events at the same time, so as to achieve a balance between benefits and adverse reactions and to explore the individualized treatment strategy of ticagrelor.
Methods
Study Design and Population
From January 2020 to June 2021, we conducted a retrospective analysis of patient data from two medical centers (Zhongshan Hospital (Xiamen), Fudan University and Zhongshan Hospital, Fudan University) who were diagnosed with diabetes mellitus and treated with ticagrelor (90 mg bid po). In addition, we prospectively collected data of diabetic patients who received ticagrelor (90 mg bid po) at Zhongshan Hospital of Fudan University between July 2023 and October 2023 for score verification. We obtained data from the inpatient medical records system for patients. In the process of hospitalization, we had been fully communicating with patients to obtain their informed consent, and then contacting and following up with patients through the Internet or telephone to obtain relevant data (Figure 1).
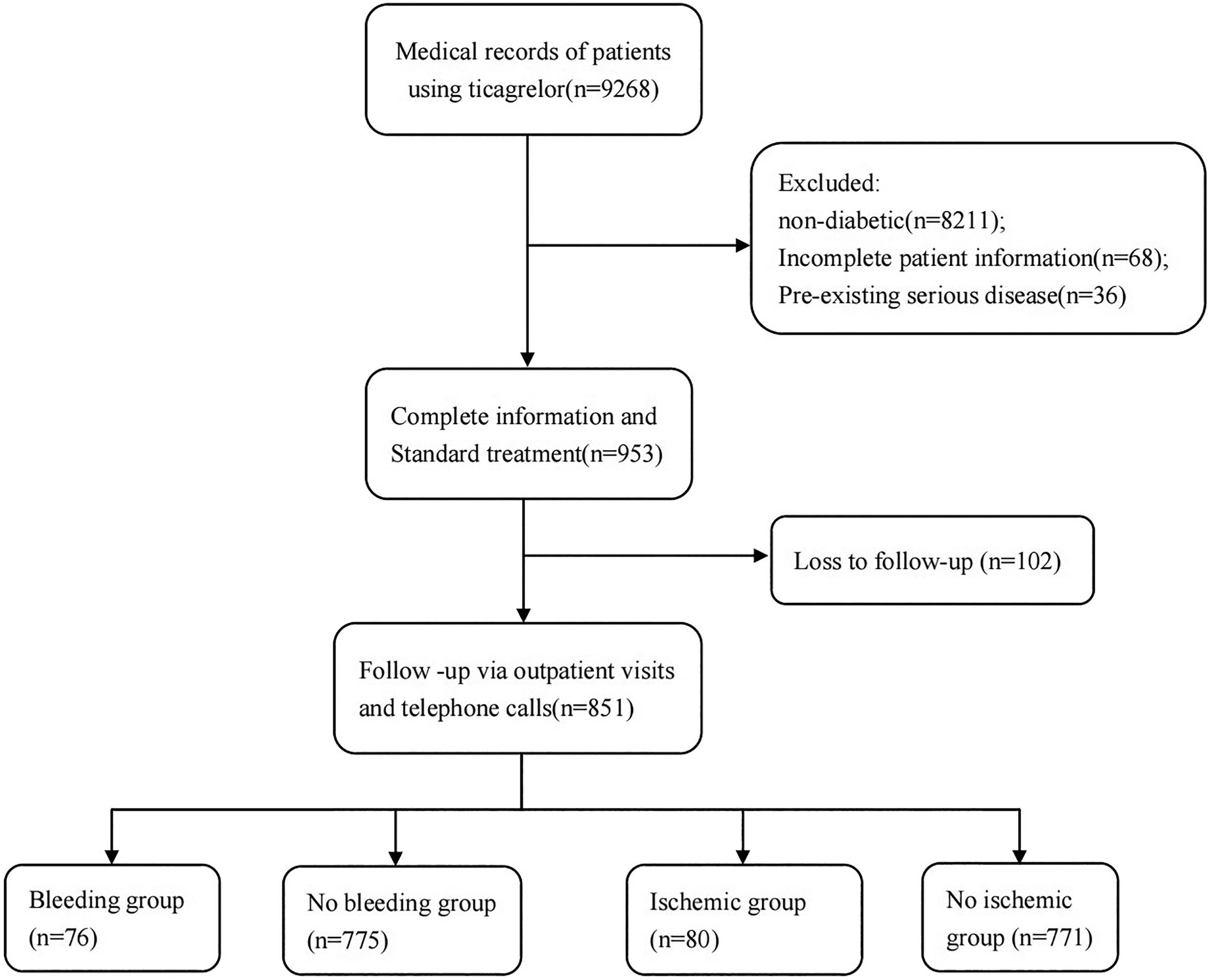
Flowchart of patient screening.
Exclusion criteria: (1) an age of <18 years; (2) Insufficient data preventing subsequent analysis; (3) Follow-up lost or less than 6 months.
The pilot program have received approval from the Ethics Committee of Zhongshan Hospital (Xiamen), Fudan University and Zhongshan Hospital, Fudan University (Ethics B2022-069; B2023-045). Through telephone follow-up, all study participants have provided their informed consent.
Data Collection
We gathered information on every patient from the electronic database of the hospital. Healthcare providers efficiently collected and documented information promptly, which encompassed details such as personal characteristics (eg, age and gender), concurrent medical conditions (eg, high blood pressure, kidney dysfunction, diabetes, previous instances of bleeding, and low red blood cell count (defined as hemoglobin levels below 13 g/dl in males or 12 g/dl in females), and additional prescribed drugs (eg, atorvastatin, aspirin, antidiabetic drug).
Endpoints and Follow-Up
For approximately 6-24 months, patients were followed up using both the clinic and telephone. Regular outpatient follow-ups were necessary to effectively monitor symptoms and clinical signs. Patients displaying a propensity to bleed were advised to undergo additional evaluation. The primary results included occurrences of bleeding events and ischemic events.
Major hemorrhage encompasses lethal hemorrhage, bleeding that poses a threat to life (surgical intervention necessitating ≥3 units of blood transfusion, permanent consequences), and profound bleeding (needing treatment or assessment like gastrointestinal bleeding, joint hematoma, retroperitoneal bleeding, basal bleeding, hematuria, hemoptysis, etc, necessitating ≥2 units of blood transfusion).
Secondary bleeding includes mild nosebleed, endoscopic hematuria, skin stasis, mild hemorrhoid bleeding, etc Patients will not have serious bleeding consequences.
Thrombosis occurrences encompass fatality, heart attack, clotting within a stent, brain attack, or the need for target vessel revascularization (TVR), among others.16,17
Statistical Analysis
The analysis of data is conducted using SPSS 25.0 software provided by IBM Corp. located in Armonk, NY, United States. A continuous variable is measured by its mean ± standard deviation or median, whereas a categorical variable is measured by its percentage. Pearson's χ2 test or Fisher's exact test are used for analyzing categorical variables individually. An analysis of the univariate variables associated with hemorrhage or ischemic damage analyzed by logistic regression confirms whether they are independent predictors (P < .20). The final model will include those with a significance level of .05. Based on the coefficients in the model, every predictor is assigned a weight. The evaluation of risk scoring models involves the calculation of the area under the curve (AUC) for the bleeding group versus the nonbleeding group (1:1) and the ischemic group versus the non-ischemic group (1:1). The assessment of the model's calibration ability is conducted using the Hosmer-Lemeshow test. Statistical significance will be considered if the p value is less than or equal to .05.
Results
Patient Characteristics
There are a total of 955 patients in this study,104 of whom were used for validation of the score (Supplemental Material Table 1). Table 1 and Supplemental Material Table 2 shows the baseline characteristics of the patients in the modeling group. Baseline includes patients’ basic information (eg, height, weight, age, etc), concomitant diseases (hypertension, anemia, kidney insufficiency, etc), and concomitant medications (aspirin, atorvastatin, etc).
Baseline Clinical Characteristics of Patients.
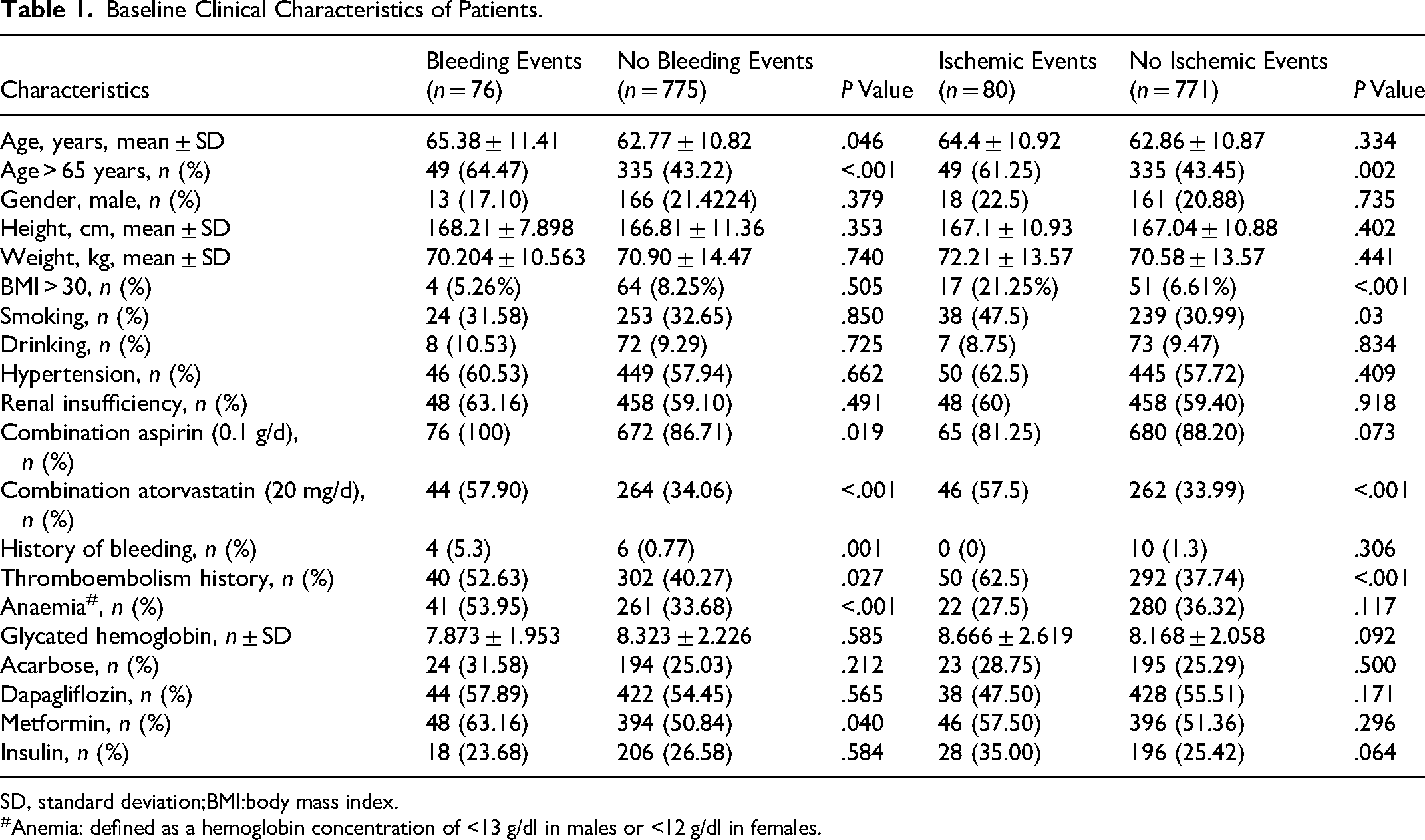
SD, standard deviation;BMI:body mass index.
Anemia: defined as a hemoglobin concentration of <13 g/dl in males or <12 g/dl in females.
Establishment and Evaluation of Bleeding Risk Factors and Risk Scoring Model
The analysis of a single variable reveals that bleeding is linked to age >65 years (P < .001), previous bleeding episodes (P < .001), history of thromboembolism (P < .001), presence of anemia (P < .001), creatinine levels (P = .043), hemoglobin levels (P = .048), use of aspirin (P = .031), use of clopidogrel (P = .002), and use of atorvastatin (P < .001), glycated hemoglobin (P = .517), metformin (P = .693), acarbose (P = .797), insulin (P = .656), dapagliflozin (P = .865). The bleeding risk scoring model is established through the analysis of multiple factors, which includes age >65 years, previous instances of bleeding, anemia, as well as the use of aspirin and atorvastatin. The A4B score is given to the risk factors based on the regression coefficient of the final model (Table 2). The final regression coefficient determines that factors such as the age of >65, having a history of bleeding, experiencing anemia, taking aspirin, and using atorvastatin are given scores of 1, 2, 1, 1.5, and 1, respectively. We define a score ≤1 as a low risk group, 2-4 is defined as a moderate risk group, and ≥4 is defined as a high-risk group. Following a 24-month observation period, significant variations in bleeding incidents (P < .05) have been observed, indicating that the high-risk group has approximately 11 times greater likelihood of experiencing bleeding compared to the low-risk group.
Bleeding Risk with a Univariate Analysis.

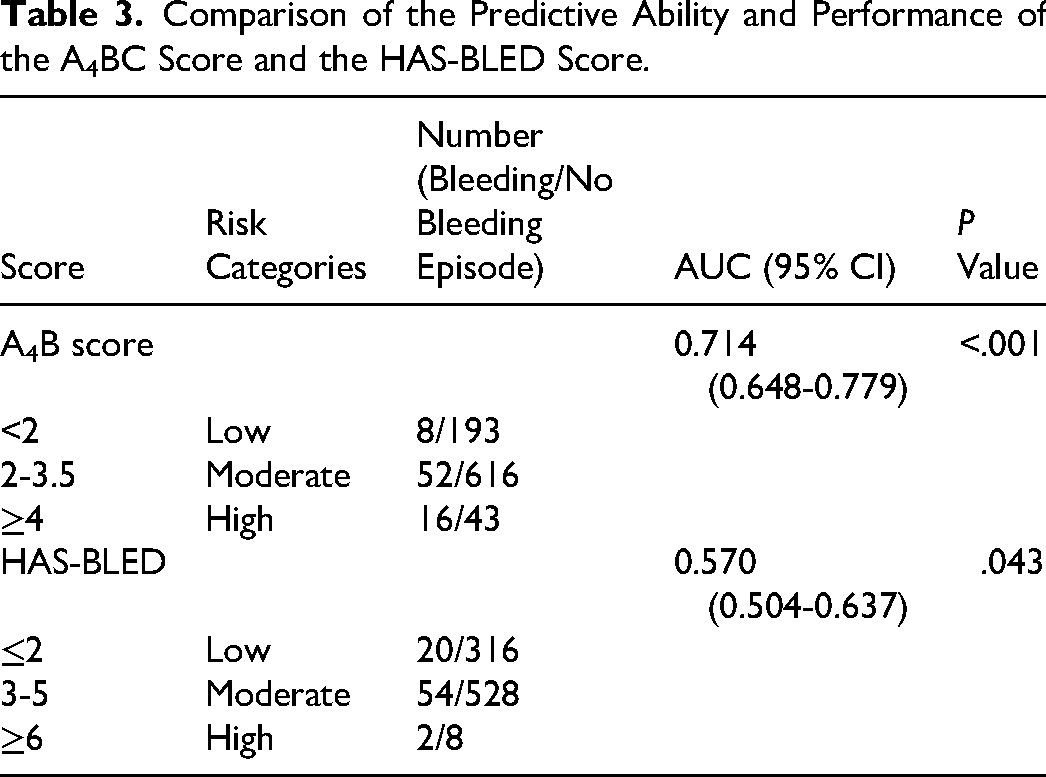
There is a higher AUC of A4B score of 0.708 than the AUC of HAS-BLED score (0.570, 95% CI 0.504-0.637;P = .043 Table 3). The P-value of the Hosmer-Lemeshow test is .200. According to the Hosmer-Lemeshow test, the risk scoring model exhibits satisfactory calibration ability with a P-value greater than .05 (Figure 2).
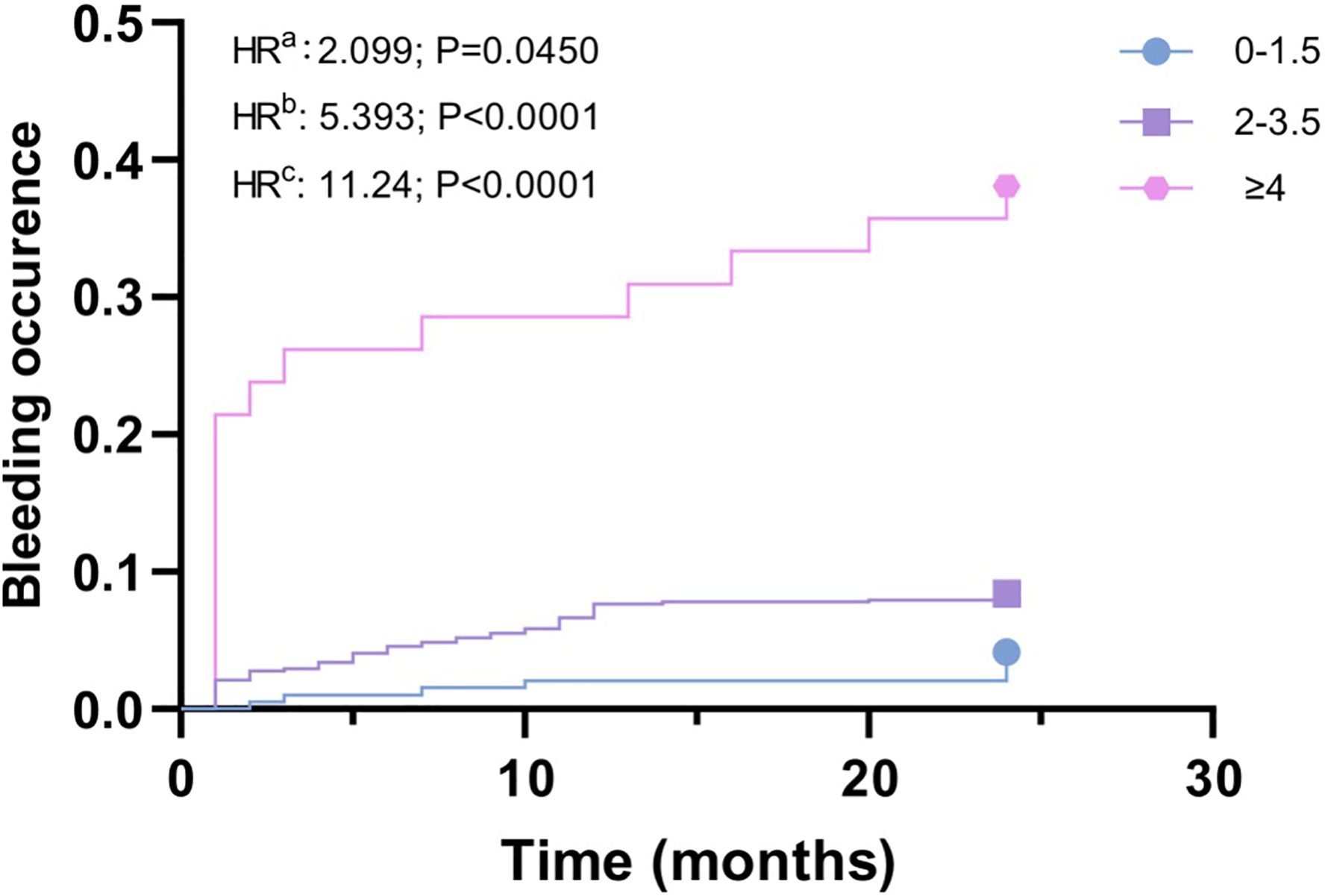
Time to bleeding (TTB) in ticagrelor treated patients, stratified into three subgroups (low risk group, moderate risk group, high risk group) according to the A4B scores. a Referred as the comparison between Low risk group and Moderate risk group. b Referred as the comparison between Moderate risk group and High risk group. c Referred as the comparison between Low risk group and High risk group.
Comparison of the Predictive Ability and Performance of the A4BC Score and the HAS-BLED Score.
Establishment and Evaluation of Ischemic Risk Factors and Risk Scoring Model
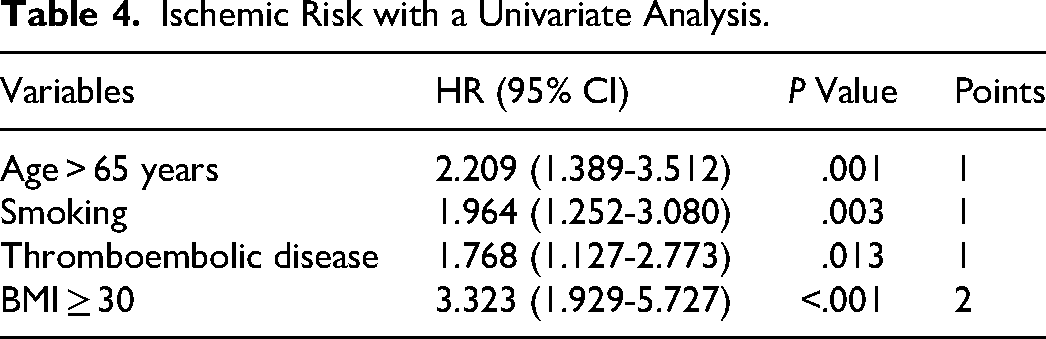
The univariate analysis reveals that risk factors for ischemic events include an age of >65 (P = .001), smoking (P = .004), having thromboembolic disease (P < .001), abnormal INR (P = .004), taking atorvastatin (P = .050), and having a body mass index (BMI) ≥ 30 (P < .001), glycated hemoglobin (P = .287), metformin (P = .752), acarbose (P = .825), insulin (P = .564), dapagliflozin (P = .952). Through multi-factor analysis, four risk factors are used to establish ischemic risk scoring model, including an age of >65 years, smoking, thromboembolic disease, BMI ≥ 30. Based on the regression coefficient of the ultimate model, we allocate scores to the risk factors (Table 4), which are referred to as the ABST score. Based on the final regression coefficient, the age of >65, smoking, thromboembolic disease, BMI ≥ 30, are assigned 1, 1, 1 and 2 points, respectively. We define a score 0-1 as a low risk group, 2-3 as a medium risk group, and 4-5 as a high risk group. After 24 months of follow-up, there are differences in ischemic events between groups (P < .05), that the risk of thrombus in the high-risk group is about 15 times than that of the low-risk group.
Ischemic Risk with a Univariate Analysis.
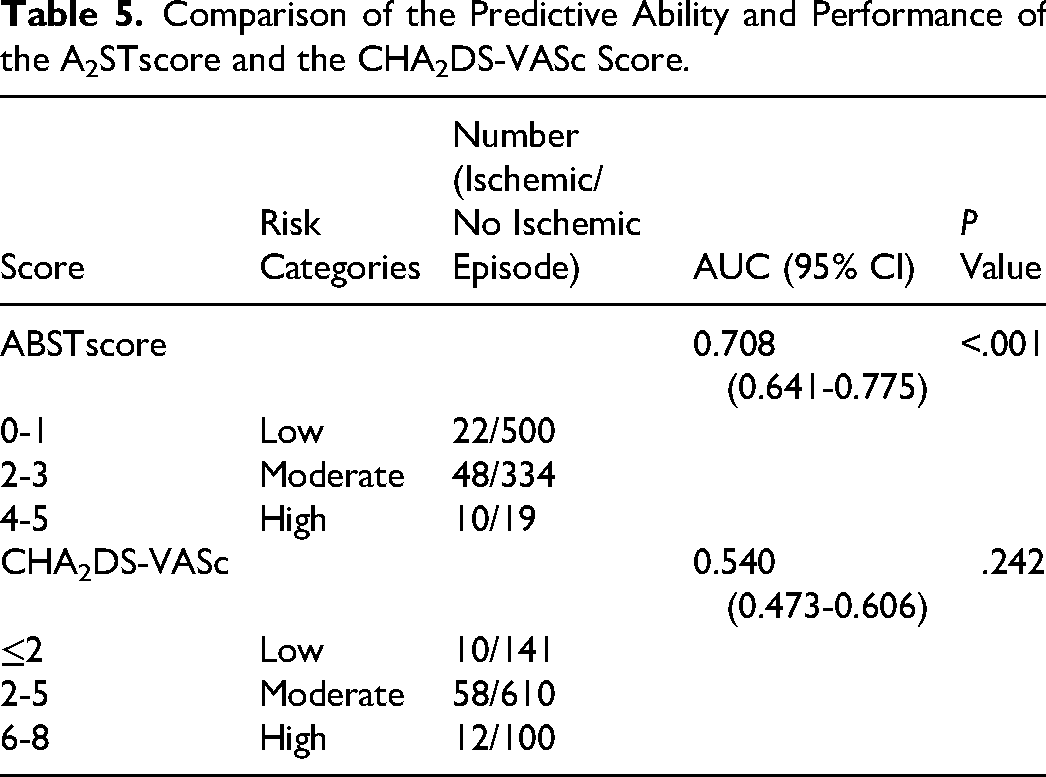
There is a higher AUC of ABST score of 0.708 than the AUC of CHA2DS-VASc (0.540, 95% CI 0.473-0.606;P = .242 Table 5). The P-value of the Hosmer-Lemeshow test is .149. According to the Hosmer-Lemeshow test, the risk scoring model exhibits satisfactory calibration ability with a P-value greater than .05 (Figure 3).
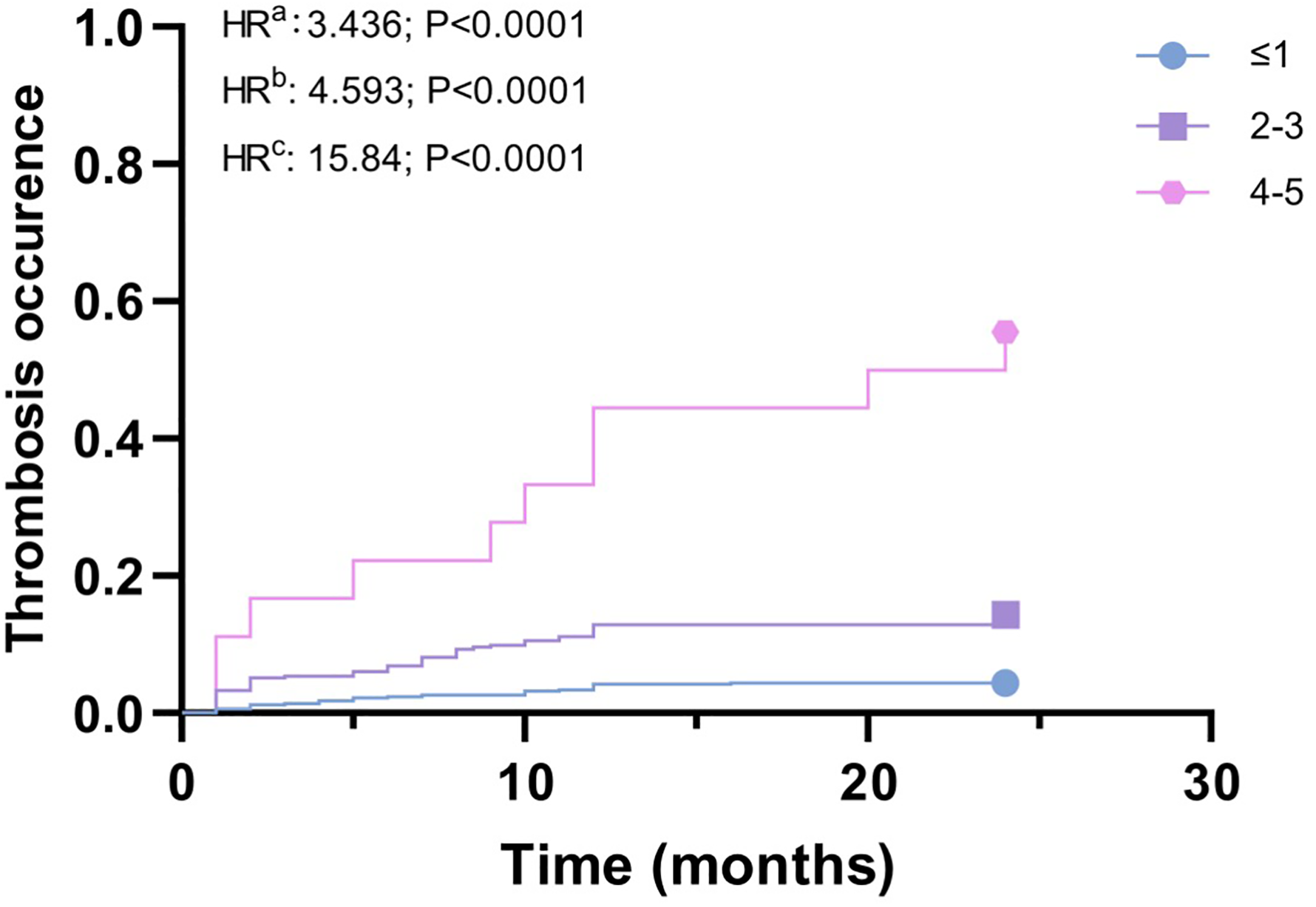
Time to thrombosis (TTT) in ticagrelor treated patients, stratified into three subgroups (low risk group, moderate risk group, high risk group) according to the ABST scores. a Referred as the comparison between Low risk group and Moderate risk group. b Referred as the comparison between Moderate risk group and High risk group. c Referred as the comparison between Low risk group and High risk group.
Comparison of the Predictive Ability and Performance of the A2STscore and the CHA2DS-VASc Score.
Validated Model
Medication education was given to these patients when they were discharged from hospital, and the questionnaire of Morisky Medication Adherence Scale (MMAS-8) was filled out recently to evaluate the compliance. 89.42% of the patients could achieve 6 points or above, 69.23% could achieve 8 points or above, and 10.58% of the patients had poor compliance. The results of compliance were statistically analyzed with bleeding and embolization events, which were P > .05. Patient information was brought into A4B score and ABST score, and ROC analysis was performed. AUC of A4B score was 0.849 (95% CI 0.749-0.949; P < .001). The AUC of ABST score was 0.898 (95% CI 0.747-1.0; P < .001) (Supplemental Material Figures 1 and 2). These results indicated that the A4B score and ABST score exhibited a high level of precision in its predictions.
Discussion
The presence of diabetes mellitus is a major risk factor for atherosclerosis. Studies have shown that individuals with diabetes exhibit elevated platelet activity and adhesion, leading to a greater occurrence of cardiovascular events and diminished effectiveness in preventing blood clot formation.18,19 Patients taking ticagrelor long-term for antiplatelet treatment, due to the role of the drug, may lead to the occurrence of bleeding events. Its resulting gastrointestinal hemorrhage, cerebral hemorrhage and other diseases are often fatal to patients. Our research focuses on examining the negative incidents that take place while diabetic patients are using ticagrelor. The aim is to investigate the factors that contribute to bleeding and ischemic events, and develop a scoring system to assess the risk of bleeding and thrombosis in diabetic patients who are prescribed ticagrelor. The examination of both single and multiple sets of data indicates that variables such as the age of >65, a previous occurrence of bleeding, anemia, the use of aspirin and atorvastatin can elevate the likelihood of experiencing bleeding. Factors like age of >65 years, smoking, thromboembolic disease, BMI ≥ 30 can increase the risk of thrombus. Therefore, if a patient's score is high, which means that the patient has a greater risk of bleeding or thrombosis, it is necessary to carefully select the patient's antiplatelet drugs and personalized administration.
In our study of bleeding scoring model, an age of >65 years and bleeding history are the same as the HAS-BLED scoring model, but the scores assigned to them are different. In our scoring model, bleeding history are assigned 2 points. In a study by Magnani et al, a history of spontaneous bleeding requiring hospitalization is independent predictors of bleeding. 20 Anemia and aspirin are also included in our model, which can lead to an increasing risk of bleeding. The 2021 study by Meina Lv et al also demonstrates that anemia and dual antiplatelet can lead to an increased risk of gastrointestinal bleeding. 21 In the Magnani's study, anemia is also an independent risk factor for bleeding. 20 Biologically, anemia reduces the mass of RBCs in the central column of flowing blood through a vessel resulting in fewer platelets coming into contact with the endothelial surface at the periphery of the flowing blood column. Thus, anemia results in impaired primary hemostasis. 22 In our bleeding group, 100% of the patients are combined with aspirin, so aspirin has a greater impact on bleeding, giving 1.5 points. Furthermore, it has been observed that the combination of atorvastatin also elevates the likelihood of experiencing bleeding. In a study by Pierre Arenco et al, it reduces the overall incidence of stroke and cardiovascular events, but increases hemorrhagic stroke incidence with 80 mg of atorvastatin per day. 23 On the one hand, it may be related to the level of cholesterol, including low-density lipoprotein, triglyceride, apolipoprotein B, etc For patients with higher risk, it is recommended that the low-density lipoprotein should be reduced to less than 1.4, and too low cholesterol level and triglyceride will increase the risk of bleeding. 24 Relevant literature has mentioned that the reduction of ApoB or LDL-C is significantly correlated with the risk of cerebral hemorrhage.25–27 On the other hand, statins themselves can directly interfere with the coagulation system and platelet activation. 28 In vitro studies have also confirmed the hypothesis that atorvastatin may directly interfere with platelet activation, as its dose-dependent (0.1-10 μmol/L) down-regulates NOX2-derived oxidative stress, ultimately leading to impaired platelet isoprostaglandin formation and phospholipase A2 activation, resulting in reduced platelet TxA2 production. Late antiplatelet effect seems to be closely related to LDL lowering, and progressive and parallel decreases in platelet TxA2 and LDL have been detected in related studies.29–31 There are other combinations, such as diltiazem and ketoconazole. In Renli Teng's study, it was shown that compared with ticagrelor alone, diltiazem (moderate cytochrome P450 (CYP) 3A inhibitor) co-administration significantly increased the mean maximum concentration (Cmax) and mean area under the plasma concentration-time curve (AUC) for ticagrelor by 69% and 174%。Cmax and AUC for ticagrelor were increased by 135% and 632%, respectively, by ketoconazole co-administration. 32 However, in this study, there were only 25 patients combined with diltiazem, and only 1 patients combined with ketoconazole, and no effect on bleeding events was found.
The AUC reports that the A4B score model demonstrates greater accuracy in predicting bleeding risk in diabetic patients compared to the HAS-BLED score (AUC 0.714 vs 0.570). A4B score model could distinguish the risk of bleeding with statistical significance (HRa = 2.099, P = .045; HRb = 5.393, P < .001; HRc = 11.24, P < .001). It can distinguish the bleeding risk of different groups.
The ischemic score model examines four separate risk factors: an age of >65, smoking, a history of thrombotic disease, and BMI ≥ 30. It is worth noting that an age of >65 and a history of thrombotic disease align with the CHA2DS-VASc score, as stated by Chizyński K et al who states that the frequency of arterial hypertension and fibrinogen level are higher in the older group than in the younger one. 33 According to Noaman's research, there is a gradual rise in the long-term death rate from the youth to middle-aged to the elderly group (6.1% vs 9.9% vs 26.8%, P < .001). 34 Cigarette smoking is widely recognized for its association with cardiovascular disease, including ischemic stroke. Multiple studies have demonstrated a robust correlation between smoking and the formation of blood clots. 35 Additionally, the act of smoking cigarettes poses a risk factor for brain infarction, specifically lacunar and atherothrombotic brain infarction, 36 aligning with our own discoveries. Comorbid conditions such as nicotine/smoking exposure and diabetes can further alter brain energy metabolism, thereby, worsening the outcome of an ischemic stroke event in a growing population of patients. 37 According to Yoo JE's research, a higher BMI and waist circumference are linked to an elevated risk of cardiovascular diseases, including myocardial infarction and ischaemic stroke. 38 Additionally, our study indicates that a high BMI can also raise the likelihood of developing blood clots. Renli Teng's study results indicate that co-administration of ticagrelor increased the digoxina (P-glycoprotein substrate) maximum plasma concentration by 75%. Digoxin had no effect on the pharmacokinetics of ticagrelor or its active metabolite, AR-C124910XX. Co-administration of ticagrelor and digoxin was well tolerated. 39 In this study, the number of cases of digoxin combined use was small, only 4 cases, and no effect on thrombotic events was found. Based on the AUC, the ABST score model demonstrates superior predictive ability for thrombosis risk compared to the CHA2DS-VASc model (AUC 0.708 vs 0.540), exhibiting statistical significance within various risk groups (HRa = 3.436, P < .001; HRb = 4.593, P < .001; HRc = 15.84, P < .001). The high-risk group has a 15-fold increased likelihood of blood clot occurrence compared to the low-risk group.
This study also has some limitations. First of all, there are only two centers in this study at present, and the representativeness of the samples is insufficient. It is hoped that in the following research, other centers will join to increase the richness and representativeness of the samples. Second, This study is retrospective and may have incomplete data. A common problem is that complications such as missed bleeding and thrombosis are easy to occur. Finally, patient compliance is a key factor. Patients’ age, knowledge level and the importance of the disease have an impact on their medication compliance, and also have a great impact on the indicators of adverse events like bleeding and ischemia.
Our expectation is that the newly developed scoring system will effectively differentiate the risk level of bleeding and thrombosis among diabetic individuals. This will serve as a valuable guide for determining the appropriate clinical treatment plan involving ticagrelor, ultimately minimizing the occurrence of negative incidents and improving the overall prognosis for patients.
Conclusions
We establish two risk score models for diabetic patients taking ticagrelor, namely, bleeding risk score and thrombosis risk score (A4B score and ABST score). The A4B score assesses the risk of bleeding by considering five factors (the age of >65, previous bleeding incidents, anemia, aspirin use, and atorvastatin use). When compared to HAS-BLED, A4B demonstrates better predictive capability. On the other hand, the ABST score evaluates the risk of ischemia by taking into account five factors (the age of >65, smoking, history of thromboembolic disease, and BMI ≥ 30). Compared to CHA2DS-VASc, ABST demonstrates better predictive capacity. This novel scoring system could aid in the identification of diabetic patients susceptible to bleeding and ischemic conditions. For high-risk patients, the dose of ticagrelor can be adjusted appropriately or the medication can be adjusted.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241254107 - Supplemental material for New Score Models for Predicting Bleeding and Ischemic of Ticagrelor Therapy in Patients with Diabetes Mellitus
Supplemental material, sj-docx-1-cat-10.1177_10760296241254107 for New Score Models for Predicting Bleeding and Ischemic of Ticagrelor Therapy in Patients with Diabetes Mellitus by Xiaotong Xia, Shu Chen, Chang Cao and YanRong Ye, Yun Shen in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contribution
YS has designed the study. Every writer has contributed to the exploration of literature and gathering of data. Statistical analysis has been performed on CC, RY and SC. TX has drafted the first edition of the manuscript. YS and TX have conducted a rigorous review and revision of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the Ethics Committee of Zhongshan Hospital (Xiamen), Fudan University and Zhongshan Hospital, Fudan University and carried out in accordance with the principles of the Declaration of Helsinki. (Ethics B2022-069; B2023-045).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
