Abstract
The objective of this study is to determine the fibrin monomer reference intervals in healthy children. This cross-sectional study was conducted in the Hematology Department at Vietnam National Children's Hospital (April 2023 to March 2024). Children without prior history of clotting disorders or anticoagulants use hospitalized in preparation for orthopedic surgery or inguinal hernia surgery were enrolled in the study. The fibrin monomer test method was the quantitative fibrin monomer test on the STA-R system (Diagnostica Stago™, France). Eighty-six children (58 males and 28 females) were enrolled in the study. The median (interquartile range, 2.5th-97.5th) fibrin monomer value of the study subjects was 2.56 (0.11-5.93) µg/mL, with no statistically significant difference in fibrin monomer values among the age groups of 1 month to 3 years, 3 years to 13 years, and 13 years to 18 years. This is the first study conducted in Vietnam to determine reference values of fibrin monomer in children. This information can help in the diagnosis and treatment of early hypercoagulation stage and disseminated intravascular coagulation in children.
Introduction
The International Society on Thrombosis and Haemostasis (ISTH) and Japanese Ministry of Health and Welfare's have issued diagnostic guidelines for disseminated intravascular coagulation (DIC), where the diagnosis of compensated phase DIC is based on soluble fibrin monomer (FM) levels.1–3 DIC is further subdivided into overt-DIC and non-overt DIC where overt DIC is characterized by obvious clinical signs and widespread coagulation that can lead to bleeding; whereas, non-overt DIC is where abnormal coagulation occurs without the severe bleeding or visible symptoms typically associated with overt-DIC. FM is created through the cleavage of fibrinogen by thrombin and the subsequent release of fibrinopeptide A during the hypercoagulation cascade.4,5 Some studies indicate that FM is superior in diagnosing DIC compared to D-dimer.6,7 Another study found that the diagnostic capability of FM was comparable to that of D-dimer for both overt-DIC and non-overt DIC groups. 8 Measuring FM can also help evaluate hypercoagulation in pregnant women, predicting the risk of thrombosis. 9
Research studies have predominantly focused on establishing FM values for the adult population, while a reference value in pediatric populations has not been adequately investigated. Establishing FM reference values for healthy children can aid in the diagnosis and treatment of hemorrhagic and thrombotic diseases. 10 This is especially beneficial for lower-middle-income countries like Vietnam, where standardized benchmarks can significantly enhance healthcare strategies and treatment outcomes for pediatric thrombotic disorders.
Materials and Methods
Patients
This cross-sectional study was conducted in the Hematology Department at Vietnam National Children's Hospital (April 2023 to March 2024).
The guardian of all participants provided written informed consent, and the study was approved by the institutional medical ethics committees of the Vietnam National Children's Hospital Ethics Council (approval no. 641/BVNTW-HDDD).
Children without prior history of clotting disorders or anticoagulants use hospitalized in preparation for orthopedic surgery and inguinal hernia surgery were enrolled in the study. The demographic variables of age (months), gender, and weight (kilograms) were recorded. Study subjects were divided into three age groups: infants from 1 month to 3 years (Group 1), children from 3 years old to 13 years (Group 2), and adolescents aged 13 to 18 years (Group 3).
Children with medical conditions such as hematological diseases, chronic liver disease, cancer, severe trauma, malnourished, and obesity were excluded from the study.
Fibrin Monomer Test Methods
Whole blood (2 mL) was collected from each patient and placed into a tube with 3.2% natricitrate anticoagulation, which was then analyzed in the Hematology Department of National Children's Hospital. Platelet-poor plasma that had separated from the whole blood was anticoagulated using sodium citrate 3.2% S-Monovette tube (Sarstedt, Australia); this was completed by combining one part sodium citrate with 9 parts of whole blood, which was then centrifuged at 3000 rpm at room temperature. Plasma was frozen and stored at −20 °C for up to 1 month in a deep freezer. Frozen plasma samples were thawed at 37 °C in a basin of water and were tested within 2 h of defrosting. The test samples were pre-analyzed for color, turbidity to eliminate suspected cases of hemolysis, hyperbilirubinemia, and hyperlipidemia. The quantitative FM test was performed using the STA-R system (Diagnostica Stago™, France). 11
This test was based on the immunological method of turbidity measurement on the automatic coagulator system. This process involves incubating a patient's plasma with a suspension consisting of latex particles bound with FM antigen antibodies. FM in the patient's plasma will bind to the latex-antibody complex, resulting in agglutination. The degree of agglutination is proportional to the concentration of FM in the patient's plasma and is measured based on changes in optical density. FM concentrations count both soluble FM complexes and free FM in patient plasma.
Statistical Analysis
Qualitative variables are expressed as percentages. Non-standard distribution quantitative variables are represented as medians and interquartile range (IQR). FM values were expressed as medians and 2.5 to 97.5 percentile according to CLSI guidelines. 12 Percentage differences were compared using Chi-square tests. Average values were compared using the Mann–Whitney U-test. FM values between age groups were compared using the 1-way ANOVA operation, followed by the Dunnett test for correction. The comparisons are double-tested and statistically significant when the P-value <.05. A scatterplot was made to visualize the changes of FM over time. The variables were processed using SPSS statistics software version 29.0.1.1 (Armonk, NY: IBM Corp).
Results
Patients
Eighty-six children (58 boys and 28 girls) were enrolled in the study. The median age was 68.5 months (range 10 months-449 months). The median weight of children was 19.05 kg (Table 1).
Demographics of the Study's Population.
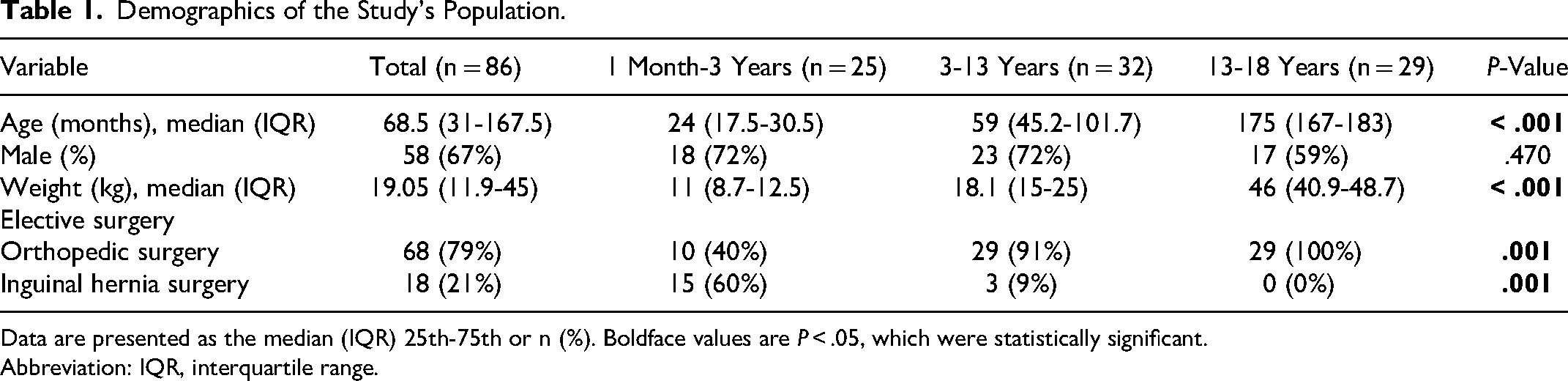
Data are presented as the median (IQR) 25th-75th or n (%). Boldface values are P < .05, which were statistically significant.
Abbreviation: IQR, interquartile range.
Laboratory Data
The median (interquartile, 2.5th-97.5th) FM value of the study subjects was 2.56 (0.11-5.93) µg/mL, with no statistically significant difference in FM values between the age groups of 1 to 3 (Table 2). The Dunnett test demonstrated no difference in FM values between the 3 age groups (between group 1 and group 2 [P = .283]; group 1 and group 3 [P = .283]; group 2 and group 3 [P = .073]).
Fibrin Monomer Laboratory.
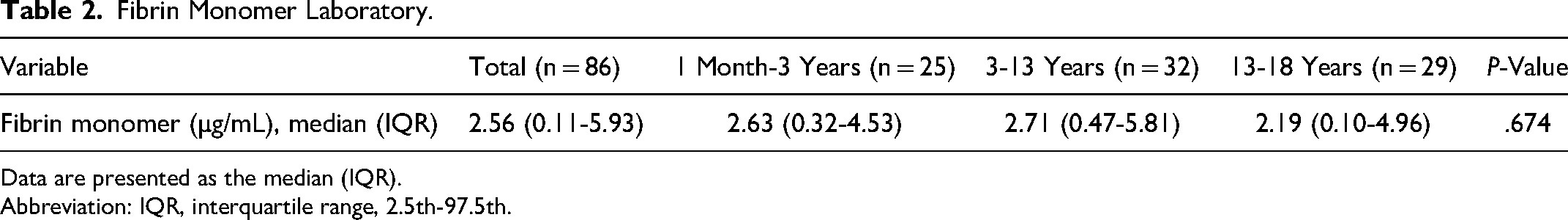
Data are presented as the median (IQR).
Abbreviation: IQR, interquartile range, 2.5th-97.5th.
FM levels between males and females also had no statistically significant difference (Table 3). The distribution of normal values of FM concentration in children is visually depicted in Figure 1. A scatterplot of FM by age is depicted in Figure 2.
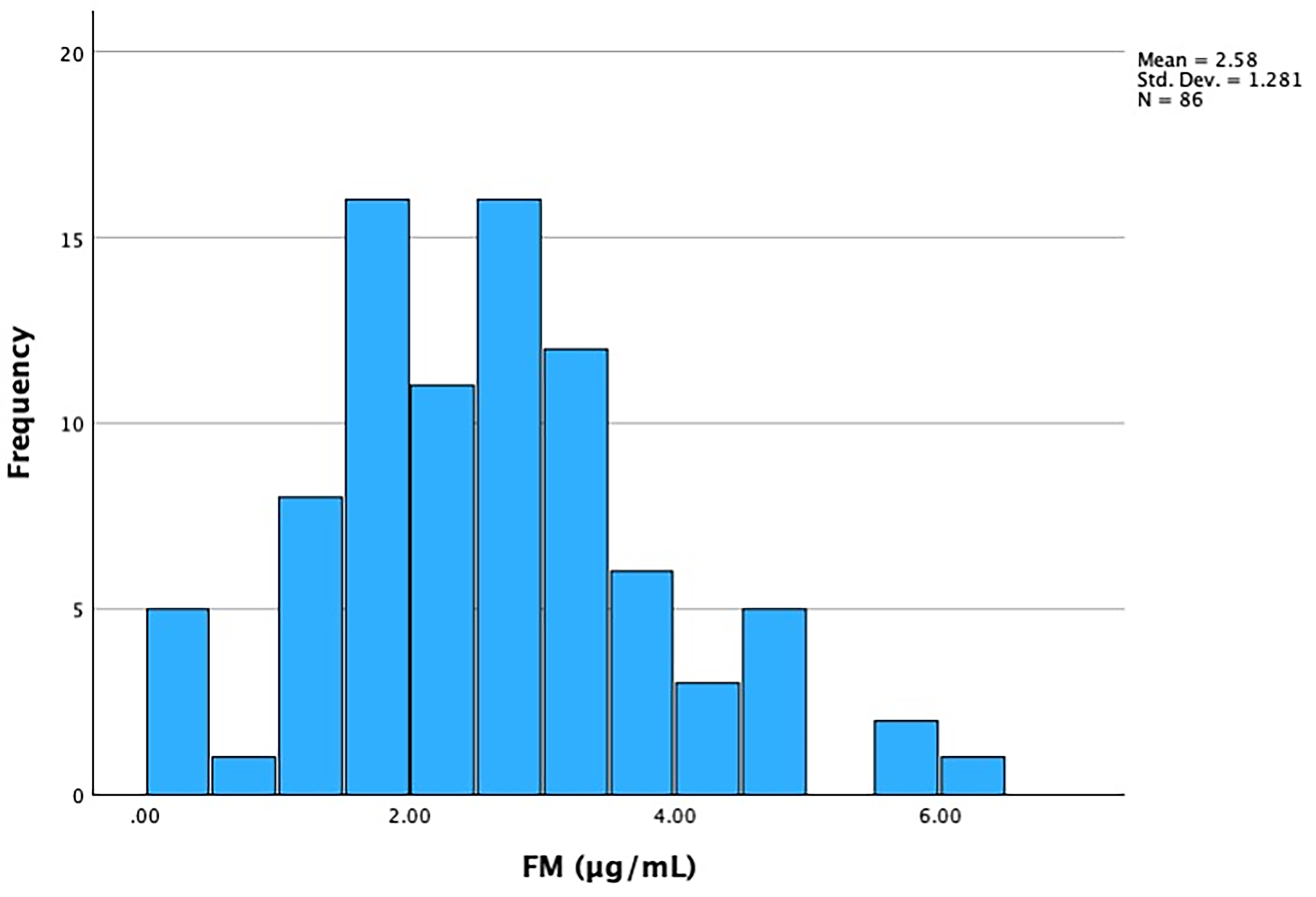
Distribution of fibrin monomer concentration of the study subjects.
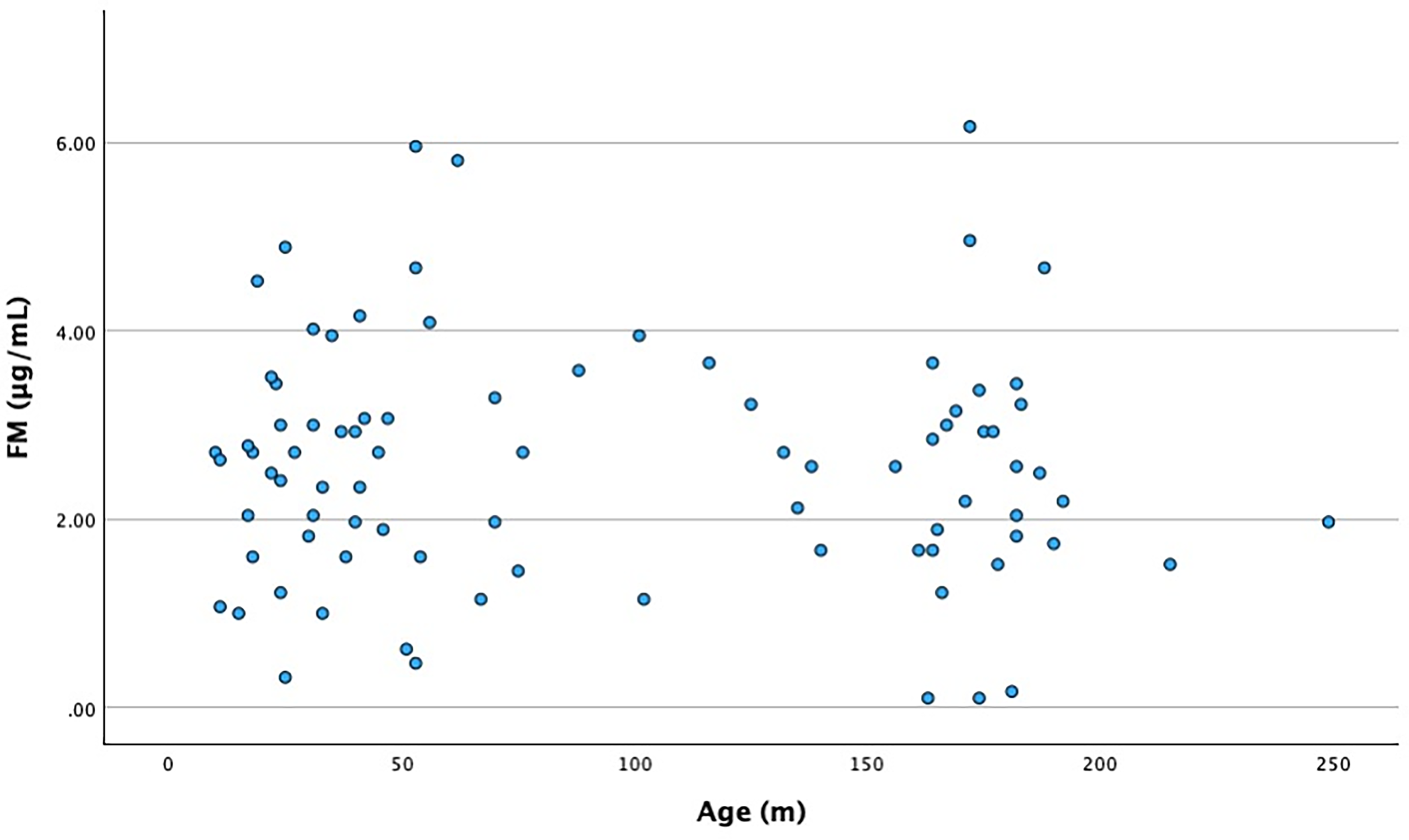
A scatterplot of fibrin monomer by age.
Fibrin Monomer by Gender.

Data are presented as the median (IQR) 2.5th-97.5th.
Abbreviation: IQR, interquartile range.
Discussion
We characterized 86 children who received a preoperative FM test at the Vietnam National Children's Hospital to establish an FM value reference interval. This is the first study in Vietnam and Southeast Asia countries to determine a FM reference range in pediatric population.
FM has been noted to play an important role in the diagnosis of thrombosis. 13 Although the ISTH uses FM on the nonovert-DIC status scale, it does not provide a clear standard for normal FM in children. 14 This study will supplement data on the normal reference values of FM in Vietnamese children being tested at the Vietnam National Children's Hospital.
In a previous study on the role of fibrinogen degradation products in the diagnosis of thrombosis, Hydeo Wada et al 7 found that the concentration of FM in normal adult plasma was less than 6.0 μg/mL. A recently report from Letunica et al 15 revealed the reference FM values in children, which was in line with our study. The results of our study showed that the median value of FM concentration was 2.56 μg/mL. There was no statistically significant difference in normal concentrations of FM in children across age groups or gender.
The value of FM in the diagnosis of DIC and the prognosis of treatment in critically ill children will be the next research direction after this study.
Limitations
This is a single-centered cross-sectional study with a small sample size. Further multicenter studies with larger sample sizes are needed to increase the accuracy of the results.
Conclusion
This is the first study in Vietnam to determine reference values of FM in children. It can help physicians to make an early diagnosis of the hypercoagulation stage and DIC in children.
Footnotes
Acknowledgments
We acknowledge the Department of Hematology staff at Vietnam National Children's Hospital. We also would like to thank Virginia Commonwealth University School of Medicine Global Research Partners, and the editing team that includes Venkat Abbaraju, Aashish Batheja, Shivani Mullapudi, Jane Choi, Sarah Durisek, and Naomi Ghahrai.
Author Contributions
Tuan, Xoay, and My contributed to the conception and design of the study. Trang, Duyen, Kien, Son, Thang, Quan, and Hung collected the clinical information. Xoay wrote the first draft of the manuscript. Tuan and My wrote sections of the manuscript. All authors have revised, read, and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
