Abstract
Fibrin-related markers (FRMs) such as fibrin and fibrinogen degradation products (FDPs),
Introduction
Disseminated intravascular coagulation (DIC) is often found in patients with infections, leukemia, and solid tumors. 1,2 In infectious DIC, microvascular dysfunction, depression of antithrombin (AT) and protein C, and elevated plasminogen activator (PA) inhibitor I (PAI-I) are frequently present. 3–5 Therefore, hypercoagulation without hyperfibrinolysis are usually observed in septic DIC. 6,7 High levels of tissue factor (TF) and PA are detected in some leukemic or solid cancer cells, 8,9 and both hyperfibrinolysis and hypercoagulation are observed in DIC due to leukemia or the presence of solid cancers. There are several differences among the pathophysiologies of DIC due to these 3 underlying diseases and the hemostatic abnormalities are different among the 3 underlying diseases.
Two diagnostic criteria for DIC have been established by the Japanese Ministry of Health and Welfare (JMHW)
10
and another by the International Society of Thrombosis and Haemostasis (ISTH).
2
The ISTH overt DIC criteria tend to focus on septic DIC, but the other criteria established by the JMHW tend to diagnose the condition based on the presence of various underlying diseases. The JMHW and ISTH diagnostic criteria include global coagulation tests, such as the prothrombin time (PT), platelet count, fibrinogen, and fibrin and fibrinogen degradation products (FDPs) or
In this study, 522 patients with infections, solid cancers, or hematopoietic tumors were prospectively evaluated using the DIC diagnostic criteria established by ISTH and JMHW, and the usefulness of FRMs were examined for DIC associated with the 3 underlying disease states (infections, solid cancers, or hematopoietic tumors).
Materials and Methods
A total of 522 patients in 9 institutes with underlying disorders known to be associated with DIC were registered for this prospective study on the DIC diagnostic criteria between January 1, 2005 and May 31, 2008. The study protocol was approved by the Human Ethics Review Committee of the Mie University School of Medicine and a signed consent form was obtained from each participant. This study has been faithfully carried out in accordance with the Declaration of Helsinki. The inclusion criteria were based on the presence of one or more of the following laboratory findings: a platelet count of less than 120 × 103/μL; FDP of more than 10 μg/mL; fibrinogen of less than 100 mg/dL; and a PT ratio of over 1.25. Any patients with symptoms associated with thrombotic thrombocytopenic purpura and heparin-induced thrombocytopenia, antiphospholipid syndrome, or with severe liver injuries were excluded from this study. There were 238 patients with infectious diseases (83 females and 155 males, median age: 71 years; age range 62-77 years), 132 with solid cancers (43 females and 89 males, 64 years; 54-72 years), and 152 with hematologic tumors (67 females and 85 males, 62 years; 45-72 years) who were included in the study. Disseminated intravascular coagulation was diagnosed using the ISTH overt DIC diagnostic criteria 2 or JMHW DIC diagnostic criteria. 10 The treatment for DIC 7 including AT, heparin, low-molecular-weight heparin, gabesate mesilate, nafamostat mesilate, or danaparoid sodium were carried out under the observation of the individual physicians after blood sampling was obtained at registration. The patients who were alive at 28 days after the registration were considered to be survivors and those who died by 28 days were considered to be nonsurvivors.
The PT, fibrinogen, platelet count, and FDP were measured at each of the institutes based on the methods reported in numerous previous studies.
11–13
An FDP assay at each institute correlated well with the LPIA FDP (Mitsubishi Chemical Medience Corporation, Tokyo, Japan). The plasma levels of SFMC
14
and
Statistical Analysis
The data are expressed as the medians (95% confidence interval [CI]). The differences between the groups were examined for statistical significance using the Mann-Whitney U test. A P value of less than .05 was considered to be statistically significant. All statistical analyses were performed using the SPSS II software package (SPSS Japan, Tokyo).
Results
Out of 522 patients, 66 patients with infections, 48 with solid cancers, and 63 with hematopoietic tumors were diagnosed to have DIC based on the diagnostic criteria of the JMHW.
10
Regarding the FRMs, FDP,
Plasma levels of fibrinogen degradation product (FDP),
Fibrin-Related Markers in Patients With DIC and Withouta
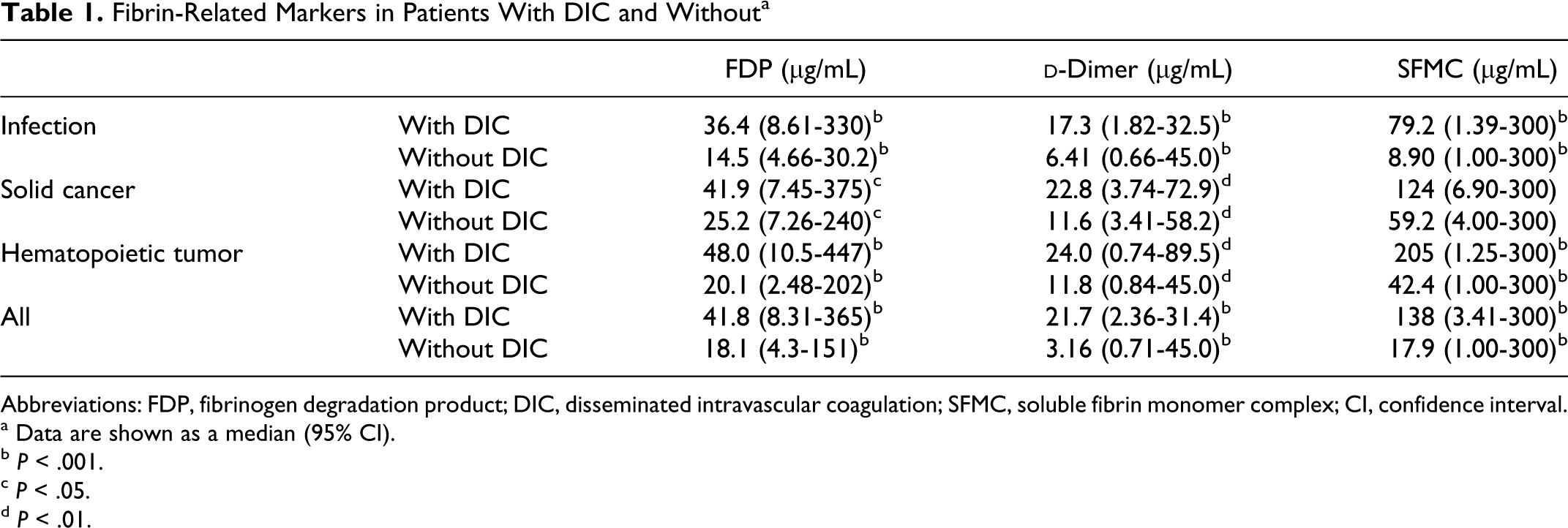
Abbreviations: FDP, fibrinogen degradation product; DIC, disseminated intravascular coagulation; SFMC, soluble fibrin monomer complex; CI, confidence interval.
a Data are shown as a median (95% CI).
b P < .001.
c P < .05.
d P < .01.
Cutoff values of 2 points (moderate increase) and 3 points (strong increase) according to the ISTH overt-DIC diagnostic criteria were adopted as the 2.5th percentile with the 95% confidence interval (CI) and median of patients with DIC (Table 1). For all FRMs of solid cancers or SFMC in each of the disease groups, the value of the 2.5th percentile was similar between the DIC group and the non-DIC group, and a cutoff value of 2 points for the FRMs could not be determined for solid cancers. In addition, the cutoff value of 2 points for the SFMC could not be determined for any of the 3 diseases (Table 2
). In the analysis of all patients, the cutoff values of 2 points for FDP,
Cutoff Value of FRM for DIC Diagnostic Criteria a
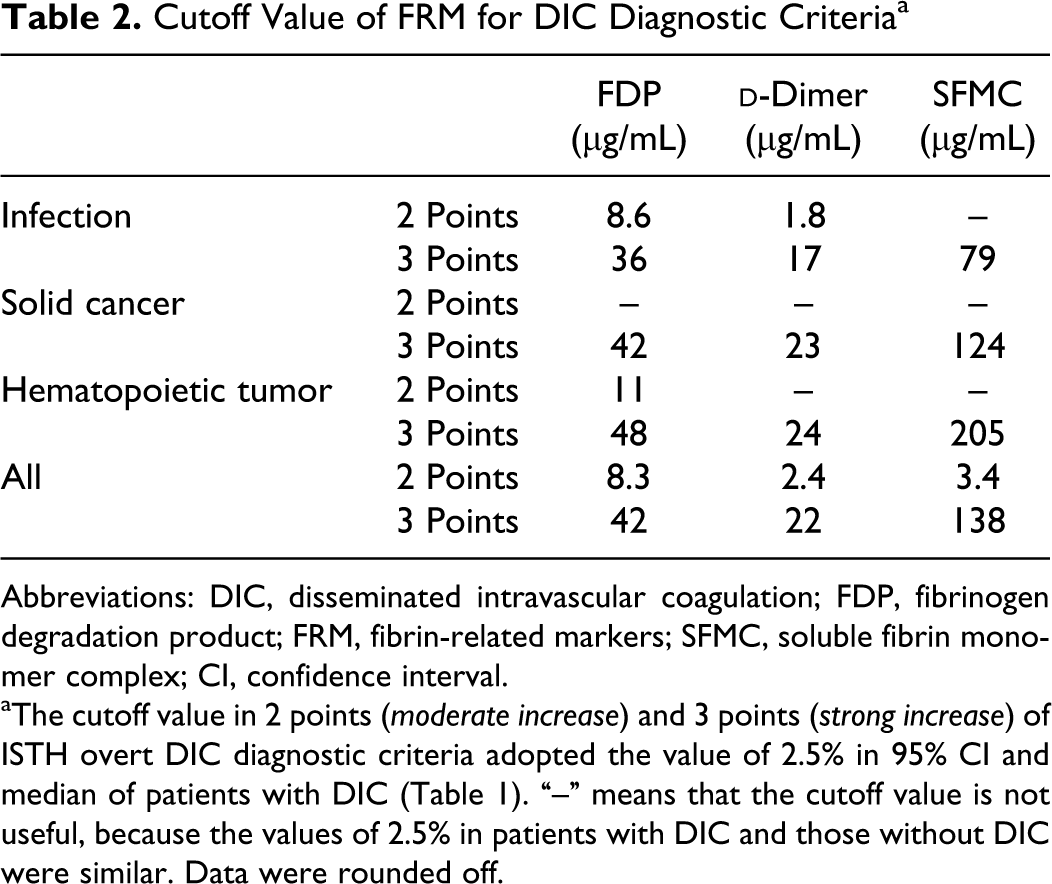
Abbreviations: DIC, disseminated intravascular coagulation; FDP, fibrinogen degradation product; FRM, fibrin-related markers; SFMC, soluble fibrin monomer complex; CI, confidence interval.
aThe cutoff value in 2 points (moderate increase) and 3 points (strong increase) of ISTH overt DIC diagnostic criteria adopted the value of 2.5% in 95% CI and median of patients with DIC (Table 1). “–” means that the cutoff value is not useful, because the values of 2.5% in patients with DIC and those without DIC were similar. Data were rounded off.
Concordance of Diagnosis for DIC Between JMHW and ISTH Overt DIC Diagnostic Criteria
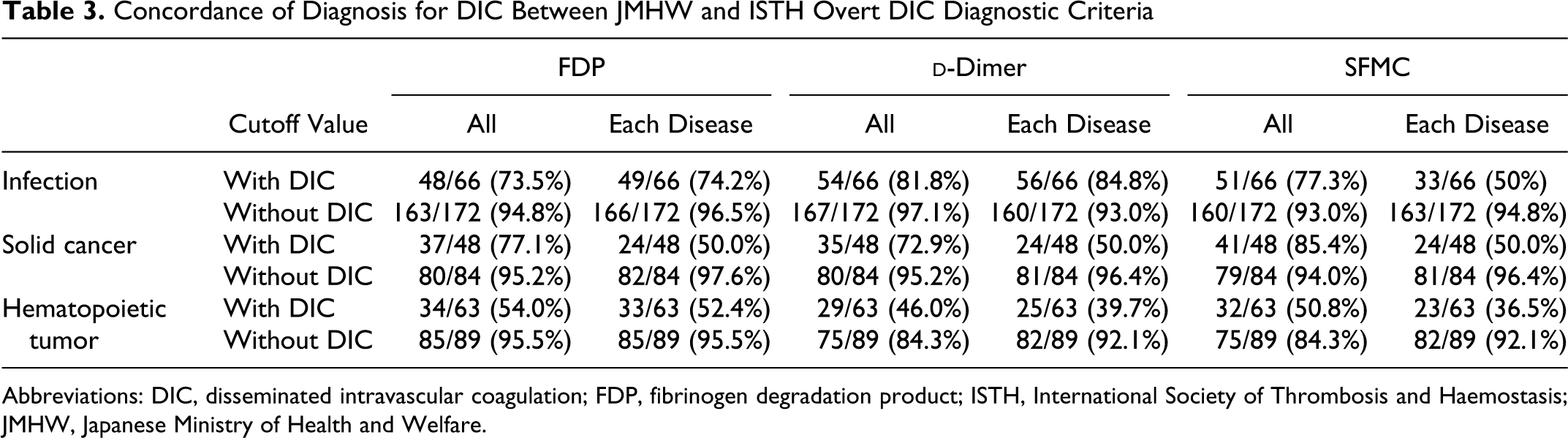
Abbreviations: DIC, disseminated intravascular coagulation; FDP, fibrinogen degradation product; ISTH, International Society of Thrombosis and Haemostasis; JMHW, Japanese Ministry of Health and Welfare.
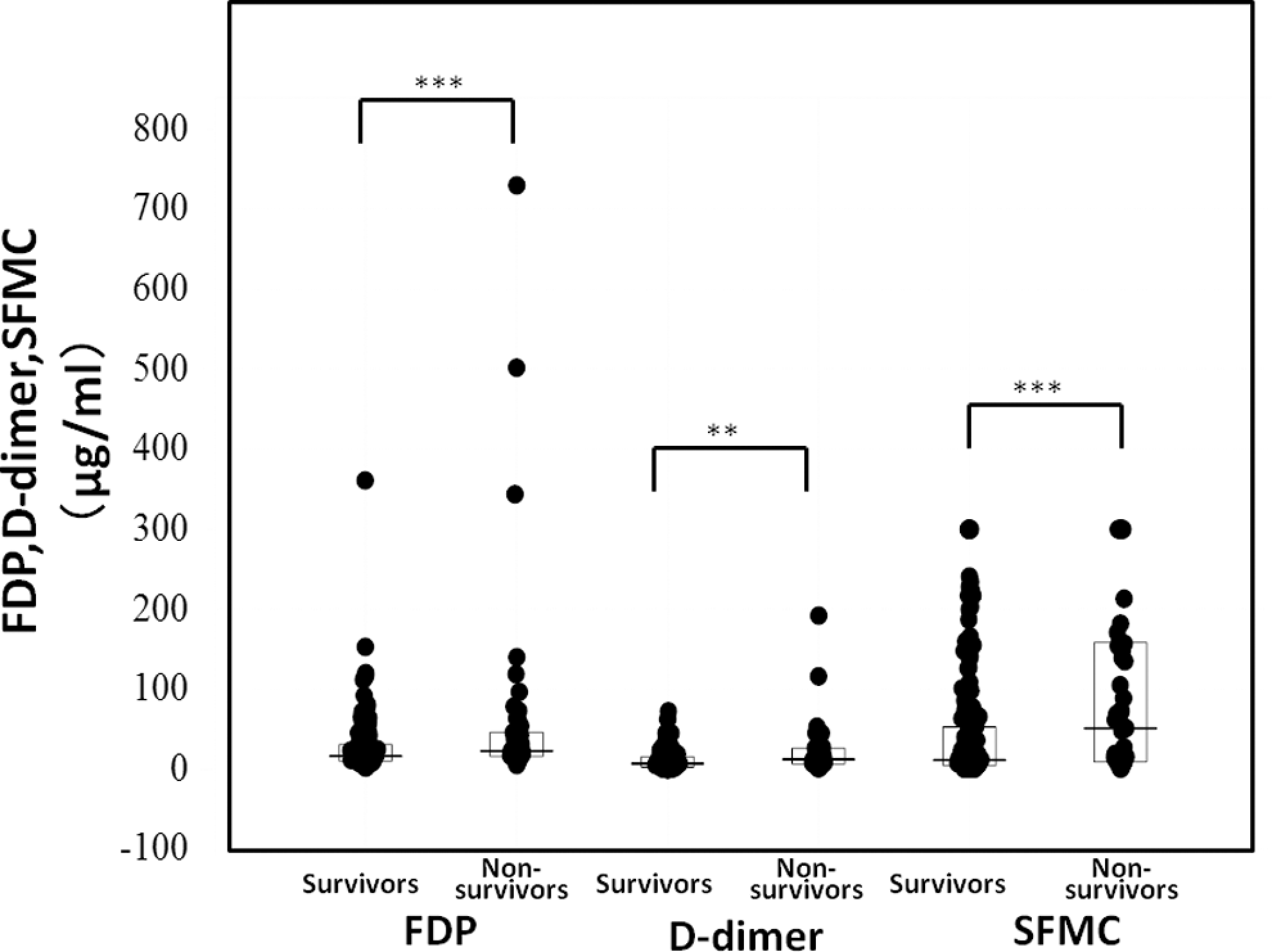
The differences in the FDP,
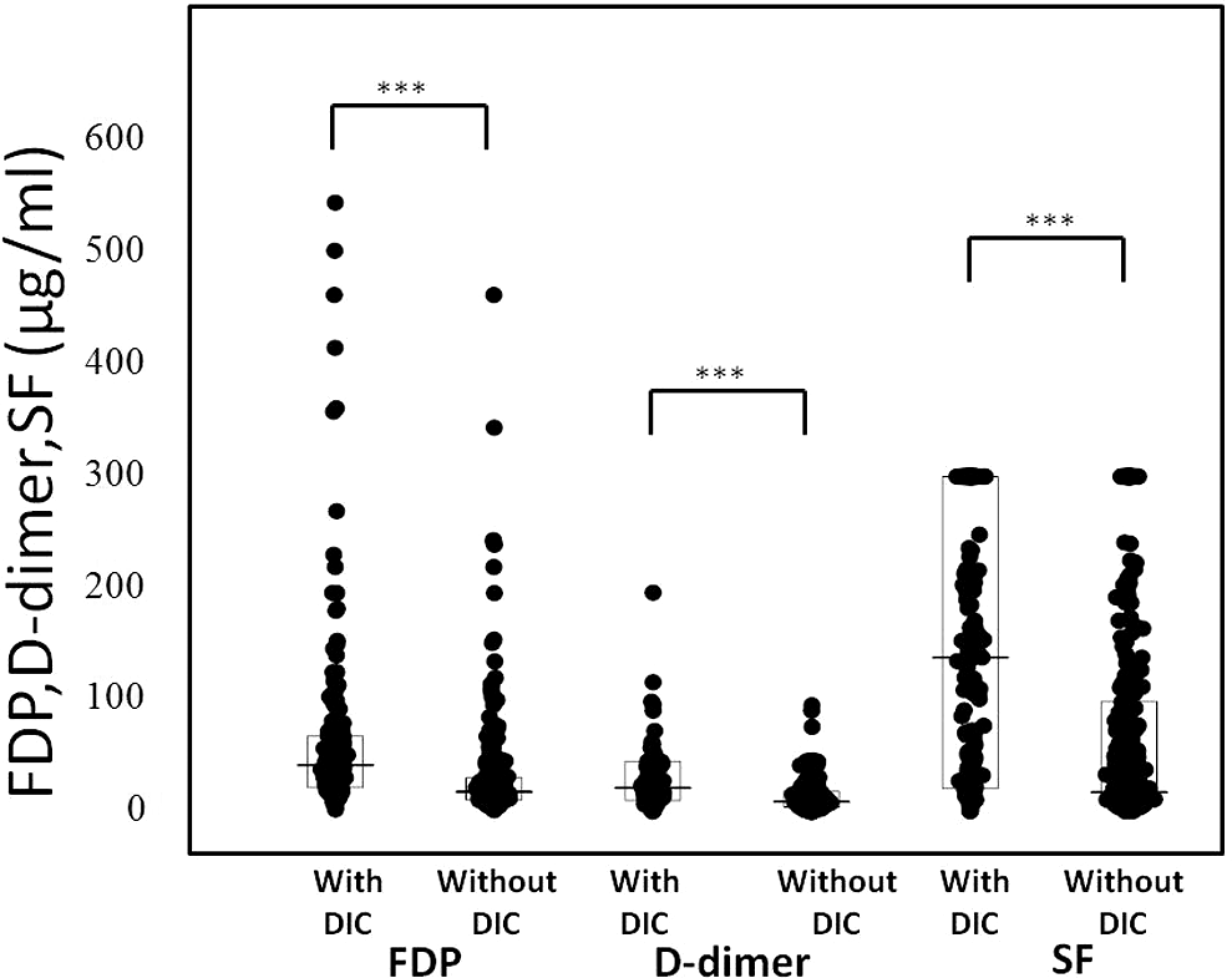
Plasma levels of fibrinogen degradation product (FDP),
Fibrin-Related Markers in Survivor and Nonsurvivor a

Abbreviations: FDP, fibrinogen degradation product; SFMC, soluble fibrin monomer complex; CI, confidence interval.
a Data are shown as a median (95% CI).
***P < .001. **P < .01.
Area Under the Curve (AUC) for Diagnosis of DIC for Prediction of Poor Outcome
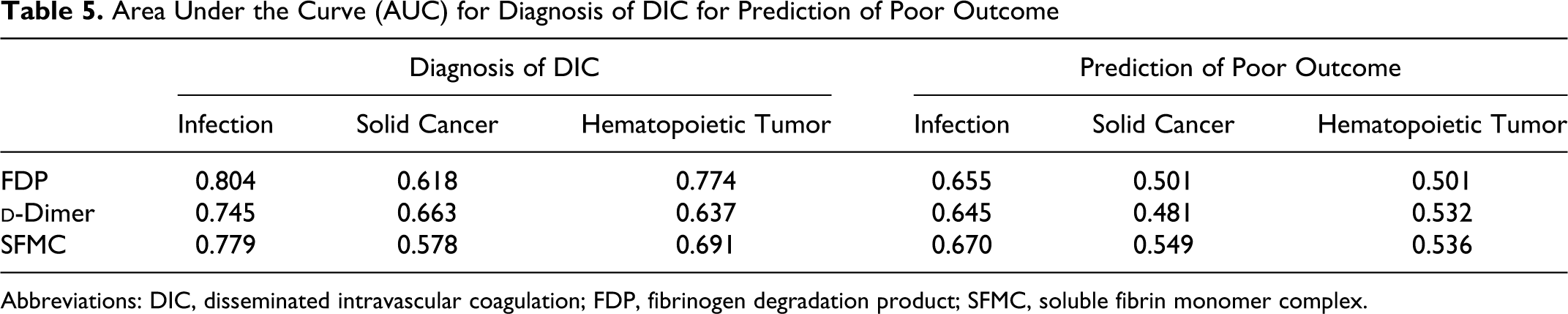
Abbreviations: DIC, disseminated intravascular coagulation; FDP, fibrinogen degradation product; SFMC, soluble fibrin monomer complex.
Discussion
Fibrin-related markers are very important markers for making a diagnosis of DIC, but the cutoff values of FRMs have not yet been clearly established in ISTH overt DIC diagnostic criteria.
2,4
In this study, FDP,
In the analysis of cutoff values of 2 points and 3 points for the ISTH overt DIC diagnostic criteria, the FDP,
Although the cutoff values of FRMs were slightly different among the patients with the 3 different underlying diseases, it is easy to use these cutoff values for the analysis of all 3 diseases. The diagnosis of DIC based on the ISTH overt DIC criteria using this cutoff value had a higher concordance with DIC based on the JMHW criteria in comparison to using the other cutoff value. Using the cutoff values from this study, the concordance of the diagnosis of DIC between the JMHW and the ISTH overt-DIC diagnostic criteria was similar to that described in previous reports, 20 and it was also relatively high in patients with infections and low in those with hematopoietic tumors, however the concordance of exclusion for DIC was high. These results confirm that the diagnostic criteria for DIC proposed by the JMHW are more sensitive than those proposed by the ISTH.
In this study, markedly elevated FRM levels were able to predict a poor outcome in the patients with infections, but not in those with solid cancers or hematopoietic tumors, thus suggesting that DIC may be related to a poor outcome in patients with infections but not in those with solid cancers or hematopoietic tumors. 4 Activated protein C and AT improved the mortality in the septic patients with DIC. 21,22 In patients with infections, the diagnosis of DIC using an adequate cutoff value of FRMs will be therefore useful for predicting the outcomes in such patients.
In conclusion, the appropriate cutoff value is slightly different for the diagnosis of DIC among the participants with the above described 3 different underlying diseases and this value will be useful for both making a diagnosis of DIC and predicting the outcome in such patients. Fibrin-related markers are more useful for the diagnosis of DIC based on infections than based on other underlying disorders.
Footnotes
Acknowledgments
This prospective study was carried out across 9 institutes, comprising the Department of Molecular and Laboratory Medicine and Emergency Medicine of Mie University School of Medicine, the First Department of Surgery of the University of Occupational and Environmental Health School of Medicine, the Department of Internal Medicine of Takasaki National Hospital, Takasaki, the Department of Internal Medicine of Teikyo University School of Medicine, the Department of Emergency Medicine and Intensive Care of the Graduate School of Medicine, Nagoya University, the Department of Anesthesiology and Critical Care Medicine of Hokkaido University Graduate School of Medicine, the Department of Emergency and Critical Care Medicine of Nippon Medical School, the Department of Internal Medicine of Shibata Hospital-Niigata Prefectural Hospital, the Research Division of Cell and Molecular Medicine of the Center for Molecular Medicine, and the Jichi Medical University School of Medicine. This work was supported in part by a Grant-in-Aid from the Ministry of Health, Labour and Welfare of Japan for Blood Coagulation Abnormalities and from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid from the Ministry of Health, Labour and Welfare of Japan for Blood Coagulation Abnormalities and from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
