Abstract
Background
The diagnosis of bleeding disorders through the Clinical Chemistry Laboratory requires specific tests. In Mexico, there are few reports on reference values (RVs) for coagulation factors (CFs). The prevalence and RVs in adults and older Mexican adults have already been described. However, it remains unknown whether these parameters differ among minors (MI), young adults (YA), and older adults (OA). The aim of this study was to determine CF levels in a group of healthy MI, YA, and OA in Mexico.
Methods
CFs were determined on a coagulation analyzer using commercial kits for the CFs: I, II, V, VII, VIII, IX, X, XI, and XII. The effect size was determined using Glass's rank biserial correlation coefficient. Comparisons between groups were performed using Dunn-Bonferroni post hoc tests following a significant Kruskal-Wallis test, and the epsilon-squared coefficient was used to estimate effect magnitude.
Results
347 participants, stratified by age group and sex, were studied. Variability in CF activity was observed. Age was found to be an important factor influencing the activity of all CFs, particularly factors II, V, VIII, IX, X, and XII, with factors V and XII proposed as biomarkers of aging.
Conclusions
CF levels observed in OA differ from those in YA and MI, as well as from the RVs proposed in commercial kits. It is important to integrate these results into established diagnostic algorithms, as well as into an evidence-based practical guide that clinical personnel can use to support a comprehensive and accurate diagnosis of patients with hemostatic disorders.
Background
Hemostasis is a delicately balanced biological system that allows the free circulation of blood through the blood vessels. A disruption of this balance can lead to hemorrhages or thrombotic events. When vascular endothelium injury occurs, cellular and protein mechanisms are activated, enabling the formation of a hemostatic thrombus (clot), tissue repair, and the cessation of bleeding to restore hemostasis. 1 For study purposes, hemostasis is divided into: primary hemostasis, which mainly involves the vascular endothelium and platelets; secondary hemostasis, or coagulation, in which coagulation factors (CFs) are involved; and finally, fibrinolysis, where the clot is dissolved. When a clot does not dissolve, it is called a thrombus. The thrombus, or part of it, may detach from the vessel, travel through the bloodstream, and block blood flow. For this reason, antithrombotic proteins (APs) participate in the regulation of coagulation: protein C (PC), protein S (PS), and antithrombin (AT). The proper balance between coagulation and the antithrombotic process allows blood to flow without thrombus formation.1,2
A defect in any phase of hemostasis can have catastrophic consequences. Hemostatic disorders, such as hemophilia, are characterized by inefficient blood clotting; they are hereditary and associated with deficiencies of CFs, FVIII and FIX.3,4 On the other hand, thrombophilia results from multiple genetic or environmental interactions that increase the risk of thrombosis, such as certain hereditary disorders that cause AP deficiencies. 5
In addition, alterations in the fibrinolytic system may occur, including abnormalities in plasminogen and FVIII, as well as hypoprothrombinemia and hyperhomocysteinemia.6–9 Other risk factors may also be acquired, such as antiphospholipid antibody syndrome, surgery, trauma, prolonged immobilization, hormone treatments, diabetes mellitus, obesity, and pregnancy, among others.9,10 Arterial and venous thrombosis are common problems faced by physicians. Some patients with thrombosis present an underlying hypercoagulable state. These states can be classified into three categories: hereditary disorders, acquired disorders, and those of mixed origin.11–13
Understanding the physiology of coagulation allows us to determine which clinical chemistry laboratory tests are appropriate to identify the cause of a hemorrhage or a thrombotic event, and to choose the right treatment to restore the lost balance in hemostasis. Clinical chemistry diagnosis requires indirect screening methods to identify hemostatic disorders, such as platelet count, bleeding time (BT), prothrombin time (PT), activated partial thromboplastin time (aPTT), and thrombin time (TT). BT is useful for assessing blood disorders such as von Willebrand disease. PT, aPTT, and TT are used indirectly to evaluate decreases in CFs, FXI and FXII concentrations, as well as factors FVIII and FIX. However, even with mild deficiencies, the results of these tests may sometimes fall within the established reference values (RVs).14,15 RVs are the ranges that healthcare professionals use to interpret the results of a set of biochemical tests. The range is generally defined as the set of values in which 95% of the normal population is found.
The RV will vary according to age, sex, race, and even the laboratory instruments used to perform population testing. Furthermore, by definition, 5% of the normal population lies outside the reference range.16–18 The RVs of clinical tests analyzed in some cases show differences compared with the intervals suggested by manufacturers of coagulation testing equipment. They may even differ from RVs suggested by international associations. Therefore, it is recommended that each clinical laboratory establish its own RVs based on its daily procedures.19,20
In Mexico, there are few studies on the RVs of CFs and AP. In 2015, Meillón et al evaluated the prevalence of alterations and RVs for proteins C, S, and AT in healthy donors. A significant difference was found between genders for the RVs of PC, PS, and AT, and PC showed variations among age groups. 21 In 2017, Hernández-Zamora et al determined the activity of CFs, APs, and plasminogen (Plg), as well as the frequency of deficiencies of these proteins in a population of healthy older adults in Mexico. Significant differences were found in some coagulation factors, and it was therefore recommended to establish RVs for this population. 22
The aforementioned studies suggest that it will also be necessary to establish RVs for hematological parameters in children and adolescents. Therefore, the objective of this study is to determine CF levels in a group of healthy minors (MI), young adults (YA), and older adults (OA) in Mexico.
Methods
Participants
All participants were recruited at the Instituto Nacional de Rehabilitación Luis Guillermo Ibarra Ibarra (INR-LGII) in Mexico City. They were healthy individuals, with no history of hematological or thrombotic disorders and no ongoing pharmacological treatment. Participants ranged in age from 3 to 89 years, included both sexes, and were unrelated. Additionally, demographic data including weight (kg), height (m), and body mass index (BMI) were collected.
Exclusion criteria included a history of cardiovascular or cerebrovascular diseases, thrombotic disorders, or liver and kidney diseases associated with hemorrhagic conditions. Individuals under antithrombotic therapy, oral anticoagulants, heparin, or vitamin K antagonists were excluded, as well as pregnant or breastfeeding women and those who had recently undergone surgical procedures.
Laboratory Analysis
Blood samples were obtained by venipuncture and collected in tubes containing 3.8% sodium citrate. Samples with insufficient volume, contamination, visible coagulation, hemolysis, or lipemia were discarded. Most samples were analyzed immediately, and all tests were completed within 4 h of collection. Samples that could not be processed on the same day were stored at −80 °C until analysis. Frozen samples were thawed rapidly in a 37 °C water bath for 5 min and thoroughly mixed before testing.
A complete blood count was performed using an automated hematology analyzer (Coulter LH 780). Citrated plasma was separated, and coagulation assays were performed on an IL ACL Elite/Pro analyzer (Werfen, Mexico City, Mexico) using commercial kits (HemosIL™) for the following determinations: TT Thrombin Time 0009758515; PT RecombiPlasTin 2G 0020002950; APTT-SP (liquid) 0020006300; Factor I 0008469810; Factor II 0008466050; Factor V 0020011500; Factor VII 0020011700; Factor VIII 0020011800; Factor IX 0020011900; Factor X 0020010000 and Factor XI 0020011300. Prior to sample testing, trueness verification was performed using in house quality controls. For each analyte was tested in three independent replicates at two concentration levels (normal and pathological). All results met predefined accuracy criteria.21,22
Statistics
The descriptive analysis was carried out using the statistical software SPSS Statistics, version 27.0 (IBM Corp., 2020, Armonk, NY, USA). The normality of score distributions was assessed with the Shapiro-Wilks test, and accordingly, the Mann-Whitney and Kruskal-Wallis tests were used to compare groups. The significance level for the P-value was set at .05. Effect sizes for the Mann-Whitney U tests were determined using Glass's rank biserial correlation coefficient (rg) and interpreted as small (rg = 0.11 to 0.28), medium (rg = 0.29 to 0.43), and large (rg ≥ 0.44). 23 Pairwise comparisons between groups were performed using Dunn-Bonferroni post hoc tests following a significant Kruskal-Wallis test, and epsilon squared was used to estimate effect sizes. 24
Ethical Aspects
All participants received oral and written information about this study and signed a letter of consent. This study was carried out in accordance with the Declaration of Helsinki and received approval from the Ethics, Research, and Biosafety Committees of INR-LGII (approval code: INR 54/16). The groups were selected under the guidelines of the Norma Oficial Mexicana NOM-253-SSA1-2012 for blood banks. All methods were carried out in accordance with relevant guidelines (CLSI C28-A3 and WS/T 402-2012 and IFCC).
Results
Demographic Data of the Study Population
A total of 347 individuals were analyzed: 172 women (49.56%) and 175 men (50.44%). The population was stratified by age ranges into: Group I, minors (MI), consisting of 109 participants aged 3 to 17 years; Group II, young adults (YA), consisting of 126 individuals aged 18 to 59 years; and Group III, older adults (OA), consisting of 112 participants aged 60 to 98 years (Figure 1).

Demographic data of the study population. Distribution by age group and sex.
Coagulation Factors Values by age Groups
Table 1 presents the measures of central tendency and the 95% confidence intervals (CI) for the CFs in the three study groups, as well as the differences between groups. Significant differences (P < .05) with a small effect size were found for all CFs among minors (MI), young adults (YA), and older adults (OA).
Reference Values (95% CI) for Coagulation Factors by Study Group (age).

*Kruskal-Wallis. **Epsilon-squared. + Dunn-Bonferroni post hoc. I MI: Minors. II YA: Young adults. III OA: Older adults.
In particular, CFs values for FI, FV, FVIII, and FIX were significantly higher in OA compared to MI, but lower for FII and FX, as shown in Figure 2. Likewise, CFs values for FII, FIX, and FX were significantly lower in the OA group compared to the YA group, but higher for FVIII and FXII in OA than in YA.
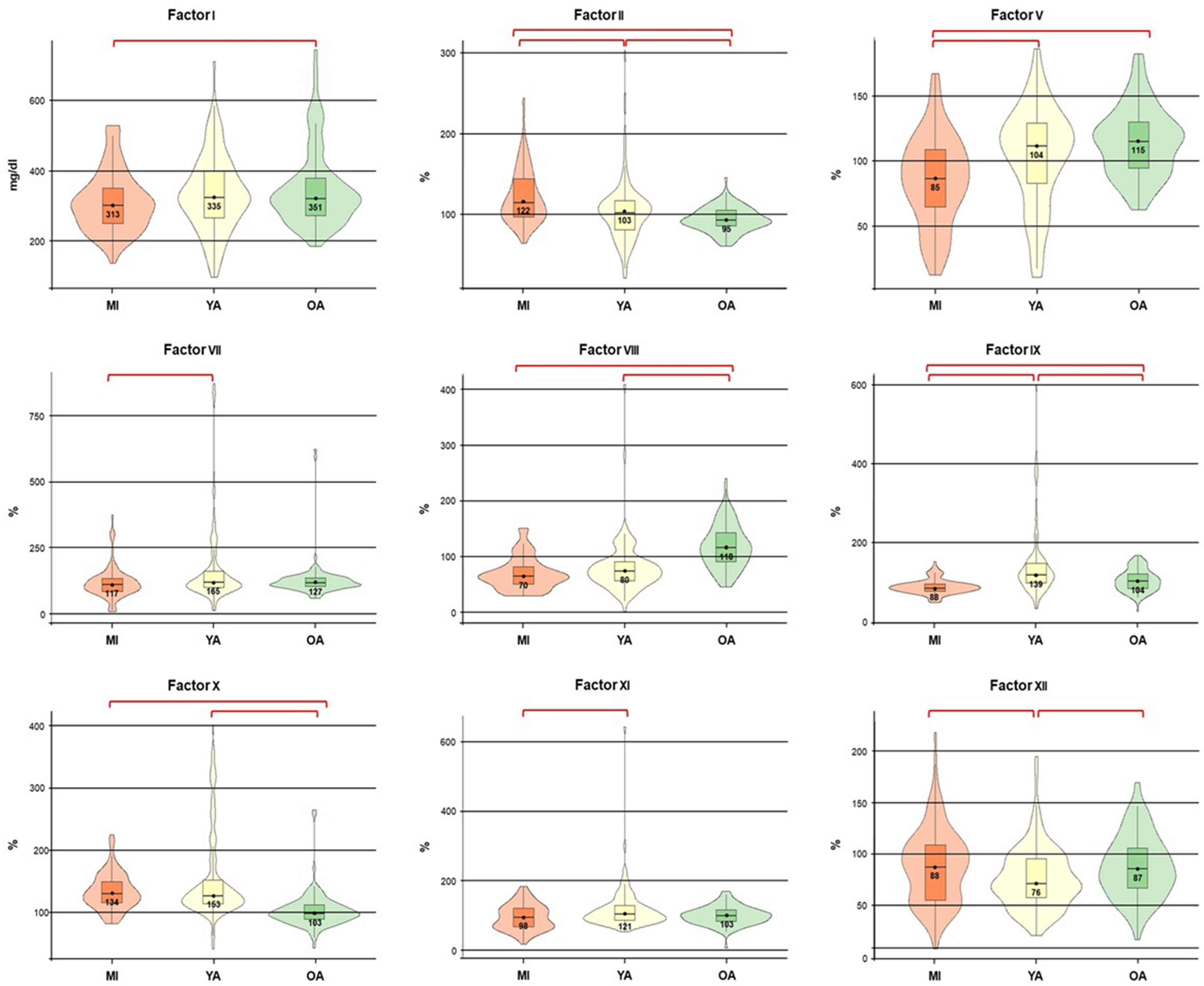
Coagulation factor values among the study groups. It is assumed that the population median parameter in each group falls within the range of values obtained from the study samples. ● Mean. ├─┤ Confidence intervals (95% CI).

Comparison of coagulation factor medians by sex and study groups. The charts show the comparison of medians and 95% confidence intervals of coagulation factor levels that exhibited significant differences (P ≤ .05) with a small effect size (rg ≤ 0.28) by gender in young adults (YA) and older adults (OA). A) In general, YA women show higher values for FI, FII, and FVIII than men; conversely, they present lower values for FVII, FX, and FXI compared to men. B) In the OA group, women show lower values for FV and higher values for FXII than men.
On the other hand, MI showed significantly lower CFs levels for FV, FVII, FIX, and FXI than the YA group, and also lower than the OA group for FV and FIX, but significantly higher for FII compared to YA and OA, and for FXII compared to YA. Young adults showed significantly higher values of FV, FVII, FIX, and FXI compared to the MI group, as well as higher FIX and FX levels compared to older adults, but lower FVIII and FXII values than those in the OA group (Table 1 and Figure 2).
Comparison of Coagulation Factor Levels by sex and age Group
By sex, significant differences with a small effect size were found between women and men in the young adult group for FI (P ≤ .002), FII (P = .002), FVII (P = .001), FVIII (P = .016), FX (P = .001), and FXI (P = .048) (Table 2). These comparisons are shown in Figure 3, where young adult women exhibited higher FI, FII, and FVIII values compared to their male counterparts, and lower FVII and FX values. Similarly, in the OA group, significant differences with a small effect size were also observed between women and men for FV (P = .044) and FXII (P = .019). In this case, OA men showed higher values than women for FV and slightly lower ones for FXII. Finally, in the MI group, no significant differences were found in coagulation factor values between sexes. When considering only the differences between men and women for each coagulation factor analyzed, according to the Mann-Whitney U test, significant differences with a small effect size were found only for FI (P = .008) and FII (P = .002) (Table 3).
Sex-Based Comparison of Coagulation Factor Reference Values Across age Groups.

Abbreviations: MI, Minors; YA, Young adults; OA, Older adults; W, Women; M, Men. —: No significant difference. Based on the Mann-Whitney U test, significant differences between men and women are shown
Comparison by Sex.
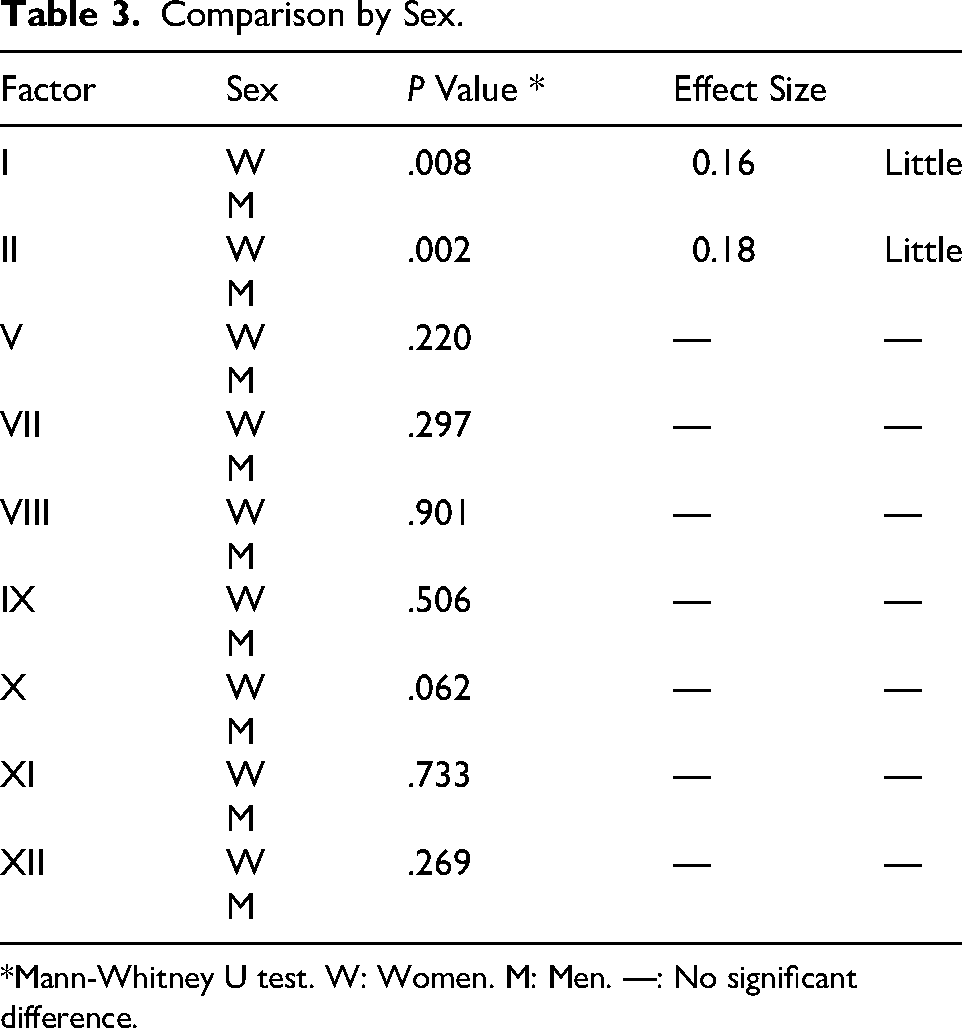
*Mann-Whitney U test. W: Women. M: Men. —: No significant difference.
Comparison Between Proposed Reference Values for the Study Population and Commercial Reference Values
Finally, a comparison is presented between the reference values provided by commercial kits for each analyzed parameter and the reference ranges proposed for the total study population. Table 4 highlights in bold the factors that showed reference ranges not included within the commercial reference values: FVII, particularly in relation to YA and OA; FIX in YA; and FX in MI and YA.
Comparison of the Proposed Reference Values for the Study Population and the Commercial Reference Values.
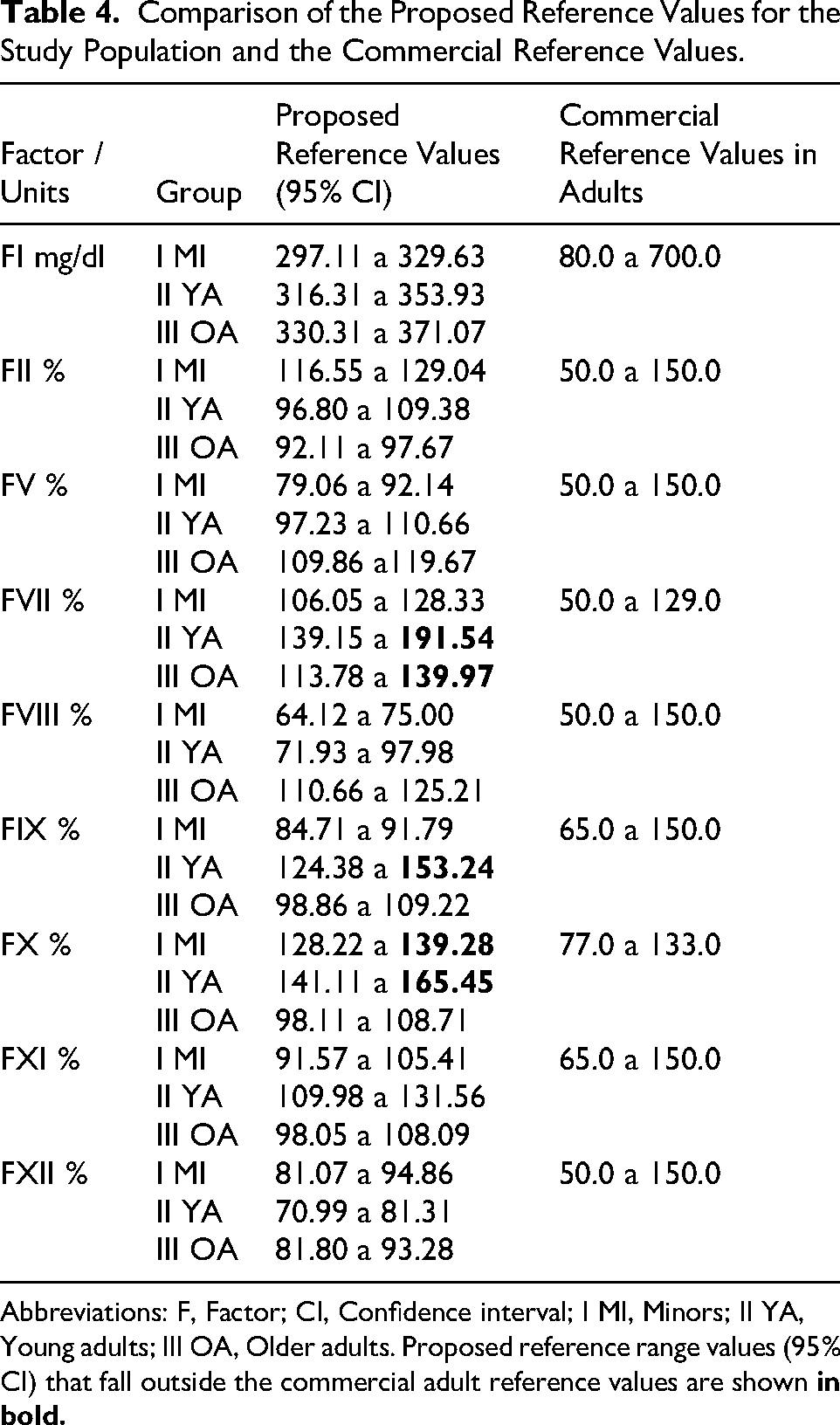
Abbreviations: F, Factor; CI, Confidence interval; I MI, Minors; II YA, Young adults; III OA, Older adults. Proposed reference range values (95% CI) that fall outside the commercial adult reference values are shown
Discussion
Physiologically, the hemostatic balance in children differs from that of adults. The development of the hemostatic system matures and changes over time, from fetal life to old age.18,25 Due to these changes, it is necessary to determine specific reference values (RVs) for each stage of life, including childhood. The characterization of the hemostatic system during childhood has been delayed, and most studies focus on adult populations.26,27 Therefore, RVs should be established for each population and clinical laboratory. Variability is influenced by several factors, such as genetics, race, lifestyle, environment, age (of the populations), or the analytical equipment and methods used. Consequently, RVs are essential for health monitoring programs, as they track physiological and pathophysiological processes.28,29
According to the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) and the Clinical and Laboratory Standards Institute (CLSI), there are three ways to establish reference values (RVs) in a population: by using determinations made in individuals from a reference population with a minimum sample size of 120 healthy individuals (IFCC recommendation); by transferring a preexisting reference interval when a method or instrument is changed, considering agreement and correlation according to the CLSI; and by validating a previously established or transferred reference interval.30,31
If control values are within RVs in a determination, patient samples can be analyzed to report their results. However, we must not forget that several of these results would be within the reference range for adults. Nonetheless, when using the RV obtained, specific for children and adolescents, they would fall outside these parameters. That is the importance of using these RVs in pediatric patients: they would receive better care.
When comparing the RVs obtained in girls and boys, results showed no significant differences in the evaluated parameters. Although it is known that sex-specific hormonal activity in adolescents has an effect on hemostatic activity, it is possible that hormonal production has not reached its peak in large part of the participants. 32
Moreover, the absence of differences could be due to the fact that both sexes have similar lifestyles, suggesting that gender-specific reference values may not be necessary. However, it would be highly valuable to increase the sample size of the 15-18-year-old population and compare it with that of the 18-21-year-old group, in order to distinguish the hormonal effect in the Mexican population and determine whether specific reference values are needed for these age ranges.
The World Health Organization estimates that the global population over 60 years of age will nearly double in the next 30 years. This change places a greater demand on health and social services, with a higher burden of morbidity in older adults, defined here as individuals aged 60 years or more. An aging population will have a higher incidence of cardiovascular diseases, partly due to higher levels of fibrinogen and some other coagulation factors in the blood, as well as increased platelet activity. All these factors lead to a hypercoagulable state that can alter hemostasis, causing an imbalance in coagulation, which plays a crucial role in the development of cardiovascular diseases. These changes in hemostasis are affected not only by age but also by gender and hormonal factors (or their absence during menopause in older women), ethnicity, comorbidities, drug interactions, and overall health as we age. It is known that throughout life, fibrinogen and coagulation factor levels vary in healthy individuals; however, the reference values (RVs) used for diagnosis and treatment are based only on two life stages: childhood and adulthood. There are no specific diagnostic guidelines based on RVs for these populations.22,33
It is important to note that the magnitude of change (effect size) should not be interpreted as an indication of clinical significance. In other words, a small effect size may still be clinically significant, while a large effect size may not be. It is increasingly recognized that traditional methods, such as statistical significance testing and effect size analysis, should be complemented by methods that assess clinically meaningful changes. 34
The medical interpretation of results is essential for clinical decision-making regarding patients. Therefore, it is necessary to compare the obtained results with the reference values calculated for the patient population. Although the Clinical Chemistry Laboratory plays a central role in diagnosis, it is a multidisciplinary process involving clinical suspicion, medical history, and laboratory findings. Consequently, for a laboratory result to be useful, it must first be medically relevant and globally comparable.35,36
With these results and a sample size of N = 347, the RVs for this population were determined, as suggested by the CLSI (n = 120), to ensure accurate evaluation. It is worth noting that conducting studies to determine any RV in a healthy population is a complex task, especially when, as in this case, it involves minors, young adults, and older adults. 36 The study of the hemorrhagic system in children and adolescents has lagged behind, and most research focuses on adult populations. In this study, it would be important to expand the sample to include minors and stratify it into children aged 1 to 12 years and adolescents aged 13 to 17 years, in order to distinguish the hormonal effect in the Mexican population and determine whether specific reference values are required for these age ranges.
Moreover, due to advances in molecular biology, it is important to note that mutations and polymorphisms associated with the predisposition to or origin of various diseases have already been described for each of these CFs. Therefore, it will be important to begin conducting studies to establish the prevalence and associations of these genetic variants and alterations.37–39 In this study, we have described how variability in coagulation factor activity may be due to different causes. Age is an important factor influencing the activity of all coagulation factors, particularly factors II, V, VIII, IX, X, and XII. Our results show that the levels of factors V and XII exhibit characteristic age-related changes among groups. Further studies are required to evaluate their diagnostic value, as they may serve as potential biomarkers of aging. In general, the coagulation factor levels observed in older adults differ from those in young adults and minors, as well as from the reference values proposed in commercial kits. Therefore, this is the first study to establish reference values for coagulation factors in a Mexican population aged 3 to 89 years.
Conclusion
It is important to integrate these results into established diagnostic algorithms, as well as into a practical evidence-based guide that clinical personnel can use to support a comprehensive and accurate diagnosis of patients with hemostatic disorders. This is particularly relevant for patients with suspected or confirmed thrombotic diseases and/or thrombophilia. Cardiovascular diseases are the leading cause of death in Mexico. Ischemic heart disease, including myocardial infarction, is the most common cause, followed by cerebrovascular accidents (strokes). Furthermore, it is important to offer these patients treatment and lifestyle options that improve their quality of life, maintain optimal health, and prevent future thrombotic events.
Footnotes
Abbreviations
Ethics Declarations
All participants received oral and written information about the study and signed a letter of consent. The study protocol was reviewed and approved by the INR-LGII Research and Ethics Committees.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data used in this study have been included in the manuscript. The corresponding author can be contacted if any further information is needed.
