Abstract
The objective of this study is to determine risk factors that may contribute to exclusion decision from recombinant tissue plasminogen activator (rtPA) in patients with acute ischemic stroke (AIS) with a combined current or history of smoking and obesity. This study was conducted on data from 5469 patients with AIS collected from a regional stroke registry. Risk factors associated with inclusion or exclusion from rtPA were determined using multivariate logistic regression analysis. The adjusted odds ratios and 95% confidence interval for each risk factor were used to predict the increasing odds of an association of a specific risk factor with exclusion from rtPA. In the adjusted analysis, obese patients with AIS with a history of smoking (current and previous) excluded from rtPA were more likely to present with carotid artery stenosis (OR = 0.069, 95% CI 0.011-0.442), diabetes (OR = 0.604, 95% CI 0.366-0.997), higher total cholesterol (OR = 0.975, 95% CI 0.956-0.995), and history of alcohol use (OR = 0.438, 95% CI 0.232-0.828). Higher NIHSS score (OR = 1.051, 95% CI 1.017-1.086), higher triglycerides (OR = 1.004, 95% CI 1.001-1.006), and higher high-density lipoprotein (OR = 1.028, 95% CI 1.000-1.057) were associated with the inclusion for rtPA. Our findings reveal specific risk factors that contribute to the exclusion of patients with AIS with a combined effect of smoking and obesity from rtPA. These findings suggest the need to develop management strategies to improve the use of rtPA for obese patients with AIS with a history of smoking.
Introduction
Stroke is one of the leading causes of death and disability and a major global problem. 1 The economic cost of stroke is huge, and the recovery requires complex and lengthy medical interventions including rehabilitation. 1 The estimated cost of stroke is more than 1.16% of the global gross domestic product. 2 The expectation is that there will be an increase in the global burden of stroke since currently there is no effective preventive measure that has been generally implemented. 3 Acute ischemic stroke (AIS) is caused by the occlusion of the cerebral artery resulting in a sudden loss of blood flow to specific regions of the brain, and loss of neurological functions. The major therapeutic goal for patients with AIS is the restoration of blood flow to salvageable ischemic brain tissue that is not already infarcted in a timely manner. 4 Reperfusion therapy using intravenous recombinant tissue plasminogen activator (rtPA) and endovascular therapies including mechanical thrombectomy (MT) are the only approved treatments for AIS. The Food and Drug Administration (FDA)-approved rtPA as the pharmaceutical agent for the treatment of AIS; however, it is not effective in patients with AIS due to large artery blockage, where the clot size is very large. 5 In such patients, MT has been reported to be more effective. In addition, the restriction on rtPA beyond 4.5 h disqualifies more than 80% of patients admitted beyond this time window. 6
Many untreated patients are excluded based on several factors, and observational studies indicate that some exclusion criteria are unreasonably restrictive. 7 For example, advanced age, rapidly improving or minor symptoms, severe hypertension, severe stroke or coma, combined history of prior stroke and diabetes, patients with unruptured intracranial aneurysms, the degree of thrombocytopenia, pregnancy, lumbar puncture, and recent surgery are all considered contraindications for exclusion from rtPA.7,8 The data regarding the safety of rtPA for patients with AIS with recent surgery are very few, 9 and given the fewness of some of the contraindications or exclusion factors and unique aspects of individual cases, generalizing results of some observational studies may be difficult when making decisions about rtPA. 7 For example, diabetes patients with stroke with a history of previous strokes when treated with rtPA may be associated with poor treatment outcomes, but this may not be applicable to all patients. 10 In general, the lack of quality evidence regarding many of the risk factors that constitute contraindications in addition to the established ones for rtPA provides opportunities for the current research to investigate risk factors that might be associated with poor outcomes and could affect decision making for rtPA therapy.
Smoking is reported to predict poor functional recovery in patients with AIS treated with rtPA. 1 This is because smoking may compromise recovery due to abnormal cardiopulmonary function, 11 with additional adverse effects on the vascular endothelium that could inhibit restorative processes. 12 An increase in hematocrit may potentially increase resistance to blood flow and oxygen supply. 13 In addition, smoking is reported to adversely influence functional recovery in patients with thrombolyzed AIS, compared with nonsmokers. 14 Obesity is closely associated with metabolic syndrome, 15 and the rate of middle cerebral artery recanalization is lower in patients with AIS metabolic syndrome when compared with those without metabolic syndrome. 16 Therefore, metabolic syndrome and hyperglycemia, both associated with obesity and higher resistance to rtPA, 17 are reported to contribute to the effect of obesity and related poor outcomes in rtPA-treated patients. In patients with body weight >100 kg (obese patients), the standard dose is reported to be insufficient in these patients and the maximum dose is limited to 90 mg. 18 Some patients with AIS that weigh ≥100 kg and received the maximum dose were associated with symptomatic intracerebral hemorrhage (sICH) and poor functional outcome. 19
While obesity can independently increase the risk of stroke, 20 it also combines with other comorbidities including diabetes mellitus, hypertension, atherosclerosis, atrial fibrillation, obstructive sleep apnea, and smoking increasing the severity of stroke resulting in poor outcomes.21–23 Therefore, investigating patients with stroke with a combined effect of smoking and obesity, including others risk factors that are associated with exclusion from rtPA is of major clinical importance. We hypothesize that risk factors associated with rtPA decisions for exclusion may not be distributed equally among patients with ischemic stroke with a history of the combined effect of smoking and obesity, smoking, or obesity. To test this hypothesis, we will identify risk factors within the populations of patients with ischemic stroke who received rtPA treatment and those who did not, and determine whether there are differences in risk factors among patients with AIS with a history of smoking and obesity, smoking, or obesity. Our second hypothesis is that individuals with a history of both smoking and obesity, smoking, or obesity may not present with the same risk factors that may affect rtPA decisions in the general stroke population. To test this hypothesis, we will determine specific risk factors that predict the exclusion from rtPA using a prospective registry of data from a regional stroke center. The objective of this study is to evaluate risk factors that may contribute to exclusion decisions from rtPA in the AIS population with a combined current or previous history of smoking and obesity. Analyzing the association between a combined effect of smoking and smoking and the rtPA exclusion decision will provide more information on specific risk factors that can be managed to improve the care of obese patients with ischemic stroke with a history of smoking.
Methods
Study Population
This study is a retrospective analysis of AIS patients’ data collected by PRISMA Health from January 2010 to December 2016. The study was approved by the IRB in addition to the PRISMA Health Ethics Committee. Inclusion criteria include patients admitted to the stroke unit of PRISMA Health from January 2010 to December 2016, diagnosed clinically and confirmed by brain imaging based on World Health Organization (WHO) criteria for diagnosis of stroke. Based on the WHO criteria, stroke is defined as rapidly developing clinical signs that result in focal or global disturbance of the cerebral function, with symptoms lasting 24 h or longer or leading to death with no known cause other than vascular origin. 24 Patients under the age of 18 years, those with incomplete medical records, and patients with a diagnosis of transient ischemic attack (TIA) were excluded from the study. Risk factors of stroke were defined as the presence of stroke risk factors such as hypertension, smoking, diabetes mellitus, alcohol abuse, heart diseases, atrial fibrillation, history of smoking, and obesity in patients with AIS. Patients with AIS with a history of smoking were defined as patients with a history of smoking of 2 and 1 cigarette per day for male and female patients. Obesity was based on the Center for Disease (CDC) definition of an adult who has a body mass index (BMI) of 30 or higher. Outcomes were variables associated with inclusion or exclusion from rtPA.
Patients’ medical records were used to extract data for sociodemographic characteristics, laboratory findings, and clinical data such as risk factors. Data on demographic variables, clinical characteristics, and laboratory values were collected. Demographics consisting of race, gender, age, ethnicity, and BMI were collected. Patients medical history included the history of atrial fibrillation (an irregular and often rapid heart rhythm), congestive heart failure (long-term condition that occurs when the heart cannot pump enough blood to meet the body's needs), carotid artery stenosis (CAS; blockage of the carotid arteries), coronary artery disease (CAD; heart condition that involves atherosclerotic plaque formation in the lumen, resulting in inadequate supply of blood and oxygen to myocardium), chronic renal disease (CRD; damage to the kidney resulting in the inability to filter waste products from the blood), depression (a serious mental illness that affect feelings), and dyslipidemia (imbalance of lipids resulting in cardiovascular diseases). We also collected data on diabetes mellitus (a metabolic disorder associated with elevated blood glucose), family history of stroke (patients whom father or mother had a stroke, especially before 65 years), hormone replacement therapy (patients who are women and received supplements of hormones that were lost during the menopausal transition), hypertension (high blood pressure when the pressure in the blood vessels is too high (eg, 140/90 mm Hg or higher) and migraine (a form of headache that can cause severe excruciating pain or a pulsing sensation, especially on one side of the head).
Data were also collected on previous stroke or TIA (a min stroke caused by a blockage in the brain similar as ischemic stroke), peripheral vascular disease (PVD; a chronic progressive atherosclerotic disorder resulting in peripheral vascular occlusion), prosthetic heart valve (a mechanical device that replaces the ventricles of the heart), sleep apnea (a condition in which the breathing stops and restarts many times while the patient sleep, and this can prevent the body from getting enough oxygen), drug and alcohol use, and tobacco use (a medical condition characterized by an impaired ability to stop or control alcohol and tobacco use despite adverse social, occupational, or health consequence). Admission data included “First Care” and was coded as either emergency department (0) or direct admission to the stroke unit (1). Laboratory data that were extracted included blood glucose (the main sugar found in the blood), total cholesterol (blood cholesterol measure of low-density lipoprotein [LDL] cholesterol, high-density lipoprotein [HDL] cholesterol, and other lipid components), triglycerides (types of fats that circulate in the blood including HDL, LDL, and lipids), serum creatinine (a normal waste product that builds up in the blood from the muscle), international normalized ratio (INR; a blood-clotting test, used to measure how quickly the blood forms a clot, compared with normal clotting), systolic blood pressure (measures of the pressure in the arteries when the heart beats), diastolic blood pressure (measures of the pressure in the arteries when the heart is at rest), and heart rate (number of time the heart beats per minute). Lastly, data were collected on ischemic stroke severity by using the National Institutes of Health Stroke Scale (NIHSS) score, which is a validated tool for assessing stroke severity. Ambulation data were collected and recorded as not documented (0), unable to ambulate (1), ambulate with assistance (2), and ambulate independently (3). Ambulation data was collected before the stroke, at admission, and discharge from the hospital. Data on improvements in ambulation was also collected. Improvement was denoted as an increase in ambulatory ability from the time of admission to the time of discharge.
Data Analysis
Statistical analysis for all data was conducted using the Statistical Package for Social Sciences version 29.0 for Windows (SPSS, Chicago, IL). The Kolmogorov-Smirnov test was used to determine if continuous variables are normally distributed. The level of significance was set at P < .05 (two-tailed). Demographic and clinical risk factor comparisons between the 2 groups were performed by student t-test for normally distributed data, the Mann-Whitney U-test for continuous variables, and the chi-square test for categorical variables. Continuous variables results were presented as means ± standard deviation (SD) while discrete variables were presented as patient count and percentage in the population of interest. These 2 groups were then separated based on whether the patient had a history of smoking, obesity, or smoking and obesity. After the univariate analysis of all variables, multivariate analysis was conducted to determine specific demographics and risk factors in obese patients with AIS with a history of smoking, obesity, or smoking alone that are associate with inclusion or exclusion from rtPA treatment.
For the multivariate logistic regression analysis, inclusion or exclusion from rtPA was used as the dependent variable while the demographics and clinical characteristics of patients with AIS who smoke are obese, or who smoke and are obese were used as independent variables. Only demographic and clinical characteristics that were initially associated with the inclusion or exclusion from rtPA treatment (P < .3) in the univariate analysis were selected for multivariate analysis. Therefore, to exclude confounding factors among clinical variables, logistic regression analysis was performed and adjusted odds ratios (OR) were calculated. For the data synthesis, we calculated ORs for each variable that is, the ratio of the odds of a specific risk factor among obese patients with AIS with a history of smoking, obese or smoking being associated with exclusion compared to the corresponding odds of inclusion for rtPA which serves as the controls. ORs with a confidence interval of 95% or higher were considered and P-values of <.05 were deemed significant. The variance in rtPA inclusion explained by covariates was determined using Cox and Snell R2.
Hosmer-Lemeshow tests were used to assess multicollinearity and interactions while the logistic regression models’ sensitivity, specificity, and accuracy were analyzed through metrics such as overall correct classification percentage and the area under the receiver operating characteristic curve (AUROC).
Results
A total of 5469 patients with AIS were either excluded or included in rtPA treatment. Of this, 4142 patients were excluded while 1327 patients were included for rtPA. The demographic and clinical factors of patients with AIS stratified by inclusion or exclusion from rtPA are presented in Table 1. Older patients with higher rates of CAS, diabetes, previous stroke, PVD, CRD with a history of diabetic medication and ambulating independently on admission were likely to be excluded from rtPA. Patients who were treated with rtPA were more likely to be Hispanics and presented with higher levels of total cholesterol, LDL, lipids, blood glucose, serum creatinine, BMI, and higher NIHSS scores. They were more likely to present with higher rates of depression, dyslipidemia, migraine, obesity, and previous TIA. They were likely to be prescribed with cholesterol reducers and antidepressants, ambulate independently before the stroke, ambulate independently at the time of discharge, be directly admitted to the stroke unit, and more likely to have improved ambulation by the time of discharge.
Risk Factors of Patients With Ischemic Stroke Divided by Those Included or Excluded From rtPA.
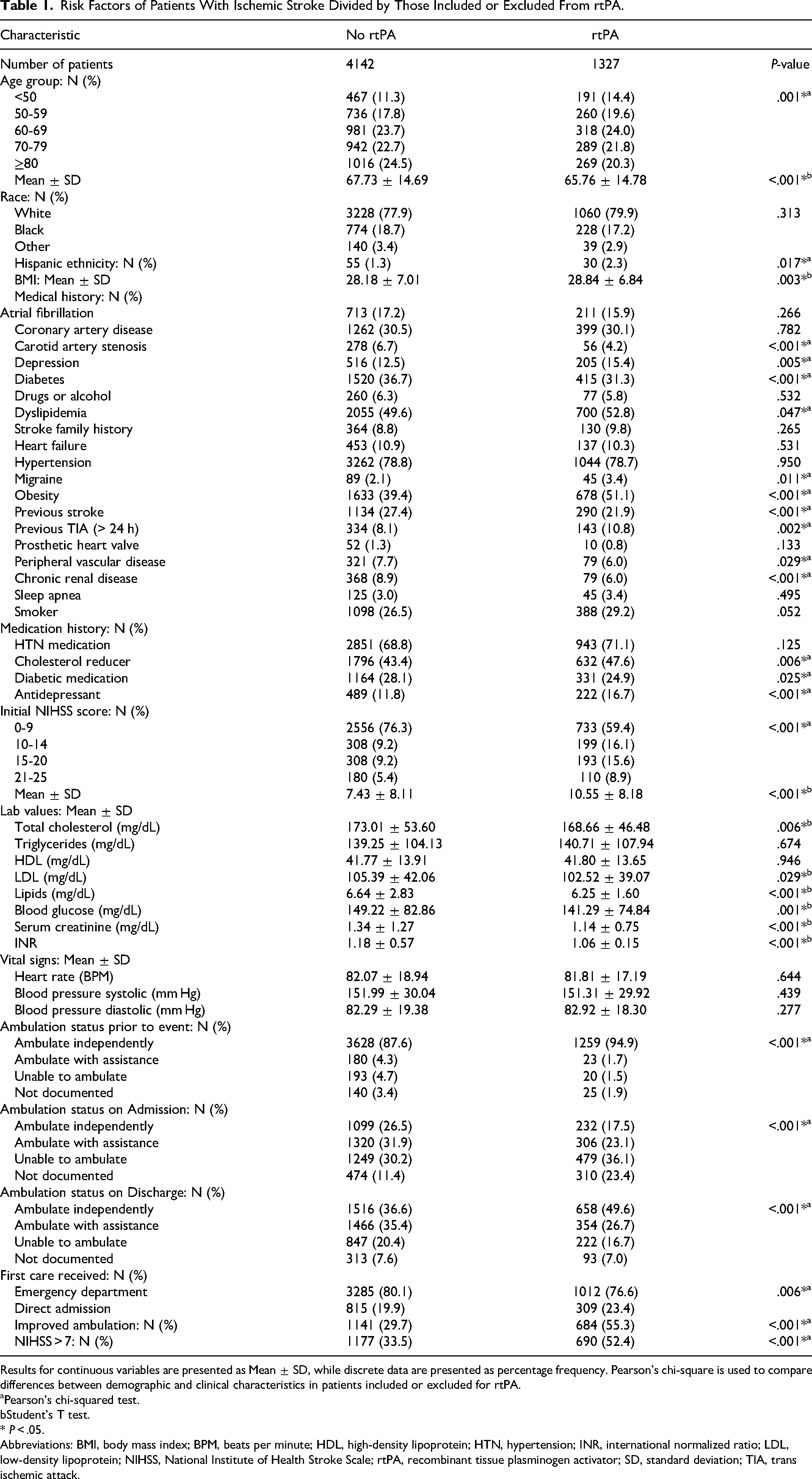
Results for continuous variables are presented as Mean ± SD, while discrete data are presented as percentage frequency. Pearson's chi-square is used to compare differences between demographic and clinical characteristics in patients included or excluded for rtPA.
Pearson's chi-squared test.
Student's T test.
* P < .05.
Abbreviations: BMI, body mass index; BPM, beats per minute; HDL, high-density lipoprotein; HTN, hypertension; INR, international normalized ratio; LDL, low-density lipoprotein; NIHSS, National Institute of Health Stroke Scale; rtPA, recombinant tissue plasminogen activator; SD, standard deviation; TIA, trans ischemic attack.
Patients with AIS stratified by smoking history or obesity, obesity plus smoking, and treatment with rtPA is presented in Table 2. Obese patients with AIS with a history of smoking, presented with CAS, diabetes, alcohol use, PVD, CRD, and higher total cholesterol, lipids, INR, ambulated independently at admission and were more likely to be excluded from rtPA. Obese patients with AIS with a history of smoking who were included for rtPA were likely to present with higher NIHSS scores and ambulated independently at discharge and they were more likely to present with improved ambulation by the time of discharge.
Risk Factors of Patients With Obese Ischemic Stroke With a History of Smoking, Obese Alone, and Smoking Alone Stratified by the Exclusion or Inclusion of rtPA.
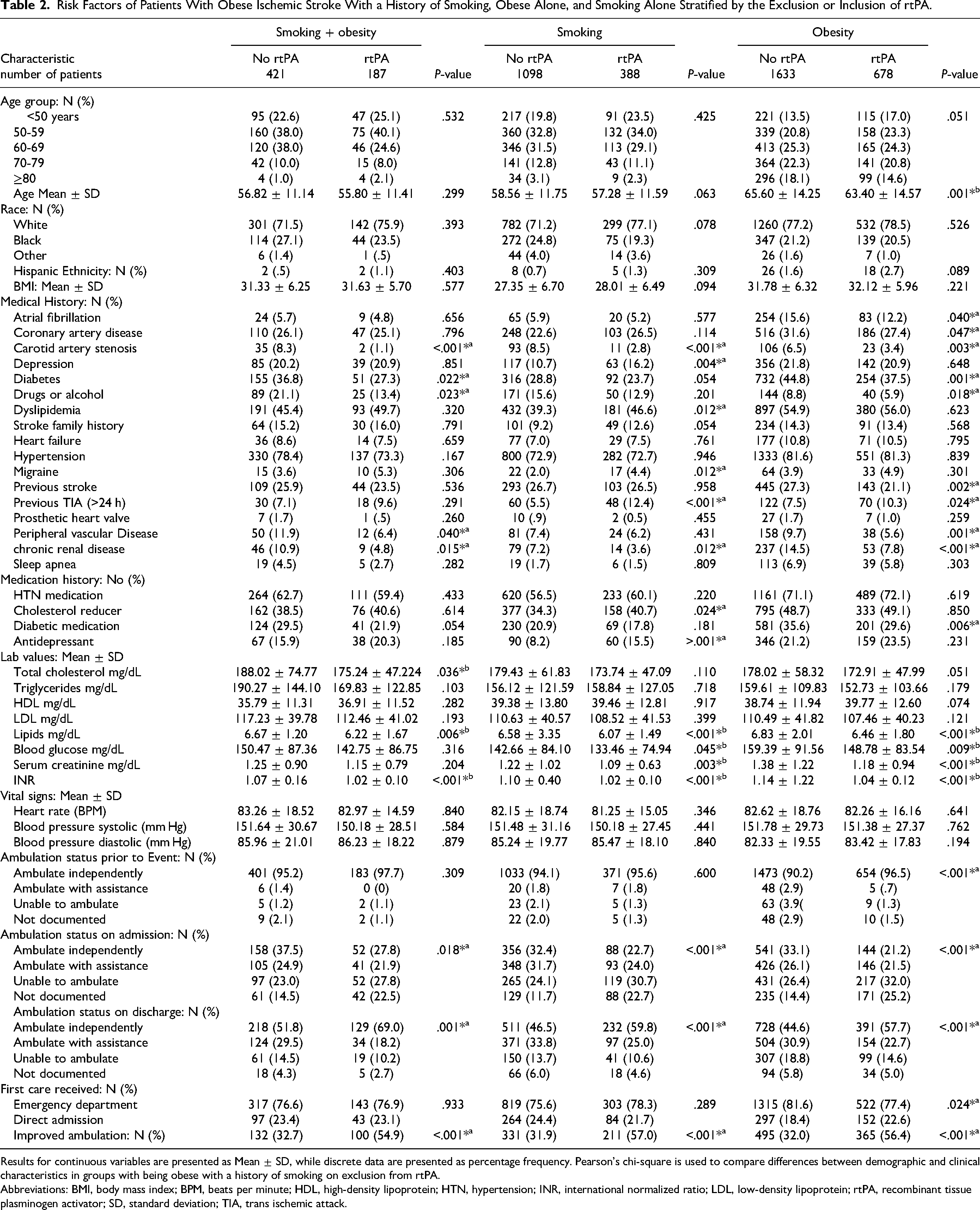
Results for continuous variables are presented as Mean ± SD, while discrete data are presented as percentage frequency. Pearson's chi-square is used to compare differences between demographic and clinical characteristics in groups with being obese with a history of smoking on exclusion from rtPA.
Abbreviations: BMI, body mass index; BPM, beats per minute; HDL, high-density lipoprotein; HTN, hypertension; INR, international normalized ratio; LDL, low-density lipoprotein; rtPA, recombinant tissue plasminogen activator; SD, standard deviation; TIA, trans ischemic attack.
For patients with AIS with a history of smoking, patients with CAS, and CRD who presented with higher lipids, blood glucose, serum creatinine, INR, and ambulated independently at admission were likely to be excluded from rtPA. Those included for rtPA were more likely to present with depression, dyslipidemia, migraine, previous TIA, and with higher NIHSS scores, prescribed with a cholesterol reducer, antidepressants, and ambulated independently at discharge. Obese patients with AIS that were excluded from rtPA were older, presented with atrial fibrillation, CAS, diabetes, alcohol use, previous stroke, CRD and presented with higher levels of blood glucose, lipids, serum creatinine, INR. They were more likely to be prescribed with diabetic medication, and ambulated independently at admission. Obese patients with AIS who were included for rtPA were likely to present with previous TIA and higher NIHSS scores. They ambulated independently before stroke, at discharge with an improved ambulation at the time of discharge, and they were directly admitted to the stroke unit.
Risk factors associated with rtPA inclusion or exclusion in the entire AIS population, correcting for confounding variables is presented in Table 3. As shown in the table, age, CAS, previous stroke, higher total cholesterol, lipids, serum creatinine, and INR were associated with exclusion from rtPA. However, Hispanics with a previous TIA, with cholesterol-reducing medication, antidepressant medication, and presented with higher NIHSS score, and directly admitted to the stroke unit were more likely to be treated with rtPA. The predictive power of the logistic regression model (Figure 1) was strong AUROC = 0.744 (95% CI 0.744-0.762).
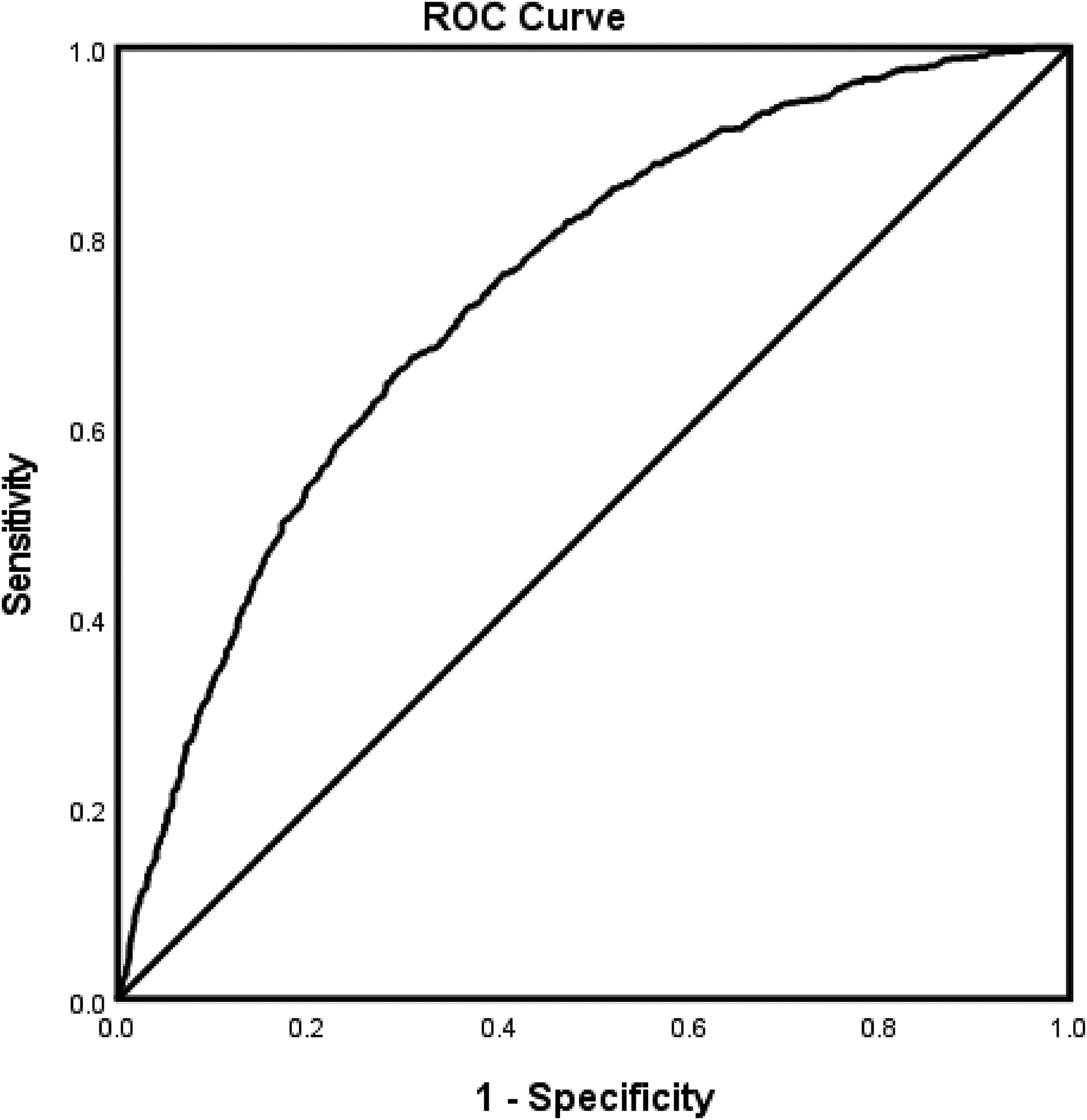
ROC curve associated with prediction of risk factors of patients with ischemic stroke that were included or excluded from rtPA for the whole stroke population. Higher AUC values in ROC analysis indicate better discrimination of the score for the measured outcome. Classification table (overall correctly classified percentage = 67.5%) and area under the ROC curve (AUROC = 0.744 [95% CI 0.744-0.762]) were applied to check model fitness. Abbreviations: AUC, area under the curve; AUROC, area under the ROC; ROC, receiver operating characteristic; rtPA, recombinant tissue plasminogen activator.
Risk Factors of Patients With Ischemic Stroke That Were Included or Excluded From rtPA for the Whole Stroke Population.
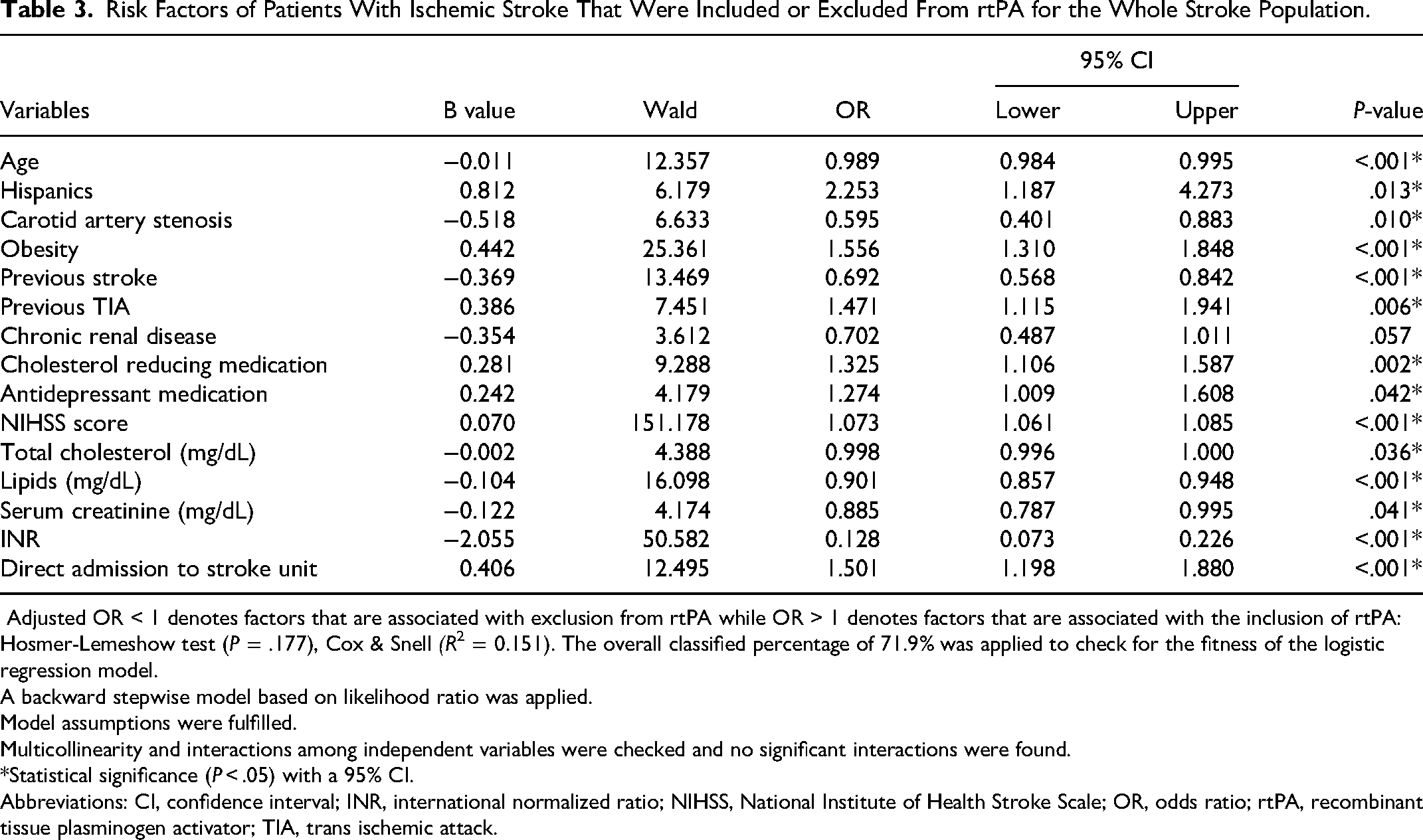
Adjusted OR < 1 denotes factors that are associated with exclusion from rtPA while OR > 1 denotes factors that are associated with the inclusion of rtPA: Hosmer-Lemeshow test (P = .177), Cox & Snell (R2 = 0.151). The overall classified percentage of 71.9% was applied to check for the fitness of the logistic regression model.
A backward stepwise model based on likelihood ratio was applied.
Model assumptions were fulfilled.
Multicollinearity and interactions among independent variables were checked and no significant interactions were found.
*Statistical significance (P < .05) with a 95% CI.
Abbreviations: CI, confidence interval; INR, international normalized ratio; NIHSS, National Institute of Health Stroke Scale; OR, odds ratio; rtPA, recombinant tissue plasminogen activator; TIA, trans ischemic attack.
Risk factors associated with inclusion or exclusion from rtPA in obese patients with AIS with a history of smoking are presented in Table 4. CAS, diabetes, alcohol use, and higher total cholesterol were associated with exclusion from rtPA, while NIHSS score, triglycerides and HDL, were linked with inclusion for rtPA. The logistic regression model (Figure 2) reveals a strong predictive power (AUROC = 0.726 [95% CI 0.677-0.774]).
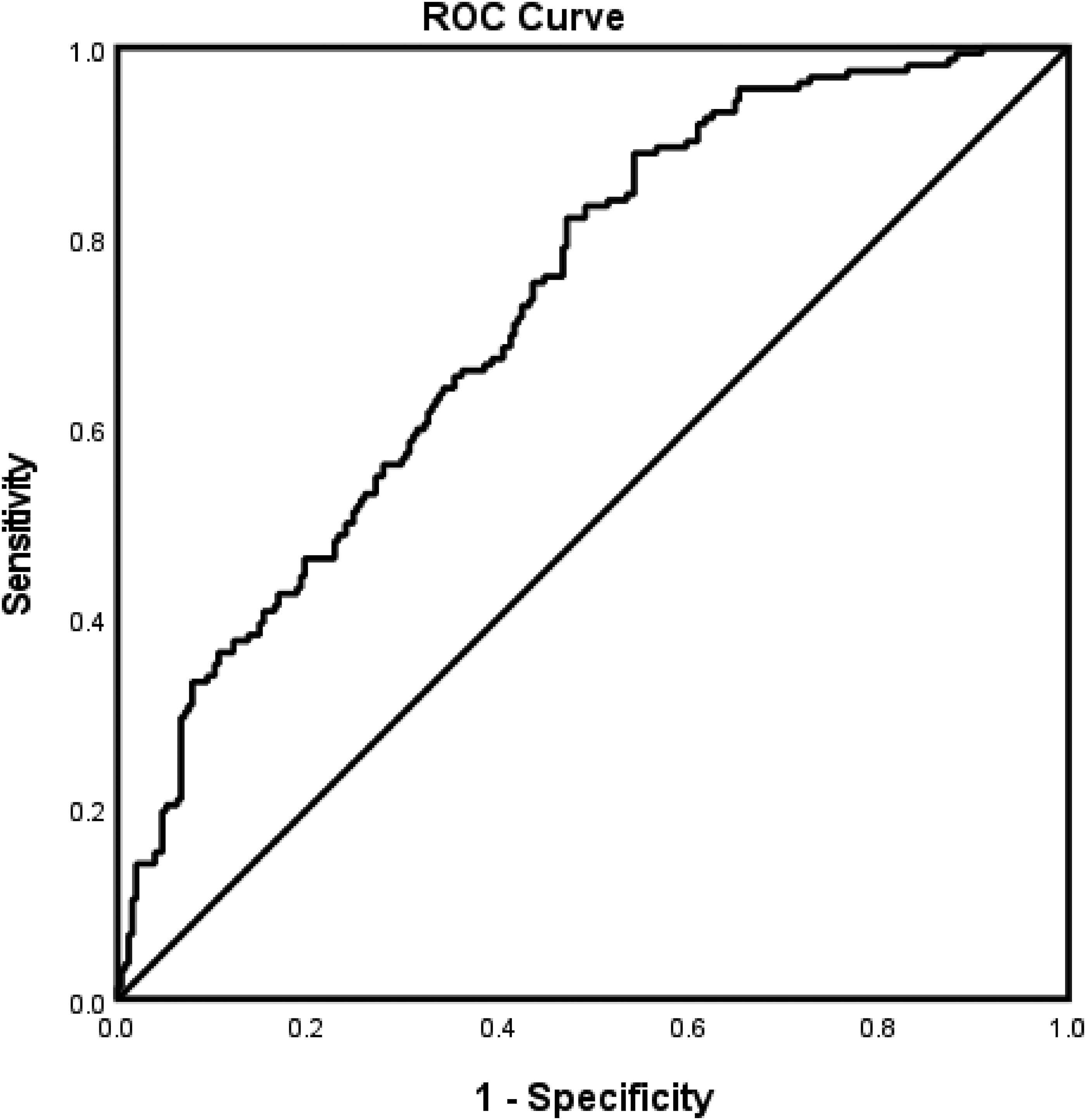
ROC curve associated with prediction of risk factors associated with inclusion or exclusion from rtPA in obese patients with AIS with a history of smoking. Higher AUC values in ROC analysis indicate better discrimination of the score for the measured outcome. Classification table (overall correctly classified percentage = 63.3%) and area under the ROC curve (AUROC = 0.726 [95% CI 0.677-0.774]) were applied to check model fitness. Abbreviations: AIS, acute ischemic stroke; AUC, area under the curve; AUROC, area under the ROC; ROC, receiver operating characteristic; rtPA, recombinant tissue plasminogen activator.
Risk Factors of Patients With Obese Ischemic Stroke With a History of Smoking for Those Excluded or Included for Thrombolytic Therapy.
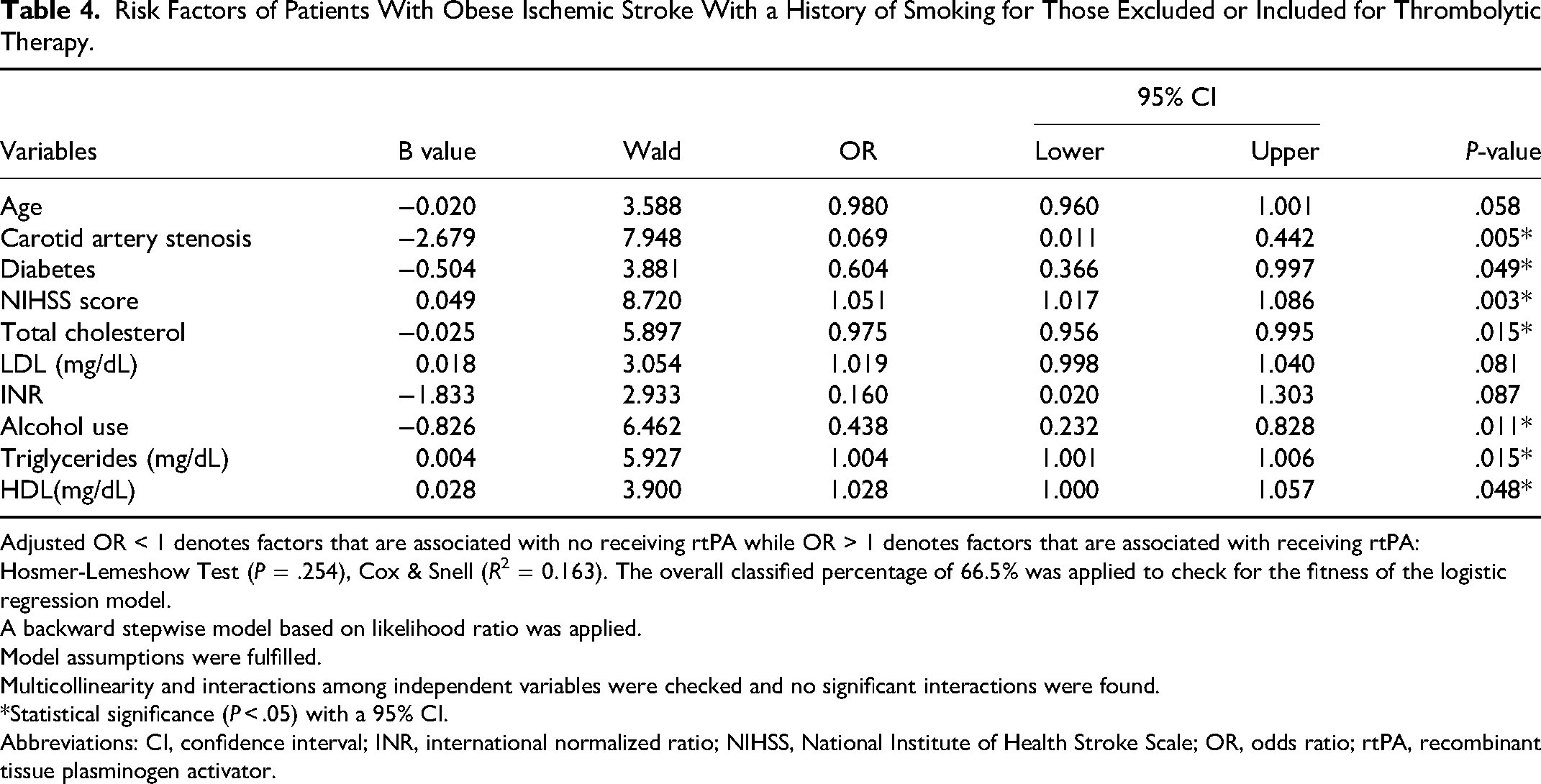
Adjusted OR < 1 denotes factors that are associated with no receiving rtPA while OR > 1 denotes factors that are associated with receiving rtPA: Hosmer-Lemeshow Test (P = .254), Cox & Snell (R2 = 0.163). The overall classified percentage of 66.5% was applied to check for the fitness of the logistic regression model.
A backward stepwise model based on likelihood ratio was applied.
Model assumptions were fulfilled.
Multicollinearity and interactions among independent variables were checked and no significant interactions were found.
*Statistical significance (P < .05) with a 95% CI.
Abbreviations: CI, confidence interval; INR, international normalized ratio; NIHSS, National Institute of Health Stroke Scale; OR, odds ratio; rtPA, recombinant tissue plasminogen activator.
For patients with AIS with a history of smoking (Table 5), age, CAS, alcohol use, higher lipids, and INR were associated with exclusion from rtPA. Caucasians, NIHSS score, CAD, dyslipidemia, and previous TIA were associated with inclusion for rtPA. The predictive power of our model (Figure 3) was strong (AUROC = 0.733 [95% CI 0.700-0.767]).
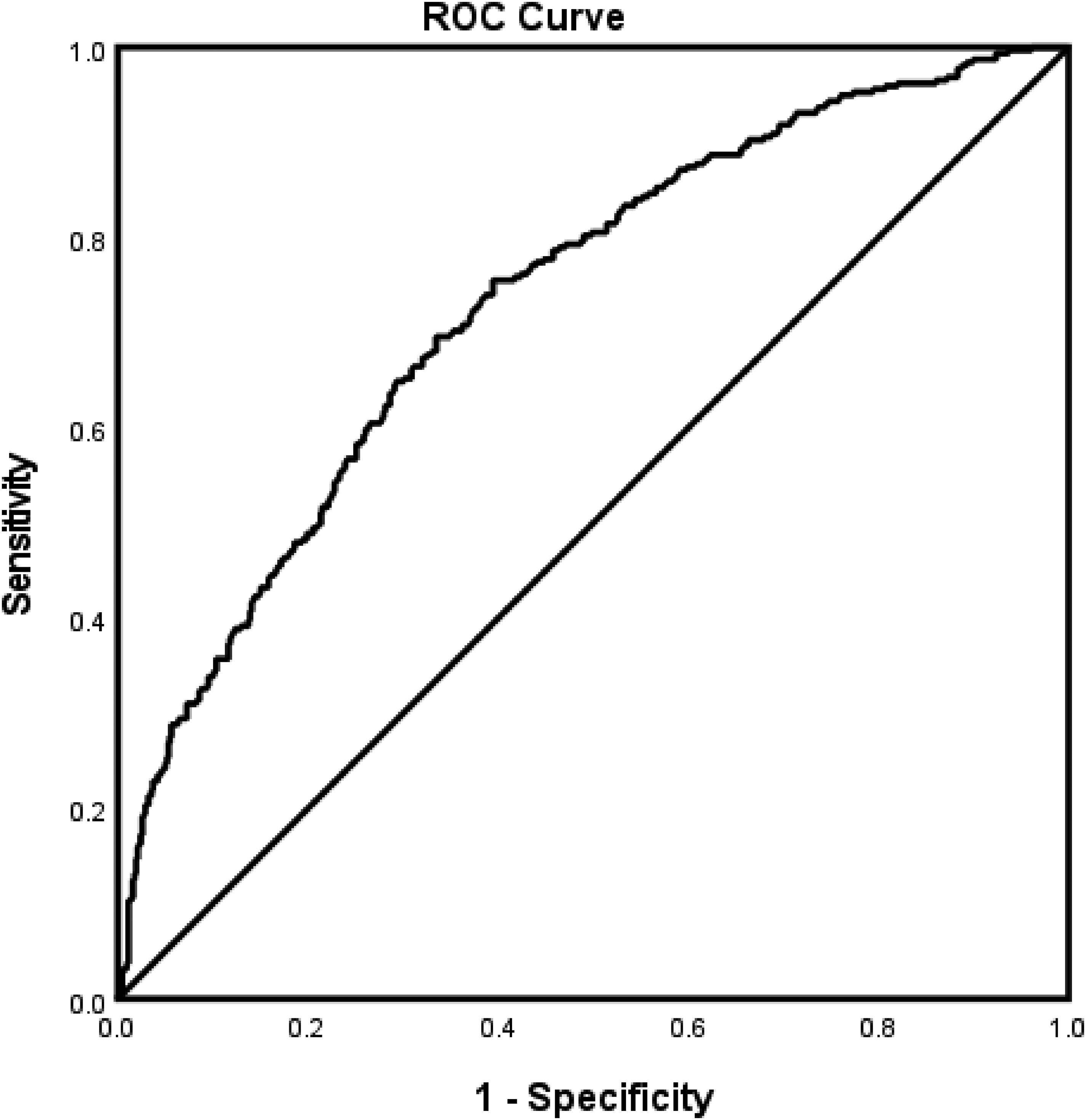
ROC curve associated with prediction of risk factors of patients with ischemic stroke that were included or excluded from rtPA in patients with AIS with a history of smoking. Higher AUC values in ROC analysis indicate better discrimination of the score for the measured outcome. Classification table (overall correctly classified percentage = 66.2%) and area under the ROC curve [AUROC = 0.733 (95% CI 0.700-0.767])were applied to check model fitness. Abbreviations: AIS, acute ischemic stroke; AUC, area under the curve; AUROC, area under the ROC; ROC, receiver operating characteristic; rtPA, recombinant tissue plasminogen activator.
Risk Factors of Patients With Ischemic Stroke With a History of Smoking Excluded or Included for Thrombolytic Therapy.
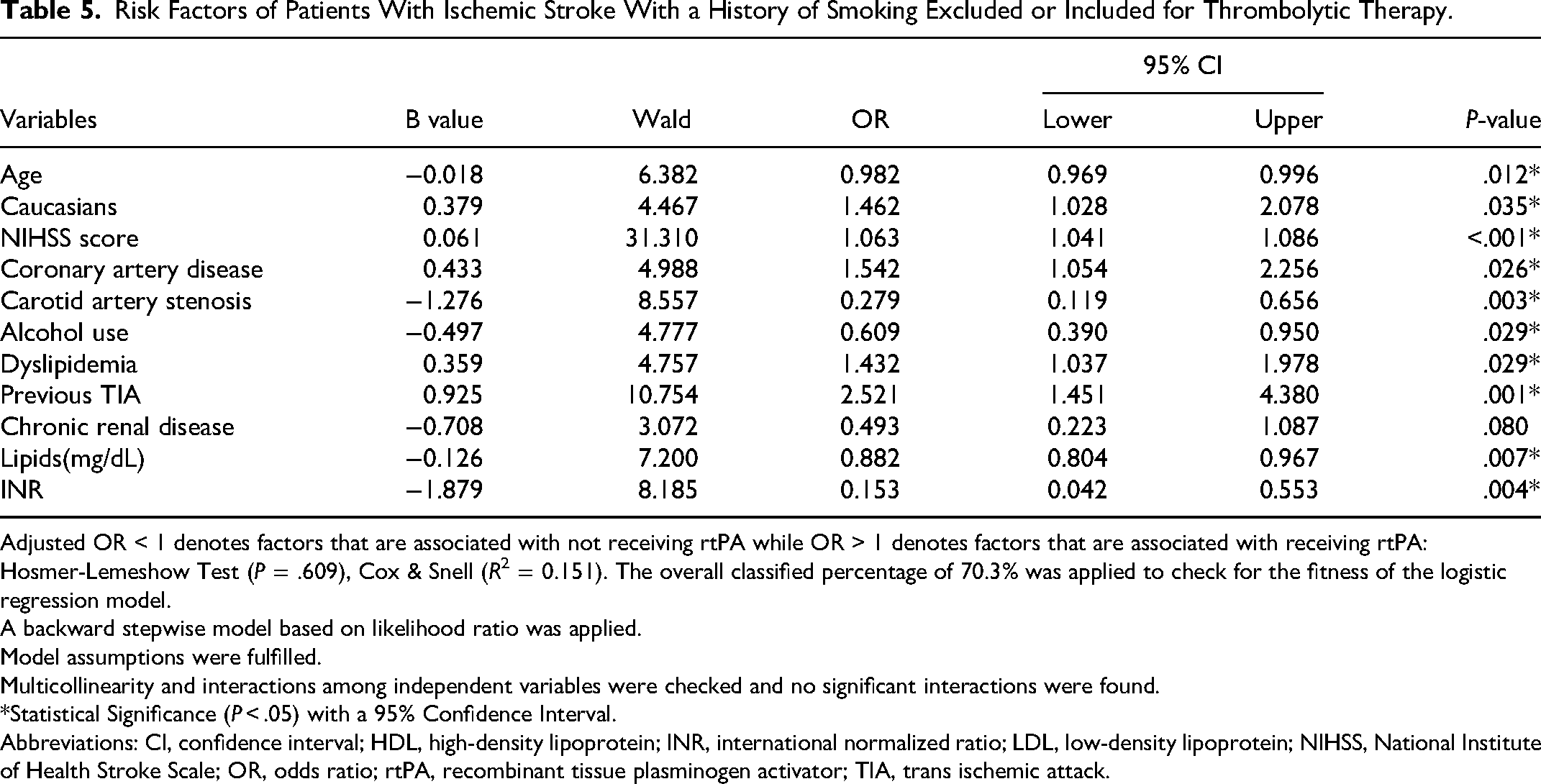
Adjusted OR < 1 denotes factors that are associated with not receiving rtPA while OR > 1 denotes factors that are associated with receiving rtPA: Hosmer-Lemeshow Test (P = .609), Cox & Snell (R2 = 0.151). The overall classified percentage of 70.3% was applied to check for the fitness of the logistic regression model.
A backward stepwise model based on likelihood ratio was applied.
Model assumptions were fulfilled.
Multicollinearity and interactions among independent variables were checked and no significant interactions were found.
*Statistical Significance (P < .05) with a 95% Confidence Interval.
Abbreviations: CI, confidence interval; HDL, high-density lipoprotein; INR, international normalized ratio; LDL, low-density lipoprotein; NIHSS, National Institute of Health Stroke Scale; OR, odds ratio; rtPA, recombinant tissue plasminogen activator; TIA, trans ischemic attack.
The risk factors associated with the inclusion or exclusion of rtPA in the obese AIS population are presented in Table 6. Age, CAS, alcohol use, CRD, higher lipids, and INR, were associated with exclusion from rtPA. However, Hispanics, NIHSS score, and direct admission to the stroke unit were associated with the inclusion of rtPA. Figure 4 presents the ROC curve for the fitness of the logistic regression, which reveals a strong predictive power (Figure 4) of our model (AUROC = 0.729 [95% CI 0.704-0.755]).
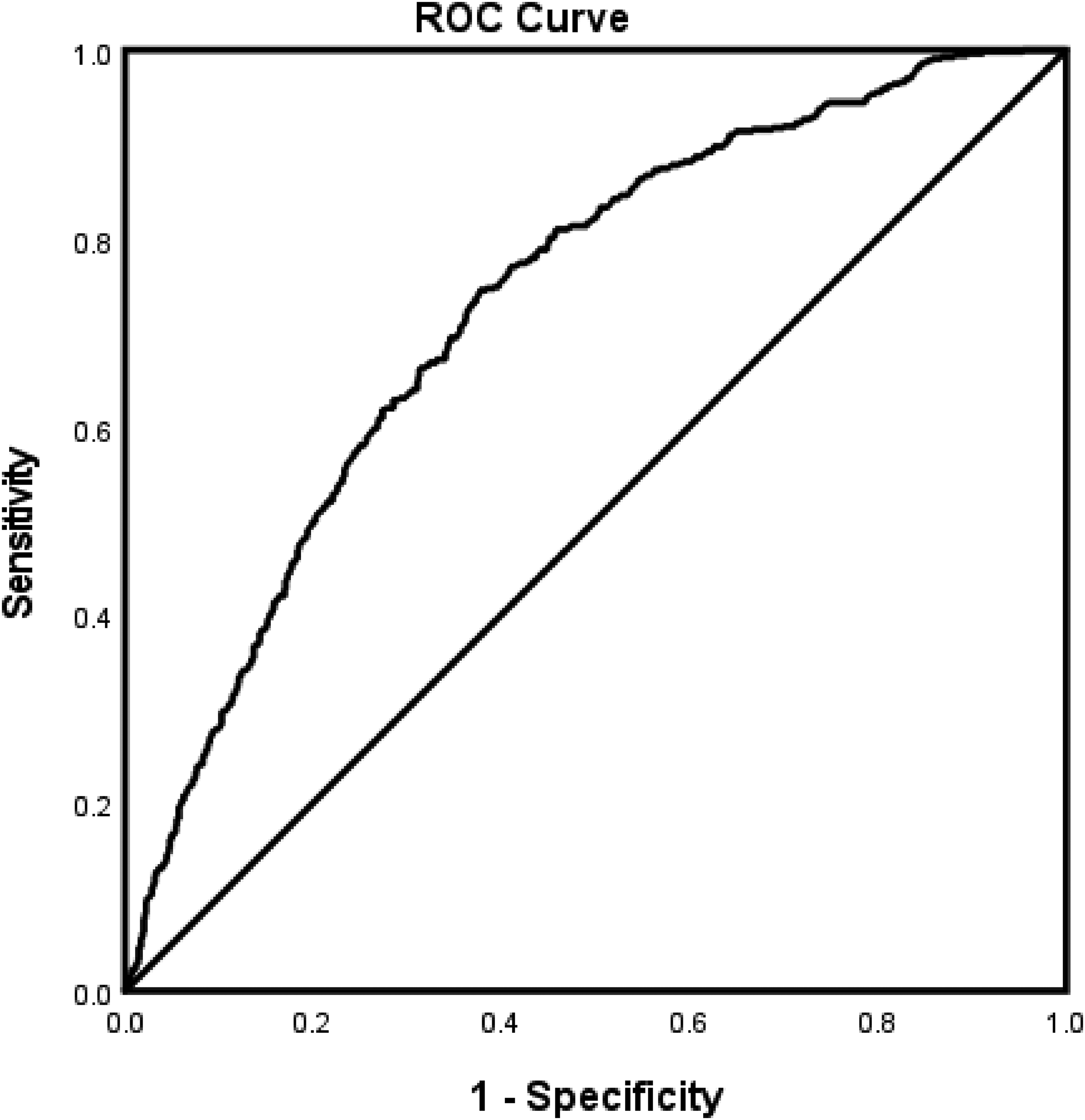
ROC curve associated with prediction of risk factors of patients with ischemic stroke that were included or excluded from rtPA in obese patients with AIS. Higher AUC values in ROC analysis indicate better discrimination of the score for the measured outcome. Classification table (overall correctly classified percentage = 62.4%) and area under the ROC curve (AUROC = 0.729 [95% CI 0.704-0.755]) applied to check model fitness.
Risk Factors of Patients With Obese Ischemic Stroke Excluded or Included for Thrombolytic Therapy.
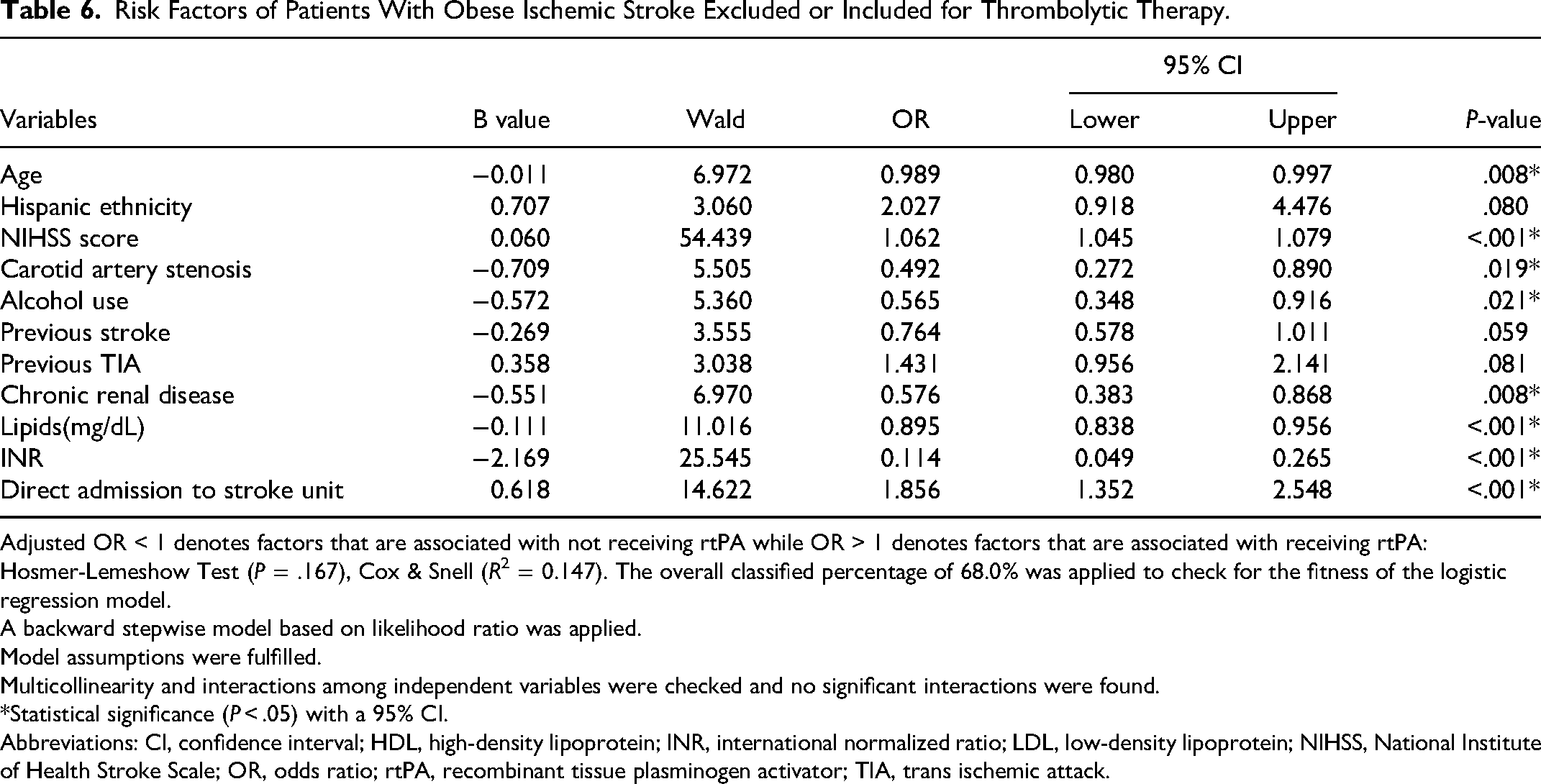
Adjusted OR < 1 denotes factors that are associated with not receiving rtPA while OR > 1 denotes factors that are associated with receiving rtPA: Hosmer-Lemeshow Test (P = .167), Cox & Snell (R2 = 0.147). The overall classified percentage of 68.0% was applied to check for the fitness of the logistic regression model.
A backward stepwise model based on likelihood ratio was applied.
Model assumptions were fulfilled.
Multicollinearity and interactions among independent variables were checked and no significant interactions were found.
*Statistical significance (P < .05) with a 95% CI.
Abbreviations: CI, confidence interval; HDL, high-density lipoprotein; INR, international normalized ratio; LDL, low-density lipoprotein; NIHSS, National Institute of Health Stroke Scale; OR, odds ratio; rtPA, recombinant tissue plasminogen activator; TIA, trans ischemic attack.
In general, our findings reveal differences and similarities (Table 7). For rtPA exclusion, CAS, and alcohol use were associated with obese patients with AIS with a history of smoking, and patients with AIS with a history of smoking or obesity. Diabetes and total cholesterol were only associated with obese patients with AIS with a history of smoking, while lipids and INR were more likely to be associated with the exclusion of patients with AIS with a history of smoking or obesity. For rtPA inclusion, NIH scores was associated with obese patients with AIS with a history of smoking, and patients with AIS with a history of smoking or obesity. Caucasians were associated with patients with AIS with a history of smoking, while Hispanics were associated with obese patients with AIS. Triglycerides and HDL were linked with obese patients with AIS with a history of smoking.
Summary of Differences and Similarities in Risk Factors Associated With Inclusion and Exclusion from rtPA for Obese Patients With AIS With a History of Smoking, and Patients With AIS With a History of Smoking or Obesity.
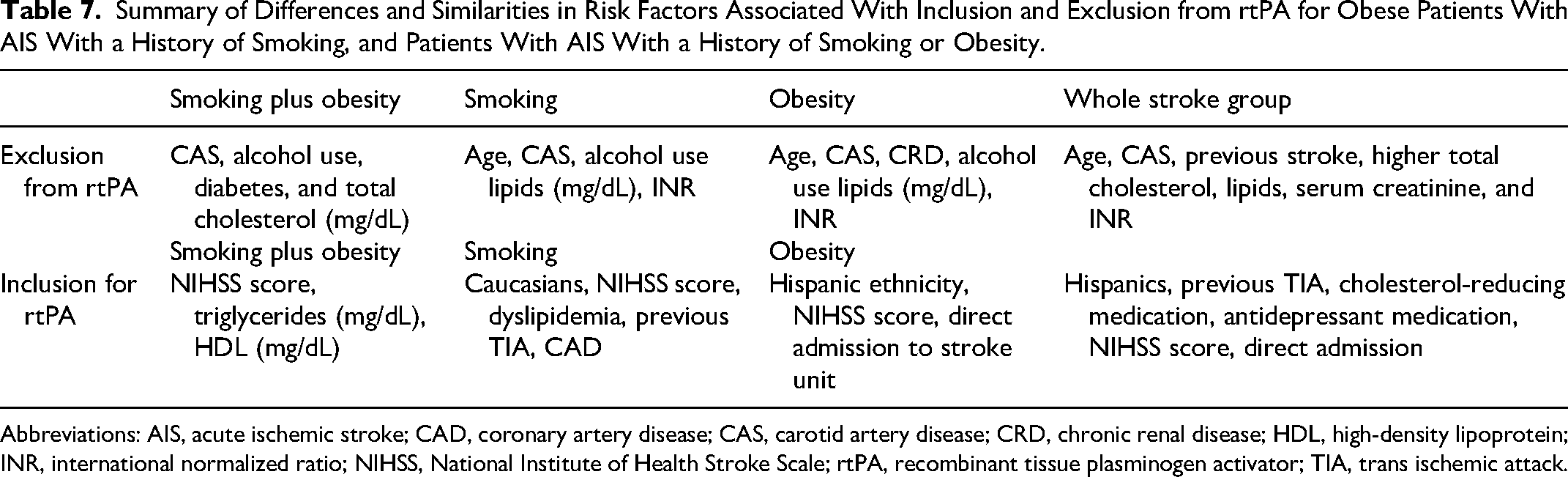
Abbreviations: AIS, acute ischemic stroke; CAD, coronary artery disease; CAS, carotid artery disease; CRD, chronic renal disease; HDL, high-density lipoprotein; INR, international normalized ratio; NIHSS, National Institute of Health Stroke Scale; rtPA, recombinant tissue plasminogen activator; TIA, trans ischemic attack.
Dyslipidemia, previous TIA, and CAD were associated with patients with AIS with a history of smoking. Direct admission was associated with inclusion for rtPA among obese patients with AIS.
Discussion
This study examined risk factors that were associated with the likelihood of being excluded from rtPA in obese patients with ischemic stroke with a history of smoking and obesity, smoking, or obesity. First, we observed that in a population of obese patients with AIS with a history of smoking, patients that present with CAS, diabetes, higher total cholesterol, and alcohol use were more likely to be excluded from rtPA, while those with higher NIHSS score, triglycerides, and elevated HDL were more likely to be included for rtPA. Second, in patients with AIS with only a history of smoking, elderly patients who present with CAS, elevated lipids, INR and a history of alcohol were more likely to be excluded from rtPA, while Caucasians with higher NIHSS scores, that present with CAD, dyslipidemia, and a previous TIA at time of admission were more likely to be included for rtPA. Finally, among the obese AIS population without a history of smoking, elderly patients with CAS, history of alcohol use, CRD, elevated lipids, and INR were more likely to be excluded from rtPA. However, patients with higher NIHSS scores at the time of admission, with a direct admission to the stroke unit for treatment were more likely to be included for rtPA.
In obese patients with AIS with a history of smoking, diabetes, alcohol use, and total cholesterol were associated with the decision for exclusion from rtPA. Diabetes is estimated to be more than 20% in patients with AIS, 25 and undiagnosed diabetes accounts for more than 26% of stroke. 26 Moreover, uncontrolled diabetes produces hyperglycemia which results in poor outcomes including mild, moderate severe intracerebral hemorrhage. 27 Therefore, the risk of hemorrhage in obese patients with AIS with a history of smoking that presents with probably uncontrolled diabetes could have resulted in the fear of hyperglycemia and contributed to the decision to exclude obese diabetic patients with AIS with a history of smoking from rtPA. Future studies on developing therapeutic strategies to reduce the hemorrhage risk with rtPA in a hyperglycemic setting will have a high translational impact on obese and diabetic patients with AIS with a history of smoking.
Our results indicate that total cholesterol (TC) was associated with exclusion from rtPA for obese patients with AIS with a history of smoking. High levels of TC are known to associated with an increased risk of ischemic stroke. 28 Moreover, a high TC level is an independent prognostic factor for thrombolysis 29 with poor outcomes, especially in patients with AIS with severe occlusion of an anterior cerebral artery. 29 Such associations are not completely surprising because cholesterol is a major risk factor for atherosclerosis in coronary artery stenosis. 30 This finding is supported by our current result of the association of higher TC levels with the exclusion of obese patients with AIS with a history of smoking from rtPA. Further studies on strategies to lower cholesterol will improve the care of obese patients with AIS with a history of smoking.
Obese patients with AIS with a history of smoking (smoking plus obesity) and smoking alone with a baseline history of alcohol use were more likely to be excluded from rtPA. Clinical trials indicated a higher baseline stroke severity in patients with AIS after chronic alcohol consumption. 31 Moreover, chronic alcohol exposure increases excitotoxicity resulting in brain damage, 32 and alcohol-induced alterations of other organs contribute to the death of brain cells in patients with stroke. 33 The explanation is that the delayed rtPA clearance by the liver after alcohol consumption could result in a deleterious increase of rtPA passage into the brain parenchyma. 34 Therefore, when this effect is combined with smoking and obesity; it might have contributed to the decision to exclude patients with AIS with the combined effect of smoking and obesity from rtPA.
In our adjusted analysis, obese patients with AIS with a history of smoking and obesity (smoking plus obesity), and smoking or obese alone who present with CAS were more likely to be excluded from rtPA. Patients with carotid stenosis-associated stroke are reported to present with high rates of poor outcomes. 35 Incidence of stroke resulting from the blockage of the carotid artery may occur with varying degrees of carotid stenosis. A severe carotid stenosis can reduce the effectiveness of rtPA, particularly in cases of carotid athero-thrombotic occlusion. 36 Successful recanalization with rtPA is only achieved in less than 30% of patients with carotid stenosis. 37 Our obese patients with AIS with a history of smoking, smoking, or obesity may have presented with severe carotid stenosis that could result in a failed rtPA recanalization. Therefore, determining the degree of carotid stenosis in obese patients with AIS with a history of smoking may help guide the choice and use of rtPA.
We observed that higher lipids and INR were associated with the decision to exclude patients with AIS with a history of smoking or obesity from rtPA. In general, the effect of individual lipid levels on clinical and functional outcomes after ischemic stroke is controversial. Some studies reported that low HDL-C was associated with the severity of stroke, 38 another study argued that this association only existed in young adult patients with stroke. 39 In our current study, the separate components of lipid levels upon admission—namely total cholesterol, LDL-C, HDL-C, and triglyceride levels were not significant in the adjusted analysis for patients with AIS with a history of smoking or obesity. Perhaps the confounding effect of individual lipids in our study population was balanced by an elevation in the overall serum lipid profile accounting for raised levels in our AIS population with a history of smoking or obesity. Moreover, cerebral ischemic patients have a more atherogenic lipid which could result in sICH after treatment of patients with AIS with rtPA resulting in poor outcome. 40 Taking all these studies together with our findings, the risk of sICH after thrombolysis could have contributed to the decision to exclude patients with AIS with a history of smoking or obesity with elevated lipids from rtPA. Future studies to understand the underlying biology that might explain the increased risk of elevated serum lipids and exclusion from rtPA for patients with AIS with a history of smoking or obesity will improve the care of patients with stroke.
INR was associated with the exclusion of patients with AIS with a history of smoking or obesity from rtPA. The current AHA/American Stroke Association guidelines indicate that INR >1.7 is a contraindication for intravenous rtPA. 41 The objective of treating patients with anticoagulant therapy with warfarin is to administer the lowest effective dose of the drug to maintain the target INR, 42 but there are concerns about the safety of using rtPA in patients taking warfarin with an elevated INR as the cutoff value of 1.7 was not determined through randomized controlled trials, and the available data are conflicting. 43 Therefore, regardless of the value of INR, it would be contraindicated if a patient was on an anticoagulant, 7 and rtPA is not recommended in patients with AIS with a platelet count of <100 000/mm, as this causes thrombocytopenia resulting in a greater risk of hemorrhage after rtPA. 44 In our study, the INR for patients with AIS with a history of smoking or obesity was <1.7, it is possible that a combination of factors including, use of anticoagulant, platelet count of <100 000/mm, and high active partial thromboplastin time (aPTT) 45 may have contributed to the exclusion of patients with AIS with a history of smoking or obesity from rtPA.
In patients with AIS with a history of smoking or obesity, increased age was associated with exclusion from rtPA. An estimated one-third of acute strokes occur among elderly people aged ≥80 years. 46 Thrombolysis has been demonstrated to be beneficial among patients aged >80 years. 47 Similar benefits of rtPA have been demonstrated among patients aged ≤80 or >80 years in a Cochrane study. 47 Inclusion of elderly patients ≥80 is advised based on their general health and baseline neurological status. 48 Thrombolysis treatment for patients presenting with AIS should be evaluated on an individual benefit-risk basis, 49 and age alone is no longer a barrier to treatment with thrombolytic therapy. 49 In our current result, the proportion of AIS with a history of smoking or obese who are ≥80 is small compared with other age groups. Therefore, the risk may outweigh the benefit in our older patients with AIS with a history of smoking who were excluded from rtPA.
We observed that CRD was only associated with exclusion from rtPA for patients with AIS with a history of obesity and was not observed in patients with AIS with a combined effect of obesity and smoking or smoking alone. Renal failure is a major risk factor for stroke, and the increased risk of stroke in CRD represents the interplay of vascular comorbidities that occur with renal impairment. 50 Thrombolytic therapy for patients with stroke with CRD is reported to show an increased risk of symptomatic intracranial hemorrhage. 51 The safety and efficacy of many acute treatments have not been demonstrated in the CRD population and similarly, most of the evidence that supports current preventative therapies in this group is from observational studies.52,53 This evidence gap is more obvious among those with advanced CRD or dialysis dependency, who are also the group with the greatest risk. 54 This might have contributed to the exclusion of patients with AIS with a history of obesity and CRD from rtPA in our current study.
Among patients with AIS that were treated with thrombolytic therapy, we observed that NIHSS score was associated with inclusion for rtPA, in (1) obese patients with AIS with a history of smoking, (2) obese AIS population, and (3) patients with AIS with a history of smoking. Severe stroke (eg, NIHSS score >25) is a relative exclusion criterion for intravenous alteplase within 3 to 4.5 h from symptom onset. 44 However, the NIHSS score for most of our patients treated with rtPA was ≤ 20 and reported to be associated with good outcomes after treatment with rtPA in another study. 55
The efficacy of rtPA in patients with AIS due to CAD is similar to nonthrombolysis treatment and is identical to the efficacy of this treatment in patients with stroke from all causes. 56 Moreover, thrombolysis in AIS due to CAD is reported to be safe, with a low rate of SICH complications and the same mortality and recurrent stroke rate as in the nonthrombolysis group. 57 This finding supports our current result indicating that patients with AIS with a history of smoking, with CAD should not be denied thrombolysis therapy.
Patients with AIS with a history of smoking and dyslipidemia were more likely to receive rtPA. While untreated dyslipidemia is generally, associated with a higher stroke risk, 58 this risk is reduced under controlled conditions. 59 Moreover, dyslipidemia is not a contraindication for rtPA. 60 Serum lipid levels on admission including elevated triglycerides and HDL-C levels, were not found to be associated with sICH or 3-month functional outcomes after intravenous thrombolysis for AIS. 61 These results align with our current findings, indicating that obese patients with AIS with a smoking history and elevated HDL, AIS with a history of smoking and elevated triglyceride levels, and those with dyslipidemia and a smoking history may not be excluded from rtPA.
Our findings indicate that individuals with AIS who have a history of smoking, and a previous TIA were more likely to be included for thrombolytic therapy. Patients with AIS with an established TIA less than 24 h exhibit distinct clinical characteristics compared to those without a recent TIA, and were more likely to be excluded from receiving rtPA than those without a TIA 24 h prior. 62 Patients with a TIA established more than 24 h before the onset of stroke typically receive treatment and may be admitted due to the TIA, facilitating the timely administration of rtPA. 63 It is possible that our patients with AIS with a smoking history, and a previous TIA presented with TIA more than 24 h before stroke, making them more eligible for inclusion in thrombolytic therapy.
The impact of ethnicity and race as a major barrier to thrombolytic therapy in the care of patients with AIS is controversial. While ethnic disparities are reported to supersede other risk factors in the use of rtPA for AIS, 64 other studies indicate that Caucasians with AIS received rtPA at a significantly higher rate than Hispanics. 65 It has been shown that inclusion for rtPA was not significantly different between Hispanics and non-Hispanics or Caucasians. 66 This finding supports our current results that both Hispanics and Caucasian patients with AIS with baseline smoking or obesity were more likely to be included in rtPA therapy. Our findings suggest the need to develop strategies to improve the use of rtPA in all patients irrespective of their race or ethnicity. The direct admission of patients with AIS for treatment reduces onset-to-need times for rtPA resulting in improved outcomes in patients with AIS undergoing thrombolysis.67–71 This is because direct admissions of patients with AIS reduces the door-to-needle time to less than 60 min in more than 65% of patients with AIS.72,73 This may have contributed to the inclusion of patients with AIS with a direct admission for rtPA in the current study due to an improvement in time, leading to expedient intervention, and resulting in improved neurological outcomes.74,75
Limitations
This study has some limitations. A major limitation is the reliance on patient-reported data. Patients may not have accurately reported their true smoking history. Additionally, there is no data on the length of time that a patient smoked for previous or current smokers before stroke. This lack of information prevented us from analyzing if the duration of smoking played a role in the risk factors associated with each group. Another significant limitation of this study is that data were obtained from a single regional stroke center. Therefore, the generalizability of the study may not be possible. Furthermore, given the retrospective nature of the study, there is the possibility of selection bias. The data did not include any control groups making it difficult for a long-term follow up. In addition, while it is also possible to treat patients with the different risk factors, the outcomes could also be worse if those patients are not excluded from thrombolysis therapy. Lastly, there were several risk factors known to be associated with stroke that were not included in this study due to inadequate data. Some of these characteristics include metabolic syndrome, moderate or heavy alcohol drinkers, and menopause. In addition, information about specific contraindications for each patient was not accessible for our data analysis. A strength of this study is the ability to analyze risk factors that contribute to the exclusion of patients with AIS with a combined effect of obesity and smoking. An important contribution of this work to stroke neurology is our findings that there are differences and similarities in risk factors associated with exclusion from rtPA in patients with AIS with a combined effect of obesity and smoking when compared with patients with AIS that are obese with a current or previous of smoking.
Conclusion
Safety of rtPA implies that patients with ischemic stroke treated with rtPA are not associated with complications related to intravenous rtPA including symptomatic intracranial hemorrhage, and major systemic hemorrhage. Patients with AIS with a combined effect of obesity and smoking or smoking alone are less likely to be treated with rtPA if they present with comorbidities or risk factors that may worsen outcomes following rtPA therapy and are therefore unable to benefit from the demonstrated advantage of rtPA reperfusion therapy. Our results reveal that obese patients with AIS with a history of smoking, patients that present with CAS, diabetes, higher total cholesterol, and alcohol use were more likely to be excluded from rtPA. In addition, among patients with AIS with only a history of smoking, elderly patients who present with CAS, elevated lipids, INR and a history of alcohol were more likely to be excluded from rtPA. Finally, among the obese AIS population without a history of smoking, elderly patients with CAS, history of alcohol use, CRD, elevated lipids, and INR were more likely to be excluded from rtPA.
The rtPA is an approved FDA therapy for the treatment of ischemic stroke. However, many untreated patients are excluded based on several risk factors. While it is also possible to treat patients with the different risk factors, the outcomes could also be worse if those patients are not excluded from thrombolysis therapy. Therefore, investigating patients with stroke with a combined effect of smoking and obesity, including others risk factors that are associated with exclusion from rtPA is of major clinical importance. Treatment of stroke risk factors might conceivably reduce the future risk of stroke. Our findings identified risk factors that can be managed to improve the eligibility of patients with AIS with a combined effect of obesity and smoking or smoking to for rtPA therapy.
Footnotes
Acknowledgment
We thank the stroke unit of Greenville Health System for helping with data collection.
Authors’ Contribution
PCB, DTO, PXB, AI-N, and TI-N designed the concept, experimental design, and data analysis. PCB and PXB participated in writing the manuscript, data collection, and editing the manuscript. All co-authors provided critical reviews on different drafts of the manuscript. All authors read and approved the final manuscript for publication.
Availability of Data and Materials
Materials are available on request from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This is a retrospective data collection. This study was approved by the Institutional Review Board of PRISMA Health institutional committee for ethics (approval number: 00052571). All data were fully anonymized before they were accessed. Data used in our retrospective analysis were from the PRISMA Health stroke data registry. This study did not require informed consent for participation by the national legislation and institutional requirements.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Institut National de la Santé et de la Recherche Médicale (grant number 5R25AG067934-04).
