Abstract
Acute ischemic stroke (AIS), characterized by high morbidity and mortality, has imposed a considerable burden on society. Despite rapid development in the treatment of AIS, there is still a high risk of recurrence. Furthermore, there is a time delay in waiting for the results of conventional coagulation tests in candidate patients for intravenous thrombolysis therapy. Heterogeneous responses to antiplatelet, intravascular thrombolysis, and endovascular therapies also worsen the situation. Thromboelastography (TEG), as a global and portable detection method for hemostasis, facilitates clinicians in disease monitoring, treatment evaluation, and prognosis prediction in AIS. In this narrative review, we provided a comprehensive summary of the clinical application of TEG in ischemic stroke and gave insights to further studies.
Introduction
Acute ischemic stroke (AIS), as one of the primary threats to human life, has imposed a considerable burden on society . 1 Early recognition of AIS patients and timely reperfusion to ischemic penumbra are the primary missions of healthcare professionals. Anti-platelet therapy is the fundamental treatment of atherosclerotic ischemic stroke. However, there is still a high risk of recurrence in patients with anti-platelet resistance. The intravenous thrombolysis could be beneficial in early reperfusion for AIS patients. However, the strict exclusion criteria and narrow time window of intravenous thrombolysis limit its general application. 2 There is a time lag in waiting for the results of conventional coagulation tests (CCTs) for those taking oral anticoagulants. Besides, the subsequent early neurological deterioration and hemorrhagic transformation (HT) make the situation more complicated. Timely efficient monitoring of coagulation hemostasis will permit early detection of these complications in AIS patients. Thus, a rapid and multi-functional clot formation assay is needed to resolve the dilemma.
TEG, first invented by Doctor Hartert in 1948, 3 is a viscoelastic hemostatic assessment technology widely used in various fields, such as surgeries, obstetrics, cardiovascular diseases, and more recently, COVID-19 infection.4–8 CCTs include parameters such as prothrombin time (PT) and activated partial thromboplastin time (APTT). CCTs ignore the comprehensive function of hemostasis, and may delay the decision-making in the emergent setting of AIS. In contrast, as a whole-blood, real-time, visualized analyzer of clot formation and lysis, TEG reflects the global function of hemostasis in point-of-care settings. The advantages provided by TEG have made it more and more popular in the management of AIS patients. However, there is a lack of large-scale research regarding the application of TEG in prognosis prediction and endovascular therapy. The main goal of this review is to systematically summarize the application of TEG in AIS, and shed new light on the potential use of TEG in further studies.
The Hemostasis status of Coagulate Function in AIS
The Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification
9
is a universal classification system that divides AIS into five major types based on disease etiology. The diverse causes of cerebral infarction contribute to the imbalance of the coagulation and fibrinolysis systems. The physiological hemostasis of these systems can be referred to
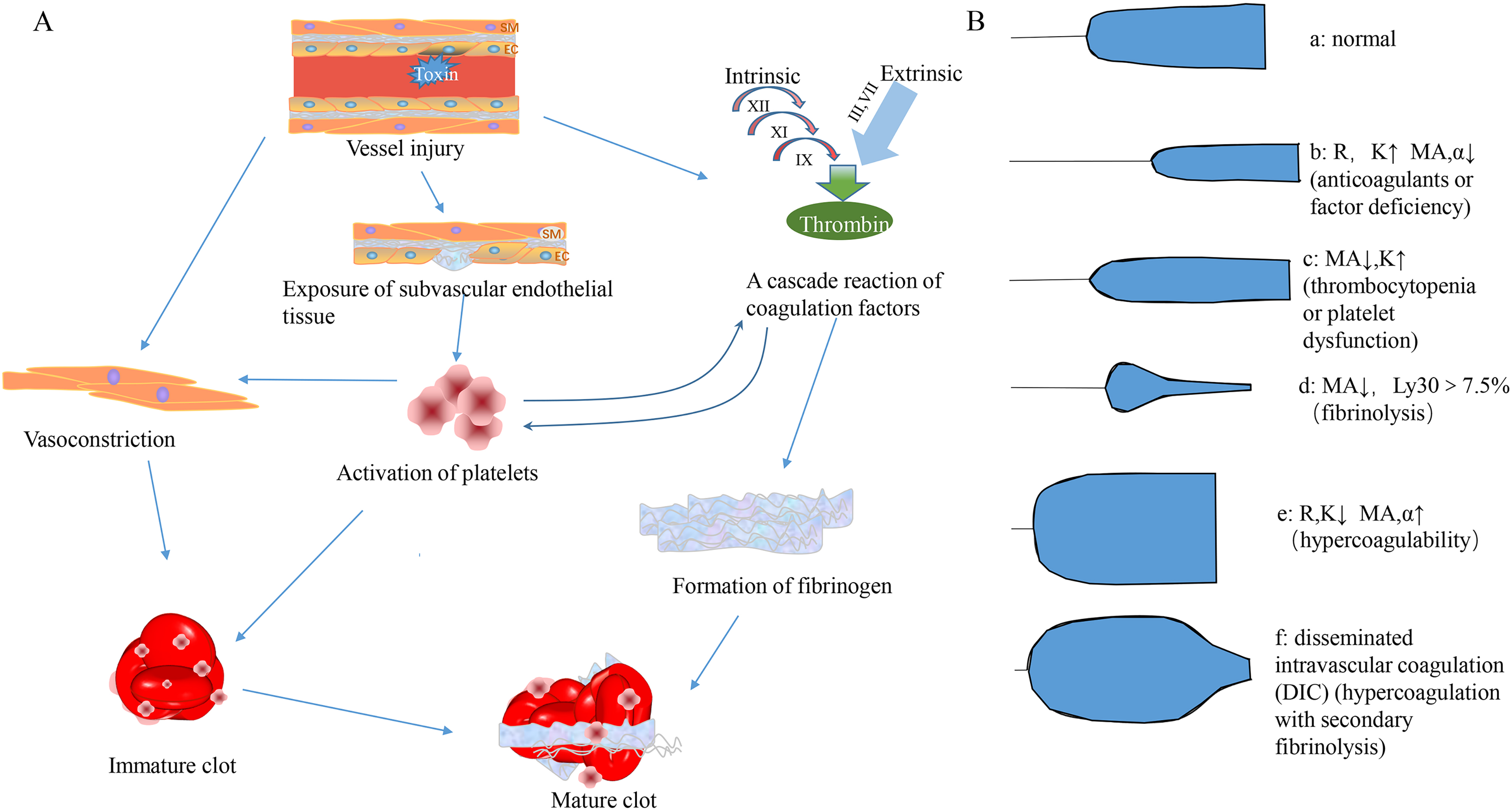
A: The physiological hemostasis. B: The representative plots of TEG under different circumstances. The figure B was adapted from the work of V Srinivasa et al 97
Accumulating evidence shows that AIS is related to hypercoagulability. The hypercoagulable state exists in 29-38% of AIS patients. 16 Higher D-dimer has been observed in cardioembolic stroke,17,18 cancer-related cerebral infarction,19,20 and stroke in the COVID-19 setting. Hyperfibrinogenemia relates to long-term mortality in AIS patients.21,22 With knowledge of coagulation change in AIS, clinicians can implement proper individual therapy for patients better.
The Introduction of TEG
The Principle
The specimen of TEG should be non-citrated or citrated whole blood. Citrated whole blood needs 30 min to equilibrate at room temperature, whereas the non-citrated one must be added into the assay within 4 to 6 min from the draw to avoid clot formation. The TEG cup that holds 0.36 ml blood can automatically control the thermostat at 37°C. Calcium is needed to trigger the coagulation process. A pin suspended by a torsion wire is submerged in the cup, which moves 4.45° every 10 s. The pin rotates with the cup, motivated by shear stress between the blood and the cup. The speed of the pin will reduce when the blood forms a clot gradually due to the resistance. The dynamic movement information of the pin is collected electronically and translated into TEG tracing by a mechanical-electrical transducer. 23
The Parameters
Referring to the instruction of TEG by the HAEMONETICS company, reaction time R (time range from the beginning of the tracing to the amplitude of 2 mm) represents the initial fibrin formation. It mainly reflects the activity of the endogenous coagulation system. R time can be shortened by hypercoagulable conditions and prolonged by anticoagulants. K (time range from amplitude 2 mm to 20 mm) reflects the process of polymerization and cross-linking of fibrin to achieve stable elastic viscosity. Angle α (an angle formed by the slope of the TEG tracing between R and K from the horizontal line) shares the same clinical significance as K. It replaces K in conditions where the amplitude fails to obtain 20 mm in a severe hypocoagulable state. Both K and α denote the speed of clot formation, which is mainly influenced by the function of fibrin. MA (the maximum amplitude on TEG tracing) indicates the absolute strength of the developed clot, which generally depends on the function of platelets. EPL (an estimated percentage of clot lysis at 30 min after MA) and LY30 (an actual percentage of clot lysis at 30 min after MA) show the process of thrombolysis or contraction of the clot. Coagulation index and shear modulus strength (G) describe the overall coagulation state in patients. Delta is the time difference between R and the time of the initial split point (SP) on the TEG tracing (R – SP), suggesting the intensity of the thrombin burst.
24
The principle and parameters of TEG are shown in
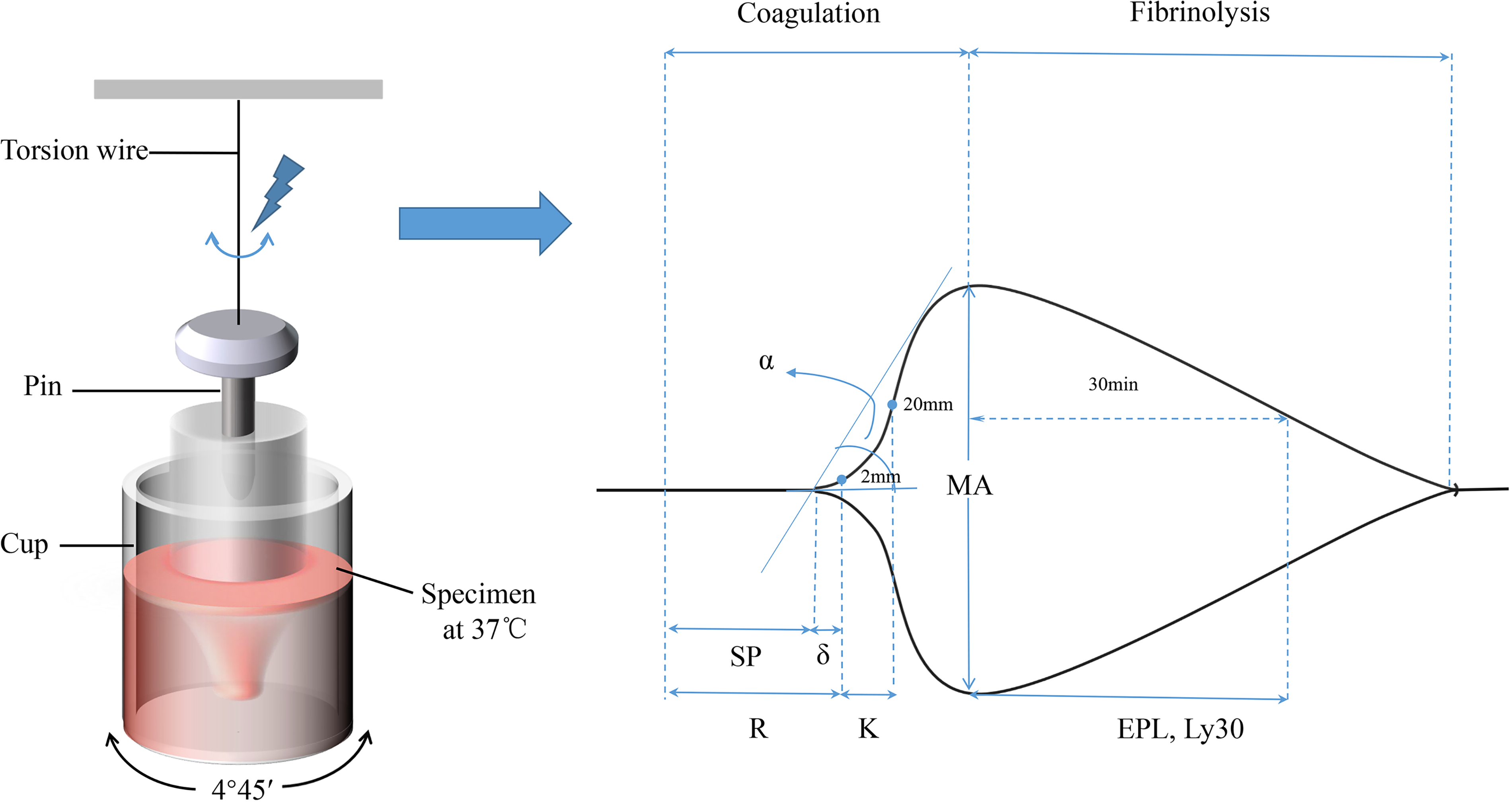
The principle and regular parameters of TEG.
Technology
There are many different technological modifications of TEG. As one of the viscoelastic hemostatic assays (VHA), TEG has evolved into several updated versions. Rotational Thromboelastometry (ROTEM) is a modern modification of the TEG. Both TEG and ROTEM operate by transforming the viscoelastic strength in developing a clot into an electromechanical signal. TEG and ROTEM reflect essentially the same information on the viscoelastic strength of the clot. However, there are some differences between them in pipetting, nomenclature, sensitivity to vibration, and the maximum number of samples that can be conducted simultaneously. 25 The newer TEG 6 s method is operated by a disposable multi-channel microfluidic cartridge, which can increase the resistance to vibration and reliability. 26 There are mainly 6 TEG assays available 27 : 1. Kaolin assay. Kaolin is an intrinsic coagulation activator and provides information similar to APTT and reflects the process of contact activation. The parameters include R, K, Angle, MA, LY30, and EPL. 2. Rapid TEG assay. Tissue factor and kaolin are activators in this assay. Rapid TEG provides analogous information with activated clotting time (ACT). The function of testing coagulation kinetics initiated by contact activation alone is lost in the Rapid TEG assay. 3. HTEG assay. It uses lyophilized heparinase as a reagent to neutralize unfractionated heparin. In combination with kaolin reagents, the efficacy of heparin is assessed by comparison with kaolin analysis alone. 4. Functional Fibrinogen assay. The reagent contains tissue factors and the GPIIb/IIIa platelet receptor inhibitor abciximab. Compared with kaolin analysis, the contribution of fibrinogen to blood clot strength can be analyzed without the effects of platelets. 5. Platelet Mapping assay. This assay contains Activator F (reptilase and factor XIIIa) in heparinized blood. Heparin is used to suppress the generation of thrombin, and Activator F transforms the fibrinogen into fibrin. In the absence of thrombin, platelet response to subsequent addition of adenosine diphosphate (ADP) or arachidonic acid (AA) is a result by comparison to Kaolin analysis. Thrombin is the strongest platelet agonist, directly activating platelets through the PAR1 receptor, which can not be inhibited by cyclooxygenase inhibitor and P2Y12 inhibitor. By Platelet Mapping assay, the degree of decrease in maximum platelet activation produced by thrombin (MAThrombin) compared with that produced by AA (MAAA) or ADP (MAADP) calculated as inhibition rate can reflect the effect of aspirin or clopidogrel, respectively. 6. Native TEG assay. This assay uses calcium as an activator and is impractical for clinical use due to the long R parameter.
The Advantages and Limitations of TEG
As the different types of thrombi mentioned before, the dedicated hemostasis balance involves the plasma factors and cellular elements. CCTs and platelet counts emphasize the isolated, partial function of hemostasis. Furthermore, they usually require plasma specimens centrifuged from the whole blood, and thus are time-consuming and problematic. Clot lysis measurements can be influenced by renal and hepatic function. Light transmittance aggregometry (LTA) is a gold standard for testing platelet function currently. Nevertheless, LTA is inefficient and requires well-trained professionals. 28 In contrast, TEG enables the analysis of global hemostatic function based on the balance from the aggregation of platelet, coagulation, and fibrinolysis. Moreover, graphic TEG results can be acquired in 11 min at the bedside, relatively less dependent on skilled staff or special equipment. TEG and CCTs are agreeable in hypercoagulable or hypocoagulable states.29,30 Although the correlation between TEG and most of CCTs is inconsistent, clot strength (MA) and clot formation (K time and α angle) have been proven to correlate with fibrinogen levels.29–31
However, TEG can not detect the conditions affecting platelets adhesion to the vascular wall. Given that TEG analysis is performed at 37°C in vitro, the effect of temperature alterations on the hemostasis in vivo should not be ignored when interpreting the TEG results. 7 From the testing principle, we can conclude that the value of TEG could be affected by oscillation and shock. Each institution should consider the methods of specimen transportation, since the TEG results might be influenced by specimen delivery. For example, the vibration of pneumatic tube system can influence the result of TEG. 23 Whether the baseline amounts of platelet and RBCs disturb the result of TEG is not known yet. 32 Notably, there is a lack of consensus on quality control in different TEG studies. The compatibility and reliability of the conclusions drawn from pooled data are questionable.33,34 Due to the nature of the assay and the variability between different TEG methods, it remains challenging to achieve standardization in multicenter studies. An ongoing global study involving nine laboratories has been performed by the TEG-ROTEM Working Group. It showed a significant inter-laboratory variance (co-efficient of variance greater than 10%), which might be caused by the operator variability. 35 This was the first study investigating the reliability and reproducibility of TEG. Meanwhile, guidelines for using TEG and ROTEM in hemophilia patients and patients with coagulopathy are available currently.36,37 In addition, detailed precautions and issues related to interpreting TEG results were summarized by Othman M et al. 38 The authors also recommend using TEG and other CCTs simultaneously to assure the effectiveness and quality of TEG evaluation. It is suggested that each patient should have a baseline TEG value to obtain their personalized reference value before treatment. 39 In summary, all laboratory assessments of hemostasis for the clinic should be interpreted cautiously, considering each piece of information from physical examination and disease history. 23
The Application of TEG in AIS
The major studies of TEG in ischemic stroke have been summarized in
The Major Studies of TEG in Ischemic Stroke.
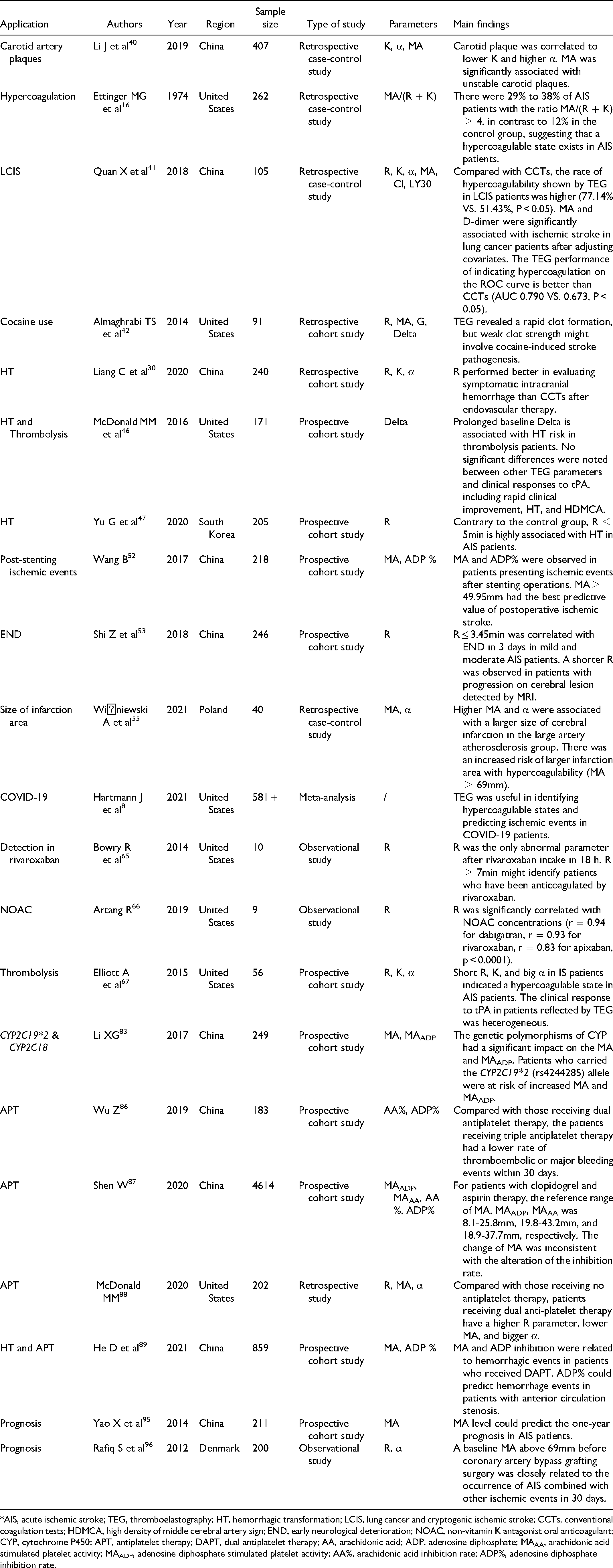
*AIS, acute ischemic stroke; TEG, thromboelastography; HT, hemorrhagic transformation; LCIS, lung cancer and cryptogenic ischemic stroke; CCTs, conventional coagulation tests; HDMCA, high density of middle cerebral artery sign; END, early neurological deterioration; NOAC, non-vitamin K antagonist oral anticoagulant; CYP, cytochrome P450; APT, antiplatelet therapy; DAPT, dual antiplatelet therapy; AA, arachidonic acid; ADP, adenosine diphosphate; MAAA, arachidonic acid stimulated platelet activity; MAADP, adenosine diphosphate stimulated platelet activity; AA%, arachidonic acid inhibition rate; ADP%, adenosine diphosphate inhibition rate.
Disease Monitoring in AIS
TEG exerts a notable role in disease management and complications monitoring in ischemic stroke. A cross-sectional study in 2019 enrolled 407 AIS patients to investigate the relationship between the TEG and carotid artery plaques. The baseline TEG information and carotid-protocol duplex ultrasound data were collected. Compared to patients without carotid plaques, a lower K and higher α-angle were associated with the existence of carotid plaques. Moreover, a lower MA was related to unstable carotid plaques, indicating that the high activity of fibrinogen and platelets is associated with unstable carotid plaques. After adjusting the baseline clinical data, no TEG parameters have been proven to predict plaque presence or stability. 40
A study collected a TEG ratio of MA/ (R + K) from AIS patients in 2 institutes, which showed that 29% to 38% of stroke patients exceeded 4, implying that a hypercoagulable state existed in ischemic stroke patients. 16 In a small group of lung cancer and cryptogenic ischemic stroke (LCIS) patients, researchers found that D-dimer and MA were independently associated with ischemic stroke. However, no difference was observed in LY30 in these patients when compared with age- and sex-matched lung cancer patients and healthy volunteers. This may denote the contribution of enhanced clot formation rather than decreased fibrinolytic activity in LCIS patients. 41 To better understand the mechanisms of cocaine use in acute stroke patients, Almaghrabi and colleagues analyzed the TEG parameters between cocaine users and non-cocaine users with either AIS or spontaneous intracerebral hemorrhage. They found that shortened median R time, reduced MA and G existed in patients of cocaine users. 42 These data revealed a rapid clot formation but weak clot strength in cocaine users.
HT is defined as bleeding in the brain tissue of the infarcted area, and TEG is expected as a supplementary way to recognize HT in AIS patients. HT is one of the most dangerous complications of AIS, especially in patients who received fibrinolysis and dual anti-platelet therapy. The occurrence rate of HT in AIS patients is about 8.5-12%.43,44 Currently, the detection of HT relies on radiology examination. Meanwhile, TEG has been wildly used in bleeding events in coronary surgeries, postpartum hemorrhage, and intracerebral hemorrhage.4,6–45 However, only a few studies investigated the role of TEG in bleeding events in AIS patients. Compared to CCTs, the parameter R is supposed to be superior to indicating the symptomatic intracranial hemorrhage after endovascular therapy. 30 Baseline delta lengthening is associated with hemorrhage transformation in AIS patients after thrombolysis therapy. 46 R <5 min was proved as an indicator of hemorrhagic transformation (odds ratio = 3.215, 95% CI = 1.153-8.969, P < 0.05) in a study that included 205 AIS patients. 47
Besides bleeding events, ischemic complications such as venous thromboembolism (VTE) are common in severe AIS patients. Pulmonary embolism and deep vein thrombosis could worsen the complicated condition of AIS patients. A negative D-dimer blood test can rule out VTE, but it could be impacted by many conditions such as cancer, infection, and trauma. 48 The slow rotation of the cylinder used in TEG is closely resemble the process of venous thrombosis at low flow rates. TEG may be a valuable tool to identify patients in hypercoagulation or at risk of VTE, especially in critically ill patients and traumatic populations. 49 A prospective, double-blind observational study in surgical ICU showed that a high thrombodynamic ratio (TDR, calculated by dividing the sum of MA and α-angle by R) was predictive of VTE occurrence. Moreover, TDR less than 10.6 could rule out VTE (100% sensitivity and 0 negative likelihood ratio). 50 However, in a study of emergency care, no TEG parameters were related to VTE. 51
Pre-stenting TEG parameters were found to be related to ischemic events after carotid stenting in a prospective study. Eighteen of 218 patients presented either a transient ischemic attack (TIA) or AIS in the area of the stented artery. After stenting operations, patients with ischemic events had higher MA and ADP inhibition rates than non-ischemic patients. Preoperative MA and ADP inhibition rates were able to predict subsequent ischemic events. MA > 49.95 mm had the best predictive value of postoperative ischemic stroke (AUC = 0.654, 95% CI = 0.520 to 0.789, P = 0.03). 52
END is defined as the NIHSS score adding 1 point or occurrence of new symptoms in 72 h from admission. 53 The occurrence rate of END is 2% to 37.5% in AIS patients. 54 A study conducted on 246 patients who presented ischemic stroke with National Institutes of Health Stroke Scale (NIHSS) score ≤14 showed that a shortened R parameter on admission was a predictive factor for END within three days after AIS onset. 53 In a cross-sectional, single-center study, higher MA and bigger α were associated with a larger size of ischemic infarct in the large artery atherosclerosis group. 55
Moreover, AIS is the most common neurologic complication of coronavirus disease 2019 (COVID-19).56,57 The incidence of AIS induced by COVID-19 ranges only from 1% to 6%, but the motility rate of 38% is relatively high. Clinical symptoms of patients who suffered both ischemic stroke and COVID-19 tended to be severer.58,59 A variety of studies has proven that TEG had an advantage in the identification of hypercoagulable state in COVID-19 patients, and was helpful for the prediction of ischemic events. 8
Treatment Guidance in AIS
Thrombolysis
Thrombolytic therapy with rt-PA is the first choice for AIS patients in the initial 4.5 h of disease onset. There have been many tests for assessing fibrinolysis effects of rt-PA treatment. However, those tests have some problems in coagulation inhibition or blood sampling,60,61 and were not suitable for clinical application. Some modified TEG assays were thus invented to better assess fibrinolysis effects of rt-PA treatment.62,63
TEG facilitates selection of patients taking anticoagulants for intravenous thrombolysis therapy. Given the limited evidence, there are some uncertainties regarding rt-PA treatment in AIS patients taking non-vitamin K antagonist oral anticoagulant (NOAC). The recommendation from American Heart Association/American Stroke Association is not to administer rt-PA to patients who took NOAC in 48 h. 64 However, it is sometimes difficult to determine the exact time of taking NOAC. For those patients taking factor Xa inhibitors, there will be a time delay when waiting for the results of CCTs. In contrast, the TEG results can be acquired at the bedside more rapidly. R value greater than 7 min could identify patients who have taken rivaroxaban for more than 18 h. 65 Taking advantage of that, TEG can provide information when the last dose of anticoagulants is taken, and is thus helpful for selecting the eligible candidates for thrombolysis in AIS patients. In a study of 9 healthy volunteers, NOAC blood concentration ≥50 ng/mL can be reflected by R value with high sensitivity and specificity. 66
TEG is also useful in the post-monitoring of rt-PA therapy. A study demonstrated that some patients had shorter baseline R, Delta, and higher α-angle after 10 min of rt-PA injection, 67 suggesting that these patients could form clots faster and more firmly. Comparing the TEG parameters before and after rt-PA therapy, this study also found that the lysis effect was heterogeneous among patients, suggesting that not all patients can achieve the same therapeutic effect by weight-adjusted rt-PA. It has been shown that over or under optimal dosing of thrombolytic therapy by wrongly estimated body weight was associated with poorer prognosis in AIS patients.68–70 Since the lysis effect could be impacted by many factors, such as the components of clots and other drug use, individualized therapy should be guided by a safer algorithm other than body weight. Although it has failed to find a correlation between TEG and clinical response to tPA, 46 it is still possible that intravenous thrombolysis could be applied based on the TEG algorithm for individual therapy. Hyperdense middle cerebral artery sign (HMCAS) on CT or blooming artifact (BA) on susceptibility-weighted imaging usually represents an RBC-rich composition of thrombi. 71 Accumulating evidence indicated that RBC-rich thrombi were related to a successful recanalization by thrombolysis72,73 and thrombectomy. 74 The parameters of TEG are associated with the different components of the clot. Thus, it is feasible to investigate whether TEG combined with image features might better predict the composition of clots in the future.
Antiplatelet therapy
TEG facilitates clinical decision-making for antiplatelet strategy in AIS patients. Antiplatelet therapy is a fundamental treatment for patients who suffer from atherosclerosis AIS. Aspirin can suppress the conversion of AA to thromboxane A2 by inhibiting COX-1 irreversibly, and clopidogrel inhibits the binding of ADP to P2Y12 receptors. Routine monitoring of platelet function in all ischemic stroke patients is not recommended, but it is necessary for patients with recurrent ischemic events.75,76 Increasing evidence showed that the responses to antiplatelet agents are heterogeneous from non-responders to hyper-responders.77,78 The phenomenon called antiplatelet resistance is associated with the recurrence of ischemic stroke. 79 However, the definition of antiplatelet resistance is under debate and depends on the testing tools used. There is a good correlation and consistency between the gold standard of LTA and TEG in the detection of antiplatelet resistance (r = -0.37, kappa = 0.634),80–82 though TEG is less sensitive. 81 One of the popular mechanisms of clopidogrel resistance is genetic polymorphism cytochrome P450 (CYP). A study including 249 patients who underwent extracranial or intracranial stenting operations was performed to investigate the relationship between genetic polymorphism and TEG parameters. Higher MA and MAADP were observed in patients with the loss-of-function CYP2C19*2 (rs4244285) allele, which may explain why they were predisposed to recurrent ischemic events. Furthermore, polymorphisms of CYP2C18 were associated with the TEG parameters MA and MAADP as well. 83
The role of TEG in guiding antiplatelet therapy in the setting of interventional neurology requires further large-scale investigation. The dual antiplatelet therapy of aspirin and clopidogrel is often used to prevent ischemic events in neuro-endovascular conditions. The solution to solve the various responses to clopidogrel due to genetic polymorphism is either increasing the dose of clopidogrel or turning to prasugrel or ticagrelor. However, both strategies lack support from guidelines due to limited evidence.84,85 In a single-center study, 183 patients from 437 patients with carotid stenting presented antiplatelet resistance to aspirin or clopidogrel as shown by TEG evaluation. These patients were given triple antiplatelet therapy (aspirin, clopidogrel, and cilostazol) or dual antiplatelet therapy (aspirin and clopidogrel). Compared with those in the dual antiplatelet therapy group, the patients in the triple antiplatelet therapy group had a lower rate of AIS or TIA in 30 days. 86 The reference ranges of MAADP and MAAA recommended for dual antiplatelet therapy (aspirin and clopidogrel) were 19.8 - 43.2 mm and 18.9 - 37.7 mm respectively. Furthermore, the change of MA was inconsistent with the alteration of inhibition rate, which should be considered when evaluating drug effect. The calculation of the inhibition rate is more reliable when the MA value is within a reasonable range. 87
In a study evaluating the efficiency of antiplatelet therapy, 202 AIS patients were divided into four groups: aspirin only, clopidogrel only, both aspirin and clopidogrel, and no antiplatelet. In the dual antiplatelet therapy group (mean R = 5.5 min, mean MA = 59.3 mm, mean α-angle = 57.8°), patients tend to have fewer and weaker clots than no antiplatelet therapy (mean R = 4.6 min, mean MA = 64.5 mm, mean angle = 64.5°). This result indicated that the rate and strength of thrombus formation were decreased in the dual therapy group. However, no difference between single antiplatelet and no antiplatelet groups was observed, which could be caused by the limited sample size and heterogeneity among groups. 88
Furthermore, TEG is also applicable in hemorrhage prediction of antiplatelet therapy. The ADP inhibition rate (ADP%) in TEG can independently predict the hemorrhage risk of antiplatelet therapy, at a cut-off point of 82.3% (AUC = 0.665, 95% CI 0.573 to 0.767, P < 0.01). ADP% at 30-82.3% is recommended in AIS patients who received dual antiplatelet therapy. 89
Anti-coagulation therapy
AF is the primary risk of cardiogenic stroke. It is recommended that anticoagulation therapy initiates according to the course and severity of cerebral infarction in patients with AF. 90 Warfarin is the most commonly used anticoagulant in the treatment of AF. Patients have to check the international normalized ratio (INR) routinely. NOAC includes factor Xa inhibitors and direct thrombin inhibitors, which are relatively safer and more effective than warfarin and do not need regular monitoring of INR. However, when treating bleeding events induced by NOAC, measuring the function of factor Xa is essential. There are several methods to measure the effect of factor Xa inhibition, and each has some inevitable defects. Taking CCTs for example, PT and APTT are inconsistent in evaluating the function of factor Xa, as the results vary with different assay reagents used. 91 Other tests, such as dilute Russell's viper venom time and factor Xa chromogenic assays, are lack specificity and hard to be used widely. 91 Similarly, interassay variability exists in different detection methods of low molecular weight heparins (LMWH). 92 A strong relationship between coagulation index and anti-Xa level has been investigated in patients who received LMWH after orthopedic surgery. 5 The R parameter rivals the gold standard test Hemoclot in evaluating the effect of dabigatran, and is tightly associated with the concentration of dabigatran. 93
However, whether TEG is suitable for evaluating the warfarin effect is under debate. The activator Kaolin used in TEG can trigger intrinsic coagulation, which mitigates the results of the extrinsic system induced by warfarin. 94 Likewise, R tended to be in the normal range at 12 h after subcutaneous injection of enoxaparin in surgical patients, 5 which limits its potential application in the evaluation of anticoagulation therapy. Moreover, platelet hypercoagulability could be identified by Delta and G parameters in TEG. A high G but a normal Delta usually indicates the hypercoagulation contributed by platelets. 24 Further research is warranted to provide more definitive evidence of TEG in the guidance of anticoagulation therapy.
Prognosis Prediction
Whether or not TEG can predict the prognosis of AIS is still under debate. Many studies demonstrated the predictive capacity of the MA parameter in the prognosis of cardiovascular disease, but it is less studied in AIS. A study in China concluded that the MA level could predict the one-year neurological function prognosis in AIS patients. 95 A baseline MA above 69mm before coronary artery bypass grafting surgery was closely related to the occurrence of AIS in 30 days. 96 On the other hand, in a study of 171 AIS patients treated with rt-PA, there was no association between TEG parameters and clinical response to thrombolysis therapy. 46 Large randomized controlled trials (RCTs) are warranted to determine the application of TEG in the prognosis prediction of AIS.
Conclusion
In general, as a rapid and global detection method of coagulate function, TEG is a portable tool in the individual management of AIS patients. However, TEG has not gotten deserved attention in ischemic stroke. Compared with the fruitful research on TEG in cardiovascular disease, there is a paucity of large RCTs to investigate the application of TEG in AIS. As a global hemostasis test, the TEG parameters in the setting of AIS seem to be unspecific. However, under certain circumstances, the trends reflected by TEG, namely hypercoagulation or hypocoagulation, could denote some specific changes in disease progression, and thus could provide complementary information for clinicians. Several questions regarding the potential role of TEG in thrombolysis, hemorrhage transformation, prognosis prediction, and endovascular therapy in AIS remain to be elucidated. To conclude, TEG has potential advantages in the management of AIS, and further research is warranted to investigate the role of TEG in AIS.
Footnotes
Acknowledgements
None
Author Contributions
Zhi Geng and Xiuzhe Wang generated the research idea and guided the work. Feng Chen drafted the manuscript. Liren Zhang and Xiaolin Bai performed the literature search and figure edition. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reviews.
Funding
This study was supported by the research grant from National Facility for Translational Medicine (Shanghai) (Grant No. TMSK-2020-122) in the decision to submit the article for publication.
Informed Consent
No patient information was involved in this study. Informed consent for patient information to be published in this article was not required.
