Abstract
Background
Cardiac arrest (CA) can activate the coagulation system. Some coagulation-related indicators are associated with clinical outcomes. Early evaluation of patients with cardiac arrest-associated coagulopathy (CAAC) not only predicts clinical outcomes, but also allows for timely clinical intervention to prevent disseminated intravascular coagulation.
Objective
To assess whether CAAC predicts 30-day cumulative mortality.
Methods
From the Medical Information Mart for Intensive Care IV (MIMIC-IV) database, we conducted a retrospective cohort study from 2008 to 2019. Based on international normalized ratio (INR) value and platelet count, we diagnosed CAAC cases and made the following stratification of severity: mild CAAC was defined as 1.4 > INR≧1.2 and 100,000/µL < platelet count≦150,000/µL; moderate CAAC was defined with either 1.6 > INR≧1.4 or 80,000/µL < platelet count≦100,000/µL; severe CAAC was defined as an INR≧1.6 and platelet count≦80,000/µL.
Results
A total of 1485 patients were included. Crude survival analysis showed that patients with CAAC had higher mortality risk than those without CAAC (33.0% vs 52.0%, P < 0.001). Unadjusted survival analysis showed an incremental increase in the risk of mortality as the severity of CAAC increased. After adjusting confounders (prehospital characteristics and hospitalization characteristics), CAAC was independently associated with 30-day mortality (hazard rate [HR] 1.77, 95% confidence interval [CI] 1.41–2.25; P < 0.001); moderate CAAC (HR 1.48, 95% CI 1.09–2.10; P = 0.027) and severe CAAC (HR 2.22, 95% CI 1.64–2.97; P < 0.001) were independently associated with 30-day mortality.
Conclusion
The presence of CAAC identifies a group of CA at higher risk for mortality, and there is an incremental increase in risk of mortality as the severity of CAAC increases. However, the results of this study should be further verified by multicenter study.
Introduction
Cardiac arrest (CA) poses a serious threat to human life with its extremely high mortality rate in the United States; the survival rate to hospital discharge is still less than 10% for out-of-hospital cardiac arrest (OHCA) and less than 20% for in-hospital cardiac arrest (IHCA). 1 Despite all our efforts to reach return of spontaneous circulation (ROSC) for CA, post-CA care remains confusing. Although some indicators associated with patients receiving cardiopulmonary resuscitation (CPR) are good predictors of outcome, 2 we are still unable to accurately predict the clinical outcome of patients after ROSC.
From pathophysiology, we can make predictions about the clinical outcome of patients from the manifestations associated with the post-CA syndrome (PCAS). PCAS was first described as a type of sepsis-like manifestation, such as coagulation disorder and systemic inflammatory response syndrome.3, 4 Dysfunction of the coagulation-fibrinolysis system plays a key role for post-CA patients. 5 Severe hypoxia and acidosis during CA and resuscitation may cause damage to vascular endothelial cells, thereby stimulating the release of tissue factors and initiating the extrinsic coagulation pathway. 6 This process seems to be very similar to the coagulation disorders caused by sepsis. In both CA and sepsis, the over-activated systemic inflammatory response causes abnormalities in coagulation.4, 7, 8 Previous studies have shown that coagulation and fibrinolysis-related indicators, including activated partial thromboplastin time, fibrinogen degradation products, and D-dimer, are closely related to the prognosis of post-CA patients and are expected to be promising biomarkers with prognostic significance.9, 10
Sepsis and trauma have long been recognized as major conditions that can occur with coagulation disorder and can lead to the progression of disseminated intravascular coagulation (DIC). Post-CA patients have the same pathophysiologic condition which could also evoke DIC. 11 Thus, the post-CA condition should be recognized as having a high likelihood of evoking DIC. 6 A previous study has shown that International Society of Thrombosis and Hemostasis DIC score is a good predictor of clinical outcome for OHCA. 12 The DIC score consists of five indicators, however, some of which are difficult to obtain quickly and accurately due to a variety of factors, making the DIC score too complex to use in clinical practice. A recent study used a simplified scoring system (including only platelet count and international normalized ratio, INR) to classify patients with sepsis-associated coagulopathy, and showed that the scoring system was a good predictor of hospital mortality. 13 Above all, after ROSC, any type of coagulopathy seems to predict a poor clinical outcome. This may be related to its further development into DIC. Therefore, we sought to determine whether this simplified scoring system (before developed into DIC) could predict clinical outcomes in patients after CA.
Materials and Methods
Study Design
A retrospective cohort study was conducted using data from Medical Information Mart for Intensive Care IV (MIMIC-IV), a large electronic database. The detail in the MIMIC-IV database is contained in four parts: Emergency Department, Admissions, Intensive Care Unit, and Follow-up. All data was extracted from the latest versions of MIMIC-IV (version 2.2) and MIMIC-IV-ED (version 2.2), which were released in January 2023. The databases contain more than 400,000 patients admitted to ICU at the single center of Beth Israel Deaconess Medical Center in the United States from 2008 to 2019. For the period from 2011 to 2019, MIMIC-IV-ED includes a free-text field which contains the patient's reported reason for presenting to the emergency department. 14 Importantly, MIMIC-IV is deidentified. Patient identifiers had been removed according to the Health Insurance Portability and Accountability Act Safe Harbor provision, and a reprogrammed system number is used to identify and access each patient's records from the database. The system includes subject_id for every patient, hadm_id for every hospital admission, and stay_id for every ICU admission; for example, a patient who has been hospitalized three times and admitted to the ICU once will have one subject_id, three ham_ids, and one stay_id. Subject_id links the information between MIMIC-IV and MIMIC-IV-ED. Before accessing this database, the CITI Data or Specimens Only Research certification was needed (our certification can be verified at http://www.citiprogram.org/verify/?k026a8801-1124-40bb-ac72-c64d66bdab42-36003834; we also needed to agree to the PhysioNet Credentialed Health Data Use Agreement 1.5.0, which contains a related ethical statement (https://www.physionet.org/content/mimiciv/view-dua/2.2/).15–17 Thus, this study did not require additional ethical approval.
Inclusion and Exclusion Criteria
This study has the following inclusion criteria: (a) all patients diagnosed with CA (icd_code9 is 4275 and icd_code10 includes I46, I462, I468 and I469. Other icd_codes of CA are associated with newborn, operation or pregnancy, so CA are associated with newborn, operation or pregnancy is excluded.); (b) age more than 18 years; (c) any initial rhythm; (d) OHCA and IHCA.
Exclusion criteria: (a) pregnancy; (b) known coagulopathy; (c) known severe liver dysfunction; (d) use of anticoagulants or/and antiplatelet drugs; (e) any malignant cancer; (f) incomplete information.
Variable Extraction
We searched the database for all patients who had CA recorded. From the Emergency Department part of the database, the vital signs, arterial blood gas, INR and platelet count at admission were extracted, along with the patient's reported reason for presenting to the emergency department from the MIMIC-IV-ED free-text field. All laboratory test results were the first results after hospital admission. From the Admissions part of the database, the age, sex, and comorbidities were extracted, as well as treatments during the hospital stay, which include coronary angiography, percutaneous coronary intervention, mechanical circulation support (extracorporeal membrane oxygenation [ECMO], impella and intra-aortic balloon pump [IABP]) and therapeutic hypothermia. The ICU length of stay (LOS) and hospital LOS were also extracted from the Admissions part. Extracted from the Intensive Care Unit part were use of the ventilator during hospitalization, medications during hospitalization, and the Simplified Acute Physiology Score II (SAPS II includes only 17 variables: 12 physiology variables, age, type of admission (scheduled surgical, unscheduled surgical, or medical), and three underlying disease variables (acquired immunodeficiency syndrome, metastatic cancer, and hematologic malignancy 14 ). Moreover, we also extracted the infusion dose of dopamine, dobutamine, epinephrine, norepinephrine, vasopressin and milrinone to calculate the maximum value of vasoactive-inotropic score. 18 Extracted from the Follow-up part were the cumulative survival days. The above variables were extracted by icd_code (version 9 or 10), subject_id, hadm_id, and stay_id, which created a unique data point number for every variable and patient in the MIMIC-IV and MIMIC-IV-ED databases.
All the variables and inclusion and exclusion criteria were extracted and determined by postgreSQL 13.0 code. The detailed code is shown in the supplementary file.
Definitions
The endpoint was defined as cumulative mortality during 30 days.
Based on INR and platelet counts in a previous study, 13 we extracted INR and platelet counts recorded for the first time after the occurrence of CA and classified cardiac arrest-associated coagulopathy (CAAC) as follows: mild CAAC was defined as INR greater than or equal to 1.2 and less than 1.4, plus platelet count less than or equal to 150,000/µL but greater than 100,000/µL; moderate CAAC was defined as either an INR greater than or equal to 1.4 but less than 1.6, or platelets less than or equal to 100,000/µL but greater than 80,000/µL; severe CAAC was defined as an INR greater than or equal to 1.6, and platelets less than or equal to 80,000/µL. Patients with an INR greater than 1.2 but no recorded platelet count, as well as those with platelet counts less than 150,000/µL but no recorded INR, were considered to have indeterminate CAAC and were treated as not having CAAC in the analysis. Whenever multiple values were available, the most abnormal values were selected for determining CAAC severity. A cross-tabular was presented to intuitively identify the CAAC severity in Supplementary Table 1.
Using the ICD-9/10 diagnosis codes for CA were all diagnoses used. In the MIMIC-IV-ED database, every patient was recorded, as a free-text field, the cause of receiving care in emergency department. Thus, we identified that the patients receiving care in emergency department for CA were OHCA. For IHCA or OHCA patients, the initial recording time starts from the time of reaching ROSC after CA. In this database, the patients who were discharged from the hospital recorded the exact time of their death within 4 years through the health insurance system.
Statistical Analysis
Continuous variable values are presented as the median (interquartile range, IQR) for skewed distributions and the mean (standard deviation, SD) for normal distributions. Total number and percentages are presented for categorical variables. Analysis of variance was used to compare normally distributed continuous variables, and the Mann-Whitney U test was used for nonnormally distributed continuous variables. The chi-square or McNemar tests were used to compare categorical variables.
The Kaplan-Meier survival curve was fit to estimate the 30-day cumulative mortality difference among different CAAC severity categories. Adjusted proportional hazards model (COX) regression models were then fit for each outcome to control for prehospital characteristics (age, sex, body mass index, all included comorbidities, OHCA, bystander CPR, time from collapse to CPR, time from collapse to ROSC, and initial rhythm shockable) and hospitalization variables (vital signs, arterial blood gas, hypothermia, coronary arteriography/percutaneous coronary intervention, invasive ventilator, ECMO, Impella, IABP, all included medications and SAPS II score). Moreover, area under curve, sensitivity, and specificity are used to assess the predictive ability of each model for 30-day cumulative survival by receiver operating characteristic curve (Supplementary Table 3).
All tests of significance used a two-sided P-value of less than 0.05. Statistical analyses were completed using IBM SPSS statistics Version 25 (Peking University, China) and R Project Version 4.2.3 (Peking University, China).
Results
From 2034 initially identified patients, the final cohort included 1485 consecutive patients (median age 67 [54–78] years; 60.0% men) with severity of illness (median SAPS score 46 [36–59]). The final cohort consisted of 233 patients (15.7%) who did not have CAAC based on preset criteria, 432 (29.1%) with mild CAAC, 219 (14.7%) with moderate CAAC, and 601 (40.5%) with severe CAAC (Figure 1).
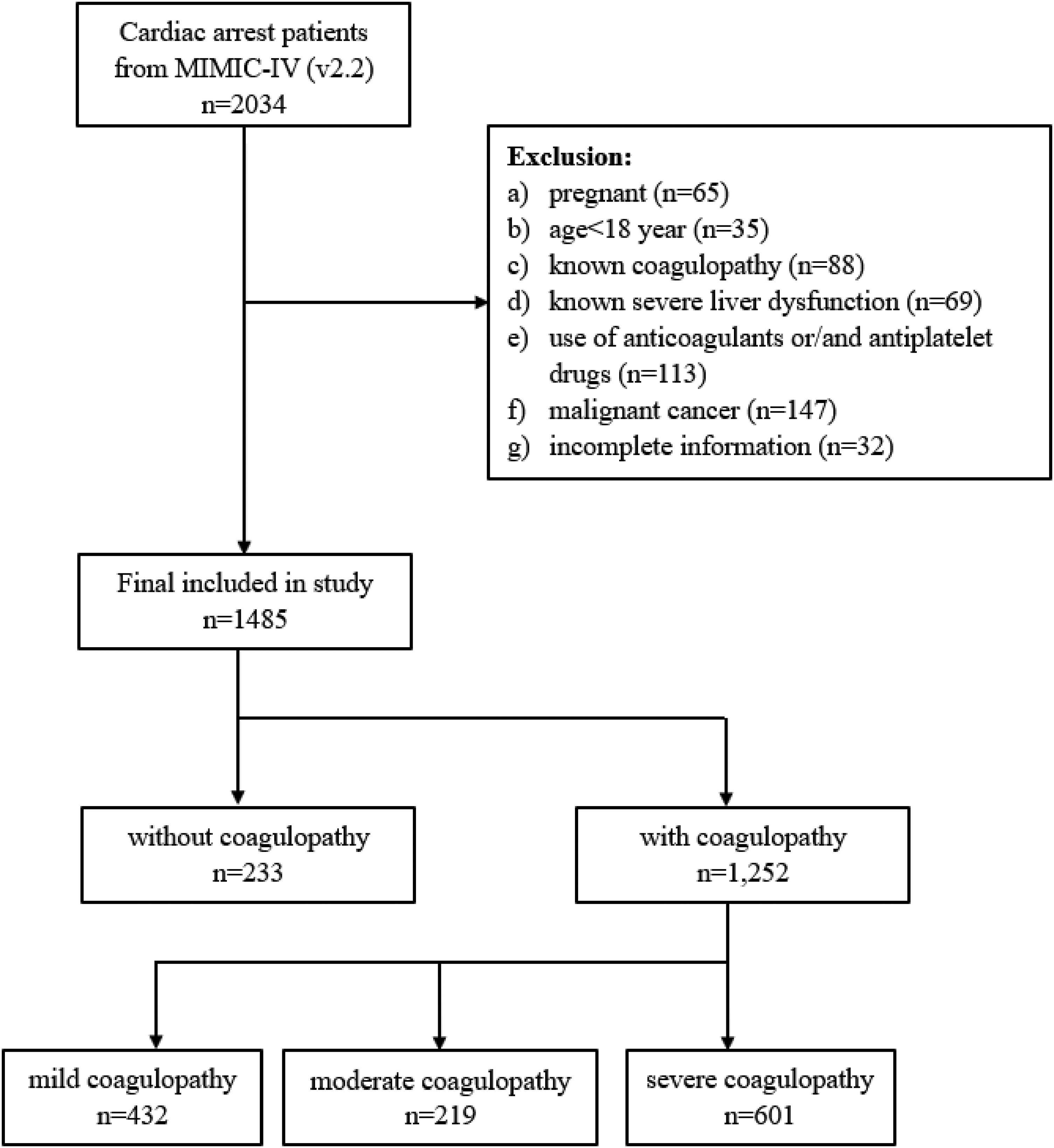
Flow chart. CAAC: cardiac arrest-associated coagulopathy.
The patients’ baseline characteristics showed that patients with CAAC were older and more often men. The rates of previous myocardial infarction, heart failure, and renal dysfunction were higher in patients with CAAC. In the severe CAAC group were more severely because they received more invasive ventilator and pressor agents and have higher SAPS II scores and poorer vital signs (Table 1).
Patients Baseline Characteristics and Clinical Outcomes.
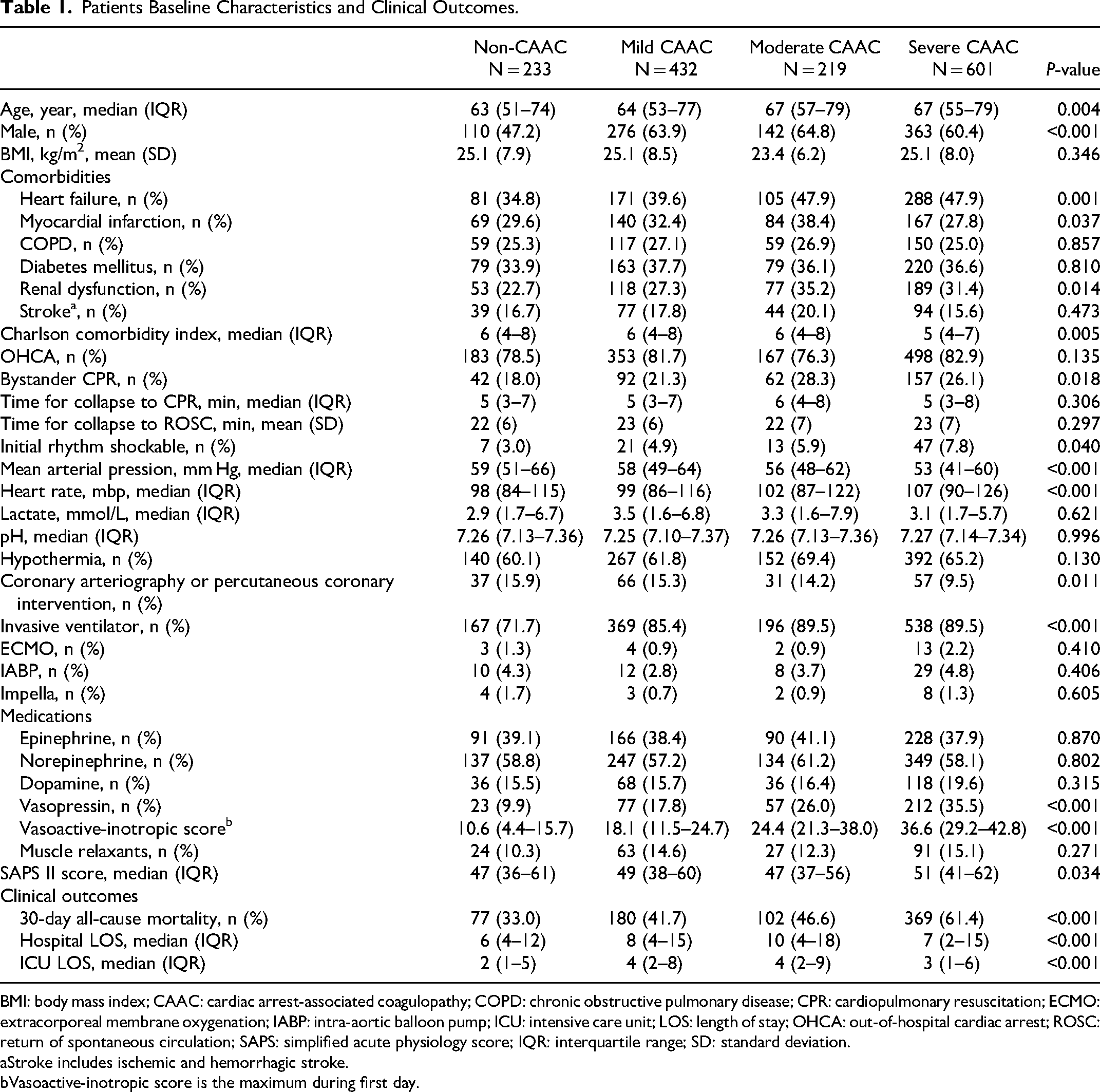
BMI: body mass index; CAAC: cardiac arrest-associated coagulopathy; COPD: chronic obstructive pulmonary disease; CPR: cardiopulmonary resuscitation; ECMO: extracorporeal membrane oxygenation; IABP: intra-aortic balloon pump; ICU: intensive care unit; LOS: length of stay; OHCA: out-of-hospital cardiac arrest; ROSC: return of spontaneous circulation; SAPS: simplified acute physiology score; IQR: interquartile range; SD: standard deviation.
Stroke includes ischemic and hemorrhagic stroke.
Vasoactive-inotropic score is the maximum during first day.
The Kaplan-Meier risk analyses showed significant differences in 30-day cumulative mortality between the without-CAAC and with-CAAC groups (P < 0.001) (Figure 2), and, in general, among the four groups (Figure 3). While the patients with any CAAC severity had higher 30-day cumulative mortality than those without CAAC, the results of two-by-two comparisons showed that there was no significant difference between the mild CAAC and moderate CAAC group (Figure 3).
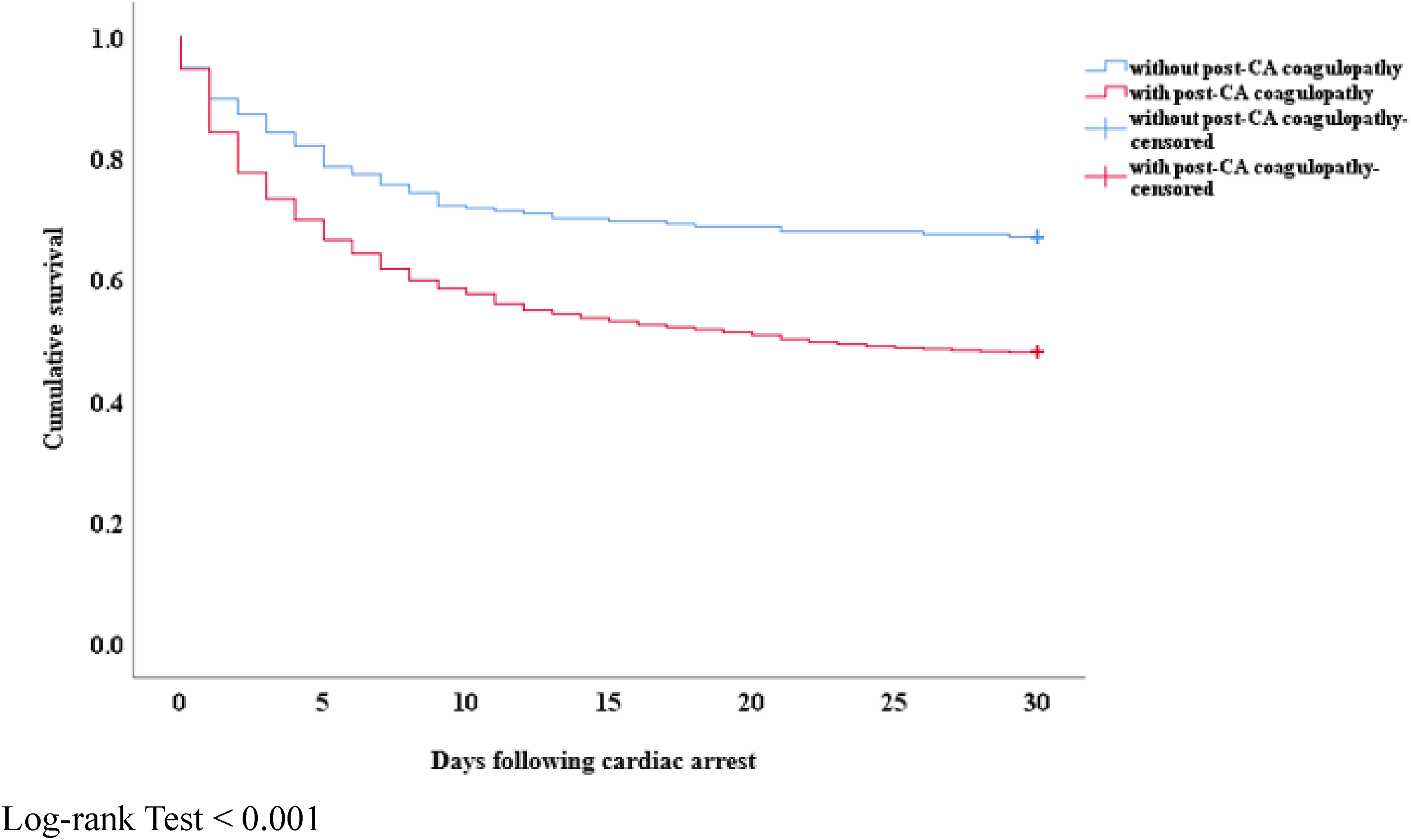
Kaplan-Meier curves for cumulative survival percentages according to the presence or absence of cardiac arrest-associated coagulopathy. Log-rank Test < 0.001.
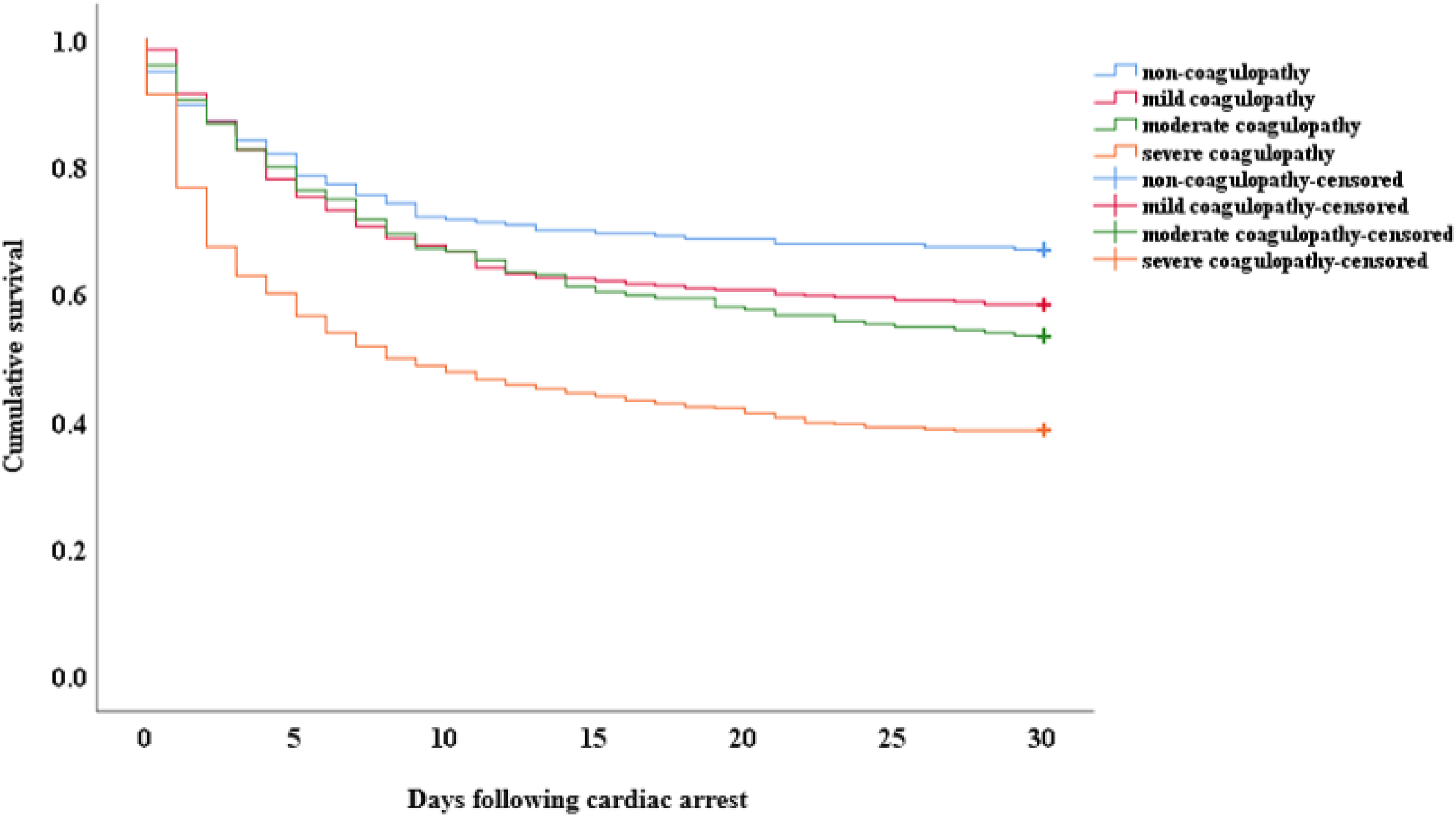
Kaplan-Meier curves for cumulative survival percentages according to cardiac arrest-associated coagulopathy severity. Statistical comparisons by log-rank test: overall comparison, P < 0.001; without coagulopathy versus mild coagulopathy, P = 0.050; without coagulopathy versus moderate coagulopathy, P = 0.009; without coagulopathy versus severe coagulopathy, P < 0.001; mild coagulopathy versus moderate coagulopathy, P = 0.336; mild coagulopathy versus severe coagulopathy, P < 0.001; moderate coagulopathy versus severe coagulopathy, P < 0.001.
Adjusted Cox regression models showed that CAAC was independently associated with 30-day cumulative mortality regardless of adjustments made for prehospital characteristics and hospitalization characteristics, with the hazard rate (HR) varying from 1.44 to 1.77. Both moderate and severe CAAC were independently associated with 30-day mortality regardless of adjustments made for prehospital characteristics and hospitalization characteristics, with HR varying from 1.46 to 1.52 for moderate CAAC and from 2.22 to 2.43 for severe CAAC. (Table 2 and Supplementary Table 2). However, linear regression did not show that CAAC severity was associated with hospital LOS (P = 0.385) and ICU LOS (P = 0.127) after adjustment for prehospital characteristics, but it might be associated with the early death in the CAAC groups (Figure 3).
Multivariable Analyses.
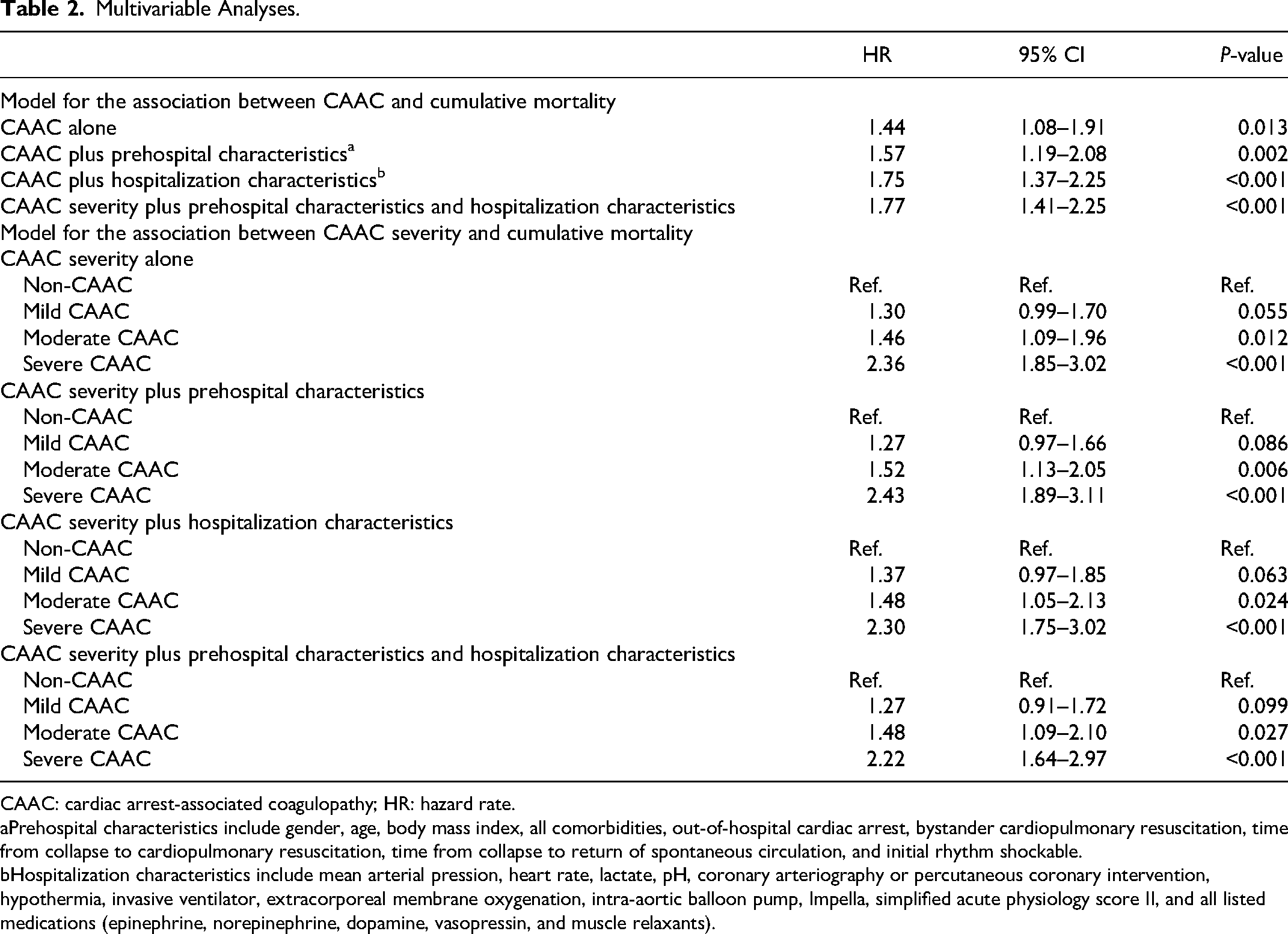
CAAC: cardiac arrest-associated coagulopathy; HR: hazard rate.
Prehospital characteristics include gender, age, body mass index, all comorbidities, out-of-hospital cardiac arrest, bystander cardiopulmonary resuscitation, time from collapse to cardiopulmonary resuscitation, time from collapse to return of spontaneous circulation, and initial rhythm shockable.
Hospitalization characteristics include mean arterial pression, heart rate, lactate, pH, coronary arteriography or percutaneous coronary intervention, hypothermia, invasive ventilator, extracorporeal membrane oxygenation, intra-aortic balloon pump, Impella, simplified acute physiology score II, and all listed medications (epinephrine, norepinephrine, dopamine, vasopressin, and muscle relaxants).
An additional sensitivity analysis of the 270 patients with pre-existing coagulopathy (known coagulopathy, known severe liver dysfunction or long-term use of anticoagulants) who were excluded from the primary analysis also demonstrated similar relationships (after adjusting all confounders, CAAC overall: HR = 1.30, [95% CI, 1.06–1.54]; P = 0.025 and severe CAAC: HR = 1.56, [95% CI, 1.12–2.01]; P < 0.001). Moreover, in order to exclude potential using anticoagulation for patients received mechanical circulation support devices, we excluded 98 patients and conducted a sensitivity analysis (after adjusting all confounders, CAAC overall: HR = 1.75, [95% CI, 1.33–2.18]; P < 0.001 and severe CAAC: HR = 2.15, [95% CI, 1.57–2.90]; P < 0.001)
Discussion
From this study, we propose a rapid and early evaluation system to assess the presence of combined CAAC and the severity of CAAC by platelet count and INR. This early assessment can be obtained before the patient develops DIC. Based on this study, this evaluation system of CAAC is a good predictor of clinical outcomes in CA patients. Moreover, as the severity of CAAC increases, it has a strong correlation with poor clinical outcomes. Therefore, this assessment system can be used not only to predict the clinical outcome of patients with CA, but more importantly, to provide evidence to support timely clinical interventions to improve the prognosis of patients with potentially adverse clinical outcomes.
Previous studies have demonstrated the relationship between coagulation changes after CA and prognosis, as well as attempts to use various indicators and models to predict the clinical outcomes,19–22 but there is still a lack of indicators and models that are accepted by most clinicians. Regardless of the coagulation indices used to assess the prognosis of CA patients, however, we cannot avoid the relevant studies of sepsis because sepsis and PCAS have very similar pathophysiological characteristics.4,23 Patients experience extreme ischemia and hypoxia during CA, and when ROSC is reached, they must face ischemia-reperfusion injury. 24 Current studies indicate that aspects such as inflammatory cytokine release, 25 platelet activation and depletion, 26 tissue factor and massive thrombin generation, 27 neutrophil activation and endothelial injury, 28 impairment of anticoagulant pathways, 22 and deterioration of fibrinolytic system 29 play key roles in post-CA coagulopathy. The presence of these factors suggests that post-CA coagulopathy and sepsis-related coagulopathy can both lead to the progression of DIC. 6 Thus, it makes sense for us to leverage our experience in sepsis.
The DIC score is indeed one of the more reliable tools used to assess the prognosis of CA. 12 However, some DIC indicators are subject to external influences and the limitations of testing conditions in different medical institutions, and their consistency is poor. The fibrinolytic product represented by D-dimer is severely limited by assay conditions and methods: a previous study has pointed to an 18–25% variation for D-dimer testing by different methods. 30 This makes the extrapolation of indicators containing fibrinolytic products inadequate. In contrast, the two indicators (INR and platelet count) selected for this study to assess CAAC have shown excellent consistency across healthcare institutions. INR is calculated according to the prothrombin time and the international sensitivity index of the assay reagent. In the majority of healthcare institutions, platelet count currently employs a combination of impedance technology and manual counting, which greatly increases its accuracy and consistency. These two indicators are far superior to other coagulation indicators, both economically and in timeliness. 31 Although the diagnostic and stratification criteria for CAAC in this study were derived from a sepsis-related study, 13 both INR and platelet count have independently shown correlations with CA prognosis in previous studies., 32
Both INR and platelet count can reflect the severity of the patient's condition after CA and might have the following causes: (a) Typically, INR levels are associated with coagulation levels. Blood is hypercoagulable after ROSC due to coagulation activation mediated by massive tissue factor release resulting from ischemia-reperfusion injury. Thus, massive consumption of coagulation factors leads to elevated INR. Previous studies have shown that the degree of depletion of coagulation factors is proportional to the degree of ischemia-reperfusion injury. 33
(b) Platelets are directly involved in the formation of thrombus. Assuming that the patient's platelet count was within the normal range prior to the onset of CA, the post-ROSC decrease in platelet count predicts the formation of massive micro-thrombosis following ischemia-reperfusion. Moreover, inflammatory cytokines released by platelets in thrombus can activate inflammatory responses in surrounding tissues and cause secondary damage. 34
Based on the above evidence, we hypothesized that the severity of CAAC is related to the severity of illness in CA. This was confirmed in our study, where patients with severe CAAC had the highest SAPS II scores and the worst 30-day cumulative survival outcomes. However, when using INR and platelet counts to predict clinical outcomes in CA patients, in addition to the need to exclude pre-existing coagulation abnormalities, attention still needs to be paid to the potential effects on coagulation that may occur in certain disease states. Especially in acute inflammation states, the main mechanisms of the coagulation derangement during systemic inflammatory activity are tissue factor-mediated thrombin generation and an imbalance or dysfunction of the normal physiologic anticoagulant mechanisms, such as the antithrombin system and the protein C system. 35 Moreover, in the setting of inflammation-induced activation of coagulation, platelets can be activated directly by endotoxin or by proinflammatory mediators, such as platelet activating factor. 36 Thus, we need to be cautious about using INR and platelet counts to diagnose CAAC when the patient is in an acute inflammatory state such as severe infection or active autoimmune disease.
This study has several important limitations. First, all data are from a single center and may not be representative of other institutions or regions. Second, this research was a retrospective cohort study, so selection bias was inevitable, and it was difficult to ensure that all variables were evenly distributed across groups. Third, the study included patients from 2008 to 2019, and such a long span of time may have affected patient outcomes due to updates in treatment and diagnosis. Third, the difference of prehospital characteristics baseline might have underlying impact for the results. Last, the diagnostic and stratification criteria for CAAC in this study were derived from a sepsis-related study, and although our study showed satisfactory results, we cannot be sure whether the cut-off values for INR and platelet count are truly reasonable for CA. Moreover, because some of the information can only be obtained through ICD code, its accuracy may be insufficient. Above all, our results should be confirmed by further multicenter studies in the future.
Conclusion
Patients with CAAC, as defined by elevated INR and platelet count, appear to have a greater risk of death compared with those without coagulopathy. Our results also suggest that the mortality risk with CAAC can be quantified according to the severity of the coagulopathy. The results of this study should be interpreted in the context of its retrospective design and study population.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231221986 - Supplemental material for Cardiac Arrest-Associated Coagulopathy Could Predict 30-day Mortality: A Retrospective Study from Medical Information Mart for Intensive Care IV Database
Supplemental material, sj-docx-1-cat-10.1177_10760296231221986 for Cardiac Arrest-Associated Coagulopathy Could Predict 30-day Mortality: A Retrospective Study from Medical Information Mart for Intensive Care IV Database by Jingwei Duan, Hongxia Ge, Wenyang Fan, Lanfang Du, Hua Zhang, Ayijiang Jiamaliding, Baomin Duan and Qingbian Ma in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgement
We thank Qinhan Yao for statistical support during the revision stage.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Clinical Cohort Construction Program of Peking University Third Hospital, (grant number BYSYDL2021008).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
