Abstract
This study investigated the efficacy and safety of pharmaco-invasive strategy with half-dose recombinant human prourokinase (PHDP) during hospitalization for patients with ST-segment elevation myocardial infarction (STEMI) to provide references for the treatment of STEMI. Patients with STEMI who fulfilled the inclusion and exclusion criteria and attended Chengde Central Hospital, Hebei Province, China, between September 3, 2019, and December 28, 2021, were included in this study. The experimental group received PHDP and the control group underwent primary percutaneous coronary intervention (PPCI). This study enrolled 150 patients with STEMI, 75 in the experimental group and 75 in the control group. Coronary angiography revealed successful thrombolysis in 64 (85.33%) patients. Compared with the control group, the experimental group had shorter first medical contact-reperfusion time (P < 0.001), less slow flow/no-reflow (P < 0.001), and a lower utilization rate of Tirofiban (P < 0.001). Validity endpoints: no statistically significant differences between the two groups. Safety endpoints: no statistically significant differences between bleeding and major adverse cardiovascular and cerebrovascular events (MACCEs), but the experimental group was more prone to arrhythmias (P = 0.040), particularly premature ventricular beats (PVB) (P = 0.008). In conclusion, the efficacy and safety of PHDP in the treatment of patients with STEMI were positive. Complete epicardial and myocardial reperfusion rates, risk for bleeding during hospitalization, and incidence of MACCEs were similar to those of the PPCI strategy. Although the PHDP group has a higher incidence of PVB, it does not increase the incidence of malignant arrhythmia. This study aimed to provide a new therapeutic strategy for the treatment of STEMI in hospitals without adequate PPCI resources condition.
Keywords
Introduction
Ischemic heart disease (IHD) is among the leading contributors to global disease burden among individuals > 50 years of age. 1 According to data from the World Health Organization in 2008, > 2 million individuals in China and India died from IHD. 2 As one of the most dangerous types of IHD, the diagnosis and treatment of ST-segment elevation myocardial infarction (STEMI) have always been the important to medical practitioners. According to United States and European STEMI management guidelines, the preferred treatment for STEMI is primary percutaneous coronary intervention (PPCI).3,4 However, based on the current medical situation in China, the ambulance use rate and effective reperfusion rate have gradually decreased from provincial to county hospitals, and most patients treated in county and sub-county hospitals cannot undergo percutaneous coronary intervention (PCI) within 120 min after electrocardiographic diagnosis of STEMI, as prescribed in the guidelines. 5 Early access to reperfusion therapy is the basis for improved patient survival and prognosis. If patients are unable to undergo PPCI within the treatment time window, guidelines recommend that they can receive pharmaco-invasive (PhI) strategy as early as possible. A PhI strategy is used for patients requiring prompt thrombolytic therapy after the first medical contact (FMC), followed by transfer to hospital for PCI, where available. Patients will undergo remedial PCI if thrombolysis fails, or coronary angiography or PCI if thrombolysis succeeds within 3 to 24 h. It can achieve timely reperfusion.
However, there are concerns regarding the efficacy and safety of thrombolysis in PhI treatment. A meta-analysis reported that PhI and PPCI have similar efficacies. 6 In terms of prognosis, PhI reduces the risk for in-hospital heart failure, but increases the risk for in-hospital stroke and bleeding. With the advent and application of new thrombolytic drugs, the efficacy and safety of pharmacological thrombolysis has gradually improved. A meta-analysis investigating the efficacy of thrombolysis using recombinant human prourokinase, a third-generation thrombolytic agent, reported that it could reduce the incidence of major adverse cardiovascular and cerebrovascular events (MACCEs), and did not increase the risk for bleeding events compared with other thrombolytic agents. 7 In addition, when the thrombolytic dose is adjusted to one-half the dose, it has been proposed that the half-dose PhI strategy is comparable to PPCI in terms of bleeding risk. 8 Other studies have confirmed the effectiveness and safety of the half-dose PhI strategy.9,10 Based on a summary of previous experience, the present study investigated the efficacy and safety of a PhI strategy using half-dose recombinant human prourokinase (PHDP) in the treatment of patients with STEMI, either before admission to hospital or in the emergency room, to provide a new reference for the treatment of STEMI.
Materials and Methods
The present investigation was a single-center, prospective, non-inferiority, and randomized controlled clinical trial. Patients with STEMI who fulfilled the inclusion and exclusion criteria and attended Chengde Central Hospital between September 3, 2019, and December 28, 2021, were included in this study. Subjects were grouped in a 1:1 ratio using the randomized numerical table method: the experimental group received PHDP; and the control group underwent PPCI. Information used in this study was obtained from hospital medical records. This study was approved by the Ethics Committee of Chengde Central Hospital and all subjects provided informed consent.
The present study included patients 18–80 years of age who fulfilled the diagnostic criteria for STEMI established by the European Society of Cardiology and experienced onset of STEMI within 12 h and had a life expectancy > 12 months. 3 In addition, patients agreed to undergo reperfusion therapy. Patients who experienced cardiac rupture, cardiogenic shock, thrombocytopenia, severe liver dysfunction, or decreased renal function (serum creatinine level > 221 mmol/L) were excluded. Additionally, patients with contraindications to iodine contrast agents, thrombolytic drugs, and antiplatelet or anticoagulation drugs were also excluded.
All patients required antiplatelet therapy before reperfusion therapy. If they had not taken antiplatelet agents within 12 h before admission, they were administered 300 mg aspirin, 300 mg clopidogrel, or 180 mg ticagrelor, followed by a regular oral dose (100 mg aspirin and 75 mg clopidogrel [once daily], or 90 mg ticagrelor [twice daily]). Before pharmacological thrombolysis, subjects in the experimental group received an intravenous loading dose of heparin (60 U/kg), followed by heparin (12 U/kg/h) for at least 48 h. They then received low molecular weight heparin (5000 U), administered subcutaneously every 12 h for 5–7 days. During pharmacological thrombolysis, each patient received 25 mg of recombinant human prourokinase (Shanghai Tasly Pharmaceutical Co.Ltd; 5 mg/piece, product no: S20110003). First, 10 mg of recombinant human prourokinase was administered intravenously over a 3 min period, and the remainder was administered via intravenous drip over a subsequent 30 min period. If thrombolysis was successful, patients were scheduled to undergo coronary angiography within 3–24 h and PCI would be performed for vessels with residual stenosis ≥ 75% confirmed by angiography. In contrast, if thrombolysis failed, the patient underwent remedial PCI. Subjects in the control group underwent PPCI within 2 h of diagnosis.
The primary endpoint of treatment effectiveness was complete epicardial and myocardial reperfusion after PCI, which fulfilled the following three criteria: post-PCI thrombolysis in myocardial infarction (TIMI) myocardial perfusion (post-PCI TMPG) grade 3; post-PCI TIMI frame count (post-PCI TFG) of grade 3; and ST-segment regression ≥ 70% within 60 min after PCI. 10 The secondary endpoint was defined according to any of the above criteria.
The examined safety endpoints included bleeding events, arrhythmias, and MACCEs. Bleeding events included major bleeding (clinically visible bleeding with a fall in hemoglobin level ≥ 5 g/dL) and minor bleeding (clinically visible bleeding with a fall in hemoglobin level < 5 g/dL). Arrhythmias included premature atrial beats (PAB), atrial tachycardia (AT), atrial fibrillation (AF), premature ventricular beat (PVB), ventricular tachycardia (VT), ventricular fibrillation (VF), and atrioventricular block (AVB), AVB II, and AVB Ⅲ. MACCEs include all-cause death, cardiac death, cardiac arrest, re-intervention, heart failure, recurrent angina pectoris, and stroke.
Sample size and power calculations were performed using PASS version 11 (NCSS Statistical Software). According to the results of the EARLY-MYO Trial, 10 the primary efficacy endpoint (complete epicardial and myocardial reperfusion after angioplasty) occurred in 34.2% of the PhI group and 22.8% of the PPCI group. Under this premise, we conducted a non-inferiority test with a pre-specified non-inferiority margin of 0.7, (1-β) and significance level α (one-sided) values of 80% and 0.025, respectively. Each group needed to include 68 STEMI patients. Assuming a 5% dropout rate during the study, each group would require at least 71 patients.
Statistical analysis was performed using SPSS version 26.0 (IBM Corporation, Armonk, NY, USA). Drawing graphs were performed using Prism version 8 (GraphPad Software, Inc. San Diego, CA). Data were tested for normality; Continuous variables that exhibited a normal distribution are expressed as
Results
This study included 150 patients who experienced STEMI (Figure 1). The mean (± SD) age of the subjects was 57.67 ± 10.89 years and 123 (85.33%) were male. The experimental group included 75 patients, with a mean age of 57.45 ± 9.69 years (66 male [88.00%]) cases. Based on clinical experience, there were 66 (88.00%) successful cases of thrombolysis in the experimental group, and 9 (12.00%) were treated with remedial PCI. Coronary angiography revealed successful thrombolysis in 64 (85.33%) patients. The control group included 75 patients, with a mean age of 57.89 ± 12.04 years (57 male [76.00%]). No in-hospital death occurred in either group.
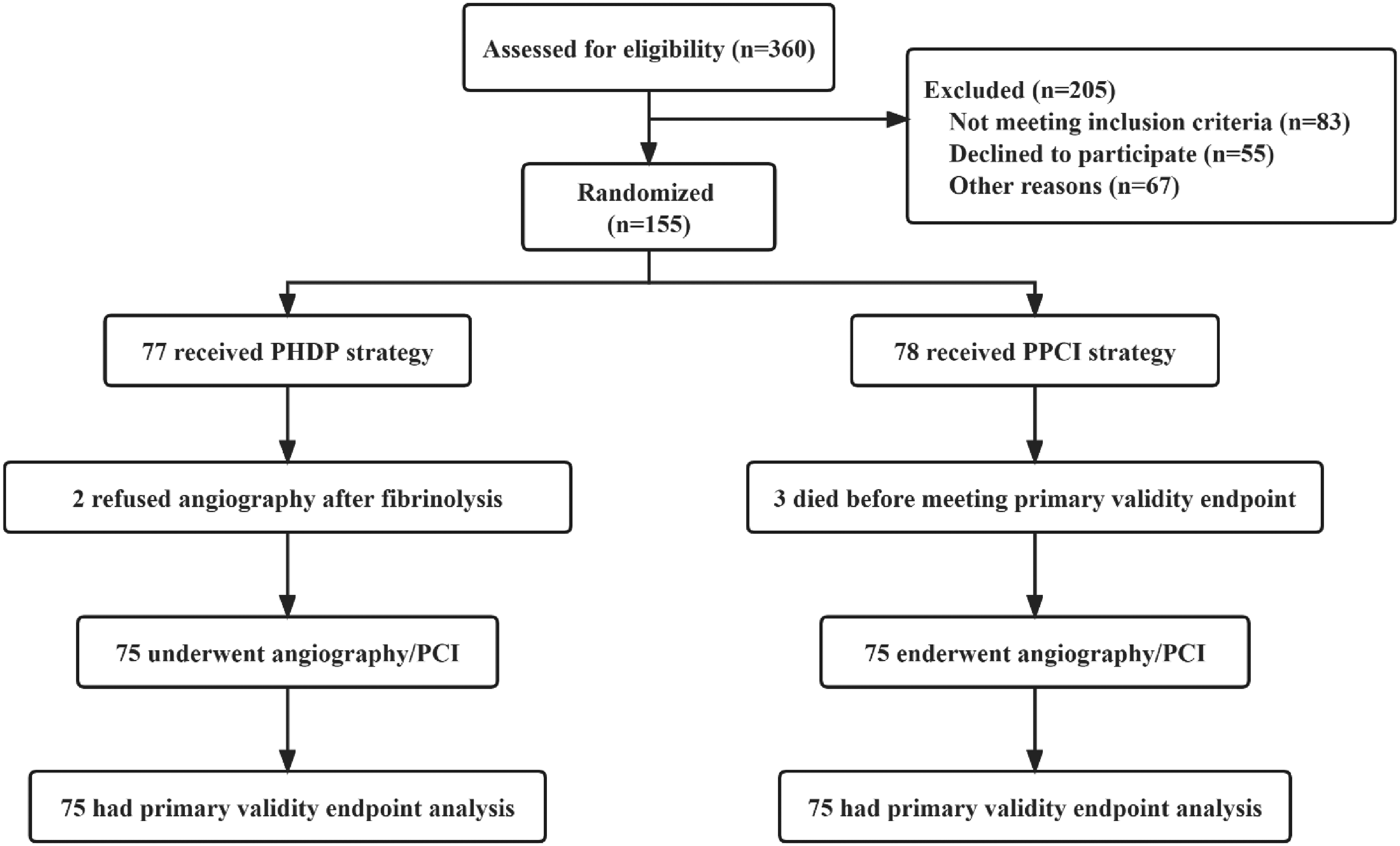
Flow diagram illustrating patient flow in the trial.
Baseline characteristics (Table 1): the differences between the two groups were not statistically significant.
Baseline Characteristics of the PPCI and PHDP Groups.
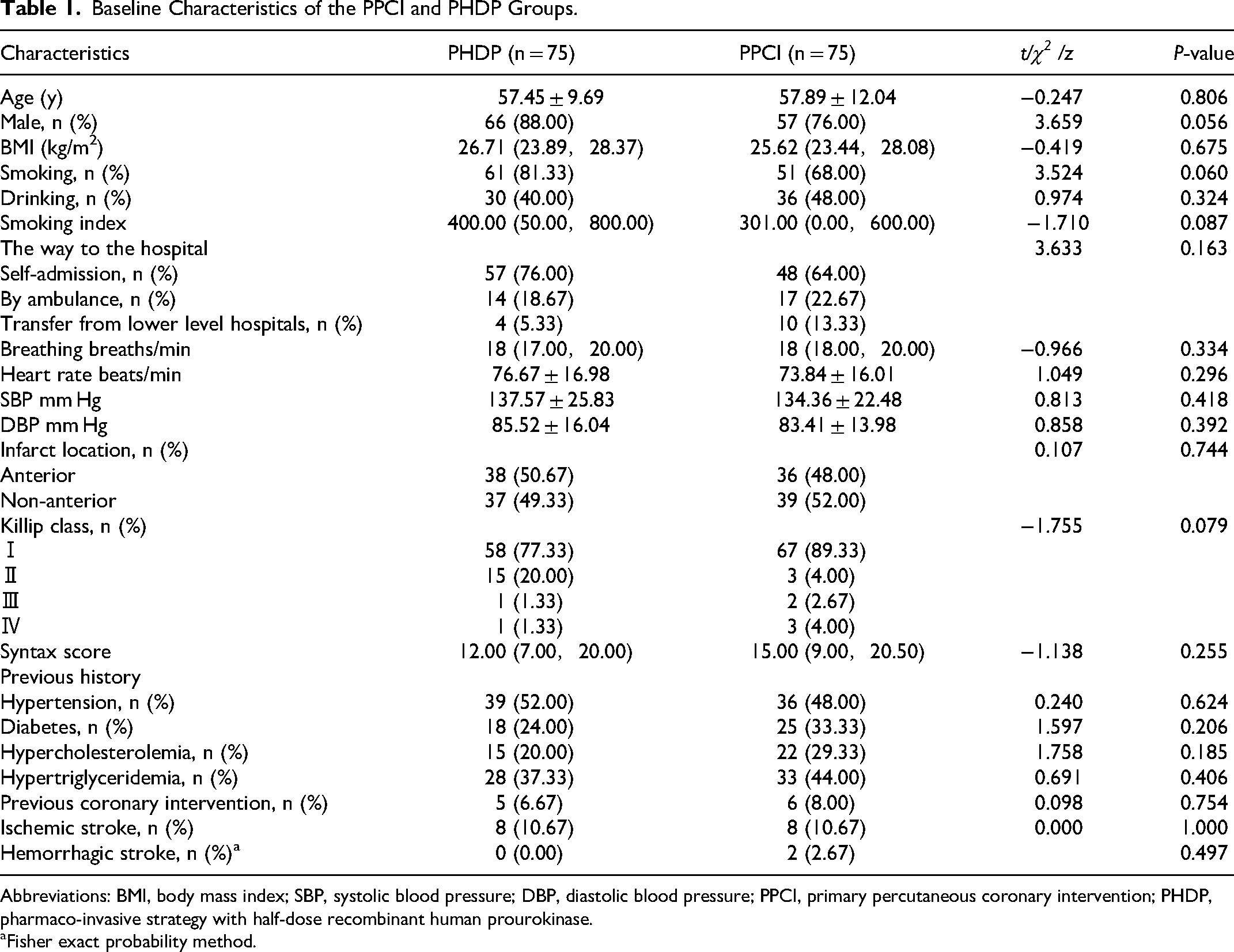
Abbreviations: BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; PPCI, primary percutaneous coronary intervention; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase.
Fisher exact probability method.
Treatment (Table 2): In rescue time intervals, there were significant differences in FMC-reperfusion (P < 0.001) and FMC-balloon dilation (P < 0.001) times. In addition, the difference in before-PCI TFG and slow-flow or no-reflow during the PCI procedure was statistically significant (P < 0.001). Furthermore, among the medications used during hospitalization, only tirofiban exhibited a statistically significant difference in usage rates (P < 0.001).
Reperfusion Therapy in the PPCI and PHDP Groups.
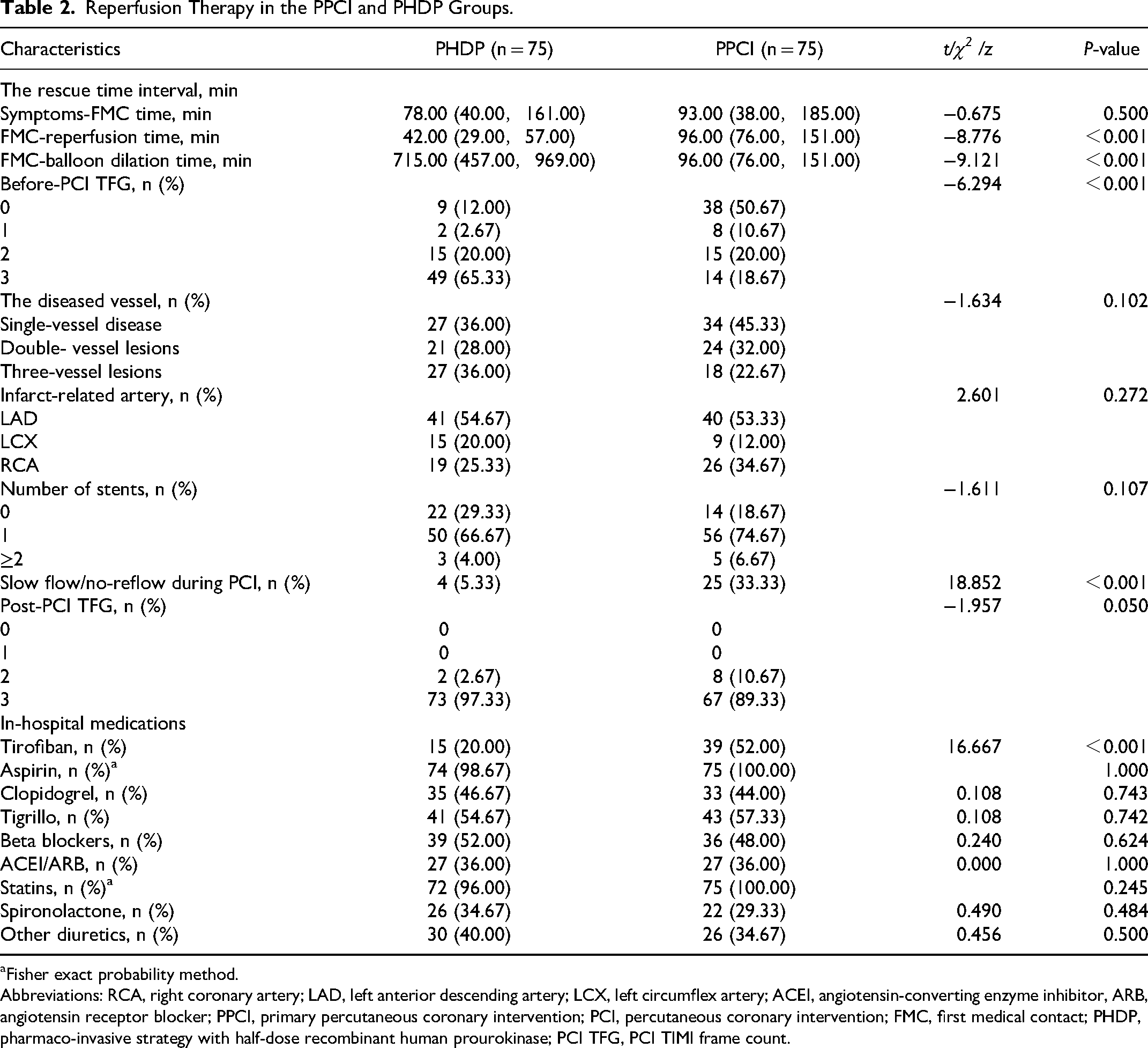
Fisher exact probability method.
Abbreviations: RCA, right coronary artery; LAD, left anterior descending artery; LCX, left circumflex artery; ACEI, angiotensin-converting enzyme inhibitor, ARB, angiotensin receptor blocker; PPCI, primary percutaneous coronary intervention; PCI, percutaneous coronary intervention; FMC, first medical contact; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase; PCI TFG, PCI TIMI frame count.
The post-PCI tests (Table 3) revealed statistically significant differences in the first postoperative activated partial thromboplastin time (APTT) (P = 0.048), fibrinogen (FIB) (P = 0.012) values, and antithrombin activity (AT) (P < 0.001).
Relevant Examination in the PPCI and PHDP Groups.
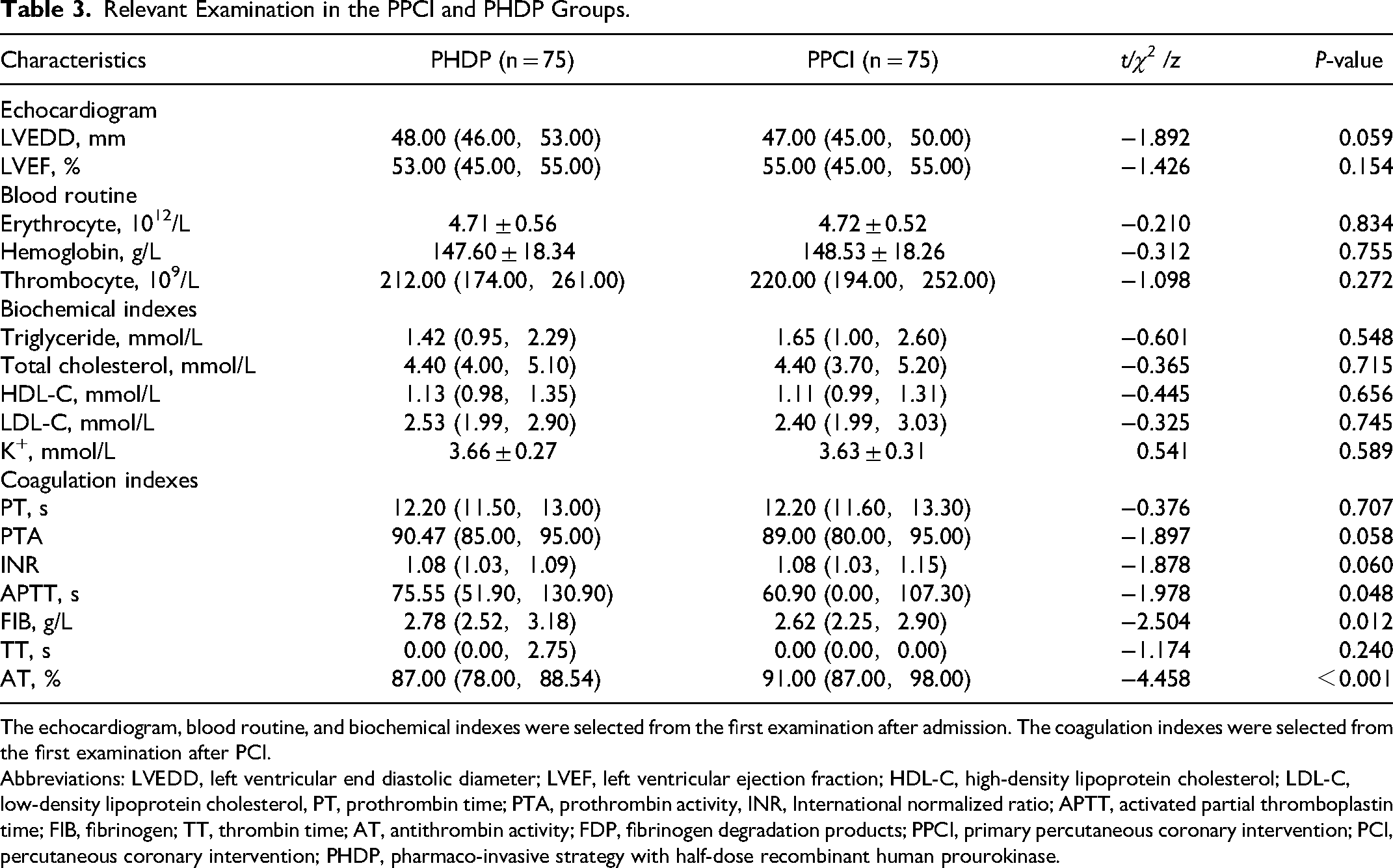
The echocardiogram, blood routine, and biochemical indexes were selected from the first examination after admission. The coagulation indexes were selected from the first examination after PCI.
Abbreviations: LVEDD, left ventricular end diastolic diameter; LVEF, left ventricular ejection fraction; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol, PT, prothrombin time; PTA, prothrombin activity, INR, International normalized ratio; APTT, activated partial thromboplastin time; FIB, fibrinogen; TT, thrombin time; AT, antithrombin activity; FDP, fibrinogen degradation products; PPCI, primary percutaneous coronary intervention; PCI, percutaneous coronary intervention; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase.
Validity endpoints (Table 4, Figure 2): no statistically significant difference between the two groups.
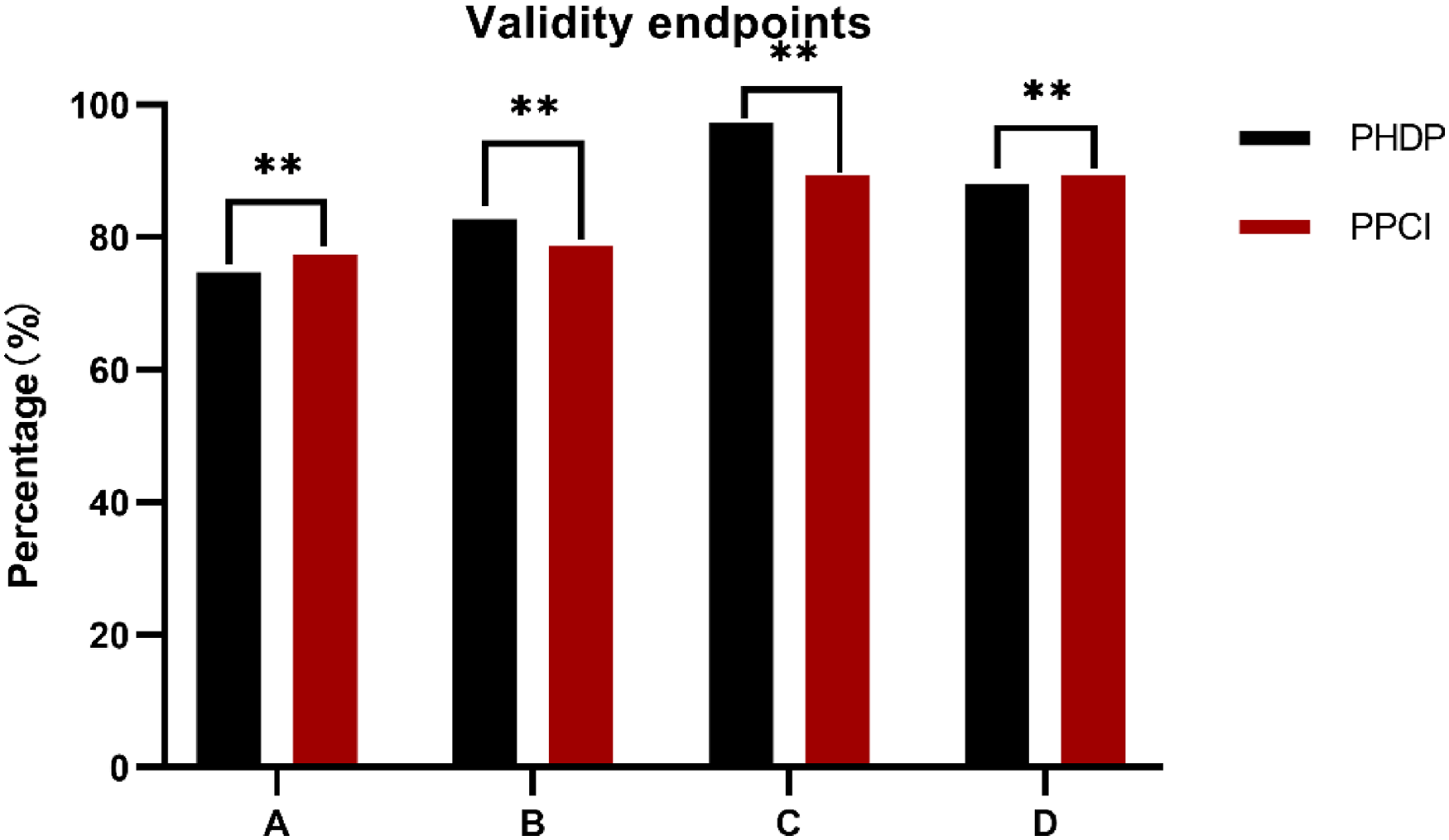
Validity endpoints in the PPCI and PHDP groups. (A) It represents complete epicardial and myocardial reperfusion, (B) it represents post-PCI TMPG = 3, (C) it represents post-PCI TFG = 3, and (D) it represents ST-segment regression of ≥ 70%. ** represents P ≥ 0.05, and ## represents P < 0.05. PPCI, primary percutaneous coronary intervention; PCI, percutaneous coronary intervention; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase; PCI TFG, PCI TIMI frame count; TMPG, TIMI myocardial perfusion.
Validity Endpoints in the PPCI and PHDP Groups.
The successful thrombolysis was confirmed by coronary angiography.
Abbreviations: PPCI, primary percutaneous coronary intervention; PCI, percutaneous coronary intervention; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase; PCI TFG, PCI TIMI frame count; TMPG, TIMI myocardial perfusion.
Safety endpoints (Table 5, Figure 3): no statistically significant differences were observed between minor bleeding and MACCEs. No major bleeding, all-cause death, cardiac death, cardiac arrest, heart failure, re-intervention, or stroke occurred during hospitalization in either group. In contrast, the difference in the incidence of arrhythmias between the two groups was statistically significant (P = 0.040), particularly for PVB (P = 0.008).
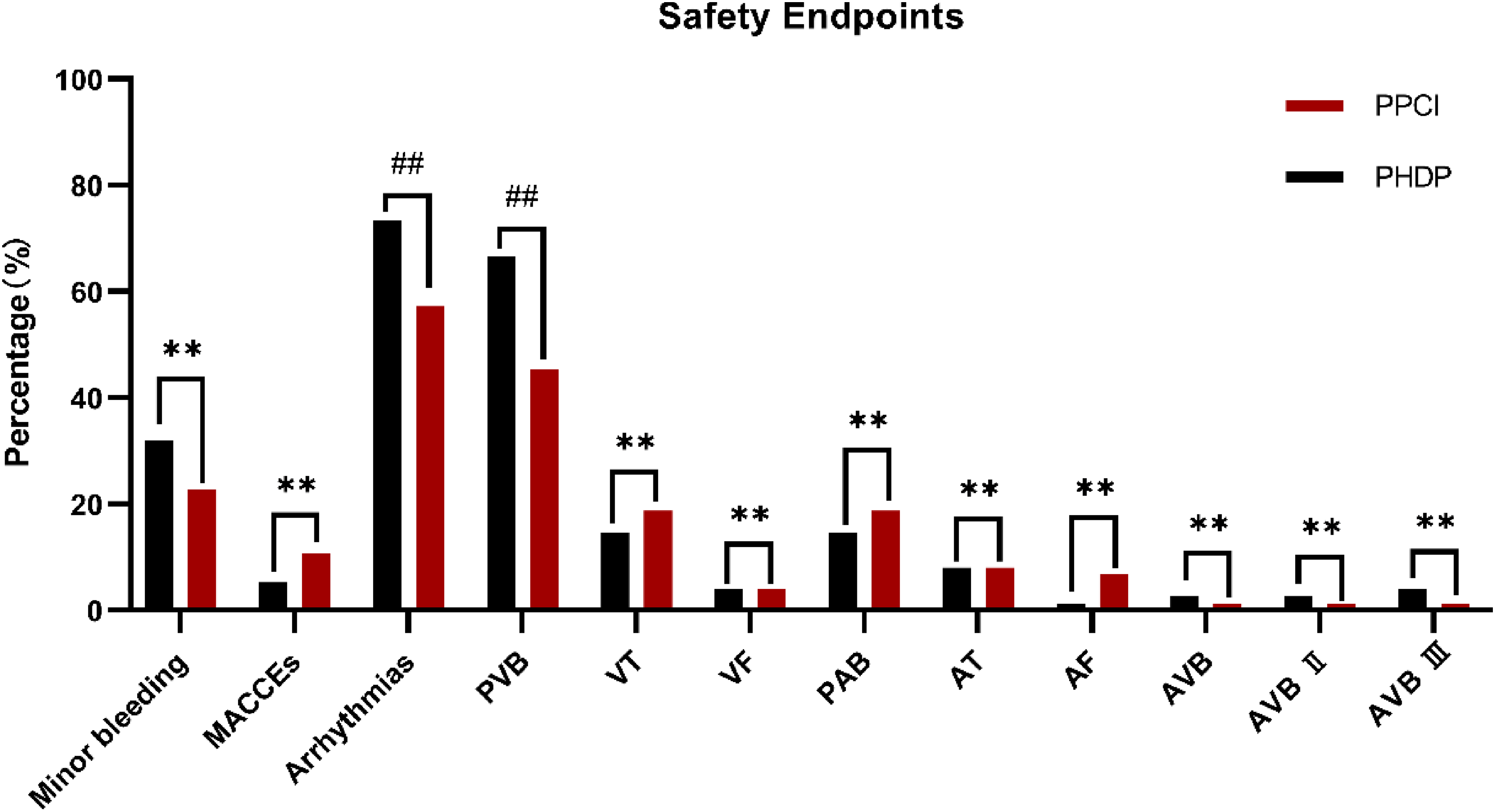
Safety endpoints in the PPCI and PHDP groups. ** represents P ≥ 0.05, and ## represents P < 0.05. PPCI, primary percutaneous coronary intervention; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase.
Safety Endpoints in the PPCI and PHDP Groups.
Abbreviations: PPCI, primary percutaneous coronary intervention; MACCEs, major adverse cardiovascular and cerebrovascular events; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase; PAB, premature atrial beats; AT, atrial tachycardia; AF, atrial fibrillation; PVB, premature ventricular beat; VT, ventricular tachycardia; AVB, atrioventricular block; VF, ventricular fibrillation.
Fisher exact probability method.
Further analysis using logistic regression was conducted to examine the potential factors that may influence the occurrence of PVB (Table 6). The results indicate that these factors are not independent risk factors for the occurrence of PVB.
Factors for Logistic Regression Analysis to Predict PVB.

Abbreviations: FMC, first medical contact; PHDP, pharmaco-invasive strategy with half-dose recombinant human prourokinase; PVB, premature ventricular beat.
Discussion
PPCI is the optimal treatment option for STEMI because of its ability to provide complete reperfusion of the myocardium in a short period; however, performing PPCI within this time window is a significant challenge. Reimer et al 11 found that the scope of myocardial necrosis gradually increased from endocardial to epicardial with prolongation of ischemic time, and that approximately one-third of cardiomyocytes were salvageable within 3 h after the onset of myocardial ischemia, while almost none were salvageable after 6 h in dog models. Therefore, myocardial reperfusion in the shortest time possible is key to treating patients with STEMI. By comparing primary angioplasty (coronary stenting within 3 h) and early routine post-fibrinolysis angioplasty (full-dose of Tenecteplase (TNK) within 3–12 h followed by coronary stenting), Fernandez-Aviles et al 12 found that early routine post-fibrinolytic angioplasty resulted in better epicardial and myocardial reperfusion (21.00% vs 6.00%; P = 0.003). Besides, the WEST study 13 compared TNK and usual care, interventional treatment within 24 h after TNK thrombolytic therapy (including rescue PCI for reperfusion failure), and PPCI, and found that the incidence of death and recurrent myocardial infarction in the TNK and usual care groups was higher than that in the PPCI group after 30 days of follow-up (P = 0.021), while there was no difference between PPCI and PCI after thrombolytic treatment with TNK (P = 0.378). In addition, Rashid et al 14 found that a PhI strategy did not increase mortality, reinfarction, or stroke rates in patients with STEMI compared with PPCI. However, in a previous study by Armstrong et al, 8 STEMI patients with symptoms within 3 h of onset and without undergoing PPCI within 1 h were randomly assigned to either the PPCI group or the TNK post-thrombolytic transfer PCI group, and the dose of TNK was adjusted to half-dose in patients ≥ 75 years of age. The results revealed that the risk for intracranial hemorrhage was higher in the full-dose post-thrombolytic transfer PCI group than in the PPCI group (1.00% vs 0.20%; P = 0.040), whereas it did not increase in the half-dose thrombolysis group (0.50% vs 0.30%; P = 0.450). Therefore, this study sheds new light on reducing the risk for bleeding associated with the PhI strategy and suggests that early TNK thrombolysis may shorten the symptom-reperfusion time (100.00 min vs 178.00 min, P < 0.001). Recently, a meta-analysis of several randomized controlled trials investigating recombinant human prourokinase revealed some advantages in its safety and efficacy, such as higher post-thrombolytic TIMI grade and TMPG grade, lower incidence of MACEs, and no significant difference in bleeding risk compared with other types of thrombolytic drugs. 7
In this study, based on previous experience, we used a half-dose of recombinant human prourokinase—a new thrombolytic agent—to compare PHDP and PPCI in the treatment of patients with STEMI and also found that PHDP shortened the FMC-myocardial reperfusion time (42.00 min vs 96.00 min, P < 0.001), which provided patients with reperfusion therapy earlier. In terms of the therapeutic effect, coronary angiography confirmed that the success rate of thrombolysis in the PHDP group reached 85.33% before PCI. This is why the before-PCI TFG was statistically significant difference (P < 0.001) in patients between the two groups. Moreover, the rate of complete reperfusion of the myocardium and epicardium after PCI was not worse than that PPCI, and the reperfusion efficiency was higher, which could provide reperfusion to the myocardium on the verge of necrosis within a short time and effectively prevent further expansion of the area of myocardial infarction. Notably, compared with PPCI, patients in the PHDP group had a lower slow or no recurrent flow rate and tirofiban use rate during the procedure and did not increase the incidence of bleeding and MACCEs during hospitalization, indicating better efficacy and higher safety of PHDP. Tirofiban is effective in blocking platelet aggregation 15 ; therefore, it was considered that low tirofiban use may be one of the reasons why PHDP did not increase the risk for bleeding during hospitalization. In the comparison of the first post-PCI coagulation indicators, the mean values of both groups were within the normal range, although the differences in FIB (P = 0.012) and AT (P < 0.001) were statistically significant. While in the PHDP group, APTT (P = 0.048) was prolonged, considering that the prolongation of APTT may be related to the use of heparin anticoagulation before thrombolysis.
Malignant arrhythmias after PCI are an important cause of poor patient prognosis. Kumar et al 16 found that the risk for VT was significantly increased if the reperfusion delay was > 5 h, and the risk for ventricular arrhythmia was significantly increased after 2 years of follow-up. Similarly, this study found that PHDP was more likely to cause arrhythmia during hospitalization (77.33% vs 57.33%; P = 0.040), especially PVB (66.67% vs 45.33%; P = 0.008). Jonathan W et al 17 conducted a 13-year follow-up study on 1139 subjects, monitoring their 24-h ambulatory electrocardiogram. The study revealed that a higher frequency of PVB was associated with an increased incidence of congestive heart failure and a higher risk of mortality, as well as a reduction in left ventricular ejection fraction. Furthermore, PVB had the potential to progress to polymorphic VT, VF, and other malignant arrhythmias. 18 However, it is important to note that in this particular study, patients who experienced these arrhythmias did not have adverse outcomes. Logistic regression analysis also confirmed that PHDP is not an independent risk factor for the occurrence of PVB. It is also a reminder that more attention should be devoted to the occurrence of arrhythmia when using this strategy to treat patients with STEMI, and routine electrocardiographic monitoring is needed to recognize PVB early to avoid malignant arrhythmia.
The present study had some limitations, the first of which was its single-center, non-blinded, randomized controlled design. The trial period was short and the number of subjects was small. Therefore, there were some biases and confounding factors. Second, subjects included patients with STEMI 18–80 years of age, with an onset time < 12 h; as such, the conclusion does not necessarily apply to elderly (> 80 years of age) or young (< 18 years of age) patients with STEMI, or those with an onset time of ≥ 12 h. Therefore, the effectiveness and safety of PHDP in elderly patients with STEMI and those with STEMI onset time ≥ 12 h need to be explored through large-sample, multi-center, and randomized controlled clinical studies. Third, the subjects were not diagnosed with coronavirus disease 2019 (COVID-19). COVID-19 leds to hemodynamic changes in the body, which may increase the prevalence of STEMI and the degree of coronary artery lesions. However, the results of this study do not apply to these patients; therefore, further clinical trials are required.
In summary, this study explored the efficacy and safety of PHDP in the treatment of patients with STEMI during hospitalization, which provides some reference for the treatment of STEMI. Through a randomized controlled analysis, compared with the PPCI strategy, this study found that applying PHDP could also provide timely and effective reperfusion therapy for STEMI patients, and it could increase the transit time for patients who cannot reach a hospital with PCI capability within the requisite time window. Notably, this study confirmed that PHDP significantly reduced the incidence of slow blood flow or no-reflow during surgery and did not increase the incidence of hemorrhage and MACCEs during hospitalization, thus reducing medical risks. Although PHDP can increase the incidence of PVB during hospitalization, it is less likely to cause malignant arrhythmias and has no adverse effects. Overall, PHDP was safe and effective for patients (18–80 years of age) with onset within 12 h. Although the effectiveness of recombinant prourokinase in treating patients with STEMI had been confirmed, the ESC guidelines still recommend that patients undergo PPCI as soon as possible (Class I). We also recommend that patients are transported to the nearest PCI hospital for treatment as soon as possible. However, this study was conducted during a period of high COVID-19 incidence, while some patients faced limited access to medical resources, resulting in difficulties reaching the hospital within 120 min for PPCI treatment. Adverse events during PPCI surgery also occurred occasionally. Presently, many primary hospitals do not have the capacity to perform PPCI, and some hesitate to perform thrombolytic therapy for patients with STEMI due to fear of complications, such as bleeding, which results in many patients with STEMI not receiving timely treatment. Based on the results of this study, the PHDP group had a lower incidence of no reflow and slow flow in the coronary artery during PCI compared to the PPCI group. Postoperative outcomes suggest that the PHDP treatment strategy is non-inferior to PPCI treatment, and the use of PHDP treatment can shorten the time to reperfusion for patients. We should consider developing PHDP treatment in primary hospitals without PPCI capacity. This program aims to overcome delays associated with PPCI and provide timely treatment for patients who cannot undergo PPCI within 2 h. By doing so, it can improve the reperfusion rate and treatment success rate for patients who experience STEMI. However, due to the limitations of this study, our conclusion still needs to be verified in further large-sample, multi-center, randomized controlled studies.
Conclusions
The efficacy and safety of PHDP in the treatment of patients with STEMI were positive. Complete epicardial and myocardial reperfusion rates, risk for bleeding during hospitalization, and incidence of MACCEs were similar to those of the PPCI strategy. Although the PHDP group has a higher incidence of PVB, it does not increase the incidence of malignant arrhythmia. This study aimed to provide a new therapeutic strategy for the treatment of STEMI in hospitals without adequate PPCI resources.
Footnotes
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Author Contributions
Donglei Luo contributed to the design and conduct of the study; project administration; funding acquisition; review and approval of the manuscript; and decision to submit the manuscript for publication. Jie Dou and Jie Gao contributed to the collection, analysis, management, and interpretation of the data; preparation and editing the manuscript; and decision to submit the manuscript for publication. Jingtao Guo contributed to the design and conduct of the study; funding acquisition; review and approval of the manuscript; and decision to submit the manuscript for publication. Huihui Yang, Ruoling Guo, and Chao Jiang contributed to the collection and management of the data; review and approval of the manuscript; and decision to submit the manuscript for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the S&T Program of Chengde (No. 201804A013, 202109A019), the Medical Science Research Project of Hebei Province (No. 20200348), and the Hebei Province Master's Graduate Innovation Ability Training Funding Project (No. CXZZSS2023147).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Chengde Central Hospital (protocol code 20181113001 and date of approval).
