Abstract
Early neurological deterioration after intravenous thrombolysis (IAT) leads to increased mortality and morbidity in patients with acute ischemic stroke (AIS). This study investigated the correlation between serum Cav-1 and NGB levels and END after IAT and explored their predictive values for poor prognosis of AIS. Totally 210 patients with AIS who underwent IAT within 4.5 h of onset were included and assigned into END group (n = 90) and Non-END group (n = 120). ELISA was used to detect serum Cav-1 and NGB levels before IAT in AIS patients. The prognosis of END patients after 3 months of treatment was evaluated using the modified Rankin Scale. Logistic multifactorial regression was used to analyze independent risk factors for END and poor prognosis after IAT. ROC curve was used to analyze the predictive effect of Cav-1 and NGB on END and poor prognosis after IAT. The area under the ROC curve was analyzed by MedCalc comparison. Compared with the Non-END group, serum Cav-1 was lower and NGB was higher in the END group. Cav-1 and NGB were independent risk factors for END after IAT. Cav-1 + NGB better predicted END after IAT than Cav-1 or NGB alone. Cav-1 and NGB were independent risk factors for END poor prognosis after IAT. Cav-1 combined with NGB better predicted poor prognosis of END after IAT than Cav-1 or NGB alone. Serum Cav-1 combined with NGB may assist in predicting the risk of END occurrence and poor prognosis after IAT in patients with AIS.
Keywords
Introduction
Stroke is the second leading cause of death and the third leading cause of disability worldwide and has become a serious global challenge. 1 Acute cerebral infarction, also known as acute ischemic stroke (AIS), is one of the two major subtypes of stroke and occurs mainly as a result of cerebral ischemia due to thrombosis of the cerebral vessels. 2 The key to treatment in the hyperacute phase of AIS is to open the occluded vessel and restore blood flow as soon as possible, thus saving the ischemic semidark zone. 3 The International Cerebrovascular Guidelines4,5 consistently recommend prompt intravenous thrombolysis (IAT) with recombinant tissue-plasminogen activator (rt-PA) as the primary form of hyperacute reperfusion therapy for AIS. This approach improves the overall prognosis of AIS patients, but there is still an early neurological deterioration after thrombolysis in 2.2%∼37.5% of patients.6,7 Previous studies have shown that post-thrombolysis END leads to an increased likelihood of mortality and morbidity and is associated with prognosis in patients undergoing IAT for AIS,8–10 as a serious adverse event after IAT. Therefore, the search for risk factors and measurable biomarkers of END after IAT in AIS patients is of great importance.
Caveolin-1 (Cav-1) is an integrin located in cell membrane vesicles and has been reported to regulate a wide range of cellular events such as proliferation, lipid metabolism, cell tracking, and signal transduction. 11 Cav-1 plays a broad regulatory role in AIS, including blood-brain barrier permeability, neuroinflammation, free radicals, apoptosis, angiogenesis, and neurogenesis, shown to be beneficial for AIS. 12 Studies have unveiled that Cav-1 regulates cerebral thrombo-inflammation in acute ischemia-reperfusion injury. 13 Reduced serum Cav-1 in AIS patients is associated with cerebral microhaemorrhage. 14 Low serum Cav-1 expression significantly increases the risk of haemorrhagic transformation following rt-PA administration. 15 In addition, a study reported that strategies to modulate Cav-1 represent a novel therapeutic method to control blood-brain barrier disruption and subsequent neurodegeneration during cerebral ischemia. 16 Neuroglobin (NGB), a member of the histone bead protein family, has a high affinity for oxygen and is highly specifically expressed in cerebral neurons. 17 A growing body of evidence clearly demonstrates the neuroprotective effects of NGB against hypoxic and ischemic injury in cultured neurons and animal brains.18–21 Studies have reported that elevated serum NGB levels after AIS can be used as a predictor of stroke severity and poor prognosis. 22 However, the relationship between serum Cav-1 and NGB levels and END after rt-PA in AIS patients is unclear. This study was to investigate whether serum Cav-1 and NGB levels are associated with END after IAT in AIS and to explore their predictive values for poor prognosis in AIS, with a view to enabling early identification of END and providing reference for clinicians to scientifically develop individualized treatment plans.
Methods
Study Subjects
The study was a retrospective analysis study and all experiments were approved by the ethics committee of Dalian Central Hospital Affiliated to Dalian University of Technology and conducted in accordance with the Declaration of Helsinki. The study selected 292 patients with AIS who underwent IAT with rt-PA within 4.5 h of onset in Dalian Central Hospital Affiliated to Dalian University of Technology between June 2020 and May 2022 as the screening study population. Inclusion criteria were as follows: (i) meeting the diagnostic criteria of AIS in the Chinese Guidelines for the Diagnosis and Treatment of Acute Ischemic Stroke 2018; (ii) referring to the American Heart Association/American Stroke Association 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke 5 and the European Stroke Organization (ESO) Guidelines for the Treatment of Intravenous Thrombolysis in Acute Ischemic Stroke, 4 standardized IAT with rt-PA therapy within 4.5 h of onset; (iii) age greater than or equal to 18 years; (iv) signed informed consent. Exclusion criteria: (i) time between onset and admission > 4.5 h; (ii) previous thrombolysis or endovascular therapy; (iii) history of hematological disease, cancer, or surgery; (iv) incomplete clinical data or inability to assess neurological deficit within 24 h of IAT; (v) severe renal or liver disease; (vi) loss to follow-up.
Of the selected 292 patients with AIS, according to inclusion and exclusion criteria: 25 were excluded for thrombolysis or endovascular treatment; 16 were excluded for a history of hematological disease, cancer, or surgery; 6 were excluded for severe renal or liver disease; 21 were excluded for incomplete clinical data; 14 were excluded since they could not be assessed for neurological deficit within 24 h of IAT. A total of 210 patients were included for final analysis (Figure 1). These patients were assigned into the END group (n = 90) and the Non-END group (n = 120) according to whether END occurred after IAT with rt-PA.
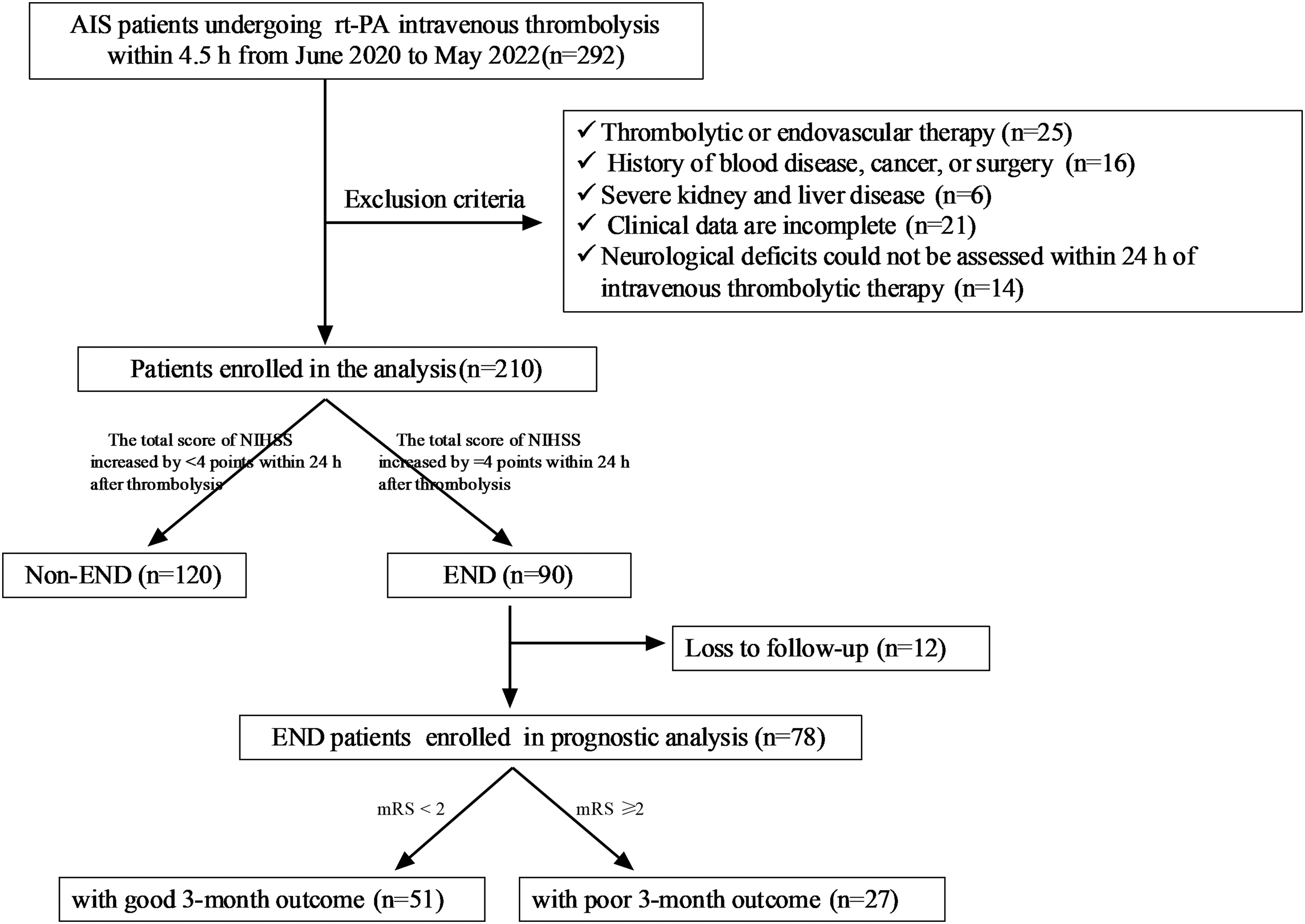
Patients flow-chart of the cohort.
Definition of END
AIS severity was assessed on the day of admission using the National Institutes of Health Stroke Scale (NIHSS) score. Stroke severity was assessed 2–3 times by two certified neurologists within 24 h of admission. END was defined as an increase in the NIHSS score by ≥ 2 points (or stroke-related death) within 5 d of admission in AIS patients compared with the initial score on admission.
rt-PA Therapy
All patients with AIS were treated with intravenous drip of Alteplase 0.9 mg/kg (50 mg, Boehringer Ingelheim Pharma GmbH, Germany, S20110052) within 4.5 h of onset, controlling the maximum dose to less than 90 mg. About 10% of the Alteplase was administered intravenously within the first 1 min and the remaining 90% was pumped intravenously within 1 h. Patients’ vital signs were closely monitored during treatment. In addition, all patients received standard treatment in the stroke unit, such as antiplatelet therapy and statins.
Clinical Data Collection
On the day of admission, demographic characteristics [age, sex, and body mass index (BMI)], vascular risk factors [smoking, alcohol consumption, hypertension, hyperlipidemia, diabetes, coronary heart disease (CHD), atrial fibrillation (AF), clinical assessments [NIHSS score, blood pressure, hemorrhagic transformation (HT), onset to needle time (ONT)], stroke subtype [largeartery atherosclerosis (LAA), cardioembolism (CE), small-artery occlusion (SAO), stroke of other determined etiology (SOE), and stroke of undetermined etiology (SUE)], site of lesion (Anterior circulation, Posterior circulation) were recorded. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured and recorded immediately on admission. The site of lesion, stroke subtype, and HT were assessed by computed tomography, magnetic resonance imaging, electrocardiography, echocardiography, carotid ultrasound, and transcranial Doppler. Stroke subtypes were classified according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) criteria. 23 Five mL of fasting elbow venous blood was drawn, of which 2 mL was used for detection of laboratory biochemical parameters, including total cholesterol (TC), triglyceride (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), fasting blood glucose (FBG), homocysteine (Hcy), hyper-sensitive C-reactive protein (Hs-CRP), and white blood cell (WBC), white blood cell (WBC), neutrophils, and lymphocytes. The remaining 3 mL of blood was centrifuged at 1500 g for 10 min within 30 min of collection and the serum was stored at −80 °C for subsequent testing of Cav-1 and NGB levels.
Enzyme-Linked Immunosorbent Assay (ELISA)
Serum Cav-1 and NGB levels of AIS patients at admission were measured using ELISA kits according to the manufacturer's instructions. Specific information on the ELISA kits is as follows: human Cav-1 ELISA kit (sensitivity 0.188 ng/mL; detection range 0.313–20 ng/mL; EH0775, Wuhan Fien Biotechnology, Wuhan, China), human NGB ELISA kit (sensitivity 0.052 ng/mL; detection range 0.156–10 ng/mL; RDR-NGB-Hu, AmyJet Scientific, Wuhan, China).
Follow-up
The prognosis of patients in the END group was evaluated by the modified Rankin Scale (mRS) after 3 months of treatment. 24 There are seven levels of mRS: 0 (completely asymptomatic); 1 (only symptoms that do not affect daily life and work); 2 (mildly disabled, does not need others to take care of daily life), but unable to perform all the activities before the disease alone; 3 (moderately disabled, partially needs help with daily life and can walk independently); 4 (moderately to severely disabled, needs help with daily life and has difficulty in walking independently); 5 (severely disabled, totally dependent on others for daily life, staying in bed); 6 (death). mRS < 2 was considered as good prognosis and was included in the good prognosis group; mRS ≥ 2 was considered as poor prognosis and included in the poor prognosis group.
Statistical Analysis
SPSS 21.0 (SPSS, Inc., Chicago, IL, USA) and GraphPad Prism 8.01 software (GraphPad Software Inc., San Diego, CA, USA) were used for statistical analysis and graphing of the data. The data were divided into counting and measurement data. Count data were expressed as number of cases and percentages. The measurement data were tested for normal distribution using the Shapiro-Wilk test. Measurement data that conformed to a normal distribution were expressed as mean ± standard deviation, and those that did not conform to a normal distribution were expressed as median [interquartile range]. Comparison between two groups: chi-square test for count data, independent sample t-test for measures that conform to normal distribution, and Wilcoxon test for measurement data that do not conform to normal distribution. Logistic multi-factor regression analysis was applied for identifying whether serum Cav-1 and NGB are independent risk factors for END and poor prognosis after IAT in patients with AIS. Receiver operating characteristic (ROC) curves were used to analyze the adjunctive predictive value of serum Cav-1 and NGB on the occurrence of END and poor prognosis after IAT, and MedCalc to compare the difference in the area under the ROC curve (AUC). Differences were considered statistically significant at P < 0.05.
Results
Comparison of Clinical Baseline Information Between the Two Groups of Subjects
A total of 210 patients with AIS who underwent IAT with rt-PA within 4.5 h of onset were included in this study from June 2020 to May 2022 in our hospital. These patients were allocated into the END group (n = 90) and Non-END group (n = 120) according to whether END occurred after rt-PA treatment. We compared the clinical baseline data of the two groups of AIS patients and the results are shown in Table 1. Compared with the Non-END group, the END group had a higher proportion of patients with diabetes, AF, and HT, a higher NIHSS score on admission, longer ONT and thrombolysis time, higher serum TG, FBG, Hcy, Hs-CRP, WBC levels, and lower lymphocyte levels (all p < 0.05), while other clinical baseline information such as age, sex, BMI, history of smoking and alcohol consumption were not significantly different between the two groups (all p > 0.05).
Clinical Baseline Data of the Two Groups of AIS Patients.

Note: END, early neurological deterioration; AIS, acute ischemic stroke; BMI, body mass index; CHD, coronary heart disease; NIHSS, National Institutes of Health Stroke Scale; SBP, systolic blood pressure; DBP, diastolic blood pressure; HT, hemorrhagic transformation; ONT, onset to needle time; LAA, large artery atherosclerosis; CE, cardioembolism; SAO, small-artery occlusion; SOE, stroke of other determined etiology; SUE, stroke of undetermined etiology; TC, total cholesterol; TG, triglyceride; HDL, high-density lipoprotein; LDL, low-density lipoprotein; FBG, fasting blood glucose; Hcy, homocysteine; Hs-CRP, hyper-sensitive C-reactive protein; WBC, white blood cell.
Significantly Lower Serum Cav-1 Levels and Higher NGB Levels in the END Group of AIS Patients
To investigate the relationship between serum Cav-1 and NGB levels and END after IAT, we first examined the changes in their expression in the two groups of patients. ELISA results showed that compared to the Non-END group, serum Cav-1 levels were notably lower in AIS patients in the END group (3.23 ± 1.85 ng/mL vs 5.51 ± 1.97 ng/mL, p < 0.001, Figure 2A) and NGB levels were clearly higher (8.15 ± 1.10 ng/mL vs 6.52 ± 1.14 ng/mL, p < 0.001, Figure 2B).

Analysis of the differences in serum cav-1 and NGB levels between AIS patients in the Non-END and END groups. ELISA was performed to detect (A) Cav-1 and (B) NGB levels in the serum of AIS patients in the Non-END and END groups. Data were expressed as mean ± standard deviation. Independent samples t-test was used for comparisons between the two groups. ** p < 0.01.
Serum Cav-1 and NGB Were Independent Risk Factors for END After IAT in Patients with AIS
We further assessed whether serum Cav-1 and NGB levels were independently associated with END after IAT in patients with AIS. Using the occurrence of END after IAT as the dependent variable, based on the analysis results on Table 1, the items with P < 0.05, including diabetes mellitus, AF, NIHSS, HT, ONT, thrombolysis time, TG, FBG, Hcy, Hs-CRP, WBC, Lymphocyte, Cav-1, and NGB were included as independent variables in a logistic multivariate regression analysis model. The results showed (Table 2) that after correction for all potential confounders, Cav-1 (OR = 0.540, 95% CI 0.388–0.751, p = 0.000), NGB (OR = 4.287, 95% CI 2.350–7.818, p = 0.000), NIHSS (OR = 1.174, 95%CI 1.001–1.377, p = 0.049), ONT (OR = 1.025, 95%CI 1.007–1.044, p = 0.007), and Hs-CRP (OR = 1.329, 95%CI 1.081–1.634, p = 0.007) were identified as independent risk factors for END after IAT.
Logistic Multifactorial Regression Analysis of Risk Factor of the Occurrence of END After IAT in Patients with AIS.

Note: END, early neurological deterioration; IAT, intravenous thrombolysis; AIS, acute ischemic stroke; NIHSS, National Institutes of Health Stroke Scale; HT, hemorrhagic transformation; ONT, onset to needle time; TC, total cholesterol; FBG, fasting blood glucose; Hcy, homocysteine; Hs-CRP, hyper-sensitive C-reactive protein; WBC, white blood cell; Cav-1, Caveolin-1; NGB, Neuroglobin.
Serum Cav-1 Combined with NGB May Help Predict END After IAT in AIS
ROC curves were plotted to analyze the predictive value of serum Cav-1 and NGB levels on END after IAT. The results are shown in Table 3: the AUC of serum Cav-1 for predicting END after IAT was 0.787 (0.727–0.847), with 44.44% sensitivity (34.62%–54.73%), 96.67% specificity (91.74%–98.70%), and optimal cut-off value of 2.570; the AUC of serum NGB for predicting END after IAT was 0.833 (0.781–0.885), with 100.00% sensitivity (95.91%–100.00%), 50.00% specificity (41.19%–58.81%) and optimal cut-off value of 6.400; The AUC for serum Cav-1 combined with NGB to predict END after AIT was 0.903 (0.863–0.943), with 82.22% sensitivity (73.06%–88.75%), 83.33% specificity (75.65%–88.94%), and an optimal cut-off value of 4.385. MedCalc comparison of the AUC showed that the AUC of serum Cav-1 combined with NGB was greatly higher than Cav-1 or NGB alone (all p < 0.001) (Figure 3). These results suggest that serum Cav-1 combined with NGB > 4.385 may be an aid in predicting END after AIT.

ROC curve analysis of predictive values of serum cav-1 and NGB levels for END after AIT in AIS patients.
ROC Curve Analysis of Predictive Values of serum Cav-1 Combined with NGB for END After IAT in AIS Patients.

Note: ROC, receiver operating characteristic; AUC, area under the ROC curve; END, early neurological deterioration; IAT, intravenous thrombolysis; AIS, acute ischemic stroke; Cav-1, Caveolin-1; NGB, Neuroglobin.
Serum Cav-1 and NGB Are Independent Risk Factors for Poor Prognosis of END After IAT in AIS
We further explored the relationship between serum Cav-1 and NGB and poor prognosis of END after AIT. We analyzed the prognosis of 90 patients with END after AIT after 3 months of treatment and excluded 12 patients who were lost to follow-up, and finally, 51 patients with good prognosis and 27 patients with poor prognosis were included in the analysis (Figure 1). Logistic univariate regression analysis (Table 4) showed that NIHSS, ONT, thrombolysis time, Hs-CRP, Cav-1, and NGB were strikingly associated with poor prognosis of END after AIT in AIS patients (all p < 0.05), while the remaining clinical indicators were not noticeably associated with poor prognosis of END after IAT (all p > 0.05). These indicators with correlations were included in a logistic multivariate regression analysis (Table 4), which showed that after correction for all potential confounders, Cav-1 (OR = 0.322, 95% CI 0.117–0.888, p = 0.029) and NGB (OR = 3.165, 95% CI 1.018–9.841, p = 0.047) were identified as independent risk factors for poor prognosis of END after AIT.
Logistic Multifactorial Regression Analysis of Risk Factor of Poor Prognosis of END After IAT in Patients with AIS.

Note: END, early neurological deterioration; IAT, intravenous thrombolysis; AIS, acute ischemic stroke; BMI, body mass index; CHD, coronary heart disease; NIHSS, National Institutes of Health Stroke Scale; SBP, systolic blood pressure; DBP, diastolic blood pressure; HT, hemorrhagic transformation; ONT, onset to needle time; TC, total cholesterol; TG, triglyceride; HDL, high-density lipoprotein; LDL, low-density lipoprotein; FBG, fasting blood glucose; Hcy, homocysteine; Hs-CRP, hyper-sensitive C-reactive protein; WBC, white blood cell; Cav-1, Caveolin-1; NGB, Neuroglobin.
Serum Cav-1 Combined with NGB May Help Predict the Risk of Poor Prognosis of END After IAT in AIS
ROC curves were plotted to analyze the predictive value of serum Cav-1 and NGB levels on the risk of adverse prognosis in patients with END after IAT. The results are shown in Table 5: the AUC of serum Cav-1 for predicting adverse prognosis of END after IAT was 0.882 (0.808–0.955), with 92.59% sensitivity (76.63%–98.68%), 74.51% specificity (61.13%–84.45%), and an optimal cut-off value of 2.945; the AUC of serum NGB for predicting adverse prognosis of END after IAT was 0.894 (0.819–0.969), with 81.48% sensitivity (63.3–91.82)%, 86.27% specificity (74.28%–93.19%), and optimal cut-off value of 8.610; the AUC of serum Cav-1 combined with NGB for predicting adverse prognosis of END after IAT was 0.915 (0.851–0.978), with 82.22% sensitivity (73.06%–88.75%), 83.33% specificity (75.65%–88.94%), and optimal cut-off value of 7.045. MedCalc comparison of the differences in AUC showed that the AUC of serum Cav-1 combined with NGB was markedly higher than both Cav-1 (p = 0.033) and NGB alone (p = 0.021) (Figure 4). Overall, serum Cav-1 combined with NGB > 7.045 may assist in predicting the risk of adverse prognosis of END after IAT.

ROC curve analysis of predictive values of serum cav-1 and NGB levels for risk of adverse prognosis of END after AIT in AIS patients.
ROC Curve Analysis of Predictive Values of serum Cav-1 Combined with NGB for Poor Prognosis of END After IAT in AIS Patients.

Note: ROC, receiver operating characteristic; AUC, area under the ROC curve; END, early neurological deterioration; IAT, intravenous thrombolysis; AIS, acute ischemic stroke; Cav-1, Caveolin-1; NGB, Neuroglobin.
Discussion
Despite recent advances in treatment, stroke still represents a leading contributor to disability and mortality; moreover, its burden is projected to increase in the coming decades owing to population aging and growth. 25 IAT is established as an effective modality for the treatment of AIS. 26 However, END is reported to occur in 10% of AIS patients after IAT, which is an ominous clinical event tightly related to poor outcomes. 27 Therefore, early identification of END is beneficial for formulating personalized treatment strategies, thus optimizing the management of AIS. In this study, we investigated the correlation of Cav-1 and NGB with post-thrombolysis END and the obtained results elucidated that serum Cav-1 combined with NGB could assist in predicting the risk of END incidence and poor outcome in AIS patients receiving IAT.
END after IAT is partly due to ischemia progression, brain edema, or HT. 28 AF is a contributor to the increased risk of END, poor prognosis, and death of AIS patients subsequent to IAT. 28 High hs-CRP level is shown to be independently linked with END in AIS individuals suffering with AF. 29 Glucose level at admission, OTT, diabetes mellitus, and AF are evidently related to END occurrence among AIS patients undergoing reperfusion therapy. 30 A nomogram composed of diabetes mellitus, AF, Hs-CRP, baseline NIHSS, and previous antiplatelet medication may be used to predict the risk of END among individuals with AIS. 31 Additionally, compelling evidence has revealed the correlation of TG and Hcy with END and the role of hypertriglycedemia as a potential predictor for END in the setting of acute lacunar stroke. 32 In this study, 210 AIS patients were included as subjects and allocated into the END and non-END groups. The comparison of clinical data uncovered that AIS patients with post-IAT END had an elevated proportion of diabetes mellitus, AF, and HT, increased NIHSS scores on admission, prolonged ONT and thrombolysis time, and raised levels of serum TG, FBG, Hcy, hs-CRP, and WBC, as well as reduced lymphocytes.
Cav-1 confers a protective function in the first days following ischemia by facilitating astrogliosis, scar formation, and neovascularization, but the absence of Cav-1 is extensively related to large lesion volumes and serious neurological deficits. 33 Cav-1 is critical in regulating blood-brain barrier (BBB) permeability, and lentiviral-mediated re-expression of Cav-1 is shown to control BBB disruption and subsequent neurological deterioration in cerebral ischemia. 16 Ischemic stroke elevates the expression of neuroprotective protein NGB, indicating that NGB probably represents a novel target for the therapy of stroke. 34 The serum NGB concentration peaks at 72 h after AIS and is notably related to admission NIHSS score. 22 Our findings unveiled that serum Cav-1 level was downregulated and NGB level was upregulated in AIS patients with post-IAT END. An existing study has unveiled that Cav-1 decreases neuroinflammation by affecting inflammatory cells and cytokines, weakens BBB permeability via downregulation of MMP9, and facilitates nerve regeneration and angiogenesis through the Cav-1/VEGF pathway following ischemic stroke. 12 However, endothelial Cav-1 suppresses neuronal differentiation in part by negatively modulating VEGF. 35 One possible explanation may be the paradoxical phenomenon between Cav-1 and VEGF: cav-1 could downregulate VEGF expression but is also required for the VEGF signaling. Therefore, the intrinsic relationship between Cav-1 and VEGF warrants further investigations in the future.
Increased serum Cav-1 concentration is notable related to a favorable alternation in modified Rankin Scale score distribution and an improved prognosis among ischemic stroke patients following mechanical thrombectomy, suggesting the potential role of Cav-1 as a prognostic biomarker for ischemic stroke. 36 Elevated serum NGB serves as a predictor of both stroke severity and poor outcome. 22 Poor functional and cognitive outcomes following aneurysmal subarachnoid hemorrhage are tightly associated with higher NGB level. 37 However, there is no report regarding the clinical significance of serum Cav-1 and NGB levels for post-IAT END. In our analyses, Cav-1 and NGB were identified as independent risk factors for END incidence and poor prognosis following IAT among AIS patients, and a combination of the above two also showed high predictive values for END occurrence and poor prognosis.
In conclusion, the present study underlined that serum Cav-1 in combination with NGB could assist in predicting the risk of END occurrence and poor prognosis after IAT in AIS patients. However, the included sample size for analysis was small. Therefore, further expanding the sample size is warranted to investigate the clinical value of serum Cav-1 combined with NGB. In the future, we shall conduct multi-center prospective studies with expanded sample size and matched controls to increase the confidence of the results.
Footnotes
Authors’ Contributions
LHZ is the guarantors of integrity of the entire study and contributed to the study concepts; CW, HQ contributed to the study design, definition of intellectual content; XSL, JPX contributed to the literature research, clinical studies, data analysis; MHZ contributed to the statistical analysis, manuscript editing; DL contributed to the data acquisition, manuscript review; All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The study was a retrospective analysis study and all experiments were approved by the ethics committee of Dalian Central Hospital Affiliated to Dalian University of Technology and conducted in accordance with the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Dalian Central Hospital “peak plan” science and technology project (Grant number: 2022ZZ204.)
