Abstract
Acute ischemic stroke patients with intravenous (IV) recombinant tissue plasminogen activator (rt-PA) thrombolysis have different outcomes. The degree of thrombolysis depends largely on the delicate balance of coagulation and fibrinolysis. Thus, our study aimed to investigate the prognostic value of routine coagulation parameters in acute stroke patients treated with rt-PA. From December 2016 to October 2018, consecutive patients treated with standard-dose IV rt-PA within 4.5 h of stroke onset were collected in Beijing Tiantan Hospital. Routine coagulation parameters, including platelet count, mean platelet volume, platelet distribution width, prothrombin time (PT), activated partial thromboplastin time, thrombin time, and fibrinogen, were measured at baseline (h0) and 24 h (h24) after thrombolysis. The change of coagulation parameters was defined as the (h24-h0)/h0 ratio. The prognosis included short-term outcome at 24 h and functional outcome at 3 months. A total of 267 patients were investigated (188 men and 79 women) with a mean age of 60.88 ± 12.31 years. In total, 9 patients had early neurological deterioration within 24 h, and 99 patients had an unfavorable outcome at the 3-month visit. In multivariate logistic regression, the (h24-h0)/h0 of PT was associated with unfavorable functional outcomes at 3 months (odds ratio: 1.42, 95% confidence interval: 1.02-2.28). While the change of other coagulation parameters failed to show any correlation with short-term or long-term prognosis. In conclusion, the prolongation of PT from baseline to 24 h after IV rt-PA increases the risk of 3-month unfavorable outcomes in acute stroke patients.
Backgrounds
Stroke, of which 69.6% is ischemic stroke, has surpassed coronary heart disease and cancer, becoming the leading cause of death and adult disability in China. 1 Early reperfusion is the key treatment for patients with ischemic stroke, as it can save ischemic penumbra tissue before irreversible neural damage occurs. With the preoperative selection of patients and improved stent retriever, endovascular thrombectomy has shown impressive benefits for stroke patients based on a meta-analysis of 5 randomized control trials in 2015. 2 Moreover, the time window for early endovascular treatment has been extended up to 16 or 24 h.3,4 These findings updated the guidelines for the early management of acute ischemic stroke (AIS) worldwide. Although it is promising in the new era of endovascular therapy, intravenous (IV) thrombolysis with recombinant tissue plasminogen activator (rt-PA) is still the preferred and fundamental treatment for AIS within 4.5 h. 5 However, early neurological deterioration after thrombolysis occurs in ∼10% of patients due to ischemia progression, hemorrhage transformation, or brain edema. 6 Only 30% of patients have a favorable outcome at 90 days. 7 Rt-PA, known as a fibrin-specific thrombolytic agent in its pharmacological characteristic, can cause plasmin activation, fibrinolysis and ultimately result in thrombolysis and vessel recanalization. The degree of thrombolysis depends largely on the delicate balance of coagulation and fibrinolysis. Data on fibrin degradation products, d-dimer, coagulation factors, α2-antiplasmin as well as thromboelastography showed significant correlations with prognosis in stroke patients treated with thrombolysis.8–11 While the value of platelet and coagulation parameters in routine laboratory examination has not been well-recognized. Besides, the 2015 American guideline recommended that IV rt-PA treatment should be administrated before the results of blood coagulation test came out when there is no suspicion of coagulopathy. 12
Therefore, we conducted this prospective consecutive cohort study to explore the prognostic value of routine coagulation parameters in AIS patients treated with IV rt-PA.
Subjects and Methods
Ethics Statement
This study was conducted in accordance with the guidelines from the Helsinki Declaration and was approved by the Ethics Committees of Beijing Tiantan Hospital to collect data without requiring individual patient informed consent under the common rule.
Study Population
From December 2016 to October 2018, we prospectively collected consecutive patients treated with IV rt-PA within 4.5 h of AIS onset using the standard dose of 0.9 mg/kg in the emergency room of Beijing Tiantan Hospital, a tertiary stroke center that serves a population of around 3 million inhabitants. A total of 336 participants met the inclusion criteria approved by the American Stroke Association and received IV rt-PA therapy. 13 Patients who met any of the following exclusion criteria were ineligible for our study: (1) with a combination of mechanical thrombectomy, (2) treated with thrombolysis before, (3) having a premorbid modified Rankin Scale (mRS) score of >1. Eventually, 267 participants with complete baseline information were enrolled. Of note, antithrombotic drugs were strictly prohibited during the first 24 h after administration of rt-PA, including aspirin, clopidogrel, tirofiban, ticagrelor, novel oral anticoagulants, heparin or low molecular heparin, warfarin, and so on.
Laboratory Assessment
Blood samples were taken from the antecubital vein at baseline and 24 h after the infusion of rt-PA. Blood cell count was performed on EDTA with an ADVIA 120 counter (Siemens Healthcare Diagnostics), focusing on the measurement of platelet count (PLT), mean platelet volume (MPV), and platelet distribution width (PDW). For coagulation parameters, blood was collected in 5 mL evacuated tubes using one-tenth of 0.129 mol/L saline sodium citrate as an anticoagulant. Platelet-poor plasma was obtained by centrifugation at 3000 g for 20 min at room temperature and then was frozen at −40 °C in aliquots until used. Prothrombin time (PT), activated partial thromboplastin time (APTT), thrombin time (TT), and fibrinogen (Fbg) were photo-optically measured using an ALC-Top coagulometer.
Assessment of Potential Confounding Variables
Demographic information including age, sex, blood pressure at emergency, onset-to-needle time (ONT), smoking status, medical history (hypertension, diabetes mellitus, stroke, coronary heart disease, atrial fibrillation), and history of antithrombotic or anticoagulant drugs was extracted from electronic medical records. ONT was further subdivided into <3 h and 3 to 4.5 h. Weight and height were measured, and body mass index (BMI) was calculated as weight (kg)/height2 (m2). The territory of infarction based on computed tomography or diffusion-weighted magnetic resonance imaging was divided into anterior circulation and posterior circulation. Based on the Chinese ischemic stroke subclassification (CISS), etiologies of stroke include large-artery atherosclerosis, penetrating artery disease, cardiogenic stroke, other etiology, and undetermined etiology. 14 Accordingly, the etiologies were further classified into cardiogenic and noncardiogenic strokes in this study.
Clinical Outcome
Neurological deficits were assessed using the National Institute of Health Stroke Scale (NIHSS) score at baseline, 2 h, and 24 h after rt-PA by neurologists who were blinded to coagulation results. Early neurological deterioration was defined as a 4-point or greater increase in NIHSS score within 24 h after thrombolysis. 5 3-month functional outcome was evaluated by mRS score, patients were categorized into favorable (mRS 0-1) and unfavorable outcome group (mRS ≥ 2).
Statistical Analysis
All statistical analyses were performed using SPSS22.0 (IBM SPSS Statistics). Continuous variables were presented as mean ± standard deviation or median (interquartile range). Categorical variables were presented as frequency and percentage. The changes between 2-time points were defined as (h24-h0)/h0 ratio. For each parameter, the values at baseline (h0), 24 h after IV rt-PA (h24), and (h24-h0)/h0 were calculated. All above were divided into 3 groups (T1, T2, and T3) according to their tertiles, the lowest tertile was defined as the reference group.
Comparisons of coagulation parameters between h0 and h24 were performed using the Mann–Whitney U test due to the abnormal distribution. Comparisons of baseline characteristics and coagulation parameters between favorable and unfavorable outcome groups were performed using Student t-test, Mann–Whitney U test, chi-square tests, or Fisher’s exact test appropriately. Logistic regression was used to evaluate the relationship between coagulation parameters and clinical outcomes, both early neurological deterioration and functional outcome at 3 months. Only factors significantly associated with the outcome at univariate comparison were included in the multivariate model with a stepwise procedure. All statistical analyses were 2-tailed, and a p-value < 0.05 was accepted as statistically significant.
Results
Study Population
A total of 267 patients were enrolled (188 men and 79 women) with a mean age of 60.88 ± 12.31 years. In total, 9 patients had early neurological deterioration within 24 h after thrombolysis, and 99 patients had unfavorable outcomes at the 3-month visit. The mean NIHSS score on admission was 6.91 ± 5.06 and the mean ONT was 189.59 ± 40.05 min. The mean systolic and diastolic blood pressure at admission was 156.33 ± 23.94 mm Hg and 91.42 ± 14.74 mm Hg, separately. There were 168 cases of anterior circulation infarction versus 99 cases in the posterior. According to CISS criteria, large-artery atherosclerosis was the leading etiology (n = 196, 73.4%), then came cardiogenic stroke (n = 53, 19.9%), penetrating artery disease (n = 14, 5.2%) and undetermined etiology (n = 4, 1.5%).
Coagulation Parameters
The value of PLT, MPV, PT, APTT, and Fbg changed significantly at h24 in comparison with h0 (p < 0.05, Table 1), and all these parameters increased except for PLT and Fbg. While no significant changes were observed in PDW and TT from h0 to h24.
Coagulation Parameters Measured at Baseline and 24 h After Thrombolysis.
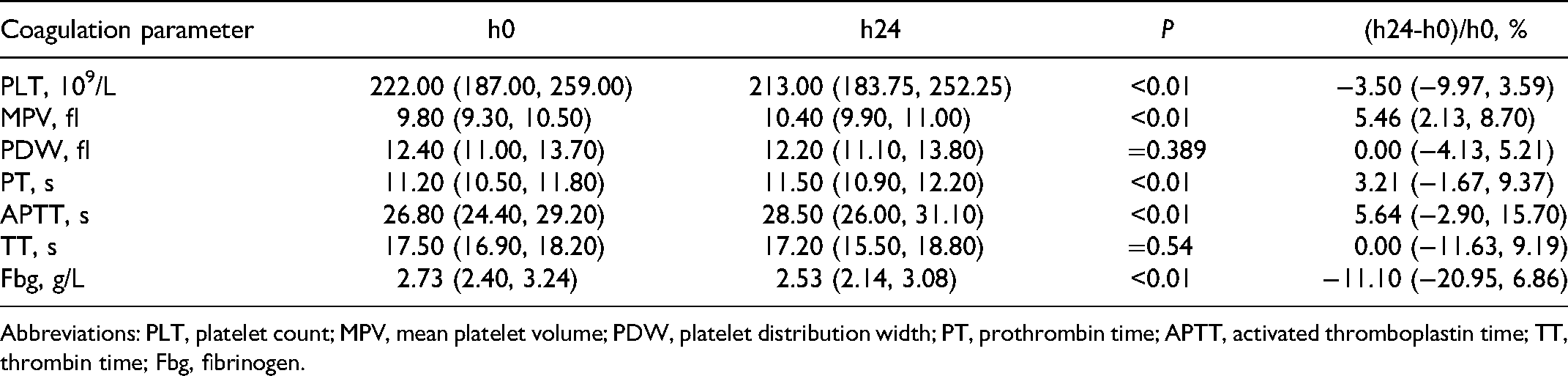
Abbreviations: PLT, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PT, prothrombin time; APTT, activated thromboplastin time; TT, thrombin time; Fbg, fibrinogen.
Correlations With Clinical Outcome
Table 2 summarizes the demographic characteristics and clinical features among the favorable and unfavorable outcome groups. There was no significant difference in age and sex between the 2 groups. Patients with favorable outcomes were more likely to have lower blood pressure, lower NIHSS score at admission, but a higher proportion of diabetes mellitus. No difference was found in BMI, ONT, the territory of infarction, etiology, smoking status, medical history of hypertension, stroke, coronary heart disease, or atrial fibrillation.
Baseline Characteristics of Participants According to the Functional Outcome.

Abbreviations: SBP, systolic blood pressure; DBP, diastolic blood pressure; BMI, body mass index; ONT, onset-to-needle time; NIHSS, National Institute of Health Stroke Scale; mRS, modified Rankin Scale.
The comparison between coagulation parameters or their changes with 3-month outcomes in univariate analyses were shown in Table 3. Patients with favorable outcome exhibited a significantly higher level of PLT at baseline as well as 24 h after thrombolysis and a lower level of PT at baseline. Besides, only the (h24-h0)/h0 of PT differed significantly among the 2 groups.
Coagulation Parameters of Participants According to the Functional Outcome.

Abbreviations: PLT, platelet count; MPV, mean platelet volume; PDW, platelet distribution width; PT, prothrombin time; APTT, activated thromboplastin time; TT, thrombin time; Fbg, fibrinogen; mRS, modified Rankin Scale.
Accordingly, we further investigated the relationship between the (h24-h0)/h0 of PT and clinical outcomes. No correlation was found between the (h24-h0)/h0 of PT and early neurological deterioration (data was not shown in the article). Whereas, the dynamic change of PT was positively associated with 3-month unfavorable functional outcome after adjusting for age, sex, and potential confounding factors including systolic blood pressure, NIHSS score at admission, and history of diabetes mellitus (odds ratio [OR]: 1.42, 95% confidence interval: 1.02-2.28, Table 4).
Odds Ratios of the PT Change for 3-Month Unfavorable Functional Outcome.

Abbreviations: PT, prothrombin time; CI, confidence interval.
Model 1: adjusted for age and sex.
Model 2: adjusted for age, sex, systolic blood pressure, National Institute of Health Stroke Scale score at admission, and medical history of diabetes mellitus.
Discussion
In this study, the change of PT after rt-PA thrombolysis was significantly related to the 3-month unfavorable functional outcome in stroke patients. Whereas, no evidence was observed between coagulation parameters and early neurological deterioration. Our study suggested that the dynamic change of PT might be a potential predictor for poor prognosis in stroke patients 3-month after thrombolysis, but not for the short-term outcome. It seems that our finding has no contradiction with the guideline recommendation that there is no need of waiting for routine coagulation results before IV rt-PA, as coagulation parameters are within the normal reference range in most cases, and the change of which has no correlation with short-term outcomes. Notably, dynamic monitoring of coagulation parameters after rt-PA thrombolysis could help to identify patients at higher risk of 3-month poor prognosis, thus allowing for early intervention.
PT, referring to the time it takes for clotting to occur after thromboplastin and calcium are added to platelet-deficient plasma, is used to reflect the efficiency of the extrinsic coagulation pathway initiated by tissue factor. Although known as a “fibrin specific” thrombolytic agent, rt-PA can produce a partial conversion of plasminogen to plasmin in the absence of fibrin. 15 The plasmin, then, destroys the integrity of the coagulation system since it is capable of hydrolyzing the coagulation factors which participated in the extrinsic coagulation pathway, including factors II, V, VII and X, leading to the prolongation of PT. The probability of hemorrhagic transformation increases with the decrease of coagulation factors in theory, while our study was unable to verify the above assumption owing to the limited number of bleeding events. Although the fluctuation of PT failed to reach the threshold of hemorrhagic transformation in the acute stage, it did increase the risk of 3-month poor prognosis among thrombolysis patients. Yet underlying mechanisms are not fully understood, we may get a hint from tenecteplase, the genetically mutated variant of rt-PA, which has a 10 to 14 times higher fibrin specificity and less impact on systematic coagulation. 15 There were several studies comparing tenecteplase and alteplase, the former thrombolytic agent was associated with higher rates of recanalization, fewer bleeding complications, and better 90-day functional outcome which ascribed to its pharmacologic properties.16–18
Although patients with favorable outcomes exhibited a significantly higher level of PLT at baseline and 24 h after thrombolysis, our study failed to show any correlation between the change of PLT and functional outcome, neither MPV nor PDW. Platelet plays an important role in cardiovascular diseases as large hyperactive platelets are generated under pathological conditions. Vizioli et al reported that the increase in MPV is associated with an elevated risk of thrombosis and also has a prognostic significance in ischemic stroke. 19 Therefore, platelet-related parameters may partially reflect platelet function, such as adhesion, aggregation, release, and contraction, while the activated processes after thrombolysis cannot be quantified. Considering the effect of rt-PA, molecular mechanisms involved remain to be further elucidated.
Previous studies indicated that the decrease of Fbg to a certain extent was a relevant cause of major bleeding complications,10,20,21 no similar result was obtained in our study. With an in-depth understanding of dynamic changes in the coagulation system after thrombolysis, we come to realize that the concentration of Fbg followed a U-shaped curve over time that bottomed at 6 h after IV thrombolysis. 20 While our study set the time point at 24 h, where the concentration of Fbg has fallen. The cost–benefit ratio should also be considered, as the measurement of coagulation parameters at multiple time points is not easy to implement in clinical practice.
There are some limitations. Our study selected the routine time-point for blood testing, times other than 0 h, and 24 h should be set to draw a more accurate curve which can better represent the prognosis of ischemic stroke. Moreover, there are markers of coagulation, anti-coagulation, and fibrinolysis more than those measured in our study which could provide a more comprehensive understanding between the coagulation system and prognosis. Besides, in a prospective cohort study, the risk ratio is a better index to demonstrate the association between exposure factors and clinical outcomes. After taking the small sample size within a single center into consideration, we chose OR rather than risk ratio in statistical analysis. Multicenter and large-scale researches are needed to provide insight into the relationship.
Conclusions
In conclusion, the prolongation of PT from baseline to 24 h after IV rt-PA increases the risk of 3-month unfavorable outcomes in stroke patients. Further large-scale, polycentric, and prospective studies with more testing points and comprehensive indicators are needed to verify our results.
Footnotes
Acknowledgments
The authors gratefully appreciate all of the participants and staff for their contributions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (2019-I2M-5-029), Beijing Natural Science Foundation (Z200016), and Beijing Municipal Committee of Science and Technology (Z201100005620010).
Ethical Approval
This study was conducted in accordance with the guidelines from the Helsinki Declaration and was approved by the Ethics Committees of Beijing Tiantan Hospital.
Informed Consent
This study was conducted without requiring individual patient informed consent under the common rule.
