Abstract
Hemostatic disturbances after cardiac surgery can lead to excessive postoperative bleeding. Thromboelastography (TEG) was employed to evaluate perioperative coagulative alterations in patients undergoing cardiac surgery with cardiopulmonary bypass (CPB), investigating the correlation between factors concomitant with cardiac surgery and modifications in coagulation. Coagulation index as determined by TEG correlated significantly with postoperative bleeding at 24-72 h after cardiac surgery (P < .001). Among patients with a normal preoperative coagulation index, those with postoperative hypocoagulability showed significantly lower nadir temperature (P = .003), larger infused fluid volume (P = .003), and longer CPB duration (P = .033) than those with normal coagulation index. Multivariate logistic regression showed that nadir intraoperative temperature was an independent predictor of postoperative hypocoagulability (adjusted OR: 0.772, 95% CI: 0.624-0.954, P = .017). Multivariate linear regression demonstrated linear associations of nadir intraoperative temperature (P = .017) and infused fluid volume (P = .005) with change in coagulation index as a result of cardiac surgery. Patients are susceptible to hypocoagulability after cardiac surgery, which can lead to increased postoperative bleeding. Ensuring appropriate temperature and fluid volume during cardiac surgery involving CPB may reduce risk of postoperative hypocoagulability and bleeding.
Keywords
Introduction
Cardiac surgeries are frequently performed worldwide, with varying degrees of complications arising from coagulation dysfunction. Coagulation dysfunction after cardiac surgery can lead to thrombosis, 1 postoperative bleeding,2–5 myocardial ischemia-reperfusion injury, renal failure, and acute lung injury.6,7 Massive postoperative bleeding, for example, affects 5%-25% of cardiac surgery patients, and approximately half of such cases are due to a hypocoagulable state, 8 in which blood clotting is compromised. Massive bleeding may necessitate blood transfusion or additional surgery. 9 Reducing the risk of bleeding and other complications after cardiac surgery requires careful perioperative monitoring of coagulation function. 10 In contrast to conventional coagulation tests, which assess only single steps in the entire coagulation process, thromboelastography (TEG) can simultaneously assess the entire coagulation pathway, detecting insufficient levels of coagulation factors, reduced activity of fibrinogen or platelets, and hyperactive fibrinolysis (Figure 1). TEG is now widely used for monitoring coagulation function as well as guiding blood transfusion11–18 and the use of anticoagulant or antiplatelet therapies.19,20
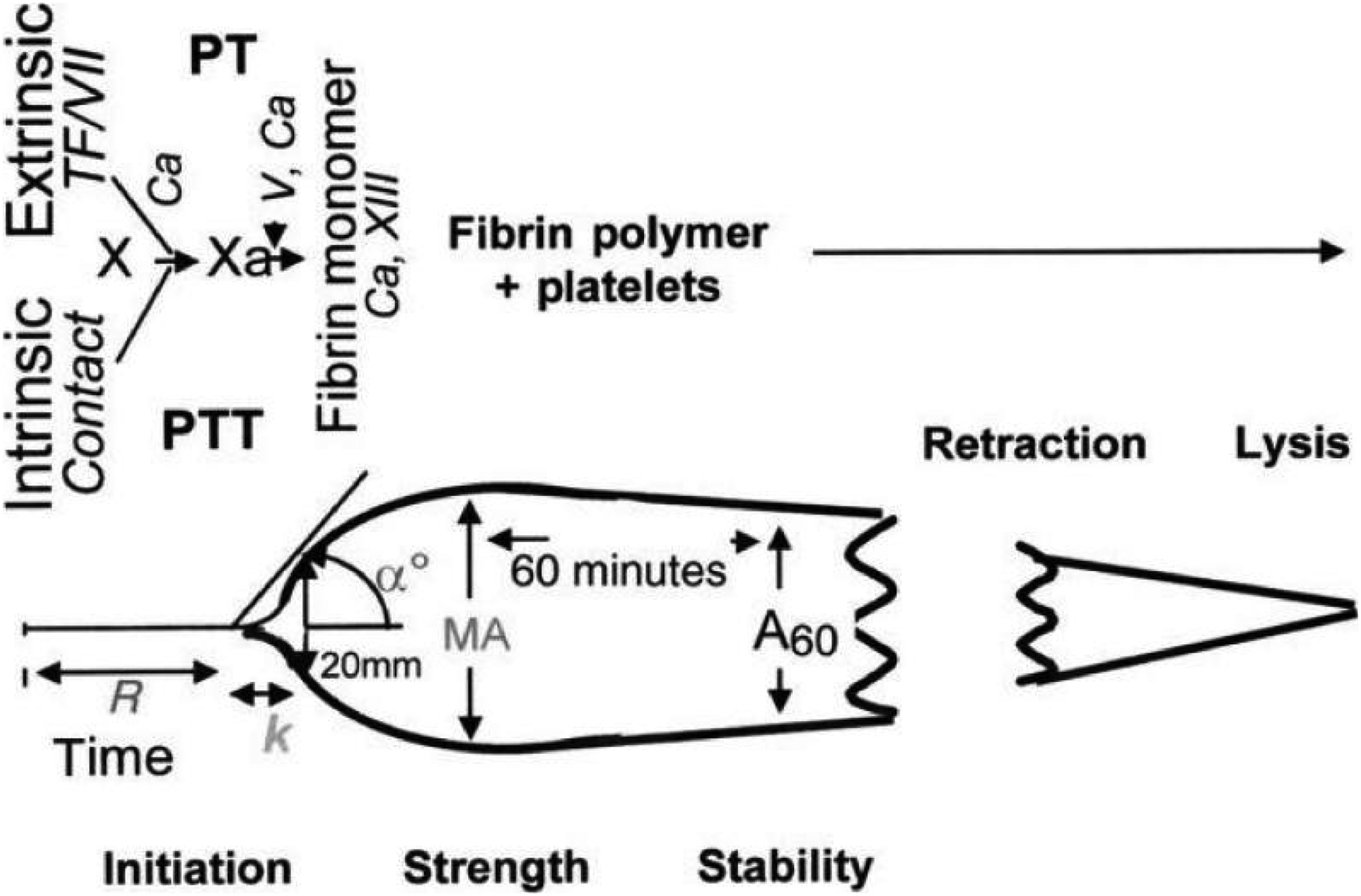
Diagram of TEG/ROTEM parameters.
Here we used TEG to assess pre- and postoperative coagulation function in patients at our medical center who underwent cardiac surgery involving cardiopulmonary bypass (CPB), and we explored potential associations between risk of hypocoagulability and clinicodemographic or perioperative characteristics in order to identify risk factors. To our knowledge, this is the first study to use TEG to analyze the occurrence of hypocoagulability after cardiac surgery and its risk factors.
Methods
This study retrospectively analyzed medical records of patients at least 18 years old who underwent elective cardiac surgery involving CPB at the Second Affiliated Hospital of Zhejiang University School of Medicine (Hangzhou, China) between March 1, 2021 and April 30, 2022. Patients were excluded if they did not undergo perioperative TEG, they had a primary blood disorder or a history of anticoagulant use, or their medical records were incomplete.
This study was approved by the Ethics Committee at the Second Affiliated Hospital (approval 20220814), which waived the requirement for informed consent because, at the time of surgery, patients or their legal guardians consented to the analysis and publication of anonymized medical data for research purposes.
Surgery
In all patients, arterial blood pressure, electrocardiography, cardiac rate, SpO2, and end-expiratory carbon dioxide concentration were monitored. After endotracheal intubation, anesthesia was induced using midazolam, sufentanil, vecuronium bromide, and etomidate, then maintained intraoperatively using sevoflurane inhalation combined with intravenous propofol anesthesia. Cis-atracurium and sufentanil were administrated intraoperatively to maintain muscle relaxation and provide analgesia. Additionally, norepinephrine, epinephrine, nitroglycerin, and other medications were used as necessary to maintain hemodynamic stability. At 10 min after anesthesia induction, blood gases were analyzed and preoperative TEG was performed (see “Assessment of coagulation function” below).
All patients received intravenous heparin at 3 mg/kg via intravenous injection. When the activated clotting time exceeded 480 s, CPB was initiated using a 1:2 (v/v) mixture of crystalloid solution and colloid solution and an S5 extracorporeal circulation machine (Stockert, München, Germany). During bypass, mean arterial pressure was maintained between 40 and 80 mm Hg, and hemoglobin concentration between 70 and 100 g/L. Patients received 40%-70% oxygen at 1.5-3.5 L/min depending on arterial blood gas levels in order to maintain PaO2 between 200 and 300 mm Hg and PaCO2 between 35 and 45 mm Hg.
After CPB, heparin was neutralized using protamine at a mass ratio of 1:1-1.5; at 5 min later, venous blood (4 ml) was collected to determine activated clotting time and arteriovenous blood gas levels. When the activated clotting time reached 70-130 s, a fresh blood sample was taken for postoperative TEG.
Assessment of Coagulation Function
TEG was performed using the TEG 5000 thromboelastometer (Haemoscope, Boston, MA, USA) according to the manufacturer's instructions. The following coagulation indicators were measured: reaction time, defined as latency from the start of the test to the beginning of fibrin formation; clotting time, defined as the time to achieve a clotting amplitude of 20 mm; α angle, which reflects the speed of clot formation; maximum amplitude, which reflects the strength or stability of the fibrin clot; LY30, defined as the percent decrease in amplitude due to fibrinolysis from the maximum amplitude until 30 min later; and coagulation index, defined as Coagulation index = 0.1227 (reaction time) + 0.0092 (clot time) + 0.1655 (maximum amplitude) − 0.0241 (α angle) − 5.0220.Coagulation index between −3.0 and +3.0 was taken to indicate normal coagulation function; coagulation index >+3.0, hypercoagulability; and coagulation index <−3.0,hypocoagulability.12,21,22
Statistical Analysis
Data were analyzed using SPSS 26.0 (IBM, Chicago, IL, USA) and a definition of statistical significance of P < .05. After checking for normal distribution using the Kolmogorov–Smirnov test, we reported continuous data as mean ± standard deviation if normally distributed, otherwise as median (interquartile range, IQR). We reported categorical data as frequency (percentage). Intergroup differences in continuous variables were assessed for significance using an independent-samples t test if the data were normally distributed, otherwise using the Wilcoxon signed-rank test. For example, the paired-sample Wilcoxon signed-rank test was used to compare pre- and postoperative coagulation index, and to compare perioperative variables between patients with normal coagulation function or hypocoagulability after surgery. Intergroup differences in categorical variables were assessed using the chi-squared test.
Based on previous studies of factors that may contribute to coagulation dysfunction after cardiac surgery involving CPB,23–25 we used univariate analysis to explore potential associations of numerous clinicodemographic and perioperative variables with risk of postoperative hypocoagulability. Potential associations between thromboelastographic parameters and total drainage volume at 12-72 h after surgery were explored using general linear correlation tests. Total drainage volume was defined as total volume from thoracostomic, mediastinal, and pericardial drains. 2 Linear relationships of clinicodemographic and perioperative variables with the difference between pre- and postoperative coagulation index were explored using univariate linear regression.
Factors potentially associated with postoperative hypocoagulability in univariate analysis (based on P < .1) were entered into multivariate logistic regression in order to correct for potential confounding by other clinicodemographic variables. Where appropriate, results were reported in terms of odds ratios (ORs) and associated 95% confidence intervals (CIs).
Results
Of the 368 potentially eligible patients, we enrolled 263 (152 males) with a median age of 62.00 years (IQR 52.25-68.00 years) and mean body mass index of 23.14 ± 3.57 kg/m2 (Figure 2, Table 1). Postoperative bleeding correlated negatively with postoperative coagulation index (Table 2). In other words, worse postoperative coagulation indices translated to greater postoperative bleeding. Across the entire sample, coagulation indices fell significantly during surgery (Table 3, Figure 3), with 43 patients (16%) developing postoperative hypocoagulability, 1 patient developing postoperative hypercoagulability, and the remaining 219 showing normal postoperative coagulation (Figure 4). A similar rate of postoperative hypocoagulability (33 patients, 15%; Figure 5) was observed among the 215 patients with a normal preoperative coagulation index.

Flow chart of patient enrollment. Abbreviation: TEG, thromboelastography.

Pre- and postoperative coagulation indices across all 263 patients.
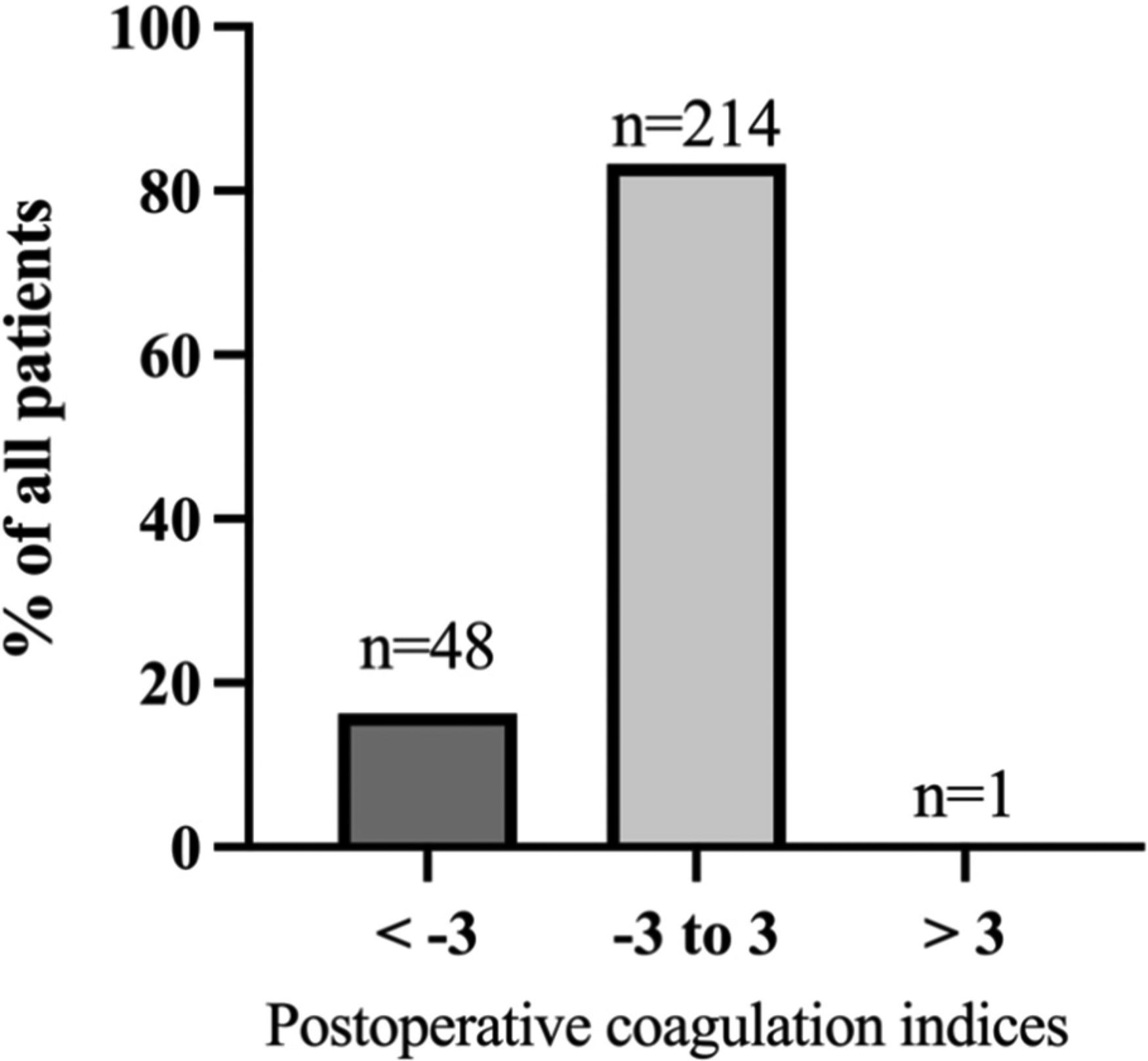
Distribution of postoperative coagulation index across all 263 patients.

Distribution of coagulation indices after cardiac surgery among 215 patients with normal coagulation indices before surgery.
Clinicodemographic Characteristics and Perioperative Variables for all 263 Patients in the Study.

Values are n (%), mean ± SD or median (interquartile range).
Correlations Between TEG Parameters and Chest Tube Drainage Volume at Different Times After Surgery.
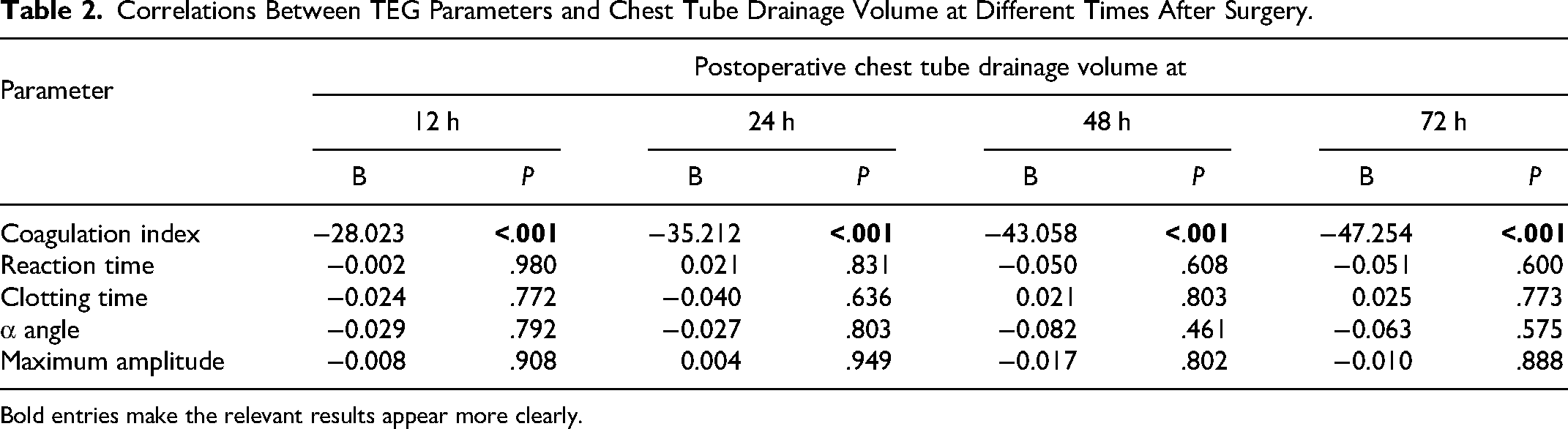
Bold entries make the relevant results appear more clearly.
Pre- and Postoperative Coagulation Indices.

Abbreviation: CI, confidence interval.
Bold entries make the relevant results appear more clearly.
When all patients were divided into a group with normal postoperative coagulation index and a group with postoperative hypocoagulability, those with hypocoagulability showed a significantly lower nadir intraoperative temperature, their CPB lasted significantly longer, and they were infused with a significantly larger fluid volume (Table 4). Multivariable logistic regression linked higher nadir intraoperative temperature with lower risk of postoperative hypocoagulability (adjusted OR 0.772, 95% CI 0.624-0.954, P = .017; Table 5).
Comparison of Perioperative Variables Between Patients who Showed Normal Coagulation or Hypocoagulability After Surgery.
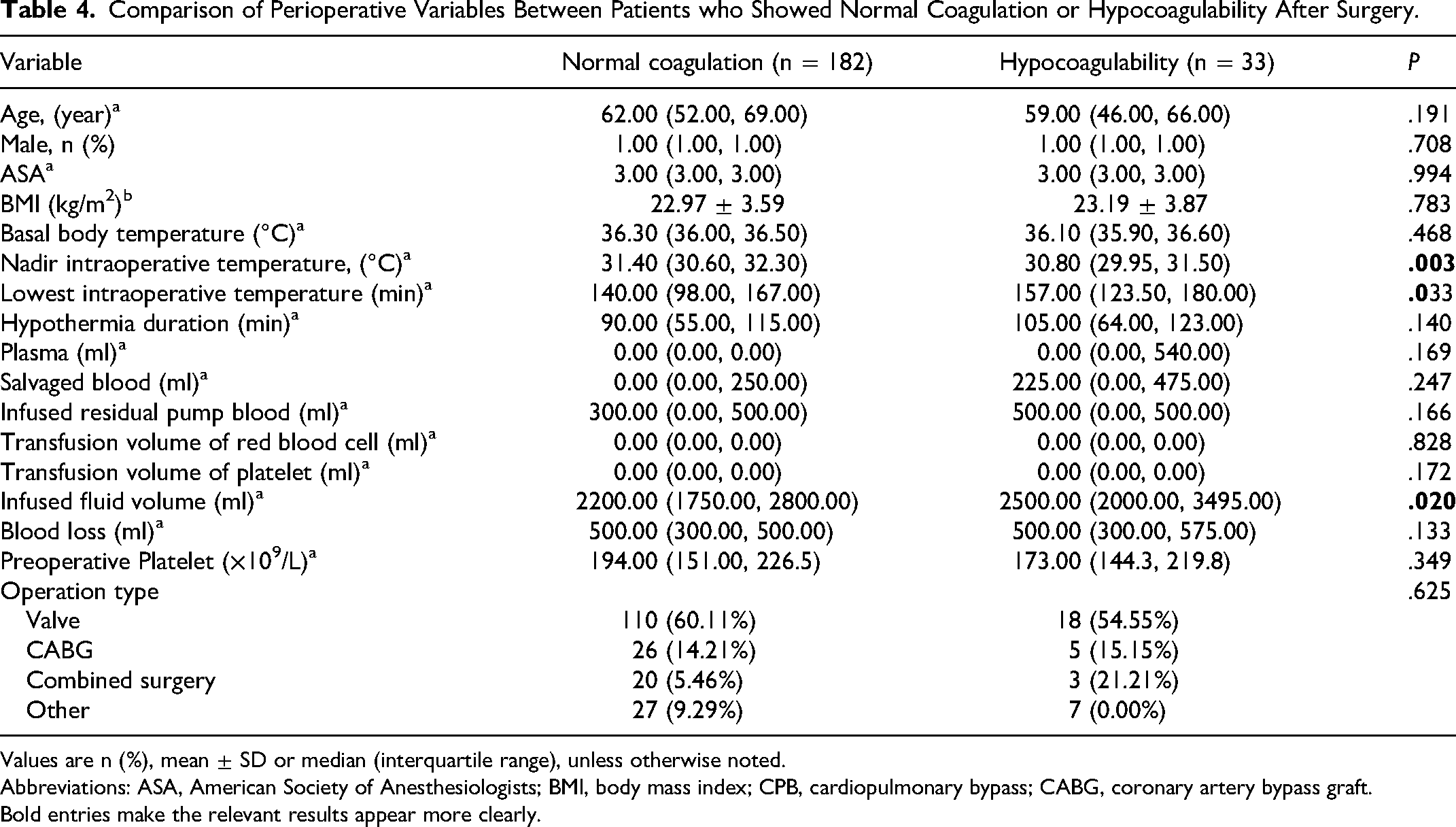
Values are n (%), mean ± SD or median (interquartile range), unless otherwise noted.
Abbreviations: ASA, American Society of Anesthesiologists; BMI, body mass index; CPB, cardiopulmonary bypass; CABG, coronary artery bypass graft.
Bold entries make the relevant results appear more clearly.
Logistic Regression to Identify Clinicodemographic or Perioperative Variables Associated with Postoperative Hypocoagulability.

Abbreviations: CI, confidence interval.
Bold entries make the relevant results appear more clearly.
Univariate linear regression of data from all 263 patients linked the following intraoperative variables to the magnitude of the difference between pre- and postoperative coagulation indices: nadir intraoperative temperature, duration of CPB, duration of hypothermia, blood loss as well as infused volumes of plasma, autologous blood, red blood cells, or fluid. After multivariable adjustment of potential confounders, the associations remained in the case of nadir intraoperative temperature and volume of infused fluid (Table 6). These results associate lower nadir temperature and greater volume of infused fluid with more severe postoperative hypocoagulability.
Logistic Regression to Identify Clinicodemographic or Perioperative Variables Associated with the Change in Coagulation index as a Result of Surgery.

Bold entries make the relevant results appear more clearly.
Discussion
Bleeding after cardiac surgery can lead to pericardial tamponade, infection, organ dysfunction, and even death.26,27 Here we identified nadir intraoperative temperature, infused fluid volume and duration of CPB as significantly associated with hypocoagulability after cardiac surgery involving CPB. Such hypocoagulability can lead to serious and even life-threatening postoperative bleeding. We identified these risk factors after using TEG to comprehensively assess coagulation function before and after surgery, 28 in contrast to conventional coagulation tests that examine only one step along the coagulation cascade. Therefore our findings are likely to be reliable.
Our data suggest that nadir intraoperative temperature, fluid transfusion volume and duration of CPB are associated with hypocoagulability after cardiac surgery. Since all three factors are modifiable, at least in principle, clinicians should pay special attention to them throughout the procedure and attempt to optimize them individually for each patient. Our analysis indicates a trend toward hypocoagulability among younger patients (as detailed in Table 4); however, this trend did not reach statistical significance. Additionally, when age was included in a multiple logistic regression model, it did not emerge as a significant factor associated with postoperative hypocoagulability.
We confirmed the validity of the thromboelastographic coagulation index for detecting hypocoagulability by demonstrating a negative correlation between the postoperative index and volume of postoperative bleeding. Similarly, another study found that TEG could predict postpartum hemorrhage. 21 Therefore, our findings not only validate the use of TEG but also underscore its usefulness in personalized patient care. Specifically, real-time monitoring using TEG allows for more accurate titration of blood products, potentially reducing the risk of bleeding-related complications. These findings could have far-reaching implications for perioperative management, particularly in optimizing coagulation support strategies, which could lead to improved patient outcomes.
Our analysis identified several additional factors that may influence risk of postoperative hypocoagulability and therefore deserve further study. In addition to the three major factors mentioned above, we also found that duration of hypothermia, plasma transfusion volume, red blood cell transfusion volume, salvaged blood transfused volume, and volume of blood lost varied linearly with the magnitude of the difference between pre- and postoperative coagulation indices. Future work should explore not only these variables but also several others that have been linked to risk of coagulation dysfunction after cardiac surgery, such as intraoperative medications and postoperative inflammatory responses.
Our findings should be interpreted with caution in light of the retrospective design, which may increase the risk of recruiting bias and prevents causal analyses; and the relatively small sample, which reduces our statistical power and prevents subgroup analysis to control for certain confounders. Our study was also limited by missing data, primarily due to the absence of TEG test or incomplete data, which may affect our findings. And we did not exclude patients with renal insufficiency, altered liver function, or insulin-dependent diabetes mellitus, which could potentially influence the study outcomes. 29 Such confounders include tranexamic acid and coagulation factor complexes that were routinely given to the study participants, reflecting standard procedures at our medical center. Our results should be verified and extended in larger, multi-site populations.
In addition to the risk factors identified in our study, it is important to note that hypothermia and dilutional coagulopathy are recognized risk factors for adverse outcomes in various clinical settings. The role of the multidisciplinary team, including cardiosurgeons, anesthesiologists, and perfusionists, is pivotal in managing these risk factors. Strategies such as active warming protocols, judicious fluid resuscitation, and real-time coagulation monitoring could mitigate these risks. Further research is warranted to explore how these strategies can be best implemented in the context of cardiac surgery.
Conclusion
Nadir intraoperative temperature, fluid transfusion volume and volume of postoperative blood loss may be associated with risk of hypocoagulability after cardiac surgery involving CPB. This work highlights the importance of careful perioperative monitoring of coagulation function to detect hypocoagulation as soon as possible, for which TEG appears to be effective.
Footnotes
Ethics Statement
This study was approved by the Institutional Review Board at the Second Affiliated Hospital of Zhejiang University School of Medicine (20220814). The requirement for informed consent was waived because of the anonymous nature and retrospective analysis of the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Commission of Zhejiang Province (grant number 2021435114).
