Abstract
Background:
Sonoclot is used to measure kaolin-based activated clotting time (kACT) for heparin management. Apart from measuring kACT, the device assesses the patient’s coagulation status by glass bead–activated tests (gbACTs; measuring also clot rate [CR] and platelet function [PF]). Recently, a new version of the Sonoclot has been released, and the redesign may result in performance changes. The aim of this study was to evaluate and compare the performance of the new (S2) and the previous (S1) Sonoclot.
Methods:
The S1 was used in the routine management of 30 patients undergoing elective cardiac surgery. Blood samples were taken at baseline (T1), after heparin administration (200 U/kg, 100 U/kg; T2 and T3), during cardiopulmonary bypass (T4), after protamine infusion (T5), and before intensive care unit transfer (T6). Kaolin-based activated clotting time and gbACTs were measured in duplicate by both the old and the new device and performance compared by Bland-Altman analysis and percentage error calculation.
Results:
A total of 300 kACT and 180 gbACTs were available. Bland-Altman analysis for kACT revealed that S2 consistently reported results in shorter time compared to S1 (overall = −14.7%). Comparing S2 and S1, the glass bead–activated tests showed mean percentage differences of −18.9% (gbACTs), +37.4% (CR), and −3.7% (PF).
Conclusion:
Since clotting is faster in the new S2 compared to S1, shorter clotting times have to be considered in clinical practice. The use of S2 kACT in heparin management will result in higher heparin and protamine dosing unless heparin kACT target values are adjusted to correct for the differences in results between S1 and S2.
Keywords
Introduction
The Sonoclot (Sonoclot Coagulation and Platelet Function Analyzer; Sienco Inc, Boulder, Colorado) is a viscoelastic point-of-care coagulation analyzer and provides information on hemostasis in a qualitative graph and as quantitative results including activated clotting time (ACT), clot rate (CR), and platelet function (PF). 1 Primarily, Sonoclot is being used to monitor the adequacy of heparin anticoagulation in the case of extracorporeal circulation. Furthermore, Sonoclot and other viscoelastic point-of-care coagulation devices play a pivotal role in perioperative hemostatic management at the bedside. 2 They have been shown to be predictive of postoperative bleeding after cardiopulmonary bypass (CPB) and cost-effective in perioperative care when combined with protocol-based transfusion strategies. 3 –5
Until now, the previous version of the Sonoclot (S1) was used mainly to measure kaolin-based activated clotting times (kACTs) as a monitor for heparin therapy, additionally allowing the assessment of the patients’ coagulation status by glass bead–activated tests (gbACTs; depicting also CR and PF). 1 Recently, a new version of the Sonoclot (S2) has been developed to perform different tests simultaneously. Although S2 shares the technical fundamentals with S1, the miniaturization of the device and the use of a metal cuvette holder instead of the plastic one in the previous S1 may affect all measurements (ACT, CR, and PF), resulting in a faster rewarming of blood samples to 37°C. Temperature differences alter the coagulation function, and therefore, any time delay incurred in rewarming the blood samples might affect the test results.
The aim of the present study was to evaluate the performance of the new S2 device by focusing on kaolin- and glass bead–activated measurements in 30 patients. The values were then compared to the previous S1 device so that the current heparin management target values may be adjusted for ensuring adequate heparinization with the new device.
Methods
Patients
With approval of the institutional research ethics board (Kantonale Ethikkommission Zurich, Switzerland; KEK Nr. StV 1-2007 SPUK Chirurgie), data of 30 consecutive patients undergoing cardiac surgery at the Triemli City Hospital, Zurich, Switzerland, were analyzed (ClinicalTrials.gov identifier: NCT02588976). In this prospective, observational clinical study, elective cardiac surgery patients requiring CPB with heparin anticoagulation were included. Emergency cardiac surgeries without the use of CPB or with CPB requiring anticoagulation other than heparin were excluded.
Perioperative Management
In all patients, perioperative routine management (ie, surgery, anesthesia, CPB, fluid and transfusion therapy) was performed according to standardized institutional protocols. Cardiopulmonary bypass was performed with a membrane oxygenator (Quadrox HMO1010; Maquet Cardiopulmonary AG, Hirlingen, Germany) under moderate hypothermia (28°C-32°C) with pump flow rates of 2.2 to 2.4 L/min/m2. The protocol of heparin management for CPB was guided by the Sonoclot analyzer (S1) using kACT test; anticoagulation was achieved with heparin 200 U/kg + 100 U/kg IV heparin (Liquemin; Drossapharm AG, Basel, Switzerland), according to Bull et al in order to maintain the kACT above 480 seconds. 6 Additional 10 000 U of heparin were added to the priming volume of the circuit. At the end of the procedure, heparin was reversed by giving protamine (Protamine; MEDA Pharmaceuticals GmbH, Wangen-Brüttisellen, Switzerland) up to a maximum dose of 1 mg/100 U of the total heparin dose. In this study, all patients received the same batches of heparin and protamine. As part of our institutional coagulation management algorithm to direct hemostatic therapy, Sonoclot’s glass bead–activated test was additionally being measured in the perioperative setting.
Sonoclot Coagulation and Platelet Function Analyzer
Detailed aspects of the Sonoclot technology have been described previously. 1 The new version of the Sonoclot (S2, model SCP1-4) was developed to convert the previous version of the single-channel Sonoclot (S1, model DP-2951) into instruments with 1, 2, or 4 output channels in order to perform different tests simultaneously. Additionally, the new S2 has several technical changes in mechanical design, electronics, and software. The S2 is considerably smaller than the S1, and a major modification in the S2 is that it is equipped with a metal cuvette holder instead of the plastic one in the S1. This helps in improved heat transfer to the blood sample, thereby resulting in a faster rewarming of blood samples to 37°C. At the beginning of the test, the temperature of blood samples may be reduced by 5°C or more due to the handling of collection syringes and dispensing cannulas. This temperature change can impair the existing coagulation function and, therefore, any time delay incurred in rewarming the blood samples can affect the test results. The faster the sample rewarms, the interference of the temperature variance in the results is reduced. The disposables, reagents, sample and reagent volumes used and the timing are identical for the old and new device. Their parts and the part numbers are the same. The reagents include separate healthy reference range values for old and new instruments. Furthermore, the manufacturer instructs the users to compare their current heparin management instrumentation and the S2 to establish control values that match the institution’s current heparin management target values for ensuring adequate heparinization. The viscoelastic output and the clot signal of both the S1 and S2 are calibrated to the same viscosity standards, providing the same viscoelastic measurement of the developing clot.
Measurement Time Points
Blood samples were taken at induction of anesthesia (T1, baseline), before initiation of CPB 3 minutes after initial heparin administration of 200 U/kg (T2), 3 minutes after the second dose of heparin 100 U/kg (T3), 20 minutes during CPB (T4), at the end of the procedure after protamine infusion (T5), and before intensive care unit (ICU) transfer (T6). All Sonoclot measurements were performed in duplicates using S1 and S2 at the same time: kACT was measured at T1 through T5; glass-bead tests were done at baseline (T1) and after heparin reversal (T5 and T6) using both the old and the new device. Apart from these measurements, further kACTs were performed with the Sonoclot S1 if clinically indicated.
Statistical Analyses
Statistical analysis was performed using IBM SPSS Statistics 2.0, release 20.0.0 (IBM Corporation Armonk, New York) and Sigmaplot 12.0 (Systat Software Inc.; San Jose, California).
Power analysis was performed to yield a potentially significant difference between measurements performed by S1 and S2: based on the extensive experience using S1, a large variation in the normally distributed values of ACT measurements before, during, and after CPB can be expected. For ACT, a mean difference of 20 ± 60 seconds, an α-error level of .05, and a sample size of 30 patients were needed to achieve an adequate test power of >80%.
Student t test was applied, and Bland-Altman analysis was performed in order to compare S1 and S2 measurements. The difference in measurements between the 2 devices was plotted against the average of the 2 measurements. The mean difference is the bias, whereas limits of agreement are obtained by subtracting and adding twice the standard deviation (SD) of the mean difference. The lower and the upper limit designate a prediction interval in which in 95% the difference between the 2 devices will lie assuming constant bias and variability. Percentage deviation (ie, percentage error) reflects limits of agreement in relation to the average measurements of the 2 devices. A P value ≤ .05 was considered to be statistically significant. Unless otherwise indicated, data are given as mean value ± SD.
Results
Sociodemographic and procedure-related data of all 30 patients undergoing elective cardiac surgery are summarized in Table 1. Three hundred duplicate kACT and 180 glass bead–activated measurements were performed using S1 and S2, thus 150 and 90 matched data sets were obtained for statistical analysis.
Sociodemographic and Procedure-Related Data.
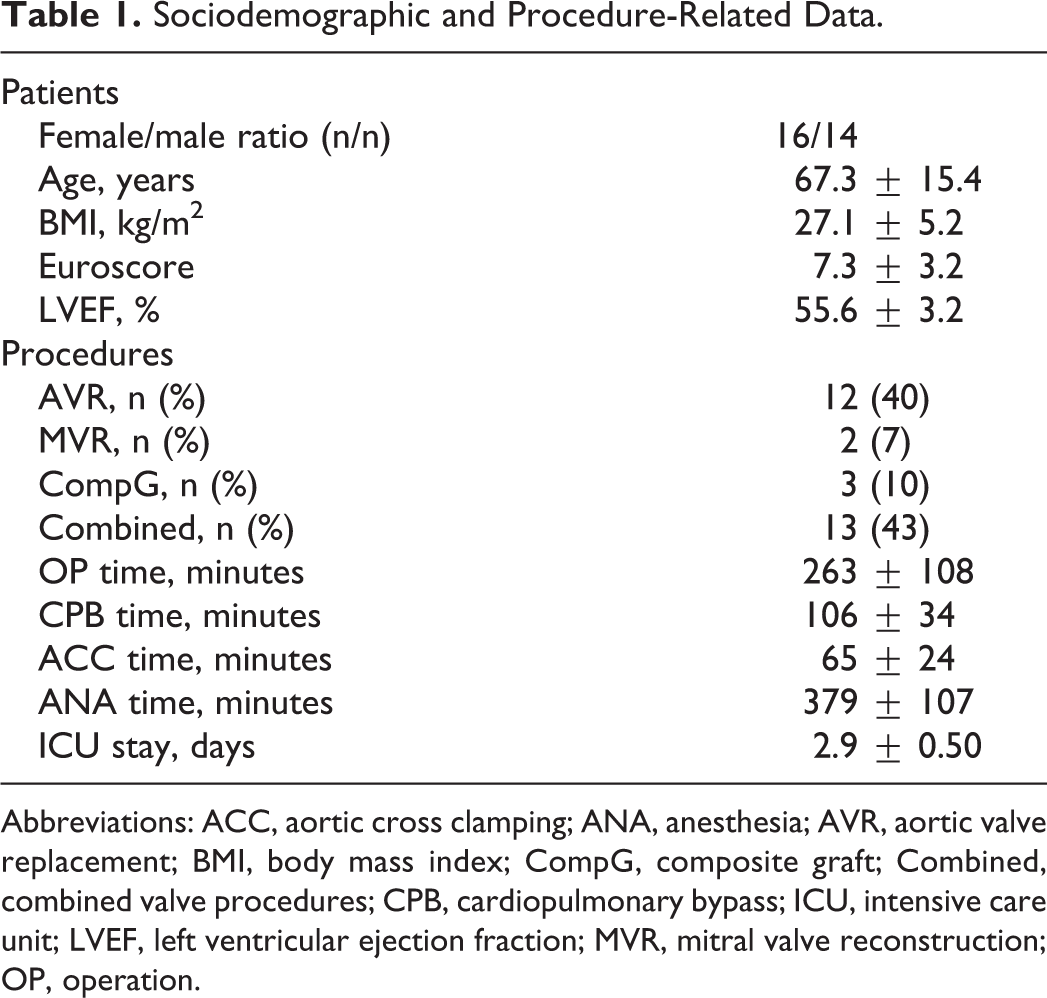
Abbreviations: ACC, aortic cross clamping; ANA, anesthesia; AVR, aortic valve replacement; BMI, body mass index; CompG, composite graft; Combined, combined valve procedures; CPB, cardiopulmonary bypass; ICU, intensive care unit; LVEF, left ventricular ejection fraction; MVR, mitral valve reconstruction; OP, operation.
Kaolin-Activated Measurements
The kACTs ranged from 60 to 950 seconds for S1 and from 60 to 701 seconds for S2, with a significant difference at each measurement point (T1-5, P < .001; Table 2). Test variability for S1 and S2 was within 6.1% to 10.6% and 3.7% to 6.3%, respectively (P = .026; Table 2). Bland-Altman analysis revealed a consistent underestimation of kACT measured by S2 as compared to S1 with a range of −14 to −104 seconds (Table 2, Figure 1). Percentage deviation (ie, percentage error) values between −10.5% and −18.5% were observed (Figure 3).
Comparison of kACT Measurements by Sonoclot S1 and S2.a
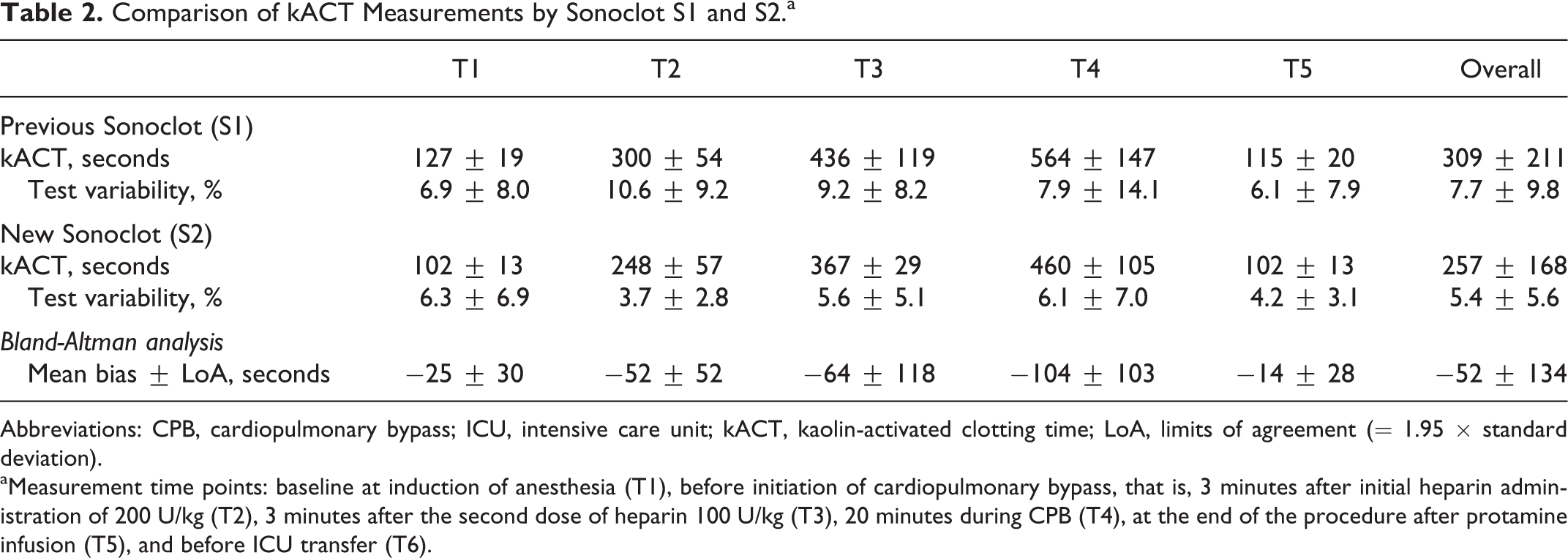
Abbreviations: CPB, cardiopulmonary bypass; ICU, intensive care unit; kACT, kaolin-activated clotting time; LoA, limits of agreement (= 1.95 × standard deviation).
aMeasurement time points: baseline at induction of anesthesia (T1), before initiation of cardiopulmonary bypass, that is, 3 minutes after initial heparin administration of 200 U/kg (T2), 3 minutes after the second dose of heparin 100 U/kg (T3), 20 minutes during CPB (T4), at the end of the procedure after protamine infusion (T5), and before ICU transfer (T6).
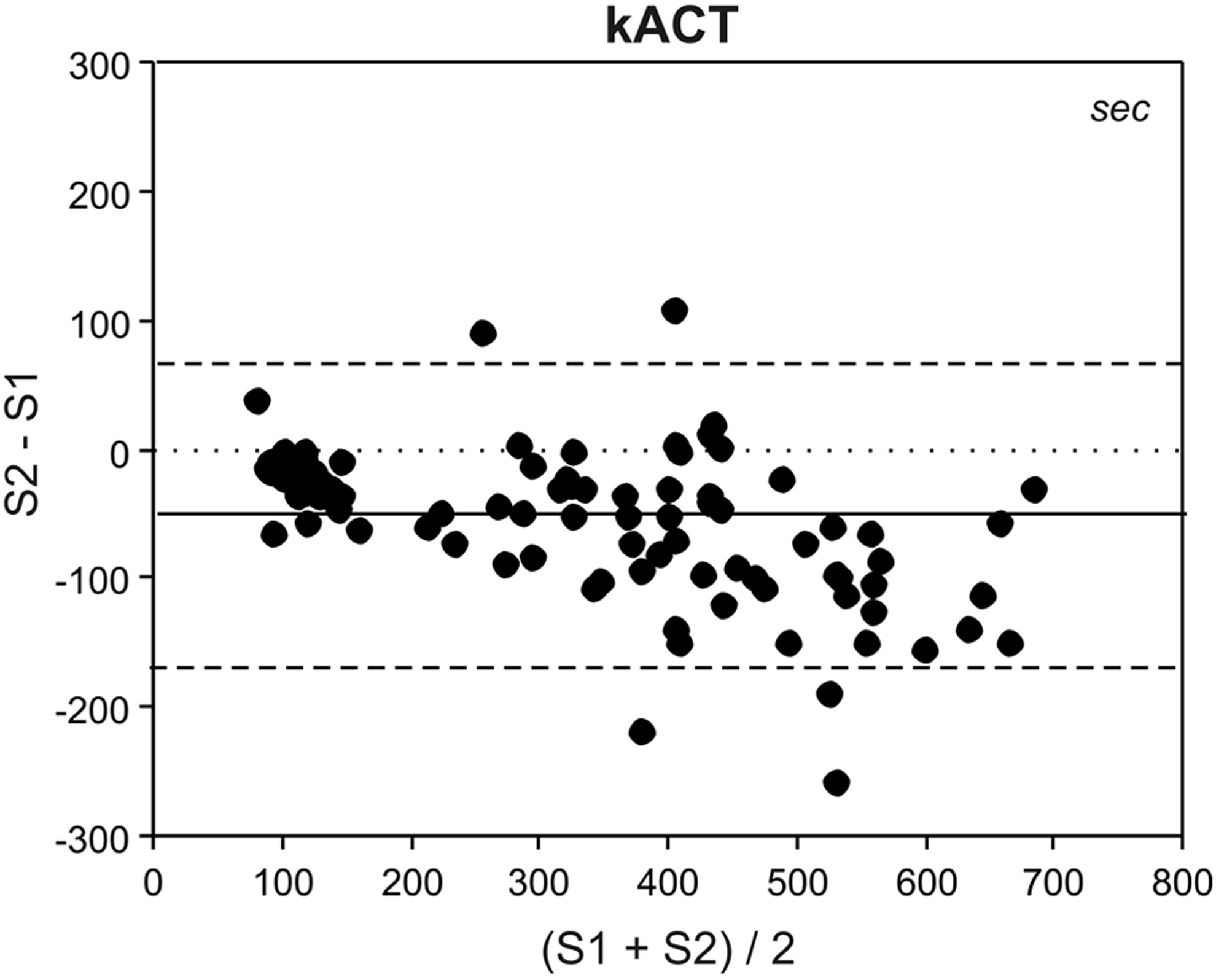
Bland-Altman analysis for kACT measurements by the previous (S1) and new (S2) Sonoclot Analyzer. kACT indicates kaolin-activated clotting time; solid line, mean bias; dashed line, limits of agreement (= 1.95 × standard deviation).
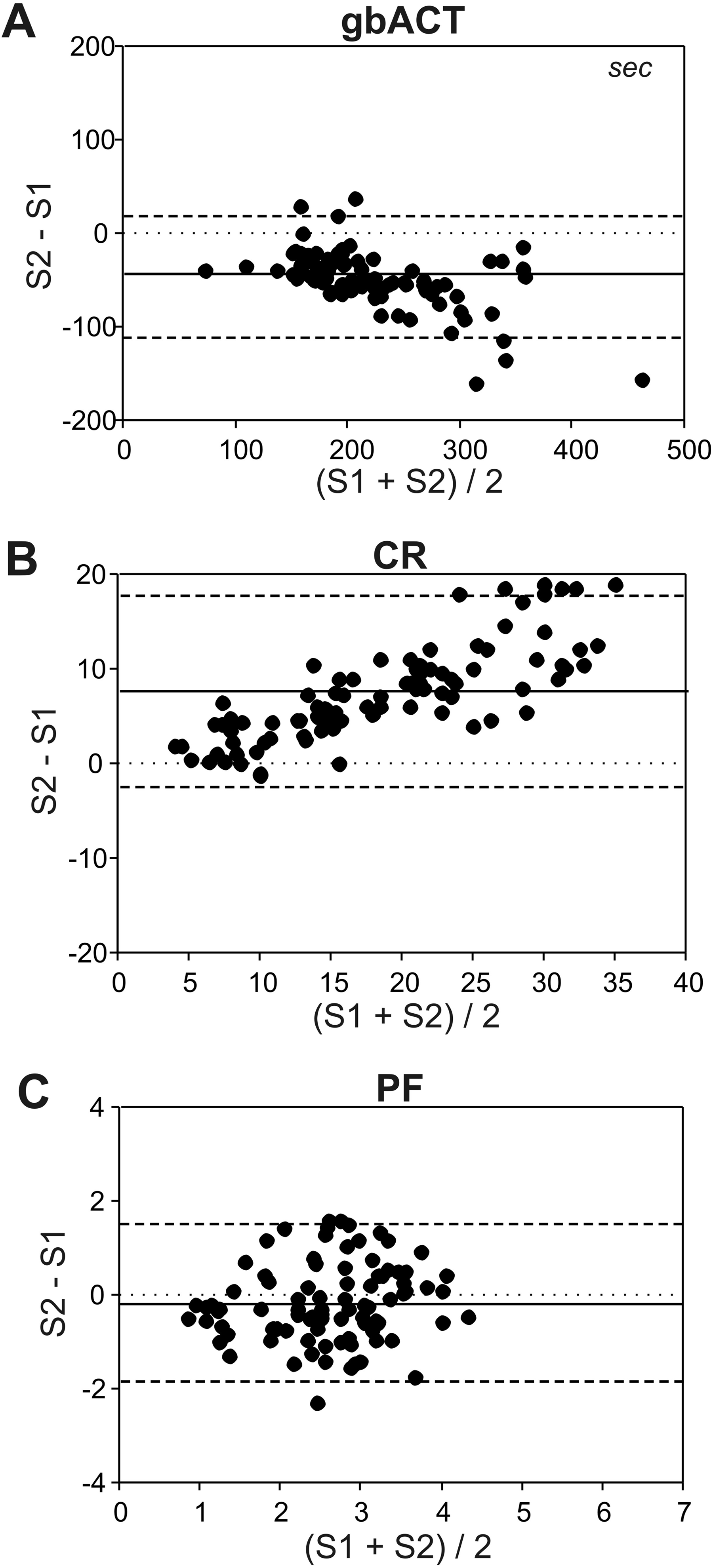
Bland-Altman analysis for glass bead–activated measurements by the previous (S1) and new (S2) Sonoclot Analyzer. gb indicates glass-bead activation; ACT, activated clotting time; CR, clot rate; PF, platelet function; solid line, mean bias; dashed line, limits of agreement (= 1.95 × standard deviation).
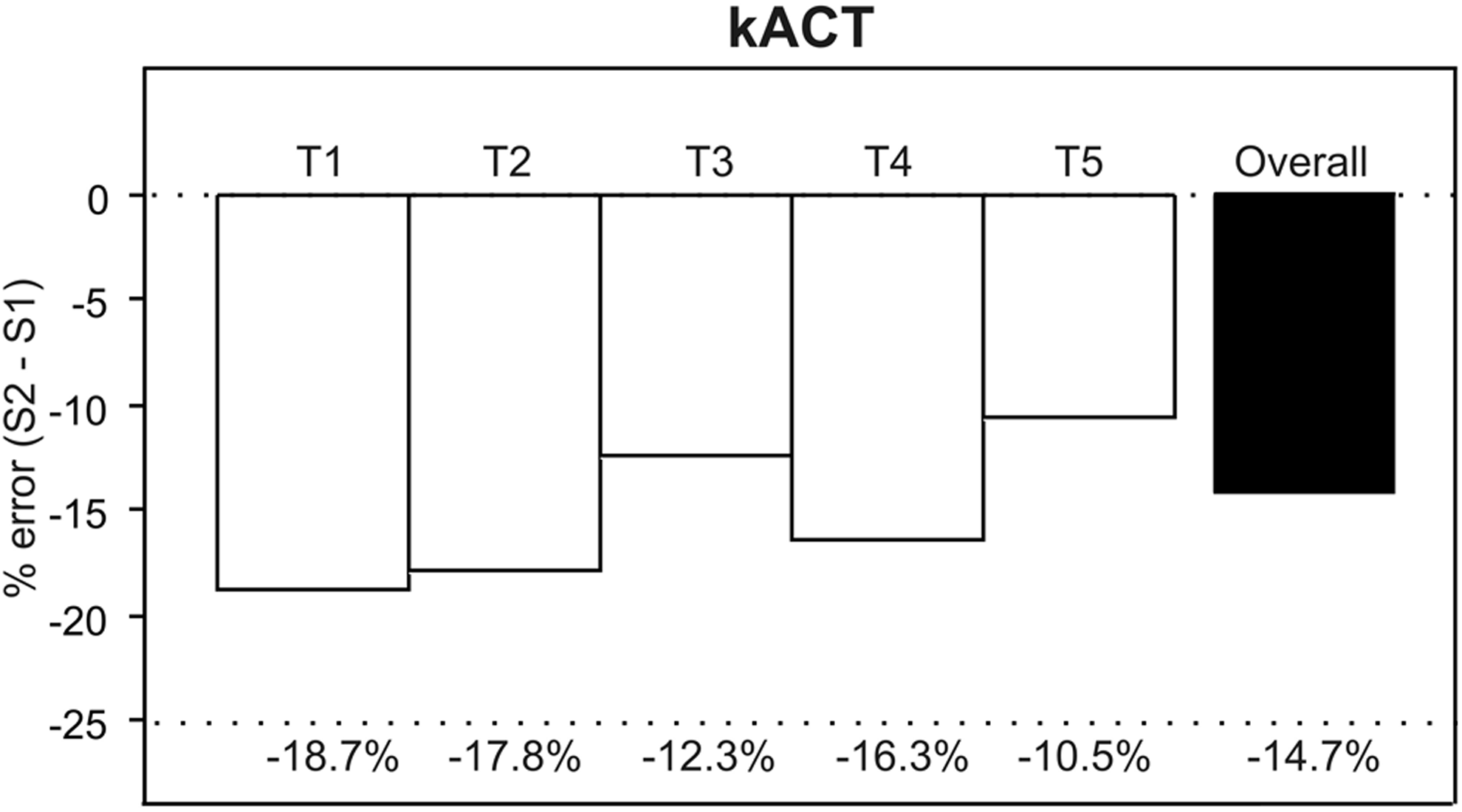
Percentage error (% error) for kACT of the new Sonoclot Analyzer (S2) compared to the previous Sonoclot Analyzer (S1). kACT indicates kaolin-activated clotting time.
Glass Bead–Activated Measurements
For gbACTs, a range of 93 to 540 seconds were observed for S1 and 54 to 384 seconds for S2. There was a significant difference between S1 and S2 at all measurement points (T1, T5, and T6, P < .001; Table 3). Clot rates were between 3.2 and 27.5 Units (S1) and between 4.9 and 44.5 Units (S2, P < .001; Table 3). Platelet function ranged from 1 to 4.5 and from 0.6 to 4.3 for S1 and S2, respectively (P < .05; Table 3). A low test variability could be demonstrated for gbACTs and CR which are assessed by both S1 and S2, whereas the variability of PF was higher (Table 3). There was no significant difference regarding the test variability of S1 and S2 (ACT: P = .113, CR: P = .780, PF: P = .831). Compared to S1, the S2 demonstrated gbACT underestimation and CR overestimation, which was observed using the Bland-Altman analysis (Figure 2). The overall effects on PF were small, but there was a tendency for overestimation at T1 and underestimation at T2 and T3. The corresponding percentage error values are presented in Figure 4.
Comparison of Glass Bead–Activated Tests by Sonoclot S1 and S2.
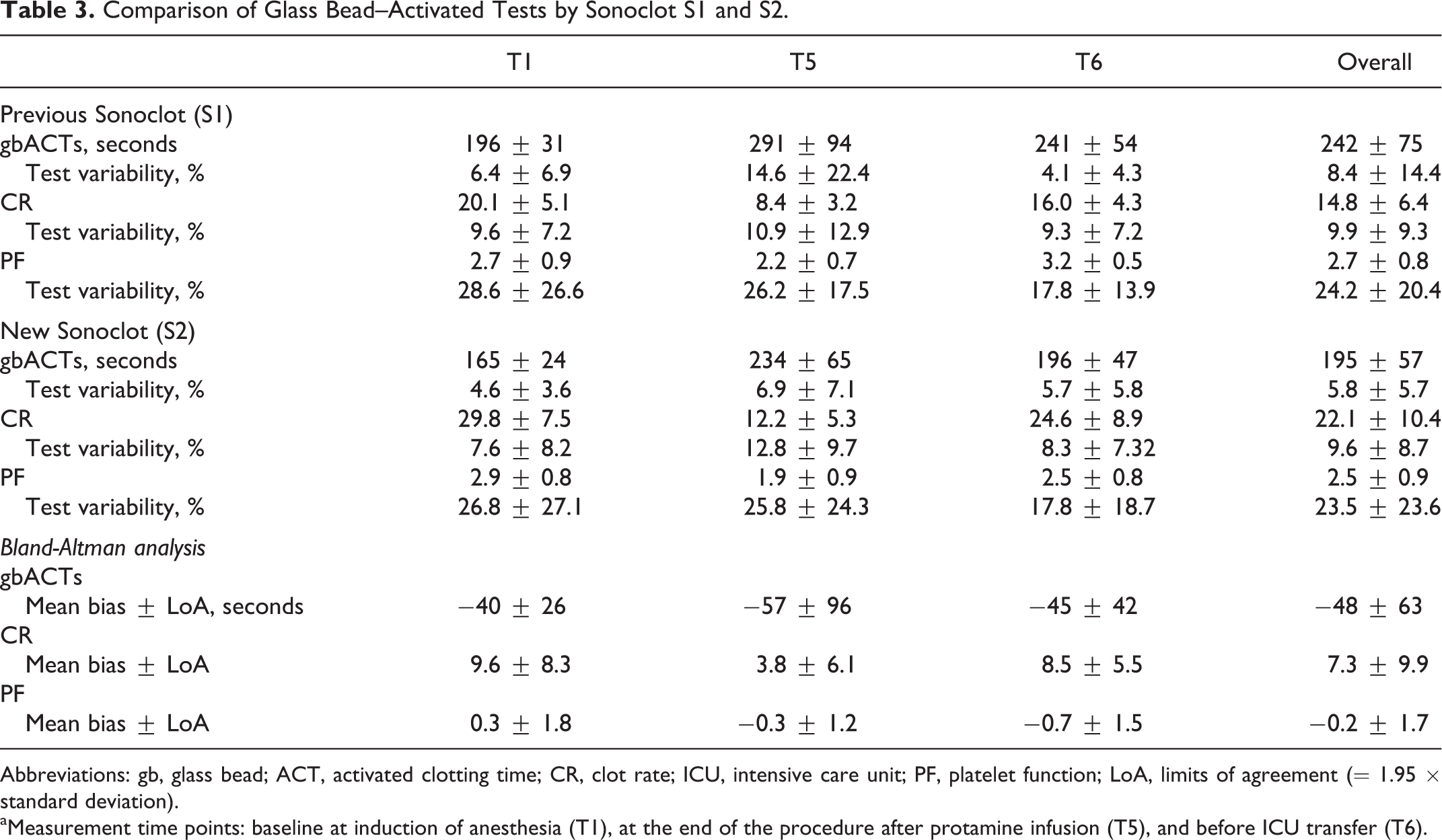
Abbreviations: gb, glass bead; ACT, activated clotting time; CR, clot rate; ICU, intensive care unit; PF, platelet function; LoA, limits of agreement (= 1.95 × standard deviation).
aMeasurement time points: baseline at induction of anesthesia (T1), at the end of the procedure after protamine infusion (T5), and before ICU transfer (T6).
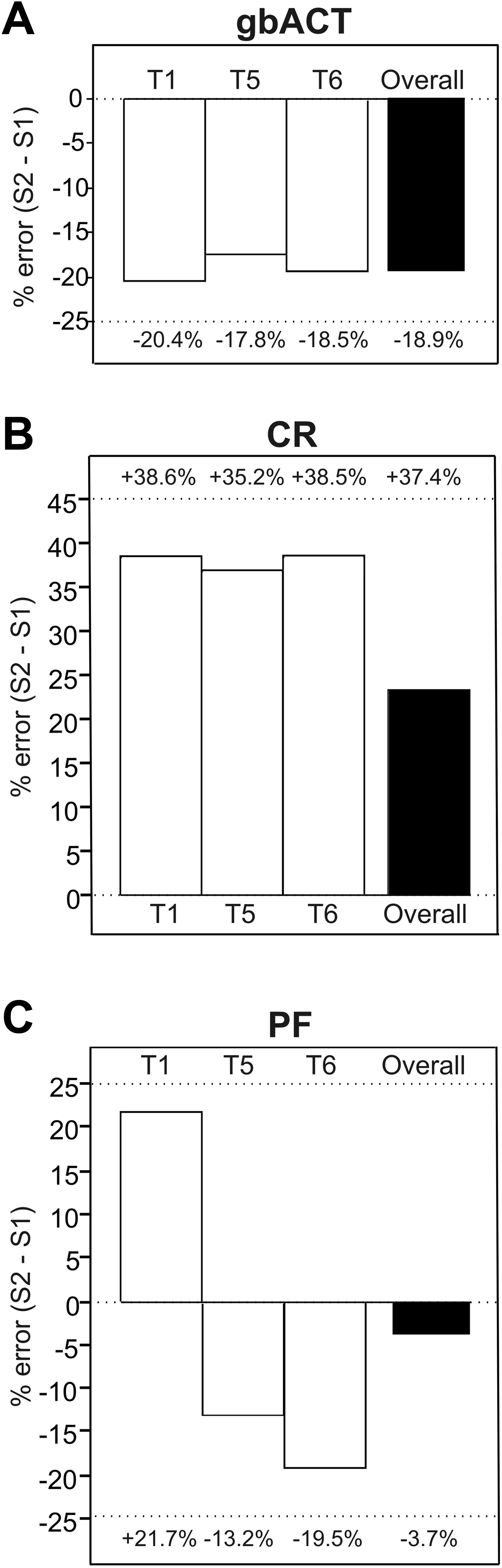
Percentage error (% error) for glass bead–activated tests of the new Sonoclot Analyzer (S2) compared to the previous Sonoclot Analyzer (S1). gb indicates glass bead; ACT, activated clotting time; CR, clot rate; PF, platelet function.
Discussion
The new Sonoclot device, S2, can rewarm the blood specimens in the test cuvette in a faster manner. Thereby, kACT values measured using S2 were lower on average (−15%) compared to that using S1, underestimating kACT by −14 to −104 seconds. This faster rewarming of blood samples using S2 showed lower gbACTs (−19%) and higher CR (+37%) values. In addition, values measured by the new S2 were more reliable as expressed by a lower test variability.
A variety of mechanisms lead to overt thrombin formation during CPB, which can activate and consume critical hemostatic components including platelets, fibrinogen, and other coagulation factors. 7 Paradoxically, inadequate anticoagulation during CPB can lead to a profound coagulopathy, likely due to disseminated intravascular coagulation, upon its termination. 8 Additionally, it has been shown that the perioperative coagulation status has a direct impact on postoperative blood loss and that coagulopathy is the main driver for excessive perioperative bleeding and blood product use. 9 Consequently, the warranty of adequate anticoagulation during CPB and targeted procoagulatory therapy thereafter is one of the basic efforts to improve perioperative outcome.
Historically, in 1957, the Mayo Clinic described prevention of the coagulation of blood for the Gibbon-type pump oxygenator, prescribing 3 mg of heparin (300 U) per kilogram of patient body weight. 10 Monitoring heparinization by the ACT of whole blood was first reported by Hattersley in 1966. 11 Bull et al then popularized the concept of the safe zone for anticoagulation during CPB in the mid-1970s, defining this as an ACT between 300 and 600 seconds. 12 In 1975, von Kaulla et al described the Sonoclot Analyzer, a device that measures the changing impedance to movement imposed by the developing clot on a small probe vibrating at an ultrasonic frequency in coagulating blood sample. 13 Based on this evolution, the practical point-of-care testing was imported into the operating department for monitoring anticoagulation.
The kACT measurement is a clinical standard for heparin management during CPB. 8,14,15 In our investigations, kACT measurements showed a significant difference between the 2 Sonoclot generations ranging from 60 to 950 seconds for the previous S1 and from 60 to 701 seconds for the new S2. Transferred to clinical practice, patients managed with kACT using S2 would receive higher heparin and protamine dosing, unless reference values were specifically adjusted for the new Sonoclot device.
Apart from heparin management, Sonoclot is being used to assess further aspects of the patient’s coagulation status such as platelet function. For example, the device has shown to reliably detect pharmacological glycoprotein IIb/IIIa receptor inhibition. 16 However, to obtain accurate results, cuvettes containing glass beads for specific coagulation and platelet activation should be used. 1 In our studies, gbACTs showed the same tendency like kACT measurements; using S1, the gbACTs ranged from 93 to 540 seconds and from 54 to 384 seconds for S2, and the CR values were between 3.2 and 27.5 Units (S1) and 4.9 and 44.5 Units (S2). For PF, this consistency could not be shown, and PF by S2 tended to overestimate S1 at the initial measurement time point but underestimated S1 at the postoperative time points.
Recently, the clinical relevance has been shown that Sonoclot might predict postoperative bleeding in patients undergoing cardiac surgery. 4 Especially glass bead measurements by Sonoclot were predictive after heparin reversal and before chest closure, which is comparable to the measurement point T5 in our study. Transferred to postoperative management in the ICU, shorter ACT and faster CR might complicate the identification of a potential postoperative bleeding tendency. Therefore, reference values for ACT, CR, and PF for daily clinical use in treatment algorithms have to be adapted for the new S2 machine.
Focusing on the technical and procedural realities, former studies described different responses of ACT under similar conditions using the same type of activator but manufactured by different companies. 17 In the present study, we used the same coagulation activator in 2 different generations of the same device. By taking the blood samples simultaneously from the same patient, classical bias such as hypothermia, inadequate specimen warming, hemodilution, platelet abnormalities, or aprotinin infusion could be reduced or completely avoided. 18 –20 Regarding the reengineered and minimized design of the S2, the question about the technical differences between S1 and S2 remains and may lead to the explanation of the aforementioned results. Beside the miniaturization of the S2, another main innovation is in replacing the plastic cuvette holder in S1 with a metal one. This change results in a faster rewarming of blood samples to 37°C and may thereby explain the faster clotting in S2. With the improved and faster temperature regulation in the new S2 instrument, the sample temperature variance as a component of test result error will significantly be reduced.
In summary, during CPB profound anticoagulation is required in order to both prevent thrombus formation within the circuit and patient and avoid the depletion of hemostatic factors by overt and uncontrolled coagulation activation. In the present study, the new Sonoclot S2 consistently reported faster kACT and gbACT results compared to the previous Sonoclot S1. These differences have clinical implications, for example, the use of S2 kACT for heparin management will result in higher heparin and protamine dosing unless kACT target values are adjusted to correct for the differences in results between S1 and S2. Further studies are needed to assess the clinical significance and outcome in patients managed by the new Sonoclot S2 machine.
Footnotes
Authors’ Note
O.D. and M.T.G. contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Study number: NCT02588976. Material support was provided by Sienco Inc, Boulder, Colorado, USA.
