Abstract
Vascular endothelial damage may activate hypercoagulation and contribute to the development of acute kidney injury (AKI). This study aimed to investigate whether early alteration in coagulation was associated with AKI onset following surgeries involving cardiopulmonary bypass (CPB) in children. This single-center retrospective cohort study included 154 infants and toddlers who underwent cardiovascular surgery with CPB. At admission to the pediatric intensive care unit, the absolute thrombin–antithrombin complex (TAT) level in each patient was measured. Moreover, the presence or absence of AKI onset in the early postoperative period was observed. Of the total participants, 55 (35%) developed AKI. A comparison within the toddler group based on the TAT cut-off value showed that both univariate and multivariable associations were found between increased absolute TAT level and AKI onset (odds ratio, 4.70; 95% confidence interval [1.20-17.90]; P = .023). The increase in absolute TAT level in toddlers during the early postoperative period following CPB was associated with AKI onset. However, a further prospective multicenter study with a larger sample size is required for validating these findings.
Background
Acute kidney injury (AKI) is a common complication following cardiopulmonary bypass (CPB) during cardiovascular surgery. The incidence of AKI is high in both adults and children and occurs in approximately 15% to 30% of cases.1,2 The onset of AKI poses a risk of poor prognosis in patients, such as increased mortality and prolonged hospitalization,3,4 highlighting the need for further efforts to predict and prevent AKI.
Cardiopulmonary bypass-induced AKI has several causes, including hypothermia, hemodilution, hemolysis, micro-thrombosis, decreased renal blood flow due to ischemic reperfusion, and renal tubular injury. 5 Recent researches suggest that capillary endothelial damage is also involved in the development of AKI.6,7 When vascular endothelial damage occurs, the coagulation cascade is activated, resulting in a state of hypercoagulation. In patients with sepsis, cytokine-induced vascular endothelial damage leads to antifibrinolytic disseminated intravascular coagulation, which has been linked to AKI onset. 8
Although CPB also causes shear stress and vascular endothelial damage, no studies have evaluated the relationship between post-CPB coagulopathy and the onset of AKI. This study aimed to investigate whether measuring biomarkers, such as thrombin–antithrombin complex (TAT) and plasmin–α2-plasmin inhibitor complex (PIC), of early postoperative change in hypercoagulation was associated with the onset of AKI following CPB in children. The findings of this study may provide useful information for earlier interventions.
Methods
Study Design and Setting
This single-center retrospective cohort study was conducted in a pediatric specialty hospital with a 10-bed pediatric intensive care unit (PICU). The study included patients admitted to the PICU following cardiovascular surgery with CPB between August 1, 2020 and December 31, 2021.
The research protocol was approved by the Clinical Ethics Committee of the Kanagawa Children's Medical Center and conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was waived due to the retrospective nature of the study and in compliance with the Personal Information Protection Law and National Research Ethics Guideline in Japan.
Participants
The study included patients who underwent cardiovascular surgery with CPB. The exclusion criteria were newborns and school children aged 73 months or older, patients with unplanned surgery, and those without necessary data for AKI diagnosis.
The following patient data were collected from electronic medical records: age in months, sex, and weight (kg). Data on primary diseases were also collected. These primary diseased comprised: hypoplastic left heart syndrome, atrioventricular septal defect, tricuspid atresia, ventricular septal defect, transposition of the great arteries, and double outlet right ventricle. They also included tetralogy of Fallot, truncus arteriosus, pulmonary atresia, coarctation/interruption of the aorta, total anomalous pulmonary venous connection, and others. Data on medical history (inborn genetic disease and chronic kidney failure) and preoperative severity (evaluated by pediatric logistic organ dysfunction-2 score and pediatric sequential organ failure assessment). Data on surgical procedures (classified by risk adjustment in congenital heart surgery system [RACHS]-1 score) were collected. operating time (min), and postoperative medications (catecholamine, diuretics, and vasodilator agent) were also extracted. Additionally, AKI risk factors (single ventricular disease, pulmonary hypertension, RACHS-1 score ≥ 3, use of vasopressin, and cardiopulmonary bypass time) were extracted as previously described9,10 and were used for analysis. Furthermore, patients were stratified into an infant and a toddler group.
Definition of AKI
In this study, AKI was defined according to the Kidney Disease, Improving Global Outcomes (KDIGO) criteria. 11 Specifically, AKI was defined as a serum creatinine level ≥ 0.3 mg/dL (≥ 26.5 µmol/L) within 48 h or an increase in serum creatinine ≥ 1.5 times the baseline within 7 days, which corresponded to the KDIGO criteria stage 1. The baseline creatinine level was defined as the stable prehospitalization creatinine level within 1 year before hospital admission.
Biomarker Measurement
The TAT level was measured as an index of hypercoagulation. Furthermore, the PIC level was measured as an index of increased fibrinolysis. Specimens were obtained from the arterial pressure line immediately following admission to the ICU. The TAT and PIC levels were measured using chemiluminescent enzyme immunoassay and latex near-infrared light nephelometry, respectively (LSI Medience, Tokyo, Japan). In addition, we calculated TAT/PIC ratio as an independent variable.
Outcome Measures
The primary outcome measure was the presence or absence of AKI onset. Three pediatricians in the hospital department made the diagnosis based on creatinine levels according to the KDIGO criteria.
Statistical Analyses
Continuous variables are expressed as medians and interquartile ranges (IQR), while categorical variables are presented as frequencies and percentages. Bivariate analysis was performed using the Mann–Whitney U test for continuous variables and Fisher's exact test for categorical variables to compare the two groups. Univariate analysis was performed for each item to identify risk factors, followed by multivariable analysis using multiple logistic regression analysis. Independent variables were selected based on their significance from the bivariate analysis. Odds ratios (ORs) and 95% confidence intervals (CIs) were obtained.
Subgroup analyses were performed separately for the infant and toddler groups using bivariate and multivariable analysis. When a significant association was found between TAT or TAT/PIC and the outcome, receiver operating characteristic (ROC) curve analysis was performed to determine an optimal cut-off point to predict the outcome. The area under the ROC curve (AUC; 95% CI), optimal cut-off value, sensitivity, and specificity were calculated. Using a binary variable of TAT or/and TAT/PIC based on the cut-off value, both bivariate and multivariable analyses were performed. Statistical significance was set at P < .05. IBM SPSS Statistics for Windows, Version 25.0. (Armonk, NY: IBM Corp.) and the package of RcmdrPlugin.EZR in R (R Foundation for Statistical Computing, Vienna, Austria) was used as statistical analysis software.
Results
This study included 264 cases of cardiovascular surgeries, 199 of which underwent CPB. After applying the exclusion criteria, 154 cases were included in the study, and 55 cases (35%) developed AKI.
Univariate analysis showed significant differences in absolute TAT level (median [IQR], 24.0 [13.2–38.4] vs 27.8 [18.7–52.2] ng/mL; P = .046), vasopressin use (n [%], 16 [16.2] vs 20 [36.4]; P = .006), and CPB time (median [IQR], 82.0 [63.0–113.0] vs 107 [77.0–148.0] min; P = .004) (Table 1). However, multivariable analysis did not reveal any significant independent variable.
Clinical Characteristics and Comparison of AKI and Non AKI Groups.

AKI, acute kidney injury; HLHS, hypoplastic left heart syndrome; AVSD, atrioventricular septal defect; TA, tricuspid atresia; VSD, ventricular septal defect; TGA, transposition of the great arteries; DORV, double outlet right ventricle; TOF, tetralogy of Fallot; Truncus, truncus arteriosus; PA, pulmonary atresia; CoA, coarctation of the aorta; TAPVC, total anomalous pulmonary venous connection; RACHS-1 score, risk adjustment in congenital heart surgery system-1 score; CPB, cardiopulmonary bypass; ICU, intensive care unit; TAT, thrombin–antithrombin complex; PIC, plasmin-α2-plasmin inhibitor complex.
Regarding the subgroup analysis, the results showed no significant difference between the absolute TAT value and AKI onset in the infant group. In the toddler group, significant differences were observed in absolute TAT level (median [IQR], 27.7 [13.1-40.8] vs 62.9 [30.2-107.2] ng/mL; P = .002), TAT/PIC ratio (median [IQR], 13.3 [4.8-23.6] vs 24.6 [18.0-50.3] %; P = .005), RACHS-1 score ≥ 3 (n [%], 19 [46.3] vs 16 [80]; P = .015), and vasopressin use (n [%], 11 [26.8] vs 13 [65]; P = .006) (Table 2). However, multivariable analysis in the toddler group showed no significant difference in any of the items.
Comparison Between AKI and Non AKI Groups in the Toddler Group.

AKI, acute kidney injury; RACHS-1 score, risk adjustment in congenital heart surgery system-1 score; CPB, cardiopulmonary bypass; TAT, thrombin–antithrombin complex; PIC, plasmin-α2-plasmin inhibitor complex.
ROC analysis was performed regarding the absolute TAT level and TAT/PIC ratio in the toddler group. The AUC (95% CI), cut-off value, sensitivity, and specificity were 0.77 [0.64-0.90], 61.2 ng/mL, 85%, and 58% for the absolute TAT level and 0.75 [0.62-0.88], 10.5%, 48%, and 100% for TAT/PIC ratio, respectively (Figure 1).
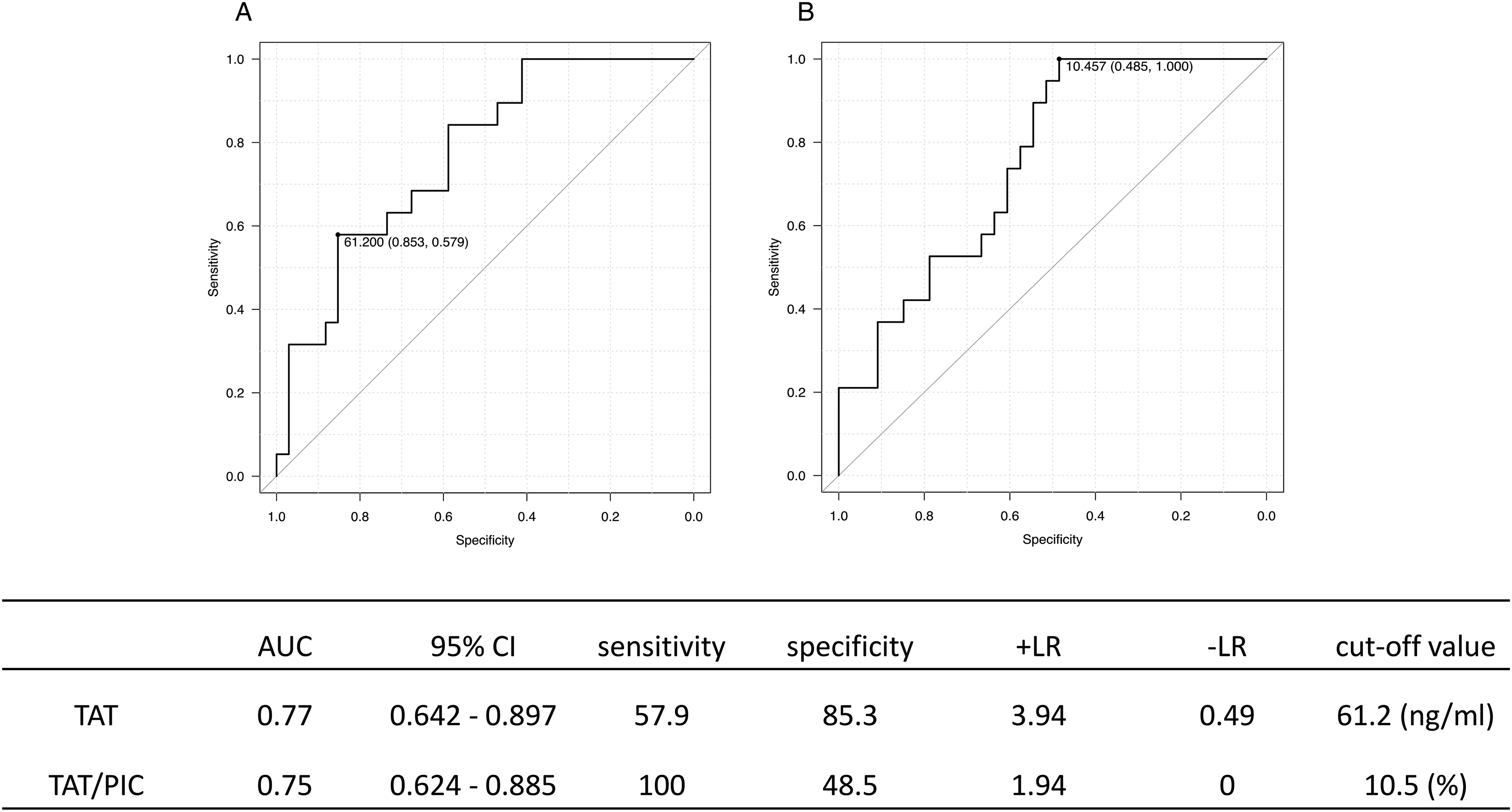
ROC analysis in the toddler group. A: Absolute TAT level; B: TAT/PIC ratio. ROC: receiver operating characteristics; TAT: thrombin–antithrombin complex; PIC: plasmin-α2-plasmin inhibitor complex.
In the toddler group, when compared based on the TAT cut-off value, significant differences were found in the TAT ≥ 61 ng/mL group for AKI onset (n [%], 9 [20.5] vs 11 [64.7]; P = .00188), RACHS-1 score ≥ 3 (n [%], 20 [45.5] vs 15 [88.2]; P = .003), and vasopressin use (n [%], 13 [29.5] vs 11 [64.7]; P = .019) (Table 3). Multivariable analysis revealed a significant difference between TAT and AKI onset (OR [95% CI]: 4.70 [1.20-17.90], P = .023) (Table 4).
Comparison Based on Cut-off Values of TAT in the Toddler Group.

TAT, thrombin–antithrombin complex; AKI, acute kidney injury; RACHS-1 score, risk adjustment in congenital heart surgery system −1 score; CPB, cardiopulmonary bypass.
Results of Multivariate Analysis Based on TAT Cut-off Values in the Toddler Group.

TAT, thrombin–antithrombin complex; RACHS-1 score, risk adjustment in congenital heart surgery system-1 score.
Discussion
In this study, we investigated the relationship between AKI onset and coagulopathy after pediatric CPB surgery. Our findings showed that there was a significant association between AKI onset and elevated TAT levels in toddlers. In addition, the predictive ability of TAT levels was moderate. To our best knowledge, this is the first study to investigate the relationship between coagulopathy and AKI onset following CPB during pediatric cardiovascular surgery. Our results suggest that combining other risk factors with elevated absolute TAT levels may be useful for predicting and preventing AKI onset.
AKI is a common complication of CPB, and its mechanism and risk factors have been extensively investigated. 12 Cardiopulmonary bypass can cause a decrease in kidney perfusion and dilution of blood with filling fluid that can reduce blood viscosity, resulting in a rapid decrease in peripheral vascular resistance. These changes can result in adenosine triphosphate deficiency and oxidative stress damage. Furthermore, the activation of neutrophils and vascular endothelial cells due to the impairment of cell adhesion factors can result in elevated levels of cytokines such as interleukin-6 (IL-6), interleukin-8 (IL-8), and tumor necrosis factor-α (TNF-α). These elevated cytokine levels can lead to vascular stasis in the renal medulla, resulting in proximal tubule cell ischemia and ultimately kidney injury. 13 Furthermore, AKI onset associated with vascular endothelial damage has also been a research interest.14,15 A study on patients with sepsis reported that elevated coagulation markers due to vascular endothelial damage were associated with AKI onset. 8 In addition, the onset of AKI due to vascular endothelial damage has also been reported to be associated with other organ diseases, such as acute respiratory distress syndrome. 16
In this study, we found that the onset of AKI was associated with CPB-induced vascular endothelial damage. During CPB, the vascular endothelium can be damaged by unphysiologically high pressure in the blood circuit, negative pressure by suction pumps, and/or shear stress caused by roller pumps. 17 Additionally, ischemic reperfusion occurs, and inflammatory factors such as cytokines, are induced. 18 These changes lead to the disruption of the intercellular barrier and the shedding of the glycocalyx. In the kidney, damage to the glycocalyx of the vascular endothelium and its detachment from the vascular surface exposes the vascular endothelium to the vascular lumen. Furthermore, the pore structure of the fenestrated capillaries is destroyed and blocked, and the connection between the capillaries and glomerular epithelial cells at the glomerulus loop is broken. Consequently, failure to maintain the three-layer structure results in renal dysfunction. 19
Damage to the vascular endothelium increases vascular permeability, induces inflammatory cells, elevates levels of coagulation factors, and results in coagulation activation. 7 TAT, a combination of thrombin and antithrombin in a ratio of 1:1, serves as an index of thrombin generation in blood vessels. Thrombin generated by the activation of the coagulation system binds to the inhibitor antithrombin to form a complex, thereby inactivating it. Detection of TAT in the blood can indicate activation of the coagulation system. In this study, we observed that TAT levels were elevated in toddlers with AKI onset, and its predictive ability was moderate. Additionally, PIC elevation was milder than that of TAT, suggesting that the patients were in a hypo-fibrinolysis state. This may result in thrombotic tendency and impair tissue perfusion, leading to organ dysfunction and AKI.
Children have immature coagulation and fibrinolytic reactions which are further affected by hemodilution due to CPB, leading to intravascular anticoagulant failure.9,20 Furthermore, due to their small body size, children receive a relatively large amount of filling fluid during CPB, often leading to shear stress, which can damage the vascular endothelium more easily compared with adults. Therefore, coagulopathy due to vascular endothelial damage is thought to be strongly related to the onset of AKI in children.
In this study, no significant differences were found between TAT value and AKI onset in the infant group. This may be due to the fact that the renal parenchyma, including the renal tubules and the vascular endothelium are more severely damaged in infants due to their immaturity.
This study has several limitations. First, the findings cannot be generalized due to the single-center and retrospective nature of the study. Therefore, caution should be taken when applying these findings to other institutes or other countries. Second, the small sample size may limit the reliability of the study for detecting significant differences and optimal cut-off values. Third, the study did not examine other markers that indicate hypercoagulation such as plasminogen, α2-plasminogen, and soluble thrombomodulin,8,20 and their interactions with TAT remain unknown. Therefore, a larger multicentered study with a prospective design is needed to validate the findings of this study.
Conclusions
The results of this study suggest that elevated absolute TAT levels in toddlers during the early postoperative period may be associated with the onset of AKI. However, further validation is needed through a prospective multicenter study.
Footnotes
Acknowledgements
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
