Abstract
Background
Data on treating idiopathic portal vein thrombosis (IPVT)—via anticoagulation (AC), systemic thrombolysis (ST), endovascular intervention (EVI), and surgery—are limited, despite the potentially severe clinical implications of this condition. This study aims to compare treatment outcomes and evaluate the prognosis.
Methods
A retrospective analysis of 77 IPVT patients was conducted. Patients were categorized according to their treatment modalities, and their demographics, clinical features, prognoses, and risk factors were analyzed. The primary endpoint was the radiological complete recanalization (CR) rate; secondary endpoints were clinical portal hypertension (CPH) incidence and overall mortality.
Results
Among the 77 patients with IPVT included in this study, 26 received AC, 23 EVI, 17 ST, and 11 surgery. During follow-up, the overall CR rate was 23.4%, CPH incidence 14.3%, and mortality 7.8%. Patients treated with EVI had a significantly higher CR rate than AC patients (HR: 0.214, 95%CI: 0.058 - 0.795, P = .011). Treatment modalities had no significant impact on long-term survival or CPH. Cox regression analysis identified elevated C-reactive protein (CRP) levels (HR 1.045, 95%CI: 1.016 - 1.075, P = .002) and splenomegaly (HR 20.325, 95%CI: 1.952 - 212.213, P = .012) at admission as mortality risk factors.
Conclusions
EVI therapy enhances the CR rate in IPVT patients compared to anticoagulation alone. Elevated CRP and splenomegaly at admission independently increase IPVT mortality.
Keywords
Introduction
Portal vein thrombosis (PVT) refers to thrombosis within the portal venous system, involving the main portal vein (PV), superior mesenteric vein (SMV), inferior mesenteric vein (IMV), or splenic vein (SV). The American Association for the Study of Liver Diseases proposed a classification system for PVT based on the duration of disease (recent: PVT present <6 months; chronic: >6 months), extent of main portal vein occlusion (a: complete occlusion [100% lumen]; b: partial occlusion [>50% lumen]; c: minimal occlusion [<50% lumen]; d: cavernous transformation with collateral circulation formation), and response to treatment or interval changes (progressive, stable, regressive). 1 This grading guides the assessment of treatment approaches and clinical outcomes. Mild cases with incomplete occlusion may be asymptomatic. When the PV is highly occlusive, with persistent or worsening thrombosis leading to intestinal circulatory dysfunction, patients typically exhibit an acute onset, manifesting as abdominal pain, abdominal distension, vomiting, hematochezia, and fever. In severe cases, this can progress to intestinal ischemia, necrosis, and infection, giving rise to septic shock, multiple organ failure, and even death. Chronic patients may exhibit clinical portal hypertension (CPH), such as esophageal/gastric varices with hematemesis/melena, ascites, splenomegaly.2–5 PVT has a low prevalence in the general population, estimated at approximately 1%, 6 and it can be categorized as either secondary or idiopathic, based on its etiology. Secondary PVT is relatively common in clinical practice, typically associated with risk factors such as liver cirrhosis, neoplasms, abdominal infections, splenectomy, inflammatory bowel disease, pregnancy, hypercoagulability-related risk factorsincluding myeloproliferative neoplasms, paroxysmal nocturnal hemoglobinuria, antiphospholipid syndrome, Factor V Leiden mutation, JAK2 V617F mutation, and G20210A mutation.2,4,5,7 In contrast, idiopathic portal vein thrombosis(IPVT) accounts for only 14% of PVT cases. 6 As the precise cause of IPVT remains unclear and its treatment outcomes are poorer than those with identifiable causes, current therapeutic strategies mainly focus on recanalization therapies, including anticoagulation (AC), systemic thrombolysis (ST), endovascular intervention (EVI) and surgery. This article highlights the current EVI techniques for the treatment of IPVT, primarily including direct thrombolysis administered via percutaneous transhepatic or transjugular approaches, as well as indirect thrombolysis delivered through the superior mesenteric artery (SMA) approach. The methods predominantly used in our center for IPVT patients are as follows: (1)SMA Angiography + Catheterization for Thrombolysis (SMA + T); (2)Portal Vein and Superior Mesenteric Vein Angiography + Thrombus Suction + Catheterisation for Thrombolysis (PV + T) and (3) Portal Vein and Superior Mesenteric Vein Angiography + Thrombus Suction + Balloon Dilatation (PV + B).
Methods
Participants and Data Collection
Medical records from the First Affiliated Hospital of Zhejiang University School of Medicine were retrospectively reviewed for patients diagnosed with PVT using International Classification of Diseases codes, spanning from January 2010 to December 2023. Relevant cases were excluded based on the following criteria: (1) patients aged younger than 18 years or older than 80 years; (2) patients exhibiting clear predisposing factors for thrombosis, such as liver cirrhosis, recent abdominal inflammation or surgery (within the last three months), active abdominal tumors, a history of splenectomy, a history oforthotopic liver transplantation, trauma, pregnancy, use of oral contraceptives, hypercoagulable states, and hematoproliferative diseases; (3) patients considered to have tumor-related thrombosis; and (4) patients with end-stage chronic diseases.
The diagnosis of PVT relied on imaging examinations, mainly including ultrasound, computed tomography (CT) scans, and magnetic resonance imaging. All the enrolled IPVT patients underwent abdominal enhanced CT scans during the diagnosis and treatment. Common laboratory assessments encompassed complete blood count, coagulation function tests, D-dimer levels, liver function tests and C-reactive protein(CRP) levels, as well as coagulation factor activity, homocysteine levels, anti-nuclear antibodies, anti-neutrophil cytoplasmic antibodies, antiphospholipid antibodies and levels of protein C and protein S. Additional procedures such as gastrointestinal endoscopy, might be performed as part of this comprehensive assessment.
Treatment strategies were developed based on the attending physician's clinical assessment of thrombus progression risk, bleeding risk, urgency of surgery, and patient preferences, resulting in the classification of patients into four groups. Anticoagulation was appropriate for most patients, whereas EVI and ST were recommended for high-risk patients with acute intestinal ischemia, severe vascular occlusion, and a need for prompt revascularization. Patients presenting with peritonitis or intestinal necrosis, active gastrointestinal bleeding (including persistent hematemesi/melena, circulatory instability, significant hemoglobin decrease exceeding 10 g/L daily, strongly positive fecal occult blood (OB) test, or imaging/endoscopic confirmation of ongoing bleeding), thrombocytopenia, or severe hepatic or renal dysfunction were excluded from ST and catheter-directed local thrombolysis. Surgical intervention was considered necessary for patients exhibiting complications of intestinal ischemia, such as bowel necrosis, obstruction, intra-abdominal hemorrhage or severe infection.
Clinical data were collected from patient medical records during hospitalization and subsequent post-discharge follow-up evaluations. To assess patient survival and disease status, follow-ups were conducted via outpatient visits and telephone. For patients newly diagnosed with IPVT and in good clinical condition, follow-ups were recommended at 1 month, 3 months, and 6 months, followed by semi-annual outpatient visits and rechecks, mainly including patient symptoms and general condition, blood tests (complete blood count, coagulation function, and D-dimer), and imaging examinations at 3 months, 6 months (contrast-enhanced CT required), and 12 months. If a patient's clinical condition deteriorates, prompt medical attention and repeated imaging should be pursued regardless of the timing. For those deciding to discontinue anticoagulant therapy, outpatient follow-ups with imaging rechecks (ultrasound or contrast-enhanced CT) every 3 months were recommended. 5
Definitions and Management
CPH: Portal hypertension was defined as increased portal venous system pressure from various causes, diagnosed when the pressure gradient (PV pressure minus inferior vena cava pressure) exceeded 5 mm Hg. Major clinical manifestations included splenomegaly, hypersplenism, esophagogastric varices, and ascites. In this study, CPH was defined as newly emerging development of esophageal/gastric varices (may with hematemesis and melena) or ascites requiring diuretic therapy.
D-dimer and fibrinogen: D-dimer and fibrinogen levels upon admission (D-dimer 1, Fibrinogen 1) and subsequently within 72 h of treatment initiation (D-dimer 2, Fibrinogen 2) were measured.
Conventional treatment: intestinal rest, parenteral nutrition, gastrointestinal decompression, administration of proton pump inhibitors, somatostatin and antibiotic, rehydration and maintenance of water-electrolyte balance.
EVI involved the following procedures:
SMA + T: This procedure entailed accessing the brachial or femoral artery through a percutaneous puncture. A sheath was then inserted, followed by systemic heparinization. Catheterization was performed to access the main trunk of the SMA, where urokinase (100 000-200 000U) was administered intra-operatively through the catheter. The catheter remained in place within the SMA to facilitate ongoing thrombolytic therapy. Postoperatively, urokinase and heparin administration could continue via the catheter for up to 3 days, with recommended dosages of 6250 U of heparin twice daily and 600 000 U of urokinase divided into two to four injections. Low Molecular Weight Heparin (LMWH) was administered every 12 h from the point of admission until the time of surgery and was halted during thrombolysis. Anticoagulation was promptly reinstated on the same day following catheter removal. PV + T: This approach involved ultrasound-guided percutaneous puncture of the PV, followed by the insertion of a sheath. Catheters were positioned in both the SMV and PV to facilitate thrombus aspiration. Urokinase (200 000-300 000 U) was sprayed into the catheter, followed by repeated thrombus aspiration. A thrombolytic catheter was retained to continue thrombolytic therapy, and a spring coil was applied at the puncture site to prevent bleeding risks. Postoperative management of urokinase and heparin followed the same regimen as outlined previously. PV + B: Similarly to the previous procedures, this involved ultrasound-guided percutaneous puncture of the PV, placement of a sheath, and catheterization into both the SMV and PV. Thrombus suction was conducted after spraying the catheter with urokinase, followed by balloon dilatation of the affected vessels. Performing re-angiography, if the imaging post-procedure demonstrates marked improvement in blood flow of both the SV and SMV, the catheter should be withdrawn, and embolization coils should be deployed at the puncture site. Anticoagulation with LMWH was maintained consistently during this period, administered every 12 h since admission.
ST: The administration of urokinase was conducted via intravenous infusion at doses ranging from 300 000 to 500 000 units once daily, or 200 000 units administered twice daily, with adjustments made based on body weight. The duration of urokinase treatment varied between 3 to 10 days, with a median treatment duration of 4 (3-3.5) days. Daily assessment of blood parameters, including complete blood count, fibrinogen levels and coagulation function should be performed, and systemic bleeding should be closely monitored. In instances where fibrinogen levels fell below 2.0 g/L, the urokinase dosage was reduced by half; if levels dropped below 1.0 g/L, thrombolysis was discontinued. LMWH was administered every 12 h from the time of admission, maintaining a consistent anticoagulation regimen throughout the treatment period.
Surgery: Surgical interventions primarily consisted of exploratory laparotomy, necrotic bowel resectionand anastomosis. All surgically treated patients initiated anticoagulation immediately postoperatively upon confirmation of no active bleeding signs, with LMWH 4100 U administered at a dosage of once daily to every 12 h, tailored to individual patient conditions.
AC: In the AC protocol, patients commenced anticoagulation immediately following the diagnosis of IPVT. LMWH was administered via subcutaneous injection during their hospital stay, with a dosing regimen of 4100 units every 12 h for patients without notable bleeding complications. If bleeding was present, the anticoagulant dosage would be reduced according to the severity of bleeding and the patient's coagulation status. If bleeding complications were to worsen, anticoagulation would be ceased, and alternative interventions including endoscopy, interventional hemostasis and surgical options would be considered. Once bleeding was stable, anticoagulant therapy should be gradually resumed until reaching a dose of 4100 units of LMWH every 12 h. For patients requiring warfarin (an oral vitamin K antagonist, VKA), the standard initiation dose was 3 mg once daily, initially bridged with LMWH for the first three days. For patients who were discharged on VKA, the subsequent dosage adjustments should be made based on coagulation monitoring to maintain the International Normalized Ratio within the target range of 2.0 to 3.0. Direct oral anticoagulants (DOACs)—including rivaroxaban, edoxaban, and dabigatran—were prescribed according to patients’ body weight and renal function. The conventional starting doses for these drugs were as follows: rivaroxaban 15–20 mg once daily, edoxaban 60 mg once daily and dabigatran 150 mg twice daily. None of these drugs required routine coagulation monitoring. It was recommended that patients continue routine anticoagulation for a minimum duration of 6 months following hospital discharge.
Statistical Analysis
Continues variables were presented as medians (interquartile range), categorical data were presented as frequencies (percentage). The distribution of continuous variables and categorical variables were compared by Mann-Whitney U test and χ2 test, respectively.
The Kaplan-Meier was applied to perform survival analyses with drawing the survival curve as well as incidence curve of CR and CPH by four different treatment methods. Log-rank method was used to test whether the univariate had significant effects on CR, CPH or death. These univariables included four different treatments, gender, age, smoking, alcohol consumption, hypertension, obesity, diabetes, abnormal liver function, gastrointestinal bleeding, ascites, days of abdominal pain, D-dimer levels, fibrinogen levels, CRP levels, comorbidities with other venous embolisms, vascular involvement by the thrombus, intestinal condition, cavernous transformation, splenomegaly, and length of hospital stay. Factors identified as significantly associated with observations in univariate analyses (P < .10) were subsequently included in a multivariate Cox proportional risk model for analysing potential risk factors for CR, CPH and death, which was used to determine adjusted HR and their corresponding 95%CI. Statistical analyses were performed using SPSS 29.0 software. All hypothesis tests were two-sided, and P < .05 were considered statistically significant unless otherwise stated.
Results
Baseline Characteristics
Between January 2010 and December 2023, a total of 77 patients with IPVT were included in the study (Figure 1). The baseline characteristics of these patients were summarized in Table 1. The incidence was higher in males (52 cases) than in females (25 cases), with a mean age of 45.0 years (range 18-80 years), a body mass index of 23.1 (range 15.4-34.9). Most patients had acute onset, with an average of 43.8 days (range 1-1460 days) of abdominal pain and an average hospital stay of 10.8 days (range 2-62 days). Upon admission, 36 patients had ascites, 21 had gastrointestinal bleeding, 16 had splenomegalyand13 patients had combined multiple venous thrombosis. The main PV was obstructed in 98.7% of cases, both left and right branches were obstructed in 84.4%, the SMV in 87.0%, and the SV in 70.1%. All patients exhibited varying degrees of elevated D-dimer and CRP levels, and no patients showed abnormalities in activated partial thromboplastin time, thrombin time, or prothrombin time. On admission, 41 patients had elevated fibrinogen levels, while 6 exhibited decreased fibrinogen levels. Within 72 h post-treatment, fibrinogen levels increased in 32 patients and decreased in 10 patients. Additionally, 15 patients had mildly elevated leukocyte counts at presentation, while two patients had mildly decreased platelet counts; One patient experienced a significant drop in platelet count during treatment. Nine cases of abnormal liver function were observed; among them, five patients had a mild increase in transaminase, and four patients had a mild increase in bilirubin. Notably, patients who presented with gastrointestinal bleeding (P = .027) or chronic abdominal pain(P = .003) were more likely to receive anticoagulant therapy. Conversely, patients with intestinal edema, ischemia or obstruction (P = .003) and those with intestinal necrosis (P < .001) were more likely to receive surgical interventions. It is expected that patients undergoing surgery would have longer hospital stays due to more severe illness and perioperative care requirements (P = .002).
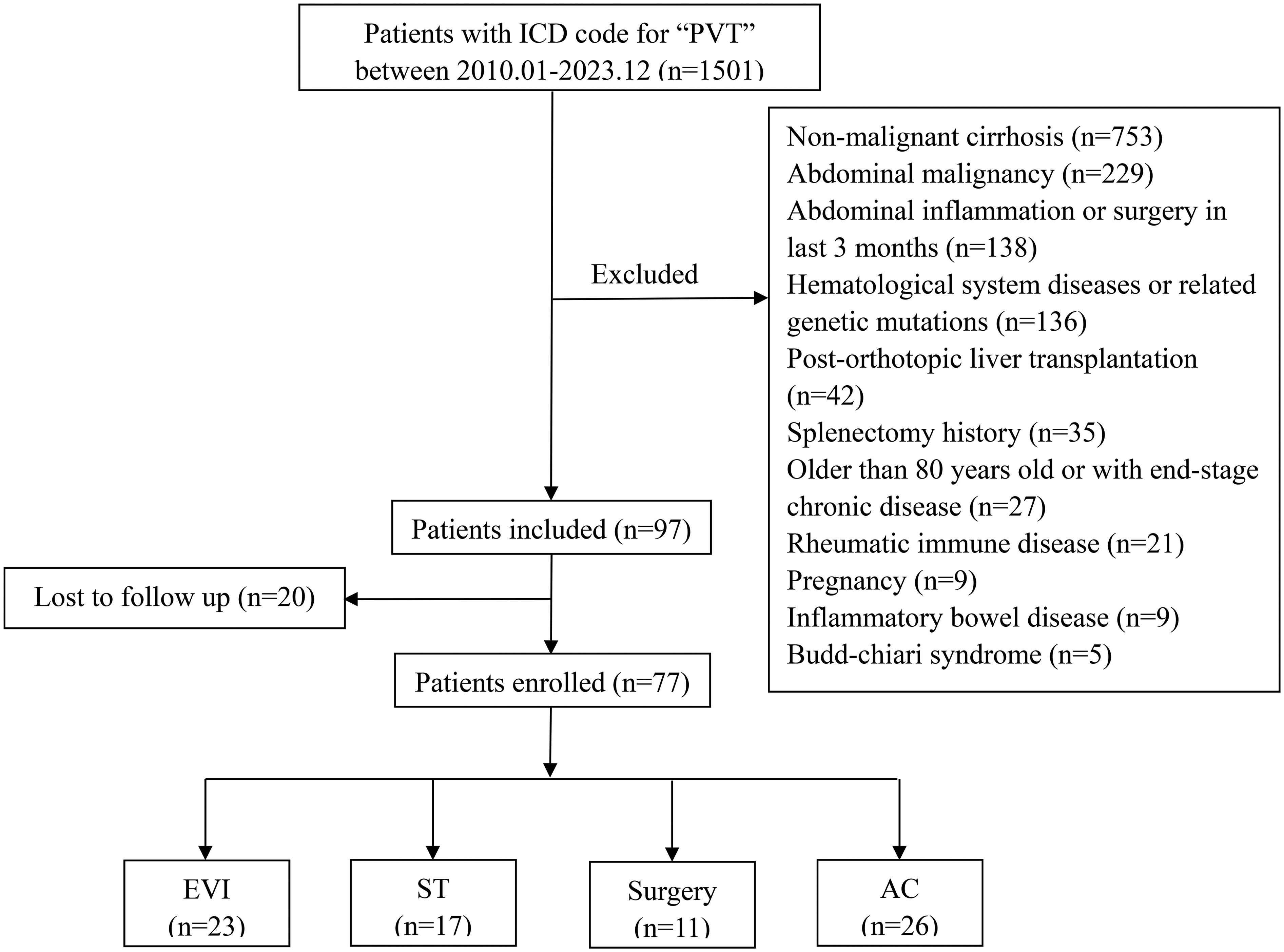
Flow Chart.
Demographic and clinical characteristics among IPVT patients with four different treatments.

Abbreviations: IPVT, idiopathic portal vein thrombosis; EVI, endovascular intervention; ST, systemic thrombolysis; AC, anticoagulation; CRP, C-reactive protein.
Long-Term Outcomes and Management
The overall survival rate for patients with IPVT was found to be 92.2%, with a median follow-up duration of 44.2 (12.7-106.3) months. Among the 6 deceased patients, three were male, with a median age of 48.5 (39.5-53) years and a median follow-up time of 49.5 (20.3-77.0) months. By follow-up, a total of 18 achieved CR with an overall CR rate of 23.4%, with 9 (39.1%) in EVI, 3 (17.6%) in ST, 3 (27.3%) in surgical group, and 3 (11.5%) in AC, P = .04. Eleven followed up with development of CPH, with an overall incidence of 14.3%, with 1 (4.3%) in EVI, 6 (35.3%) in ST and 4 (15.4%) in AC, P = .240. The overall mortality rate was 7.8%, 3 (17.6%) in ST, 3 (11.5%) in AC, P = .389. Three cases were treated with ST: one 53 year old female died of pulmonary embolism at 70.4 months, one 44 year old male died of sepsis at 79.2 months, and one 38 year old male with co-morbid cardiomyopathy died of multiorgan failure at 102.6 months. Three cases were treated with anticoagulant therapy: one 26 year old male died of refractory ascites complicating severe abdominal infections at 2.1 months. one 53 year old female died of liver failure at 28.6 months. one 80 year old female died of multi-organ failure at 17.6 months.
EVI: A total of 23 cases involving intravascular treatment were analyzed. Among these, there were 12 cases of SMA + T, 6 cases of PV + T, and 5 cases of PV + B. Figure 2 illustrated that a middle-aged female patient successfully achieved CR of the PV following 35 days of SMA + T treatment combined with sustained anticoagulation therapy. One patient with SMA + T experienced a recurrence of PVT, which was subsequently treated with Transjugular Intrahepatic Portosystemic Shunt (TIPS) and thrombolysis. At present, this patient remains in remission. In another case of PV + T, the patient developed worsening intestinal ischemia, ultimately presenting with necrosis post-surgery. This necessitated a second surgical intervention, consisting of thrombolectomy of the PV and SMV, along with resection of the necrotic intestinal segment three days later. The patient displayed satisfactory postoperative recovery.

Displays the Enhanced CT Images of the Patient's Abdomen. Image A, Acquired at the Time of Diagnosis, Demonstrates Thrombosis in the Main Portal Vein (A). Following Endovascular Intervention and Sustained Anticoagulation Therapy, Complete Recanalization of the Portal Vein (B) was evident on day 35.
ST: A total of 17 cases were analyzed, among which 3 cases had bleeding complications. One case presented with hematuria, one with subcutaneous ecchymosis, and one with gingival bleeding. However, all symptoms were effectively controlled through appropriate treatment.
Surgery: A total of 11 cases underwent emergency exploratory laparotomy and resection of necrotic intestine as an initial procedure with two patients undergoing thrombectomy with PV trunk dissection. Postoperatively, three patients developed short bowel syndrome as a complication. Another patient experienced back pain, gastrointestinal bleeding, dyspnea, and an inability to lie flat following surgery. Subsequent examinations revealed intestinal necrosis and pulmonary embolism; thus, a second resection of the necrotic intestine was performed on the 27th day post-initial surgery. The patient demonstrated a positive recovery following the second operation.
AC: The initial anticoagulation strategies were adjusted due to bleeding complications during the treatment process in 19 patients, 16 of which were attributed to gastrointestinal bleeding. Additionally, one patient experienced a significant decrease in platelet count following LMWH therapy, which was diagnosed as heparin-induced thrombocytopenia (HIT). Consequently, LMWH administration was promptly discontinued, and the patient was transitioned to warfarin for anticoagulation management. Two patients had received rivaroxabanfor anticoagulation before IPVT diagnosis: one for atrial fibrillation and one for pulmonary embolism. Among the cohort, 25 patients were discharged on warfarin, 46 on rivaroxaban, 5 on edoxaban, and 1 on dabigatran. Among 77 patients, 31 terminated anticoagulant therapy before the last follow-up or the occurrence of a death event. Of those who continued anticoagulant therapy, 11 were on warfarin and 35 were on DOACs.
Analysis
In the univariate analysis, the type of treatment was the only factor significantly associated with the CR, as evidenced by an overall log-rank P value of .040, which suggested that EVI is beneficial for achieving recanalization. Figure 3 presented the stratified log-rank P values for comparisons among different treatment groups, suggesting that EVI confers a benefit in recanalization compared to the AC group. Additionally, five factors demonstrated a significant association with patient mortality in the univariate analysis, indicated by log-rank P values of less than .10. These factors included: CRP levels (dichotomized at 50 mg/L, P = .022), gastrointestinal bleeding (P = .039), intestinal edema/ischemia/obstruction (P = .066), splenomegaly (P = .089), and length of hospital stay (P = .059). The survival curve of CRP level, which was significantly associated with outcomes (P < .05) in the univariate analysis and retained significance after multivariate adjustment, was presented in Figure 4. The aforementioned factors were incorporated into the multivariate analysis using the Cox proportional hazards model, suggesting that high levels of CRP were associated with splenomegaly and the risk of patient death as shown in Table 2. No risk factors associated with CPH were found in the univariate analysis.

Kaplan-Meier Curves Comparing the Probabilities of CR Among IPVT Patients with Four Different Treatments. Abbreviations: CR, complete recanalization; EVI, endovascular intervention; ST, systemic thrombolysis; AC, anticoagulation; IPVT, idiopathic portal vein thrombosis.

Kaplan-Meier Curves Comparing the Effect of CRP Levels on Mortality in Patients with IPVT.
Multivariable Cox regression in predicting the risk of mortality in patients with IPVT.

Abbreviations: IPVT, idiopathic portal vein thrombosis; CRP, C-reactive protein.
Discussion
Prompt diagnosis and treatment of IPVT is associated with an improved prognosis. 8 Anticoagulation serves as the cornerstone of IPVT treatment, regardless of the initial therapeutic strategy, and is recommended for the majority of patients. Given that intestinal blood flow obstruction and intestinal ischemia are the key pathological characteristics of IPVT, gastrointestinal bleeding is a frequently observed clinical manifestation. Therefore, gastrointestinal bleeding is not considered an absolute contraindication to anticoagulation, EVI, or ST. Patients who exhibit no signs of persistent active bleeding, maintain stable hemodynamic parameters and hemoglobin levels, and demonstrate only weakly positive fecal OB results may still undergo the aforementioned treatments under close clinical monitoring, with potential adjustments in drug dosing as clinically indicated. Compared with patients who did not receive anticoagulant therapy, the AC group had a higher recanalization rate, fewer thrombotic recurrence events, and a lower hemorrhage rate.9,10 Initial anticoagulation with LMWH is preferred over conventional heparin which has a higher incidence of HIT, followed by VKA and DOACs.11,12 Several studies have suggested that DOACs may be associated with better PVT outcomes.13,14 For instance, a retrospective analysis involving 330 patients with recent non-cirrhotic PVT (NCPVT) indicated that the recanalization rate with DOACs was comparable to that observed with LMWH and superior to that achieved with warfarin. 15 The study by Plessier et al showed that rivaroxaban 15 mg once daily can reduce the recurrence of thrombotic eventsor death without increasing the incidence of major bleeding events. 16 In terms of duration of anticoagulation, some studies have suggested a treatment time of at least 6 months consistent with guidelines for unprovoked venous thromboembolism, 10 while others have suggested that lifelong anticoagulation should be considered for those with IPVT, those with a previous or multiple recurrences of thrombosis, and those with hypercoagulable states.6,12,17
However, the goal of CR remains unattainable for the majority of IPVT patients despite the implementation of anticoagulation therapy. In our institution, the CR rate for IPVT was documented at 23.4%, which is lower than the rates reported for NCPVT by other centers,8,11,18,19 because the latter encompasses a wider array of potentially treatable, correctable, or removable causes, such as inflammation, abdominal surgery, etc. To improve CR rate in patients with IPVT, EVI therapy has emerged as a viable treatment option.9,19 The main techniques include thrombectomy, balloon angioplasty, and stent placement, which are effective in removing or reducing thrombus in the main PV and SMV, but not in removing microscopic peripheral thrombus; therefore, EVI approaches combining thrombectomy with catheter-directed local thrombolysis has shown greater efficacy in reestablishing portal circulation.1,20,21 Compared with ST, EVI offers advantages such as shorter pathway for faster drug delivery to the thrombus site, enabling optimal therapeutic effects with lower drug dosages and reduced bleeding risk. Urokinase was selected as the thrombolytic agent in this study due to its rapid onset and superior efficacy against fresh thrombi. A study 22 involving 653 cases of catheter-directed thrombolysis showed that compared with recombinant tissue-type plasminogen activator, urokinase was safer with a lower risk of bleeding. In the present study, patients undergoing EVI exhibited significantly superior long-term recanalization outcomes compared to those treated with AC. Multiple studies have confirmed the efficacy of EVI, including a prospective cohort study involving 65 patients with acute non-malignant NCPVT. The results demonstrated that the remission rates with EVI (17% partial remission and 54% complete remission) were approximately double those with AC (7% and 30%, respectively). This indicates that EVI has a significantly stronger positive effect on recanalization (OR 4.32, P < .016), albeit at the expense of an increased risk of bleeding complications (P < .001). 23 Furthermore, a retrospective study evaluating EVI in 19 patients with complete occlusive acute-subacute PV and SMV thrombosis showed that 7 patients (36.8%) and 11 patients (57.9%) achieved complete and partial recanalization, respectively. 21
Overall long-term survival of IPVT patients is good, as evidenced by a reported survival rate of 92.2% in this study. Identified risk factors for mortality include elevated CRP levels and the presence of splenomegaly at the time of diagnosis. CRP serves as a marker of inflammation, elevated levels indicate the presence ofinfections secondary to intestinal ischemia and necrosis in patients, suggesting extensive thrombus involvement and rapid disease progression. Elevated CRP levels may also induce transient hypercoagulability, which is positively correlated with venous thrombosis, contributing to poorer outcomes in affected patients. 24 In this study, patients with known cirrhosis were excluded, leading to the inference that splenomegaly was primarily attributable to thrombus involvement in the SV, which suggests that severe occlusion of the SV vasculature and obstruction of blood return were associated with a poor prognosis for patients. Another study has reached similar conclusion that thrombus involvement of the SV was a risk factor for recanalization failure. 9
Conclusion
EVI demonstrates superior efficacy in achieving CR compared to anticoagulation alone in patients with IPVT. The occurrence of CPH is not high and the overall survival outcomes are favorable in IPVT patients with appropriate treatment. Elevated CRP levels and splenomegaly upon admission are independently associated with increased mortality risk, highlighting their potential role as prognostic markers in IPVT management. Further studies are required to explore the long-term efficacy of different treatment strategies and the prevention and management of complications in IPVT patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval to report this case was obtained from Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (ITT20240558B).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
