Abstract
Patients treated with edoxaban may require diagnostic and therapeutic procedures that involve edoxaban interruption. Although heparin bridging strategies are not recommended, heparin is frequently used in clinical practice. However, whether heparin use decreases thromboembolic risk remains unclear, and the potential for increased periprocedural bleeding remains a concern. Here, we report factors predicting edoxaban interruption and the use of heparin bridging strategies and associated clinical events from Global EMIT-AF/VTE, a multicenter, prospective, noninterventional study (Clinicaltrials.gov NCT02950168). Eligible patients are adults with atrial fibrillation or venous thromboembolism treated with edoxaban who underwent a diagnostic or therapeutic procedure. Edoxaban interruption, heparin bridging strategies, and clinical event data were collected from 5 days before procedure through 29 days afterwards. Edoxaban was interrupted in 1222/2089 procedures (58.5%); a heparin bridging strategy was used during 178 (14.6%) of these interruptions. Patients who received periprocedural heparin had higher baseline HAS-BLED (2.4±1.0 vs 1.9±1.1, P <0.0001) scores and similar CHA2DS2-VASc (3.6±1.6 vs 3.4±1.6, P = 0.09) scores versus patients who did not. HAS-BLED score >3 and high EHRA procedural risk predicted both edoxaban interruption and the use of a heparin bridging strategy, whereas CHA2DS2-VASc scores did not predict either. Bleeding and ischemic event rates were low; the all-bleeding rate was higher with the use of a heparin bridging strategy versus without (6.2% vs 3.1%, P = 0.04). Periprocedural heparin use was associated with higher bleeding rates, but not with lower thromboembolic risk. Individual patient and procedural bleeding risks appear to contribute more than stroke risk to clinicians’ consideration of a heparin bridging strategy.
Introduction
Approximately 10% of patients receiving chronic anticoagulation therapy undergo diagnostic or therapeutic procedures each year; however, there is no standard practice for periprocedural interruption of anticoagulants.1,2 Oral anticoagulant interruption may be accompanied by the use of a bridging strategy, defined as the administration of a short acting anticoagulant, such as low molecular weight heparin (LMWH), before the time of the surgery/procedure when the patient's normal anticoagulant regimen is interrupted. Bridging strategies are used more often with patients receiving vitamin K antagonists (VKAs), such as warfarin, than with patients receiving non-vitamin K antagonist oral anticoagulants (NOACs). 3 The primary aim of heparin bridging strategies is to reduce the risk of thromboembolic events periprocedurally, but this approach may increase the risk of bleeding. 3 A randomized study showed that in patients with mild to moderate risk of stroke receiving VKAs, bridging with heparin increased bleeding risk without reducing the risk of stroke. 4 Therefore, the use of a heparin bridging strategy is not recommended for most patients treated with VKA.4,5
Compared with VKAs, the rapid onset of action, shorter half-life, and improved safety of NOACs result in shorter durations of periprocedural interruptions or no interruptions at all, reducing the rationale for the use of a heparin bridging strategy. 1 There are limited data on the practice of bridging with LMWH in patients receiving NOACs who undergo periprocedural anticoagulant interruption, and the current European Heart Rhythm Association (EHRA) practical guide recommends against the use of periprocedural heparin (ie, a heparin bridging strategy) in patients receiving NOACs undergoing planned procedures. 2
In randomized controlled trials and prospective registries of patients with atrial fibrillation (AF) who required anticoagulant interruption due to a diagnostic or therapeutic procedure, the administration of periprocedural heparin was not associated with superior protection against thromboembolic events.4,6–8 Additionally, bridging strategies are more commonly associated with an increased risk of bleeding.4,7,8 None of these studies, however, evaluated the use of a heparin bridging strategy in patients receiving edoxaban or assessed characteristics that could potentially predict anticoagulant interruption or the use of a heparin bridging strategy.
In the primary analysis from the Global EMIT-AF/VTE study, preprocedural and postprocedural edoxaban interruption occurred in 67.6% and 26.7% of patients, respectively; 24.2% of patients had both pre- and postprocedural interruption. 9 Results from this analysis indicated that periprocedural edoxaban interruption did not always adhere to guideline recommendations; individual patient characteristics were likely taken into consideration for interruption decisions. The objective of this subanalysis from the Global EMIT-AF/VTE study was to investigate individual patient characteristics as predictive factors of edoxaban interruption and the use of a heparin bridging strategy and report associated event rates in real-world clinical practice.
Patients and Methods
Design
The Global EMIT-AF/VTE study is a multicenter, prospective, noninterventional study integrating data from several European and Asian countries (NCT02950168). The detailed design and overall clinical event rates were published previously.9,10 The study is being conducted in accordance with the Declaration of Helsinki and received approval from all local Institutional Review Boards. Written informed consent was obtained from participants prior to enrollment. The periprocedural management of edoxaban therapy is at the discretion of the investigator, including interruption of edoxaban therapy and the timing/duration of the interruption and the use of a heparin bridging strategy. No attempt was made to influence patient management by the study authors, study team, or the sponsor.
Patients
Patients included in this analysis were enrolled between December 2016 and April 2020 in 389 centers in Belgium, Germany, Italy, Netherlands, Portugal, Spain, the UK, Korea, Taiwan, Hong Kong, and Thailand. Eligible patients were ≥18 years of age, had AF or venous thromboembolism as the indication for chronic oral anticoagulation, were treated with edoxaban according to the local labels, underwent a diagnostic or therapeutic procedure, and were not enrolled in any other concurrent study. Patients who did not provide informed consent, did not meet the inclusion criteria, or had incomplete documentation or data were excluded from the study. 9
Observations
The Global EMIT-AF/VTE study collects baseline patient characteristics, periprocedural management, and clinical event data from patients receiving edoxaban in routine clinical practice. Observations begin 5 days before the procedure (Day −5) through 29 days afterwards, with Day 0 as the day of the procedure. To enhance data capture, patients receive a memory aid booklet at study enrollment, which is reviewed at the end of the study. In this analysis, edoxaban therapy was considered uninterrupted if treatment was administered on each day of the observation period, including at any time on the day of the procedure. Interruption was defined as any single day without edoxaban within the window from Day −2 to Day +3. Heparin bridging strategy was defined as the administration of heparin during the periprocedural period for patients with edoxaban interruption, excluding heparin use during cardiac procedures.
The EHRA classification according to bleeding risk was determined for each procedure. 2 Clinical characteristics such as HAS-BLED (hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition labile international normalized ratio, elderly, and drugs/alcohol concomitantly) score, CHA2DS2-VASc (congestive heart failure, hypertension, age ≥75 [doubled], diabetes, stroke [doubled], vascular disease, age 65–74 years, and sex [female]) score, concomitant medications, and details of edoxaban treatment were documented for each patient at baseline and during the protocol-defined pre-, peri- and postprocedural periods.
Clinical Event Outcomes
In this analysis, the primary outcome was the incidence of all-bleeding events. Other outcomes included the incidence of major bleeding (International Society of Thrombosis and Haemostasis definition), 11 clinically relevant non-major bleeding (CRNMB; defined as overt bleeding that required medical attention, but did not fulfill the criteria for major bleeding), all-cause mortality, and ischemic events (the combined incidence of acute coronary syndrome, ischemic stroke, transient ischemic attack, and systemic embolic events). All major bleeding, CRNMB, acute coronary syndrome, and acute thromboembolic events were reviewed and unanimously adjudicated by the Steering Committee.
Statistical Analyses
All comparative analyses presented in this subanalysis were prespecified, except for analyses stratified by the use of a heparin bridging strategy. 10 Binary, categorical, and ordinal parameters were summarized in absolute and percentage numbers. P-values were obtained from analysis of variance for numerical parameters and from Fisher's exact test for categorical parameters. The Wilcoxon test was used to obtain the P-value for the difference in median duration of interruption. A logistic regression model with stepwise selection of predictors was performed to investigate characteristics that predicted edoxaban interruption or the use of a heparin bridging strategy. P-values for the differences in clinical event rates were obtained using the chi-square test when event numbers were sufficient for analysis. The statistical analyses were performed using SAS® version 9.3 or higher (SAS Institute, Cary, NC, USA).
Data Sharing Statement
The data underlying this article cannot be shared publicly at this time as the Global EMIT-AF/VTE study is currently ongoing.
Results
Clinical Characteristics
Of 2089 procedures, edoxaban treatment was interrupted in 1222 (58.5%); 867 procedures (41.5%) proceeded without edoxaban interruption (Table 1). Patients with versus without edoxaban interruption were more likely to have undergone high-risk procedures according to the EHRA bleeding risk classification (29.8% vs 13.6%; P < 0.0001) and be older (mean ± standard deviation [SD], 72.8 ± 9.5 years vs 70.2 ± 10.4 years; P <0.001) and female (35.6% vs 30.9%; P = 0.03). Patients with edoxaban interruption also had more comorbidities, such as dyslipidemia (44.5% vs 39.7%; P = 0.03), coronary artery disease (22.8% vs 14.0%; P < 0.0001), valvular disease (19.7% vs 14.1%; P = 0.0008), and congestive heart failure (15.5% vs 10.6%; P = 0.001), and were more likely to have reduced creatinine clearance (≤50 mL/min; 20.0% vs 15.9%, P = 0.02) compared with patients who did not have edoxaban interruption (Table 1). Patients who had their edoxaban therapy interrupted had higher HAS-BLED (mean ± SD, 2.0 ± 1.1 vs 1.8 ± 1.0; P = 0.0005) and CHA2DS2-VASc scores (mean ± SD, 3.4 ± 1.6 vs 3.0 ± 1.6; P <0.0001) and were less likely to be using antiplatelet medication (14.6% vs 22.3%; P <0.0001) than patients who did not have their edoxaban therapy interrupted (Table 1).
Patient Demographics and Clinical Characteristics.

For demographic and clinical characteristic summaries, procedures were counted once per patient using the procedure with the highest EHRA risk score category.
BMI, body mass index; CHA2DS2-VASc, congestive heart failure, hypertension, age, diabetes, prior stroke/transient ischemic attack-vascular disease and sex category; CrCl, creatinine clearance; EHRA, European Heart Rhythm Association; HAS-BLED, hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalized ratio, elderly, drugs or alcohol; OAC, oral anticoagulant; SD, standard deviation.
Of the 1222 procedures with edoxaban interruption, a heparin bridging strategy was used in 178 (14.6%) of these interruptions; 1044 (85.4%) interruptions did not utilize a heparin bridging strategy (Table 1). Compared with patients who were interrupted without the use of a heparin bridging strategy, those with the use of a heparin bridging strategy were less likely to use antiplatelet agents at baseline (7.3% vs 15.8%; P = 0.003) and were more likely to have valvular disease (26.4% vs 18.6%; P = 0.02) and higher HAS-BLED scores (mean ± SD, 2.4 ± 1.0 vs 1.9 ± 1.1; P <0.0001). CHA2DS2-VASc scores were similar between patients who received periprocedural heparin versus those who did not (3.6 ± 1.6 vs 3.4 ± 1.6, P = 0.09). Additionally, for patients who underwent high bleeding risk procedures, significantly more procedures had edoxaban treatment interrupted with the use of a heparin bridging strategy compared with those who did not receive heparin (64.0% vs 23.9%; P <0.0001; Table 1).
Predictors of Interruption and of the Use of a Heparin Bridging Strategy
Logistic regression analysis revealed that coronary artery disease, valvular disease, a HAS-BLED score >3, and high EHRA procedural risk category predicted edoxaban interruption (Figure 1A). Valvular disease, dyslipidemia, a HAS-BLED score >3, and high EHRA procedural risk category were most predictive of the use of a heparin bridging strategy (Figure 1B).

Predictors of edoxaban interruption (A) and the use of a heparin bridging strategy. (B) EHRA risk category denotes procedural bleeding risk. CI, confidence interval; EHRA, European Heart Rhythm Association; HAS-BLED, hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalized ratio, elderly, drugs/alcohol concomitantly.
Duration of Interruption
Procedures with a heparin bridging strategy had a significantly longer total duration of edoxaban interruption than those without a heparin bridging strategy (median, 5 days vs 2 days, P <0.0001; Figure 2). For high-risk procedures, edoxaban interruption was longer with a heparin bridging strategy compared with those without heparin use (median, 5 days vs 2 days; Figure 2). For those who received periprocedural heparin, median edoxaban interruption was longest when heparin was used postprocedure (9 days) or both pre- and postprocedure (10 days) versus preprocedure (2 days; Table 2). The use of a heparin bridging strategy appeared to correlate with the length of edoxaban interruption; the procedures most likely to use a heparin bridging strategy were those with edoxaban interruptions lasting 5 days or longer (Table 3). The 2 most common procedure types with periprocedural edoxaban interruption and a heparin bridging strategy were orthopedic procedures (n = 39) and vascular access and transcatheter diagnostics and interventions (n = 36; Supplementary Table 1).
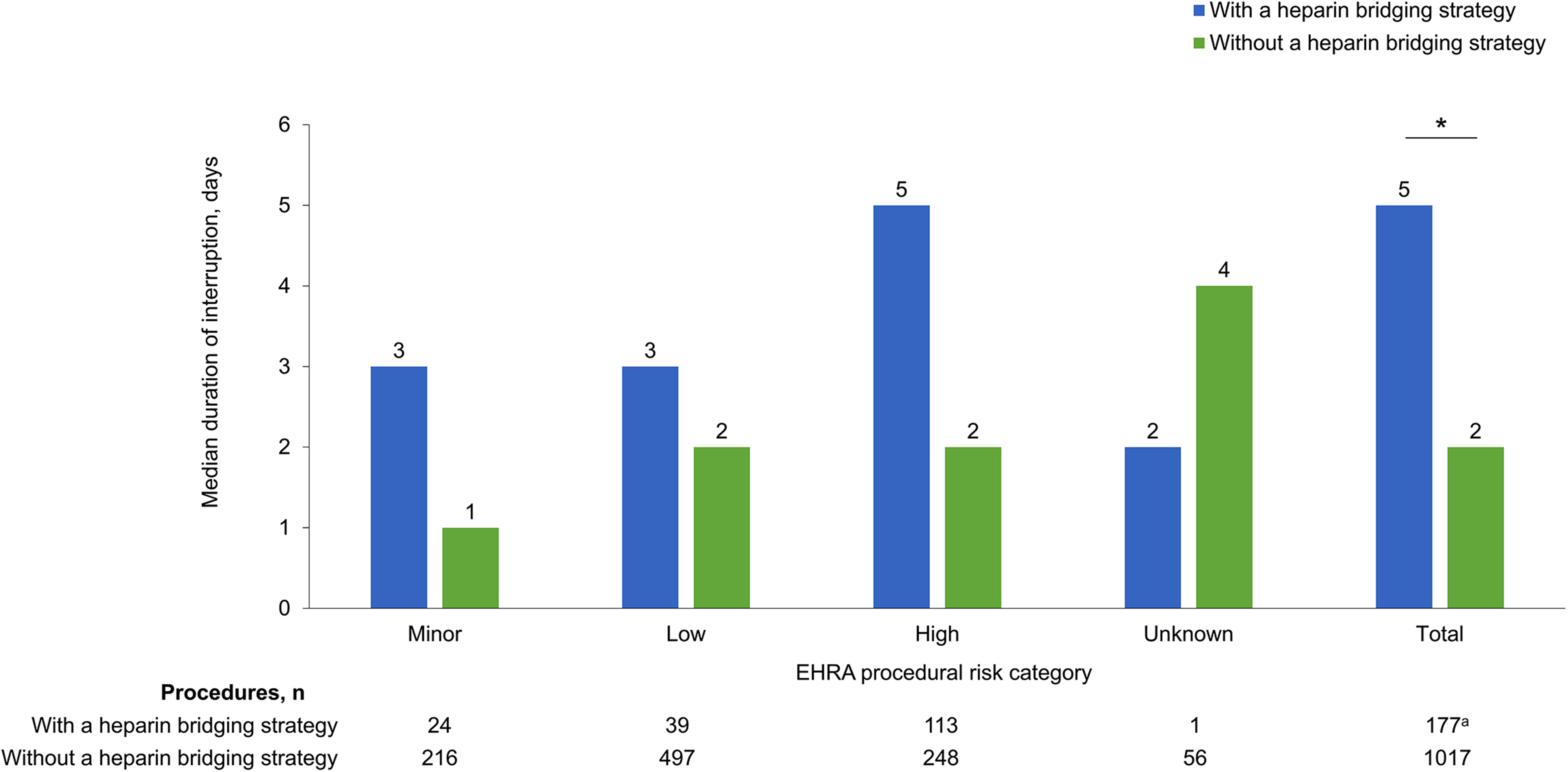
Duration of edoxaban interruption by EHRA risk and the use of a heparin bridging strategy. aDuration could not be calculated for one patient. *P <0.0001, with a heparin bridging strategy versus without a heparin bridging strategy. EHRA, European Heart Rhythm Association.
Duration of Edoxaban Interruption by Timing of the Heparin Bridging Strategy.

Use of a Heparin Bridging Strategy by Length of Edoxaban Interruption.
Clinical Events
Overall, clinical event rates were low. Patients with versus without edoxaban interruption did not differ in the incidences of all bleeding (3.5% vs 2.4%, P = 0.1518) or ischemic events (0.7% vs 0.5%; Figure 3). Patients with versus without periprocedural heparin had higher incidences of all bleeding (6.2% vs 3.1%, P = 0.0371); incidences of ischemic events were 1.7% and 0.5%, respectively (Figure 3). All-cause mortality and cardiovascular (CV) mortality were similar, independent of edoxaban interruption. However, all-cause mortality was numerically lower with versus without the use of a heparin bridging strategy during treatment interruption (0% vs 0.5%; Figure 3).

Clinical events with and without edoxaban interruption (A) or the use of a heparin bridging strategy (B). *P = 0.0371, with a heparin bridging strategy versus without a heparin bridging strategy; statistical analyses were only performed on all bleeding due to low event numbers. CRNMB, clinically relevant non-major bleeding; CV, cardiovascular; MB, major bleeding.
Discussion
The Global EMIT-AF/VTE study is the first real-world evidence study to report periprocedural interruption of oral anticoagulation and heparin bridging strategies in patients receiving edoxaban who underwent diagnostic and therapeutic procedures. This study includes data from patients who experienced interruption with or without heparin use as per local clinical practice. Here, we analyzed patient characteristics that could predict edoxaban interruption and the use of a heparin bridging strategy and reported clinical event rates.
The baseline patient characteristics observed in the Global EMIT-AF/VTE study were similar to the prospective, noninterventional Dresden registry study investigating periprocedural anticoagulant interruption in patients receiving rivaroxaban, dabigatran, or apixaban. 8 Across both studies, patients were 70–74 years old, 19%–20% had coronary artery disease, and 14%–18% had renal impairment. 8 The Dresden study, however, reported interruption in 78.3% of procedures, whereas the Global EMIT-AF/VTE study reported interruptions in 58.5% of procedures. 8 Despite a higher rate of interruption, there were fewer major or high-risk procedures in the Dresden study versus the current study (10.1% vs 23.1%), and the median duration of interruption was slightly longer in Dresden (3 days vs 2 days). 8
In the prospective, observational PAUSE study, all patients receiving rivaroxaban, dabigatran, or apixaban had their treatment interrupted; 33.5% of procedures were high risk. 12 Regardless of the type of NOAC, patients undergoing high-risk procedures were assigned a longer interruption period compared with patients undergoing low-risk procedures. 12 Overall, major bleeding and thromboembolic event rates were low (<2%) in the Dresden and PAUSE studies, but these studies did not include edoxaban.8,12 Results from the current study support low clinical event rates with edoxaban in real-world clinical practice, indicating that physicians were able to navigate periprocedural interruption for patients receiving edoxaban in clinical practice.
Heparin bridging strategies aim to reduce the risk of thromboembolic events during interruption of anticoagulation therapy and are typically reserved for patients at high risk for thrombosis; however, the potential for triggering more bleeding events remains a serious concern. 3 Notably, the randomized, controlled BRIDGE trial found no benefit in the prevention of arterial thromboembolism for patients receiving warfarin who were bridged with LMWH. 4 The proportion of procedures using a heparin bridging strategy in the current study (8.5%) is much lower than the proportion reported among patients in the Dresden registry study (29.8%) and among patients receiving warfarin or dabigatran in the prospective, observational ORBIT-AF registry study (23.7%).7,8 This difference in the rate of heparin bridging strategies may be related to the fact that the Dresden and ORBIT-AF data were collected many years before the current study; as guideline recommendations have developed in more recent years, bridging strategies are less often utilized in clinical practice.13,14 Furthermore, there may be regional differences in heparin use, as ORBIT-AF and Dresden were registry studies in the US and Germany, respectively.
The Global EMIT-AF/VTE study provides insight into predictors of the use of a heparin bridging strategy in edoxaban-treated patients undergoing invasive procedures. This study found the CHA2DS2-VASc score was not a significant predictor of edoxaban interruption or the use of a heparin bridging strategy. In contrast, the HAS-BLED score and the EHRA procedural risk level were significant predictors of the use of a heparin bridging strategy. These findings suggest that the bleeding risks associated with the individual patient (HAS-BLED score) and the procedure (EHRA procedural risk category) may play larger roles than stroke risk (CHA2DS2-VASc score) in decision-making when physicians consider the use of heparin bridging strategies. As demonstrated in the current study, the use of a heparin bridging strategy was more frequent in patients who had a longer interruption of edoxaban. In patients with a higher perceived bleeding risk, physicians may opt for a longer preprocedural interruption of edoxaban and may be more hesitant to restart therapeutic oral anticoagulation, resulting in longer anticoagulation-free intervals. These results suggest that more conservative management is observed when maintaining or initiating heparin to bridge anticoagulant interruption during EHRA high bleeding risk procedures or in cases where the patients have more risk factors for bleeding.
Clinical event rates associated with the use of a heparin bridging strategy were low and generally similar between ORBIT-AF, Dresden, and PAUSE studies, and this subanalysis of the Global EMIT-AF/VTE study despite differences in study designs. Similar to the current study, there were no differences in the rates of CV events between procedures with and without the use of heparin bridging strategies in the Dresden and ORBIT-AF registries.7,8 The rates of major bleeding and CRNMB were higher among patients in the Dresden study compared with the current study (6.6% vs 2.8%). 8 However, this could be attributed to differences in reporting or slight differences in clinical characteristics among the patients being bridged. In the ORBIT-AF registry, statistical analyses were performed to adjust for known confounders and baseline differences in patient characteristics; bleeding events were still higher with the use of a heparin bridging strategy in their multivariate-adjusted analysis. 7
Similar to previous studies, the results of this analysis follow a similar trend of higher bleeding rates with the use of a heparin bridging strategy. Periprocedural heparin use after edoxaban interruption was associated with a higher risk of bleeding and no benefit for ischemic event prevention in the general population. This same conclusion was reached in previous prospective registries and randomized control trials with both NOACs and VKAs and is aligned with the current guideline recommendation to not utilize heparin bridging strategies with NOAC interruption.2,4,6–8,12,13,15 However, the low overall event rate in the current study suggests a heparin bridging strategy may be best utilized on an individual basis by taking patient characteristics and procedural risk into consideration. The use of a heparin bridging strategy is most often reserved for patients at high risk of stroke, but a recent randomized trial found no significant benefit in the use of a heparin bridging strategy to prevent major thromboembolism in patients with AF at high risk of stroke or with mechanical heart valves. 6
There are notable strengths of this subanalysis of the Global EMIT-AF/VTE study. These include the prospective, observational, multicenter, multinational design and the large, unselected cohort of patients. The Global EMIT-AF/VTE study is a unique comprehensive data set that complements previously published data from the Dresden and PAUSE studies, which did not include patients receiving edoxaban.8,12 Taken together, the EMIT-AF/VTE, Dresden, and PAUSE studies offer a comprehensive data set of real-world periprocedural NOAC use.
As this is a prospective registry study, there is no comparator arm for patients receiving edoxaban. Additionally, the current analysis does not differentiate potential regional differences in interruption and bridging strategies or provide details of the dose of LMWH (therapeutic or prophylaxis). These are potential topics for future study.
In conclusion, this Global EMIT-AF/VTE study subanalysis demonstrates that patient bleeding risk (HAS-BLED) and EHRA procedural risk category are significant predictors of edoxaban interruption and the use of a heparin bridging strategy. Individual patient and procedural bleeding risk appear to play a larger role than a patient's stroke risk in clinicians’ consideration of utilizing a heparin bridging strategy. While the use of a heparin bridging strategy was associated with a higher rate of all-bleeding events, clinical event rates in this subanalysis of the Global EMIT-AF/VTE study were low.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231200223 - Supplemental material for Predictive Factors and Clinical Events Associated with Edoxaban Interruption and Heparin Bridging Strategy: EMIT-AF/VTE
Supplemental material, sj-docx-1-cat-10.1177_10760296231200223 for Predictive Factors and Clinical Events Associated with Edoxaban Interruption and Heparin Bridging Strategy: EMIT-AF/VTE by Amparo Santamaria, Cathy Chen and Paolo Colonna, Christian von Heymann, Manish Saxena, Thomas Vanassche, James Jin, Martin Unverdorben in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Medical writing and editorial support were provided by Lauren Hanlon, PhD, CMPP, of AlphaBioCom, a Red Nucleus company, and were funded by Daiichi Sankyo, Inc.
Contributions
All authors were responsible for interpretation of results;
Conflict of Interest Disclosure
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Daiichi Sankyo, Inc.
?>
These data were presented in part at the 2021 Congress of the International Society on Thrombosis and Haemostasis on July 20, 2021, in Philadelphia, PA, USA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
