Abstract
Background
Underdosed edoxaban was prevalent in Asian clinical practice, yet its outcomes in patients with non-valvular atrial fibrillation (AF) remain unclear.
Methods
We conducted a retrospective cohort study at a Chinese tertiary-care hospital to investigate the real-world effectiveness and safety of underdosed edoxaban in patients with AF. According to international dosing criteria, patients were stratified into recommended dose group and under dosed group. After inverse probability of treatment weighting, the effectiveness endpoints (ischemic stroke (IS) or systemic embolism (SE), all-cause mortality) and safety endpoints (major bleeding and clinically relevant non-major [CRNM] bleeding) were evaluated between the two groups.
Results
Of 616 included non-valvular AF patients, 31.5% received an underdosed edoxaban regimen. The under dosed group showed no significant differences in the risk of IS or SE (HR 0.78, 95%CI 0.18–3.32), major bleeding (HR 0.19, 95%CI 0.01–2.12), or all-cause mortality (HR 0.60, 95%CI 0.28–1.31) compared to the recommended dose group. However, a significant reduction in CRNM bleeding was observed in the under dosed group (HR 0.47, 95%CI 0.23–0.95).
Conclusion
In this real-world Chinese tertiary-care cohort, underdosed edoxaban was administered to nearly one-third of patients with non-valvular AF. Compared with the recommended dose, this dosing strategy was associated with comparable risks of IS/SE and major bleeding, as well as a potentially lower incidence of CRNM bleeding. These findings should be considered preliminary and warrant validation in larger, multi-center prospective studies.
Introduction
Atrial fibrillation (AF) is the most prevalent cardiac arrhythmia in daily clinical practice, affecting an estimated 37 million individuals globally.1,2 Thromboembolic stroke or systemic embolism (SE) are the potential complication in patients with atrial fibrillation, and oral anticoagulation therapy is the principal priority of AF Management. 3 Although vitamin K antagonists have been used for decades, direct oral anticoagulants (DOACs) are now recommended as the preferred choice of anticoagulants for non-valvular AF patients. 4
Edoxaban, a DOACs supported by the Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation–Thrombolysis in Myocardial Infarction 48 (ENGAGE AF TIME-48) trial, 5 was recommended for stroke prevention in patients with non-valvular AF at a standard dose of 60 mg once daily, with a reduced dose of 30 mg once daily recommended for patients with a body weight ≤ 60 kg, creatinine clearance 15–50 ml/min, or concurrent use of certain P-glycoprotein inhibitors. Despite a recommended dosing regimen for edoxaban, non-recommended prescription of edoxaban remains a major problem in the real-world setting. 6 The global Edoxaban Treatment in Routine Clinical Practice for Patients with Non-Valvular Atrial Fibrillation (ETNA-AF) study revealed that 17.4% of patients received non-recommended dose of edoxaban, with 64.4% of those patients being underdosed, and Asian populations were more likely to receive underdosed exdoxaban than Europe populations (19.1 vs 8.5%). 7
Given that Asian populations have smaller body sizes and a higher risk of bleeding complications (eg intracranial hemorrhage), compared to western populations,8,9 Asian clinicians are more inclined to prescribe underdosed exdoxaban than their counterparts in western countries. Nonetheless, the clinical outcomes of under-dosed edoxaban in Asian populations has yet to be definitively established. Recently, the Global ETNA-AF study examined the clinical outcome of edoxaban in patients with atrial fibrillation. 7 However, the study explicitly excluded individuals receiving the 15 mg regimen—an edoxaban dose frequently prescribed to Asian patients. It remains uncertain whether underdosed edoxaban is as effective as the recommended dose in preventing the thrombotic events and whether it offers a safer profile in terms of bleeding risk in Asian populations. Therefore, we aimed to evaluate the efficacy and safety of under dosed edoxaban in Asian patients with non-valvular atrial fibrillation under routine clinical practice conditions.
Ethics Approval
The study protocol was approved by the Medical Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine (NO. 2023-1236 and 2024-0619).
Methods
Study Design and Subjects
This was a retrospective observational cohort study conducted at the Second Affiliated Hospital of Medical, Zhejiang University School of Medicine. Using an electronic medical records database, patients who were discharged with an edoxaban prescription from July 1, 2021 to August 31, 2023 were identified. The inclusion criteria were: (1) new to edoxaban (without a previous history of edoxaban prescriptions before admission); (2) patients who were diagnosed with atrial fibrillation. The exclusion criteria were: (1) patients with valvular AF (moderate-to severe mitral stenosis or mechanical heart valve); (2) patients with a creatinine clearance <15 ml/min before receiving edoxaban; (3) aged <18 years; (4) patients who received overdose edoxaban therapy (edoxaban at a daily dose of 60 mg for patients with a body weight ≤ 60 kg, creatinine clearance 15-50 ml/min, or concomitant use of certain P-glycoprotein inhibitors); (5) patients who were lost to follow-up.
Data Collection and Definition
A chart review was conducted for each patient included in the study. Patients’ information was collected, including basic demographics, laboratory data, and clinical data during hospitalization. Demographic data included sex, age, weight, and height. Laboratory data included alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin, hemoglobin and serum creatinine at baseline of edoxaban therapy. Clinical data included edoxaban dosage, comorbidities (hypertension, diabetes mellitus, congestive heart failure, coronary heart disease, peripheral artery disease, liver dysfunction, malignancy and anemia), prior ischemic stroke (IS), prior major bleeding, smoking and drinking history, concomitant drugs (eg, antiplatelet agents such as aspirin, clopidogrel, ticagrelor; β blocker; amiodarone; digoxin; P-glycoprotein inhibitors). Liver dysfunction was defined as AST or ALT more than 3 times the upper limit of normal or total bilirubin more than 2 times the upper limit of normal. 10 Prior major bleeding was defined as a history of any major bleeding event, gastrointestinal hemorrhage, intracranial hemorrhage, or prior blood transfusion. 11 Anemia was defined as hemoglobin concentration less than 130 g/L in males or less than 120 g/L in females. 12 Creatinine clearance (CrCl) was calculated by the Cockcroft Gault equation. 13 The CHA2DS2-VASc and HAS-BLED score were used to predict the risk of thromboembolism and bleeding in patients with non-valvular AF treated with oral anticoagulants.14,15
According to the edoxaban dosing criteria of European Society of Cardiology (ESC) guideline for the management of atrial fibrillation,
16
edoxaban is recommen
Follow-up and Outcomes
All included patients received follow-up telephone interviews from January 2023 to March 2024 by trained clinical pharmacists. The primary efficacy outcome was the rate of IS or SE. IS was defined as a new focal neurological deficit of sudden onset, lasting ≥24 h, and confirmed by brain imaging (CT or MRI) consistent with ischemia. 17 SE was defined as an acute vascular occlusion of an extracerebral arterial territory, objectively confirmed by imaging (angiography, computed tomography angiography, or magnetic resonance angiography) or surgical embolectomy. 18 The primary safety outcome was the rate of major bleeding, as defined by the International Society of Thrombosis and Haemostasis: fatal or symptomatic bleeding event requiring transfusion of ≥ 2 units of red blood cells, involving hemoglobin drop of ≥ 2 g/dL, or occurring in a critical area/organ (intraspinal, pericardial, intracranial, retroperitoneal, intraocular, intra-articular, or intramuscular with compartment syndrome). 19 Secondary clinical outcomes were clinically relevant non-major (CRNM) bleeding and all-cause mortality. CRNM bleeding was defined as overt hemorrhage that did not meet the criteria for major bleeding but required medical intervention, unscheduled physician consultation, medication discontinuation or disruption of routine daily activities. 20
All potential endpoint events were identified through a two-stage process involving initial screening and independent adjudication. Initially, two trained clinical pharmacists performed the initial screening via structured telephone interviews using a standardized questionnaire (Supplementary Table 1) and a subsequently reviewed of electronic medical records, which included cross-verification of outpatient and inpatient data. To ensure the capture of out-of-network events, patients were specifically asked during interviews about hospitalizations or visited an emergency department at other facility. For any such event reported, pertinent documents including the medical records, discharge summaries and imaging reports, were systematically requested for review. Finally, all identified potential events were adjudicated by two physicians, who were blinded to patient group assignment and not involved in study conduct, based on pre-defined criteria and a comprehensive review of source documentation, including imaging reports, physician notes, and laboratory data.
Statistical Analysis
Continuous variables are expressed as mean ± standard deviation. Categorical variables are expressed as frequencies or percentages. To balance baseline characteristics between the under dosed group and recommended group, the stabilized inverse probability of treatment weighting (IPTW) approach were performed. The propensity scores were calculated for each patient using a multivariate logistic regression model that included the following specified baseline covariates: age, sex, body weight, creatinine clearance, CHA2DS2-VASc score, HAS-BLED score, coronary heart disease, liver dysfunction, prior IS, concomitant use of antiplatelet agents, amiodarone and digoxin. Stabilized weights were calculated for each patient and subsequently truncated at the first and 99th percentiles to minimize the influence of extreme weights and improve the stability of estimates. Standardized mean differences (SMD) was used to evaluate baseline characteristics balance, where an SMD < 0.1 indicated negligible intergroup disparity.
For outcome analysis, weighted incidence rates were estimated by dividing the weighted number of events during the follow-up period by 100 person-years. The follow-up period was calculated from the index date until the occurrence of study outcome, death, or the end date of the study period (March 30th, 2024), whichever occurred first. The risks of clinical outcomes between the two groups were compared using Cox proportional hazard regression models incorporating the truncated stabilized IPTW weights and a robust variance estimator to obtain accurate confidence intervals. Hazard ratios (HR) with 95% confidence intervals (CI) were calculated, using the recommended dose group as the reference. In addition, Kaplan–Meier survival analysis was performed to evaluated the cumulative incidence of clinical outcomes by the log-rank test. P < 0.05 were considered to be statistically significant.
Stratified analysis was performed by categorizing patients into two strata based on the presence of dose reduction criteria: (1) patients without dose reduction criteria (under dosed: 30 mg vs recommended dose: 60 mg), and (2) patients with dose reduction criteria: (under dosed: 15 mg vs recommended dose: 30 mg). Furthermore, Subgroup analysis was conducted within the overall weighted population based on age (<75 vs ≥75 years), body weight (>60 vs ≤60 kg), CrCl (>50 vs ≤50 ml/min), and concomitant antiplatelet use. P for interaction of 0.05 was used to determine the significance of interactions between the two groups and each subgroup. All statistical analyses were performed using R software (version 4.1.0, R Foundation for Statistical Computing).
Results
Patient Characteristics
As show in Figure 1, from July 1, 2021 to August 31, 2023, a total of 1578 inpatients received edoxaban at discharge, of who 1410 were first-time users. Among patients receiving edoxaban for the first time, 681 patients were diagnosed as atrial fibrillation. Patients who are younger than 18 years of age (1 case), with a creatinine clearance <15 ml/min before receiving edoxaban (6 cases), received overdose edoxaban therapy (33 cases), and were lost to follow up (25 cases), were excluded. Finally, a total of 616 patients were included.
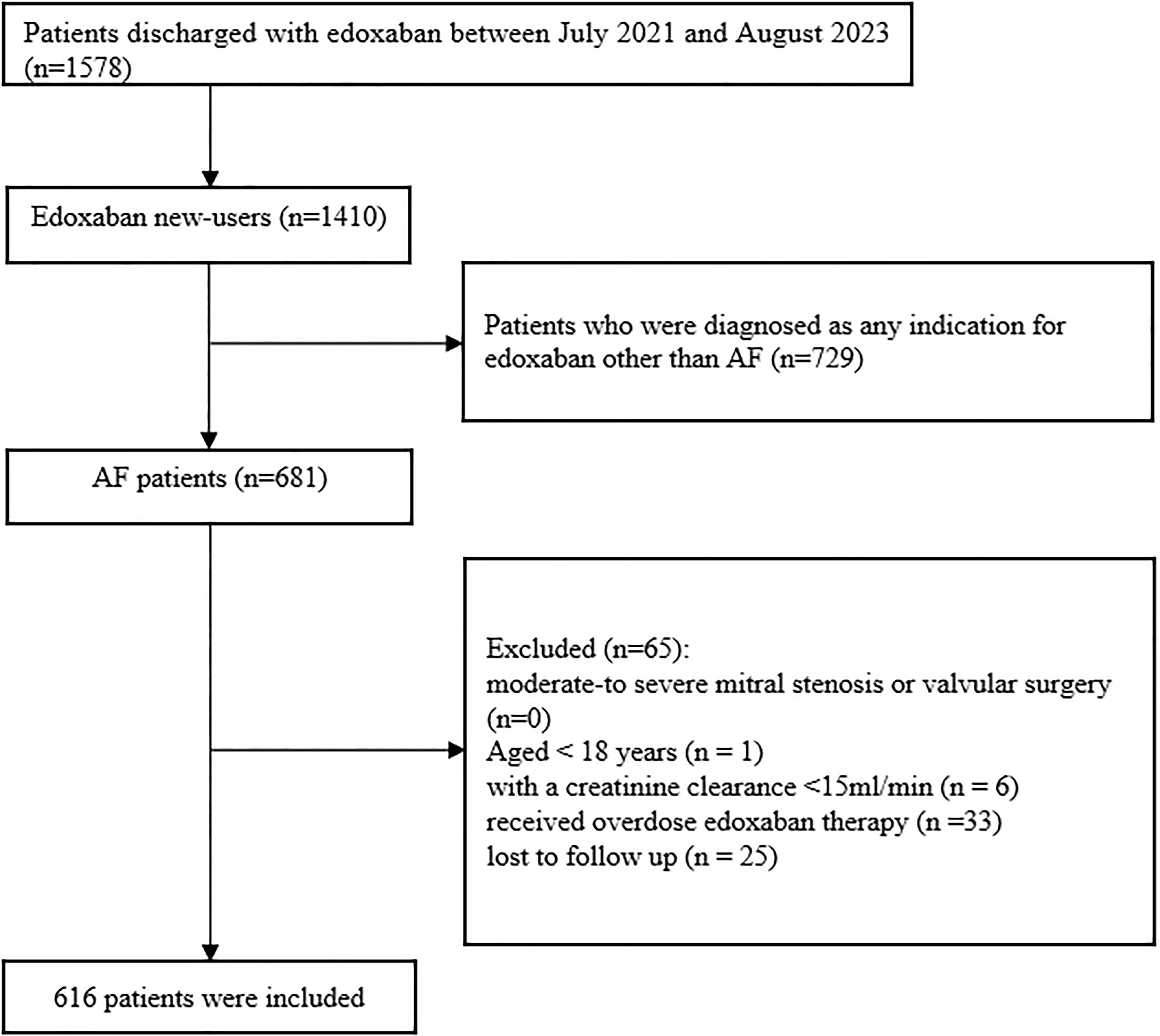
Study population and flow chart. Abbreviations: AF, Atrial Fibrillation.
Overall, 422 (68.5%) of patients received a recommended dose of edoxaban (60 mg: 16.2%; 30 mg: 52.3%), whereas 194 (31.5%) of patients received an under-dose of edoxaban (30 mg: 26.6%; 15 mg: 4.9%) (Figure 2). The mean age of the patients enrolled was 72.11 ± 11.46 years, and there were 244 female patients (40.2%). Permanent discontinuation of edoxaban occurred at comparable rates between the under dosed and recommended dose groups (15.5% vs 16.6%). Substantial imbalances of baseline characteristics were observed between the groups before weighting, as detailed in Table 1. Patients in the under dosed group had higher body weight, higher creatinine clearance, and a greater prevalence of antiplatelet use. In contrast, they displayed with lower CHA2DS2-VASc score and HAS-BLED score, as well as less diabetes mellitus, peripheral artery disease, anemia, and use of digoxin. To address these imbalances and the influence of extreme weights, we applied stabilized IPTW with truncation of the weights at the first and 99th percentiles. Following this procedure, excellent covariate balance was achieved, with all absolute SMDs falling below the 0.10 threshold (Table 1). The distribution of the stabilized weights after truncation and the improvement in covariate balance are visually confirmed in Supplementary Figure 1 and Supplementary Figure 2, respectively.

Study population grouping. Dose reduction criteria: body weight ≤ 60 kg, creatinine clearance 15–50 ml/min, or concomitant use of certain P-glycoprotein inhibitors (cyclosporine, dronedarone, erythromycin, and ketoconazole).
Baseline Characteristics for Recommended Dose Group and Under Dosed Group Before and After Inverse Probability of Treatment Weighting.

Data are presented as n (%) or mean ± SD. Abbreviations: BMI, body mass index; CrCl, creatinine clearance; IS, ischemic stroke; IPTW, inverse probability of treatment weighting; SMD, Standardized mean difference. *Age, sex, body weight, creatinine clearance, CHA2DS2-VASc score, HAS-BLED score, coronary heart disease, liver dysfunction, prior IS, concomitant use of antiplatelet agents, amiodarone and digoxin were the covariates listed in the table used to calculate the propensity score for weighting.
Clinical Outcomes
The overall median follow-up duration was 422 days (interquartile range, 282–548). The corresponding incidence rates (per 100 person-years) were 1.36 for IS/SE, 1.59 for major bleeding, 7.86 for CRNM bleeding, and 5.72 for all-cause mortality (Table 2).
Absolute Number and Incidence Rates of Outcomes Between Recommended Dose Group and Under Dosed Group Before Inverse Probability of Treatment Weighting.

IS, ischemic stroke; SE, systemic embolism; CRNM bleeding, clinically relevant non-major bleeding; #Per 100 person-years.
The weighted cumulative incidence curves and weighted incidence rates for IS/SE, major bleeding, CRNM bleeding and all-cause mortality in the recommended dose group and under dosed group are presented in Figure 3 and Figure 4. No significant difference was observed in the incidence rate of IS/SE (HR 0.78, 95% CI 0.18–3.32) or all-cause mortality (HR 0.60, 95% CI 0.28–1.31) between the two groups. Compared with the recommended dose group, underdosed edoxaban showed no significant association with reduced major bleeding (HR 0.19, 95% CI 0.01–2.12), while a significant reduction in the risk of CRNM bleeding was observed (HR 0.47, 95% CI 0.23–0.95).

The weighted cumulative incidence curves of ischemic stroke or systemic embolism (A), major bleeding (B), CRNM bleeding (C), and all-cause mortality (D) in the recommended dose group and under dosed group. Abbreviations: CRNM bleeding, clinically relevant non-major bleeding.

Weighted incidence rates, Hazard ratio (95% CI) and p value of clinical outcomes in the recommended dose group and under dosed group. Abbreviations: IS/SE, ischemic stroke or systemic embolism; CRNM bleeding, clinically relevant non-major bleeding; IR, incidence rate; PY, person-years; HR, Hazard ratio; CI, confidence interval. *Reference as recommended dose group.
Stratified analyses demonstrated that the risks of IS/SE, major bleeding, CRNM bleeding, and death were generally consistent between the recommended dose and under dosed groups across both strata defined by the presence or absence of dose reduction criteria (Supplementary Table 2). Due to the low number of observed events for the primary outcomes of IS/SE and major bleeding, meaningful subgroup analyses were precluded. In subgroup analyses for secondary outcomes, a reduced risk of CRNM bleeding with under dosed edoxaban was observed in patients with CrCl>50 ml/min. Notably, no significant interactions were observed for either CRNM bleeding or all-cause mortality across any of the specified subgroups (age, body weight, CrCl, or concomitant antiplatelet use) (Supplementary Figure 3).
Discussion
This study evaluated the clinical impact of underdosed edoxaban in a real-world cohort of patients with non-valvular AF. The main findings were as follows: first, underdosed edoxaban was administered to 31.5% of Chinese AF patients in this cohort; Second, no significant differences were observed in the rates of major bleeding or IS/SE among patients receiving underdosed and recommended dose edoxaban; and third, underdosed edoxaban was associated with a lower rate of CRNM bleeding in patients with non-valvular AF.
The frequency of the underdosed exdoxaban prescription in China was unclear. We found the prevalence of under-dose edoxaban prescription for AF was 31.5%. This is different from the ETNA-AF Europe study, under-dose edoxaban of 30 mg was observed in 8.5% patients. 21 Elsewhere, whereas 55.9% of patients were under-dosed in Global Anticoagulant Registry in the FIELD-AF (GARFIELD-AF). 22 Notably, edoxaban patients in GARFIELD-AF study was confined mostly to the Japanese. The discrepancy in the prevalence of under-dose edoxaban prescription may be mainly due to the country/regional differences. Compared with non-Asians counterparts, Asian physicians tend to prescribe under-dosed DOACs for patients with AF. Several potential reasons exist for this practice. First, the risk of major bleeding (eg, intracranial hemorrhage) is generally higher in Asians than in Western populations, 23 which affects clinicians’ dosage decisions for DOACs. Second, Asian populations typically have lower body weights than non-Asians, 23 who constitute the majority of participants in pivotal DOACs randomized controlled trials. Finally, the proportion of the elderly population in Asia is significantly higher and continues to rise compared with other regions. 8
An important clinical concern is whether underdosed edoxaban compromises clinical efficacy in real-world practice. Contrary to prior concerns of increased thromboembolic risk, our study did not observe a higher incidence of IS/SE with underdosed edoxaban (HR 0.78, 95%CI 0.18–3.32). This finding contrasts with the post hoc analysis of ENGAGE AF TIME-48 study, which showed that patients randomized to receive a reduced dose of edoxaban had a significantly higher stroke/SE risk (HR 1.31, 95% CI 1.12–1.52, p < 0.001) than those on the standard dose. 24 Notably, Asian participants consititued only 13.8% of that trial's population. Moreover, patients in our cohort were notably lighter (64 vs 82 kg), and presented with a lower burden of comorbidities (including hypertension, heart failure, and prior stroke) than those in ENGAGE AF TIME-48 study. The extent to which these racial and baseline differences account for the discrepant IS/SE risk estimates remains unclear. Of note, current studies suggested that underdosed edoxaban may effectively prevent thromboembolic events in Asian patients with AF. The ELDERCARE-AF trial, conducted in Japan, demonstrated that a once-daily 15 mg dose of edoxaban was superior to placebo in reducing stroke/SE risk in elder AF patients (HR 0.34, 95% CI 0.19-0.61). 25 Another study utilizing data from Taiwan's multi-center healthcare system showed that, regardless of whether patients met the dose reduction criteria, underdosed edoxaban (30 mg or 15 mg once daily) was associated with a lower risk of IS compared to warfarin (HR 0.39, 95% CI 0.17-0.86). 26 A similar phenomenon has been observed with other DOACs in Asian populations. 27 Collectively, these data provide reassuring evidence that underdosed edoxaban may maintain efficacy for thromboembolism prevention in Asian patients with AF. Nevertheless, the wide confidence interval in our analysis reflects statistical imprecision, likely due to the limited number of IS/SE events. Therefore, while these results are hypothesis-generating and support current regional prescribing practices, they underscore the need for larger studies to confirm the efficacy of underdosed edoxaban in this demographic.
In terms of safety, our study found that no statistically significant difference in the risk of major bleeding between under dosed and recommended dose edoxaban users. This contrasts with the pre-specified analysis of ENGAGE AF TIME-48 trial, which demonstrated a significantly lower risk of major bleeding in patients randomized to reduced-dose edoxaban (HR 0.64, 95% CI 0.55–0.74, p < 0.0001) compared to standard-dose therapy. 24 The discrepancy may be attributed to the limited statistical power in our cohort, where low event rates resulted in imprecise effect estimates, as reflected in the wide confidence interval (HR 0.19, 95%CI 0.01–2.12). Although the point estimate suggests a potential protective effect, the data remain consistent with both a substantial risk reduction and possible harm. Therefore, the findings regarding major bleeding should be interpreted with considerable caution. Notably, a lower incidence of CRNM bleeding was observed in the under dosed group. Although CRNM bleeding is less severe than major bleeding, its clinically significance is substantial due to a higher incidence and the potential for serious consequences, including hospitalization and discontinuation of anticoagulation therapy, which may ultimately lead to worse clinical outcomes. 28 However, given the borderline statistical significance of this finding (HR 0.47, 95%CI 0.23–0.95), it should be regarded as exploratory and warrants confirmation in future studies.
Besides, our study revealed there is no significant difference in all-cause mortality between patients receiving under-dosing edoxaban and those receiving recommended dose edoxaban. This finding was consistent with the ENGAGE AF TIME-48 study. 24 In contrast, the Global ETNA-AF study reported that patients who received the under-dose of 30 mg edoxaban had a higher all-cause mortality than those received recommended 60 mg dose. 7 However, baseline characteristics was not adjusted in that study; patients in the under dosed group were older, had a lower creatinine clearance, and presented more often with cardiovascular comorbidities than those in recommended dose group. This higher burden of advanced age and comorbidities may, in part, contribute to the increased all-cause mortality. 29 In another analysis from the ORBIT-AF II registry, patients who received non-recommended reduced-dose NOACs were at an increased risk of death. 30 However, this association was no longer significant after adjusting for differences in patient characteristics. These findings suggest that the observed all-cause mortality was driven predominantly by baseline patient characteristics rather than by the edoxaban dose.
Several limitations of this study should be acknowledged. First, this was a single-center study with a small sample size and a median follow-up duration of 422 days, which restricted the accurate observation of low-frequency adverse events and led to underpowered analyses with wide confidence intervals for primary outcomes. To address this, we strictly adhered to standardized inclusion/exclusion criteria to ensure cohort homogeneity, combined structured telephone interviews with electronic medical record reviews (including outpatient-inpatient cross-verification) to maximize endpoint capture, and applied stabilized IPTW to optimize baseline covariate balance. Second, although we integrated a comprehensive covariate into the IPTW model to adjust for confounding, residual bias from unmeasured factors cannot be ruled out due to the observational nature of the study. Third, the analysis was based on the initial edoxaban dose, as subsequent dose adjustments during follow-up could not be fully captured due to incomplete documentation in outpatient electronic records. To mitigate this, we specifically inquired about medication adherence and dosage changes during the follow-up interviews, but valid information remained limited. Thus, we acknowledged this limitation when interpreting dose-outcome associations, while noting that initial doses reflect real-world clinical decisions and retain clinical relevance. Finally, the study was conducted in a single tertiary-care center in China, and the findings may not be generalizable to other ethnic groups or primary/secondary care settings. To improve representativeness, we detailed the cohort's baseline characteristics for comparison with other populations, and highlighted the need for multi-center, multi-ethnic prospective studies to validate results. Despite these limitations, this study provides valuable real-world insights into the clinical outcomes associated with underdosed edoxaban in Chinese patients with AF. We look forward to future multi-center prospective studies to confirm these results and to establish edoxaban dosing strategies tailored for Asian patients with AF.
Conclusions
In this real-world Chinese tertiary-care cohort, underdosed edoxaban was administered to nearly one-third of patients with non-valvular AF. Compared with the recommended dose, this dosing strategy was associated with comparable risks of IS/SE and major bleeding, as well as a potentially lower incidence of CRNM bleeding. These findings should be considered preliminary and warrant validation in larger, multi-center prospective studies.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251414427 - Supplemental material for Underdosed Edoxaban and Clinical Outcomes in Patients with Non-valvular Atrial Fibrillation in Real World: A Retrospective Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296251414427 for Underdosed Edoxaban and Clinical Outcomes in Patients with Non-valvular Atrial Fibrillation in Real World: A Retrospective Cohort Study by Wenjun Yang, Jieyi Gao, Jie Chen, Jiali Zhang, Lulu Liu, Hongyi Yao and Huimin Xu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We gratefully acknowledge Doctors Yifeng Pan and Xiangkun Meng for their blinded adjudication of all clinical endpoint events.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Clinical Medical Special Research Program of Zhejiang Medical Association, Special Research Program of Pharmacoeconomics and Health Technology Assessment Committee of Zhejiang Pharmaceutical Association, (grant number 2022ZYC-A128, 2023ZYJ10).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
