Abstract
Thrombotic complications after allogeneic hematopoietic stem cell transplantation (allo-HSCT) significantly impact transplant outcomes. We focused on high mobility group box-protein (HMGB)1, one causative agent of thrombotic lesions in allo-HSCT, and investigated its association with platelets. We statistically analyzed available data from 172 patients with hematopoietic malignancies receiving allo-HSCT. A significant enhancement of monocyte-chemotactant protein-1, HMGB1, and platelet-derived microparticle (PDMP) levels was observed at day 0 after transplantation as compared to pre-transplantation. Multivariate analysis of the association among HMGB1 and 16 factors on day 0 revealed a significant correlation of HMGB1 levels with thrombin–antithrombin complex, interleukin-6, and PDMPs. High mobility group box-protein 1-induced procoagulant platelet induction and PDMP generation were performed in vitro using healthy platelets. High mobility group box-protein 1-induced PDMP generation was suppressed by toll-like receptor inhibitors and recombinant thrombomodulin. These results suggest that HMGB1 contributes to platelet activation in patients after allo-HSCT and is associated with PDMP-related thrombotic complications.
Introduction
Platelets are non-nucleated blood elements that play an important role in completing hemostasis by forming a clot at injured physiological sites. At the site of vascular injury, exposed extracellular matrix proteins, such as collagen and von Willebrand factor, initiate platelet activation, resulting in the secretion of secondary mediators, such as adenosine diphosphate (ADP) and thromboxane A2, that rapidly activate nearby platelets. 1 The activated platelets promote coagulation and are, therefore, called procoagulant platelets.2–4 Additionally, procoagulant platelets release small circulating membrane fragments termed platelet-derived microparticles (PDMPs) from their surface.5,6 Procoagulant platelets and PDMPs function as a normal hemostatic response when vessels are injured.4,7 Their mechanism contributes to thrombus formation through tissue factor–mediated thrombin generation.5,7 Therefore, PDMPs have been advocated as an evaluation tool for thrombogenesis. 8 In fact, PDMPs are suggested to be a biomarker for cancer-related thrombosis. 9
High mobility group box-protein 1 (HMGB1) is a nuclear protein with a molecular weight of 30 kDa and is expressed in a variety of cells. 10 High mobility group box-protein 1 binds to nucleosomes to promote DNA binding and participates in the growth of supraspinal processes in neurons. 10 It also acts as a transcriptional regulator within cells. High mobility group box-protein 1 is released extracellularly when cells undergo apoptosis and exhibit other functions thereafter. For example, HMGB1 also has important associations with platelets. 11 High mobility group box-protein 1 acts on toll-like receptor (TLR) 4 in platelet membranes, causing platelet activation and thrombus formation.12,13 Thus, HMGB1 acts as a cytokine, playing an important role in cellular responses to tissue damage.14,15 High mobility group box-protein 1 is sequestered in vivo by thrombomodulin (TM). 16
Hematopoietic stem cell transplantation (HSCT) is a beneficial treatment for blood cancers, but there is concern when it causes serious transplant-related complications, such as graft-versus-host disease (GVHD), venous occlusive disease (VOD), and thrombotic microangiopathy (TMA).17–19 Specifically, VOD, TMA, and stroke are principal complications of transplantation-associated thrombosis (TAC). 20 We conducted a multicenter study of acute complications in post-HSCT patients (SIGHT study) and have reported on some of the data obtained to date.21–25 For example, we demonstrated that HMGB1 levels are significantly elevated after conditioning treatment for HSCT, peaking at day 0. 21 This strongly suggests that elevated HMGB1 levels are closely associated with HSCT-related complications. However, the mechanism of HMGB1-induced VOD/TMA pathogenesis remains poorly understood.
In this study, we retrospectively examined the correlation between HMGB1 and other parameters using patient data from the SIGHT study. Additionally, we performed new in vitro experiments on HMGB1 and platelet activation to assess if platelet activation by HMGB1 contributes to the development of complications after HSCT.
Methods
Study Population
The SIGHT study is a clinical study conducted at 25 centers in Japan and included 289 patients who underwent allogeneic HSCT (allo-HSCT) from January 2010 to April 2016.21–25 The study protocol was approved by the Institutional Review Board of Kansai Medical University. Written informed consent was obtained from all enrolled patients through faxed documents submitted to Kansai Medical University prior to HSCT. Clinical and biochemical data were collected before allo-HSCT (day 7) and at day 0 after allo-HSCT. The present subanalysis included data from 172 patients for whom targeted laboratory data were obtained. Patients were divided into 2 subgroups based on their HMGB1 levels at day 0 after allo-HSCT. Group A was defined as patients showing >1.5-fold increase in plasma HMGB1 levels after allow-HSCT relative to levels before beginning the conditioning regimen (day 7). Group B was defined as those with <1.5-fold increase in plasma HMGB1.
Blood Sampling and Detection of Molecular Markers
Blood samples were collected from enrolled patients in tubes containing sodium citrate or without anticoagulant. Serum or citrate plasma was aspirated after centrifugation at 1000 × g for 20 min at 4 °C. Serum was frozen at −30 °C. High mobility group box-protein 1 was measured using an HMGB1 enzyme-linked immunosorbent assay (ELISA) Kit II (Sinotest). ELISA kits for soluble vascular cell adhesion molecule-1, plasminogen activator inhibitor, interleukin-6 (IL-6), tumor necrosis factor-α, and monocyte-chemotactant protein (MCP)-1 were purchased from BioSource International, Inc. Blood for PDMP samples was collected from a peripheral vein with a 21-gauge needle, placed in a vacuum container containing ethylenediaminetetraacetic acid (EDTA) to inhibit platelet activation, and mixed by inversion. The samples were kept at room temperature for 2 to 3 h, and 200 μL of the supernatant fluid was collected immediately after centrifugation at 8000 × g for 5 min to prevent platelet contamination. Collected samples were stored at −40 °C until analysis. 26 The ELISA kit used to measure PDMP was obtained from JIMRO Co. All ELISA kits were performed according to the manufacturer's instructions.
Reagents
Recombinant TM (rTM) was purchased from Asahi Kasei Pharma; ADP, prostaglandin I2 (PGI2) sodium salt, apyrase, thrombin receptor activating peptide (TRAP), SB203580 (p38MAPK inhibitor), BYL719 (PI3K inhibitor), BAY1125976 (AKT/PKB inhibitor) were purchased from Sigma-Aldrich, eritoran (TLR inhibitor) and HMGB1 were purchased from R&D System, and the collagen reagent HORM was purchased from Funakoshi Pharmaceuticals.
Flow Cytometry
Blood samples were collected in 8.6-mL sterile acid-citrate-dextrose (ACD) tubes (Becton Dickinson) and inverted so that the whole blood and ACD were thoroughly mixed before flow cytometry (FACScan) analysis. Platelet activation was measured under both unstimulated and TRAP-activated conditions. Fluorescently labeled antibodies were obtained from BD Biosciences. For detection of unstimulated and ADP- or HMGB1-activated PAC-1 binding, freshly collected whole blood (20 μL) was first diluted with buffer (180 μL) and costained (10 μL) with PAC-1, a fluorescein isothiocyanate (FITC) antihuman mouse monoclonal antibody recognizing CD41-PE and GP IIbIIIa, to measure PAC-1 expression. 27 The same type of IgG1-FITC and IgG2-PE antibodies was used as controls for nonspecific fluorescence. Blood samples were incubated at room temperature (20°C-25°C) for 10 min in the dark and then fixed by adding 250 μL of FACS buffer. For measurement of PAC-1 binding, a total of 10,000 CD on a 2-parameter dot plot displaying both side scatter and forward scatter CD41 positive events were selected by gating platelets.
Generation of PDMPs From Platelet-Rich Plasma by HMGB1
Platelet-rich plasma from healthy subjects (n = 5) was treated with purified HMGB1. After treatment with various concentrations (100-1200 ng/mL) for 60 min, PDMPs were collected, their concentrations were measured 5 times by ELISA, and the average value was calculated. Various inhibitors (SB203580; p38MAPK inhibition, BYL719; PI3K inhibition, BAY1125976; AKT/PKB inhibition, eritoran; TLR inhibition) were also added prior to HMGB1 addition, and similar experiments were performed.
Statistical Analysis
We performed a stratified analysis of patient characteristics by high and low HMGB1 levels on day 0 using the Chi-square test. Data for several biomarkers were expressed as mean ± standard deviation and were analyzed using multivariate regression analysis, as appropriate. Between-group comparisons were examined using the Newman-Keuls test and Scheffe's test. The correlation between HMGB1 concentration and continuous variables was assessed using multivariate linear regression analysis. All analyses were performed using StatFlex (version 7; Artec Corporation). Statistical significance was set at P < .05.
Results
Table 1 displays patient and treatment characteristics. Patients consisted of 108 males and 64 females aged 18 to 77 years, with an average age of 45 years. Of the patients, 76, 41, 28, and 27 patients were diagnosed with acute myelogenous leukemia, acute lymphoblastic leukemia, myelodysplastic syndrome, and other conditions, respectively. Forty-seven patients (27.3%) received a related donor transplant, and 125 (72.7%) received an unrelated donor transplant. Ninety-eight patients (57.0%) had an HLA-matched donor, and 74 (43.0%) had an HLA-mismatched donor. One hundred-nine patients (63.6%) received a myeloablative conditioning (MAC) regimen, and 63 (36.4%) received a reduced-intensity conditioning (RIC) regimen. The most common source of stem cells was the bone marrow (53.5%). In patients stratified by HMGB1 value of day 0, the trend toward higher HMGB1 values in MAC as compared to RIC was observed (Table 1).
Baseline Patient Characteristics (N = 172).a
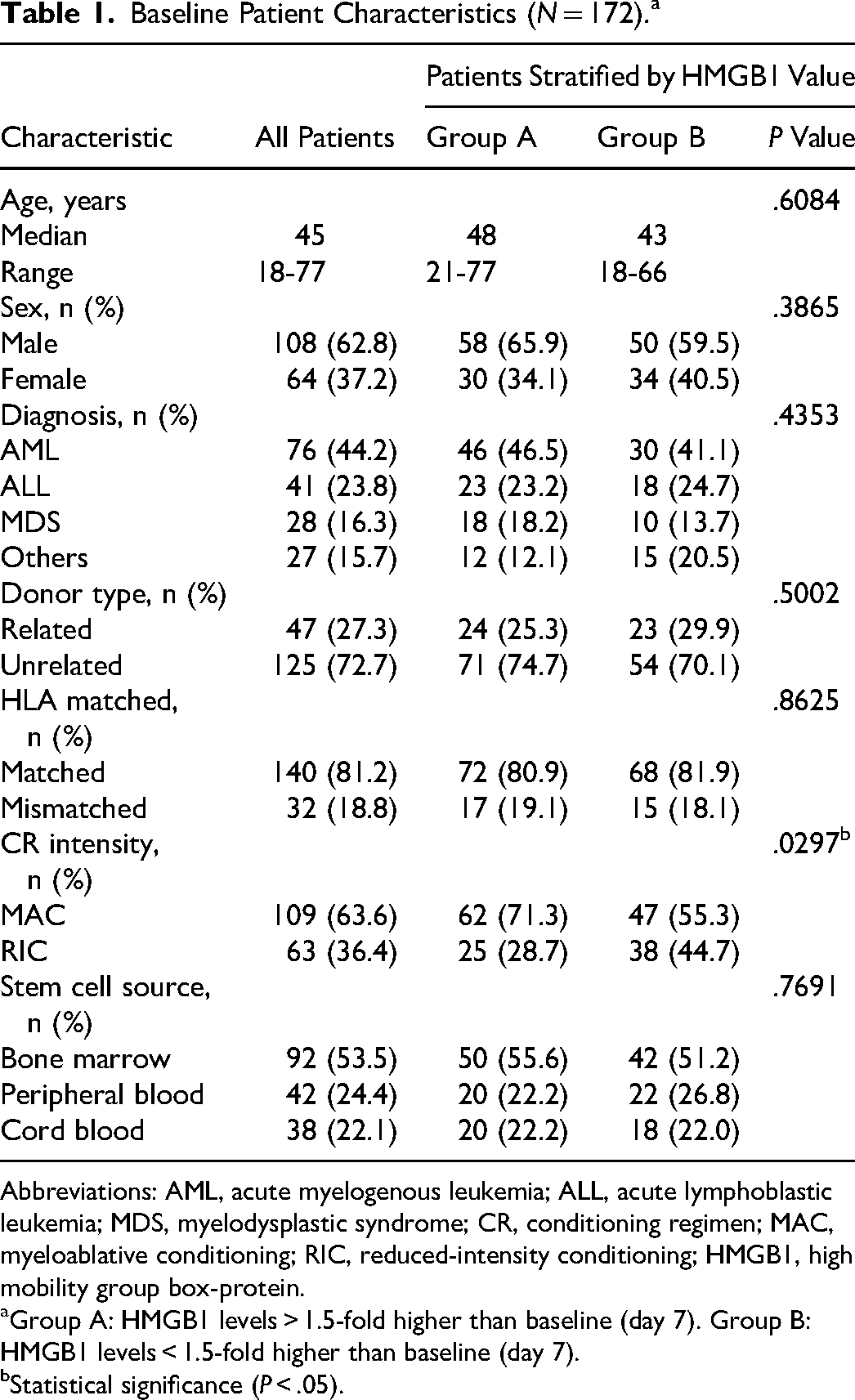
Abbreviations: AML, acute myelogenous leukemia; ALL, acute lymphoblastic leukemia; MDS, myelodysplastic syndrome; CR, conditioning regimen; MAC, myeloablative conditioning; RIC, reduced-intensity conditioning; HMGB1, high mobility group box-protein.
Group A: HMGB1 levels > 1.5-fold higher than baseline (day 7). Group B: HMGB1 levels < 1.5-fold higher than baseline (day 7).
Statistical significance (P < .05).
Table 2 compares the results of measurements of special molecular markers before and on day 0 after transplantation. However, a significant increase in MCP-1, HMGB1, and PDMP was observed at day 0 after transplantation compared to pretransplantation. The increase in HMGB1 was particularly marked (P < .0001).
Comparison of Molecular Markers Before and After Receiving HSCTa.
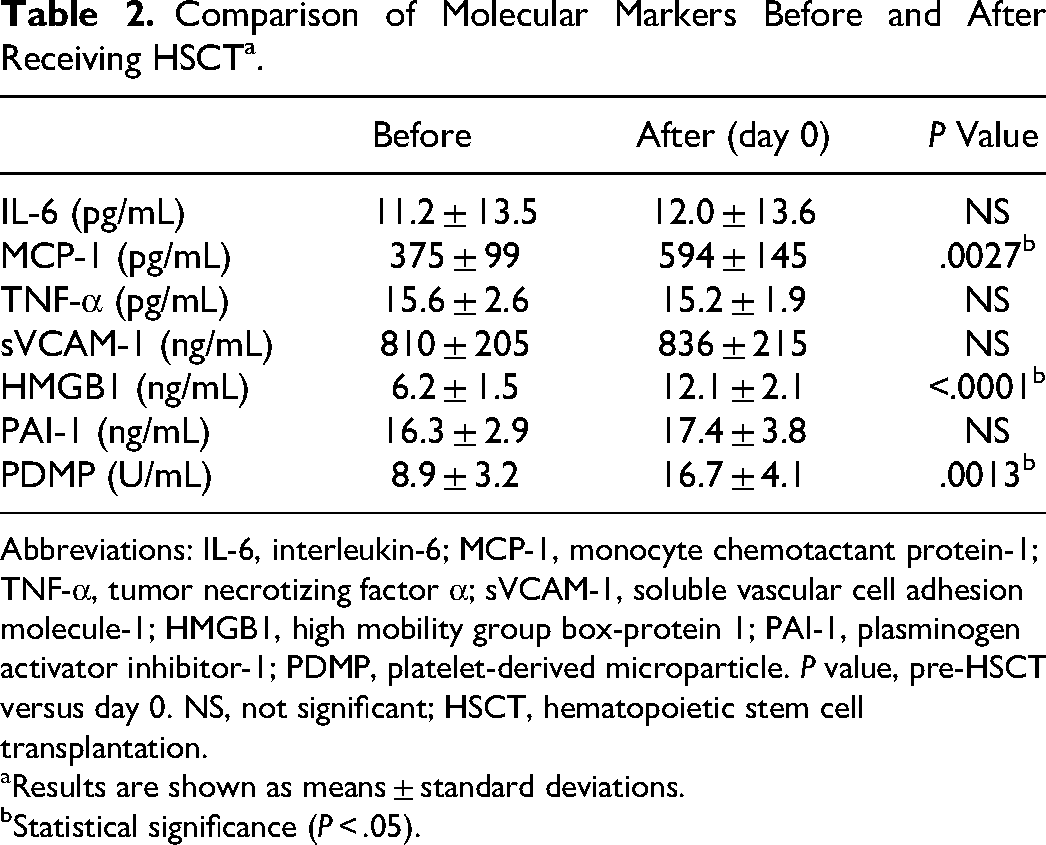
Abbreviations: IL-6, interleukin-6; MCP-1, monocyte chemotactant protein-1; TNF-α, tumor necrotizing factor α; sVCAM-1, soluble vascular cell adhesion molecule-1; HMGB1, high mobility group box-protein 1; PAI-1, plasminogen activator inhibitor-1; PDMP, platelet-derived microparticle. P value, pre-HSCT versus day 0. NS, not significant; HSCT, hematopoietic stem cell transplantation.
Results are shown as means ± standard deviations.
Statistical significance (P < .05).
We used univariate and multivariate regression analyses to investigate the association between 16 variables and HMGB1 concentrations (Table 3). In the univariate analysis, lactate dehydrogenase, total bilirubin,
Multivariate Analysis on HMGB1 in HSCT Patients.
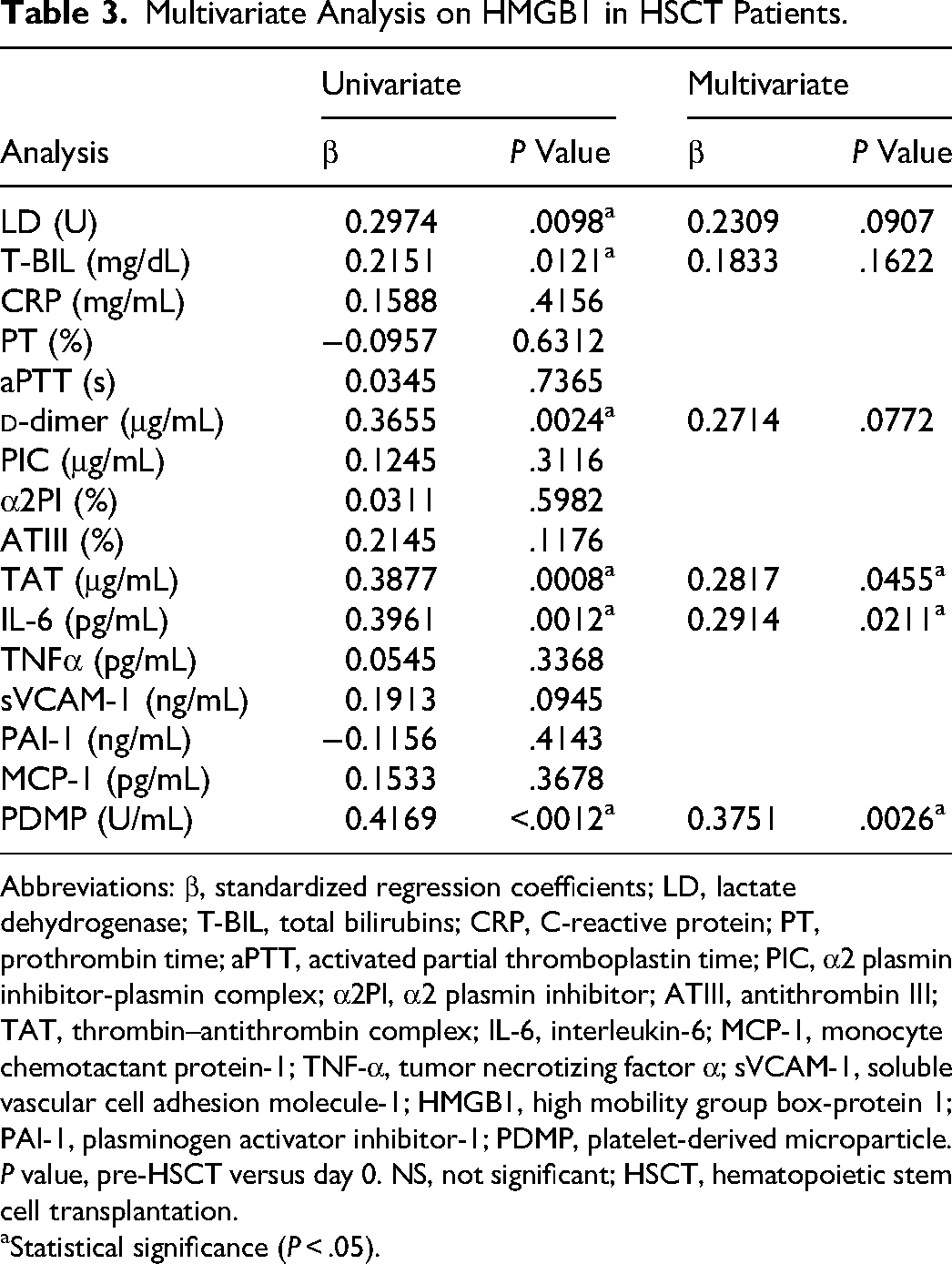
Abbreviations: β, standardized regression coefficients; LD, lactate dehydrogenase; T-BIL, total bilirubins; CRP, C-reactive protein; PT, prothrombin time; aPTT, activated partial thromboplastin time; PIC, α2 plasmin inhibitor-plasmin complex; α2PI, α2 plasmin inhibitor; ATIII, antithrombin III; TAT, thrombin–antithrombin complex; IL-6, interleukin-6; MCP-1, monocyte chemotactant protein-1; TNF-α, tumor necrotizing factor α; sVCAM-1, soluble vascular cell adhesion molecule-1; HMGB1, high mobility group box-protein 1; PAI-1, plasminogen activator inhibitor-1; PDMP, platelet-derived microparticle. P value, pre-HSCT versus day 0. NS, not significant; HSCT, hematopoietic stem cell transplantation.
Statistical significance (P < .05).
Figure 1 illustrates flow cytometry of PAC-1 binding to platelets stimulated with ADP and HMGB1. In contrast, the binding of PAC-1 to HMGB1-stimulated platelets was very slight.

PAC-1 responsiveness to adenosine diphosphate (ADP)- or HMGB1-stimulated platelets. (A) ADP-stimulated platelets (B) HMGB1-stimulated platelets.
Next, the results of HMGB1-stimulated PDMP production from healthy platelet-rich plasma are exhibited in Figure 2, where stimulation with HMGB1 at a concentration of 1200 ng/mL resulted in PDMP production of 42.3 ± 19.1 U/mL. Pretreatment with various inhibitors and rTM for this reaction had no effect on PDMP generation with SB203580 (p38MAPK inhibition), but PDMP production was markedly inhibited by BYL719 (PI3K inhibition), BAY1125976 (KT/PKB inhibition) and eritoran (TLR inhibition), (TLR inhibitor) markedly inhibited PDMP production.
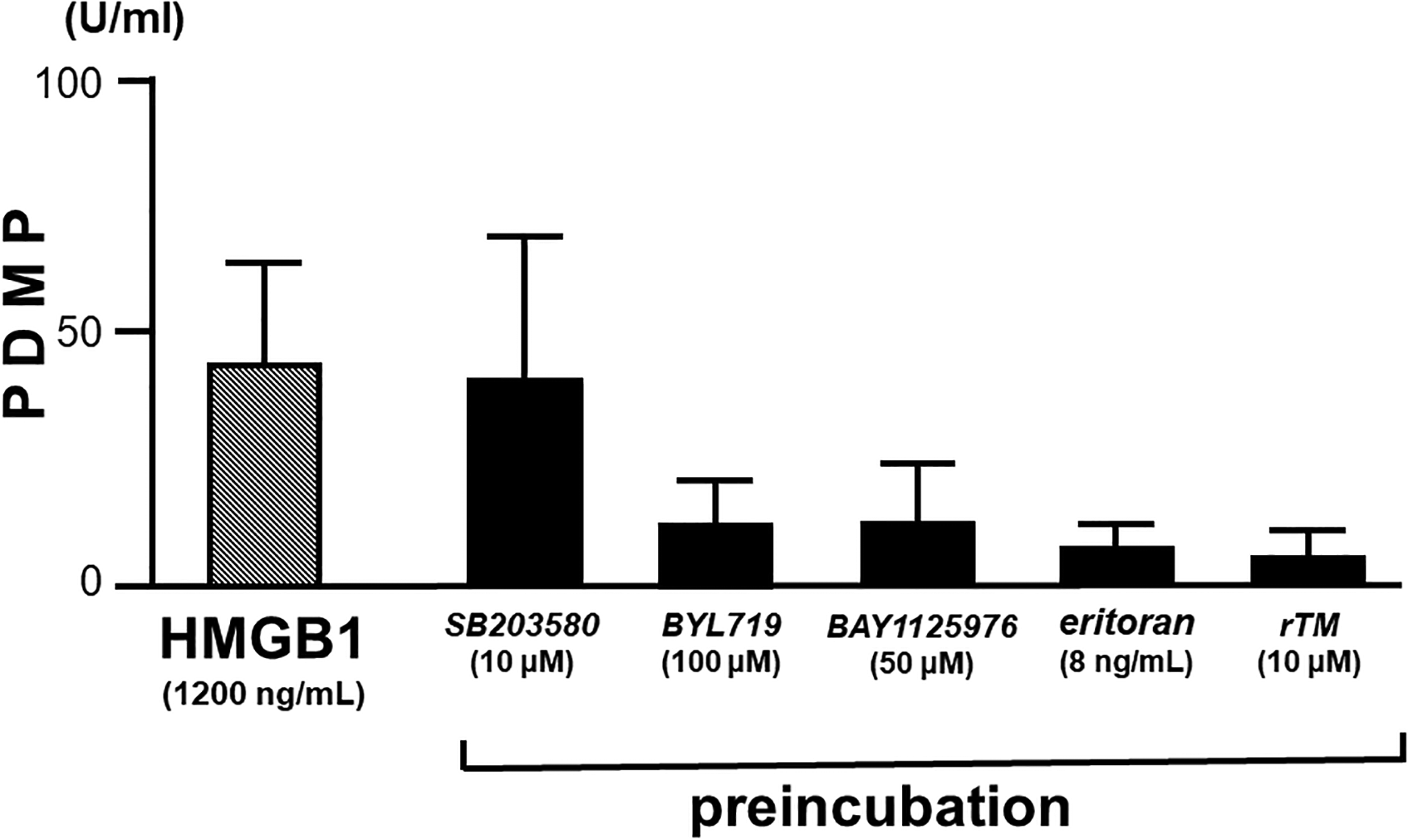
Platelet-derived microparticle (PDMP) generation by HMGB1 and its effect by various inhibitors. Five experiments were performed using platelets from healthy subjects. Results are shown as means ± standard deviations.
Discussion
Free HMGB1 in the blood is important as a tissue damage factor (DAMPs) and causes hypercoagulable pathologies.15,28,29 In this study, we observed elevated HMGB1 levels on day 0 after transplant. This finding suggests that HMGB1 may be involved in TAC development after conditioning in HSCT. 21 Cutler et al 30 reported that vascular augmentation engages in TAC development. Although the etiology of VOD is still unknown, endothelial cell dysfunction is a predictive indicator of VOD. 31 High mobility group box-protein 1 is reportedly involved in the pathophysiology of various inflammatory and immunological diseases.16,29 Additionally, the multivariate analysis indicated that HMGB1 may participate in inflammatory changes and thrombogenicity after transplantation. Several studies have investigated the role of HMGB1 in HSCT,32–34 and Tagami et al 32 described that HMGB1 is implicated in mobilizing stem cells from the bone marrow and circulating them throughout the body. Yujiri et al 33 discovered that HMGB1 can be a useful marker for acute GVHD. Furthermore, Kornblit et al 34 reported that genetic variants in HMGB1 are associated with post-HSCT outcomes. However, these previous reports do not discuss the association between HMGB1 concentrations and platelets after HSCT. For our post-conditioning HMGB1 levels, the association with PDMPs was remarkable. The association between HMGB1 and PDMPs may be determined in the thrombogenicity after transplantation.
To clarify the association between HMGB1 and PDMPs, several in vitro experiments were performed on healthy platelets. We examined the binding of PAC-1 (an antibody recognizing the conformational change in GPIIb/IIIa) to platelets and established that PAC-1 binding to HMGB1 was very mild as compared to ADP-stimulated platelets. This suggests that HMGB1-stimulated platelets are in a nonactive state and therefore cannot exhibit complete platelet aggregation. The retention of platelet aggregation capacity is a strict difference between fully activated platelets and procoagulant platelets. Platelets that demonstrate procoagulant activity are classically referred to as procoagulant platelets, a concept that has been further elucidated in recent years.35–37 Procoagulant platelets also share the following 5 common characteristics: (1) a change in mitochondrial polarity within platelets, (2) an increase in intracellular calcium concentration, (3) expression of phosphatidylserine on the platelet surface, (4) deactivation of platelet membrane glycoprotein GPIIb/IIIa, and (5) increased expression of procoagulant-associated proteins. Specifically, GPIIb/IIIa deactivation indicates that procoagulant platelets are not actively involved in platelet aggregation itself, which is considered a robust platelet response. 35 A low frequency of procoagulant platelets (<20%) leads to a bleeding tendency, whereas a high frequency (>50%) leads to a thrombotic tendency. 38 Controlling platelet procoagulant activity may potentially control thrombosis and hemostasis in many diseases. 38 Thus, the induction of procoagulant platelets by HMGB1 is an important cause of thrombotic diseases. In contrast, procoagulant platelets are also closely related to PDMP production.38,39 That is, when procoagulant platelets are generated, PDMPs are simultaneously generated.
We also examined HMGB1-stimulated PDMP generation. We previously demonstrated that purified HMGB1 generates PDMPs in a dose-dependent manner in in vitro experiments. 9 In this study, PDMP generation of 42.3 ± 19.1 U/mL was also observed when stimulated with HMGB1 at a concentration of 1200 ng/mL. Furthermore, this generation was not affected by p38MAPK inhibitors but was markedly inhibited by PI3K, KT/PKB, and TLR4 inhibitors. In addition, pretreatment with rTM also markedly inhibited PDMP generation. This result indicates that platelet activation is partially occurring, and although thrombin is produced, GPIIb/IIIa is still not activated, making it unable to reach full platelet aggregation capacity. 37
Procoagulant platelets and PDMPs are important in normal hemostasis, but can also cause serious thrombotic disease in some cases.16,40 We demonstrated that HMGB1 concentrations increased significantly after HSCT-conditioning treatment, with a peak at day 0. In particular, the trend toward higher HMGB1 values in MAC as compared to RIC was observed in patients stratified by HMGB1 day 0 values. Furthermore, HMGB1 levels at day 0 correlated with PDMP levels. This association between HMGB1 and PDMPs may induce hypercoagulability after HSCT. Recombinant TM was suggested to inhibit these responses by a different mechanism than inhibition of thrombin function, one of which could be HMGB1 inhibition by a lectin domain. We previously stated that rTM can potentially improve overall survival in HSCT cases. 24 However, the mechanism by which HMGB1 causes complications and whether rTM effectively prevents them remains to be fully elucidated. This study demonstrated that HMGB1 responded to quiescent platelets and promoted the formation of procoagulant platelets and PDMPs, which sheds some light on the mechanisms underlying the development of certain complications that occur in vivo after HSCT. Unfortunately, however, we were unable to determine a relationship between the actual VOD/TMA that developed and the findings of HMGB1 and PDMP in the present study. The VOD/TMA cases had different onset times, and the frequency of VOD/TMA cases may have differed depending on whether anticoagulation therapy was administered. Further studies employing larger patient cohorts are needed to confirm whether changes in the levels of these markers contribute to TAC following aHSCT.
This study has some limitations. First, we were unable to directly examine the association of procoagulant platelets with posttransplant thrombogenicity. The detection of procoagulant platelets in patient blood using flow cytometry would be necessary. Second, this study did not conduct a multivariate analysis of factors influencing the treatment effect on HMGB1 or PDMP following rTM therapy. Further investigation of these factors is necessary in subsequent studies with additional cases. Third, as the study exclusively enrolled Japanese participants, the generalizability of the findings to other populations is limited. Lastly, no evidence was obtained for a direct relationship between VOD/TMA and PDMP due to the presence or absence of anticoagulation therapy used, which may have had an impact. Although previous research has demonstrated a significant association between rTM therapy and TMA, 20 the participation of rTM therapy among patients in this study was unclear. Thus, further examination of this issue is warranted.
Footnotes
Authors’ Note
Institutional Review Board approval for this investigation was granted by the Medical Ethics Committee of Kansai Medical University (study number 2022021).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
