Abstract
We measured high mobility group box 1 protein (HMGB1) and platelet-derived microparticles (PDMP) in blood samples from patients with untreated type 2 diabetes mellitus (T2DM). We examined the effects of a combination of sodium/glucose cotransporter 2 (SGLT2) inhibitors and dipeptidyl peptidase-4 (DPP-4) inhibitors. Multiple regression analysis of HMGB1 was conducted on data from 252 patients in our previously reported T2DM-related clinical study. The results revealed significant correlations between HMGB1 and PDMP, soluble CD40 ligand, plasminogen activator inhibitor-1, and soluble E-selectin in multivariate analysis. Based on the HMGB1 levels before treatment with combination, 46 T2DM patients in the study were classified into two groups, high and low. The high HMGB1 group showed a significantly lower adiponectin level and higher PDMP production than the low HMGB1 group. T2DM risk significantly and positively correlated with HMGB1 and PDMPs. HMGB1-induced PDMP production was simulated in vitro using healthy platelets. Furthermore, The combination of a SGLT2 inhibitor and a DPP-4 inhibitor significantly reduced HMGB1 and PDMP levels. These results suggest that in addition to abnormal glucose metabolism, HMGB1-dependent PDMP production and the resulting development of atherosclerosis are also a concern in patients with T2DM.
Introduction
High mobility group box 1 (HMGB1) is known to regulate diverse biological processes depending on its intracellular or extracellular localization sites. 1 It broadly detects cellular stress and is important in balancing cell death and the survival responses that are essential for cellular homeostasis and tissue maintenance. 2 We recently reported that HMGB1 stimulates platelets to induce the production of procoagulant platelets and platelet-derived microparticles (PDMPs). 3 PDMPs produced from procoagulant platelets activate the coagulation system and play an important role in thrombogenesis.4–7 PDMPs also induce the expression of cell adhesion molecules in leukocytes and vascular endothelial cells, enhancing the adhesion between them and accelerating the formation of early atherosclerotic lesions. 8
Type 2 diabetes mellitus (T2DM) is a condition in which blood glucose levels are chronically elevated due to insufficient insulin activity. T2DM is the most prevalent disease globally, accounting for more than 90% of DM cases.9,10 The etiology of T2DM is not fully understood, but the ferroptosis of pancreatic β cells leading to glucose-stimulated insulin secretion (GSIS) failure and arsenic-induced pancreatic injury has been considered as one pathogenic cause. 11 Ferroptosis, first reported by Stockwell and Dixon in 2012, is a type of iron-dependent programmed cell death characterized by the accumulation of lipid peroxides. 12 It is a genetically and biochemically distinct from apoptosis, and glutathione peroxidase 4 (GPX4) and oxidoreductase (FSP1) are necessary for this cell death process.12,13 Ferroptosis of pancreatic β cells are consequently mobilizes HMGB1, which leads to diabetic complications such as myocardial ischemia and diabetic cardiomyopathy.14–16
Reactive oxygen species (ROS) are generated in cells by various factors, including mitochondrial metabolic activity and inflammatory responses.17,18 Since excess ROS can be harmful to the organism, superoxide dismutase (SOD) plays an important role in maintaining ROS balance.17–19 In T2DM, ROS are generated due to excessive cellular metabolism and the lack of antioxidants, which induce oxidative stress. 20 Pancreatic β cells are particularly vulnerable to ROS and thus increasingly susceptible to oxidative stress. Oxidative stress in T2DM causes multiple diabetic complications, including cardiovascular disease, neuropathy, renal damage, and retinopathy. 20 Therefore, in managing T2DM, controlling oxidative stress is also a key medical issue. 21
Oral antidiabetic drugs, such as sodium-glucose co-transporter 2 (SGLT2) inhibitors and dipeptidyl peptidase-4 (DPP-4) inhibitors, have shown efficacy in improving hemoglobin A1c (HbA1c) levels by lowering circulating blood glucose levels.22–24 SGLT2 inhibitors primarily function by inhibiting glucose reabsorption in the renal proximal tubules, which leads to reduced circulating blood glucose. 22 On the other hand, DPP-4 inhibitors potentiate the effect of incretin and induce insulin secretion, contributing to its hypoglycemic effect. 24 SGLT2 inhibitors are expected to have cardiovascular protective effects in addition to their hypoglycemic effects.23,25 DPP-4 inhibitors have been reported to have anti-inflammatory effects in addition to promoting insulin secretion.24,26 In our recent K-CAM-1 study, we investigated the effects of both drugs on markers related to glucose metabolism and atherosclerosis. We found that the combination reduced the level of soluble lectin-like oxidized LDL receptor (sLOX)-1, a biomarker of atherosclerosis. 27 In this study, we quantified HMGB1 and PDMP in untreated T2DM patients using patient samples from four previously reported clinical studies on DM.28–31 We examined the effects of the combined SGLT2 inhibitor and DPP-4 inhibitor therapy on HMGB1 and PDMP levels in patients with T2DM based on the K-CAM-1 study.
Methods
Study Population
The study included 252 patient samples from four previously reported clinical studies28–31 on diabetes and 46 patients with T2DM from the K-CAM-1 study. 27
For the study of untreated T2DM patients, HMGB1 was set as the objective variable, and univariate and multivariate analyses were conducted with each biomarker. In addition, T2DM patients in the K-CAM-1 study 27 were classified into two groups based on their pretreatment HMGB1 levels, high and low, and the characteristics of the high HMGB1 group were examined. The patients in the K-CAM-1 study, those on treatment with once-daily DPP-4 inhibitors, were entered. For the first 12 weeks, monotherapy consisted of tofogliflozin (TOFO) 20 mg once daily and anagliptin (ANA) 100 mg twice daily. The same volume of both drugs was then used in combination for 12 to 48 weeks for observation. Regarding the treatment effect of TOFO/ANA, we measured markers related to glucose metabolism and atherosclerosis before and after treatment in the 46 T2DM patients in the K-CAM-1 study. 27
Blood Sampling and Detection of Molecular Markers
Blood samples were collected from enrolled patients in tubes containing sodium citrate anticoagulant or without anticoagulant. Serum or citrate plasma was aspirated after centrifugation at 1000 × g for 20 min at 4 °C. Serum samples were frozen at −30 °C. HMGB1 was measured using an HMGB1 enzyme-linked immunosorbent assay (ELISA) Kit II (Sinotest, Kanagawa, Japan). ELISA kits for soluble vascular cell adhesion molecule (sVCAM-1), plasminogen activator inhibitor (PAI-1), interleukin-6 (IL-6), soluble E-selectin (sE-selectin), regulated on activation, normal T cell expressed and secreted (RANTES), soluble P-selectin (sP-selectin), soluble CD40 ligand (sCD40L) and monocyte-chemoattractant protein (MCP)-1 were purchased from BioSource International, Inc. (Camarillo, California, USA). The plasma adiponectin level was determined using the adiponectin ELISA kit from Otsuka Pharmaceuticals Co. Ltd (Tokyo, Japan). Serum sLOX-1 levels were evaluated using the sLOX-1 ELISA kit (R&D Systems, USA). Blood for PDMP samples was collected from a peripheral vein with a 21-gauge needle, placed in a vacuum container containing ethylenediaminetetraacetic acid (EDTA) to inhibit platelet activation, and mixed by inversion. The samples were stored at room temperature for 2-3 h, and 200 μL of the supernatant fluid was collected immediately after centrifugation at 8000 × g for 5 min to prevent platelet contamination. Collected samples were stored at −40 °C until analysis. 32 The ELISA kit used to measure PDMP was obtained from JIMRO Co. All ELISAs were performed according to the manufacturers’ instructions.
Production of PDMPs from Platelet-rich Plasma by HMGB1
Platelet-rich plasma from healthy subjects (n = 8) was treated with purified HMGB1. After treatment with various concentrations (50-1600 ng/mL) for 60 min, PDMPs were collected, their concentrations were measured eight times using ELISA, and the average value was calculated.
Statistical Analysis
Data for the serum biomarkers are expressed as the mean ± standard deviation (SD) and analyzed by multivariate regression analysis, as appropriate. Between-group comparisons were examined using the Newman–Keuls test and Scheffe's test. The correlation between HMGB1 concentration and continuous variables was assessed by multivariate linear regression analysis. All analyses were performed using StatFlex (version 7; Artec Corporation, Osaka, Japan). Statistical significance was set at P < .05.
Results
In a multiple regression analysis of HMGB1 using data from 252 untreated patients in our previously reported T2DM-related clinical study, significant correlations were found between HMGB1 and PDMP, sCD40L, PAI-1, and sE-selectin in multivariate analysis (Table 1). The analysis of 46 T2DM patients in the study with pretreatment HMGB1 levels classified into two groups, high and low, showed a significant decrease in adiponectin as well as a marked PDMP production in the high HMGB1 group (Table 2). Furthermore, a significant positive correlation was noted among these 46 T2DM patient samples between HMGB1 and PDMPs (r = 0.5872, P < .001) (Figure 1).
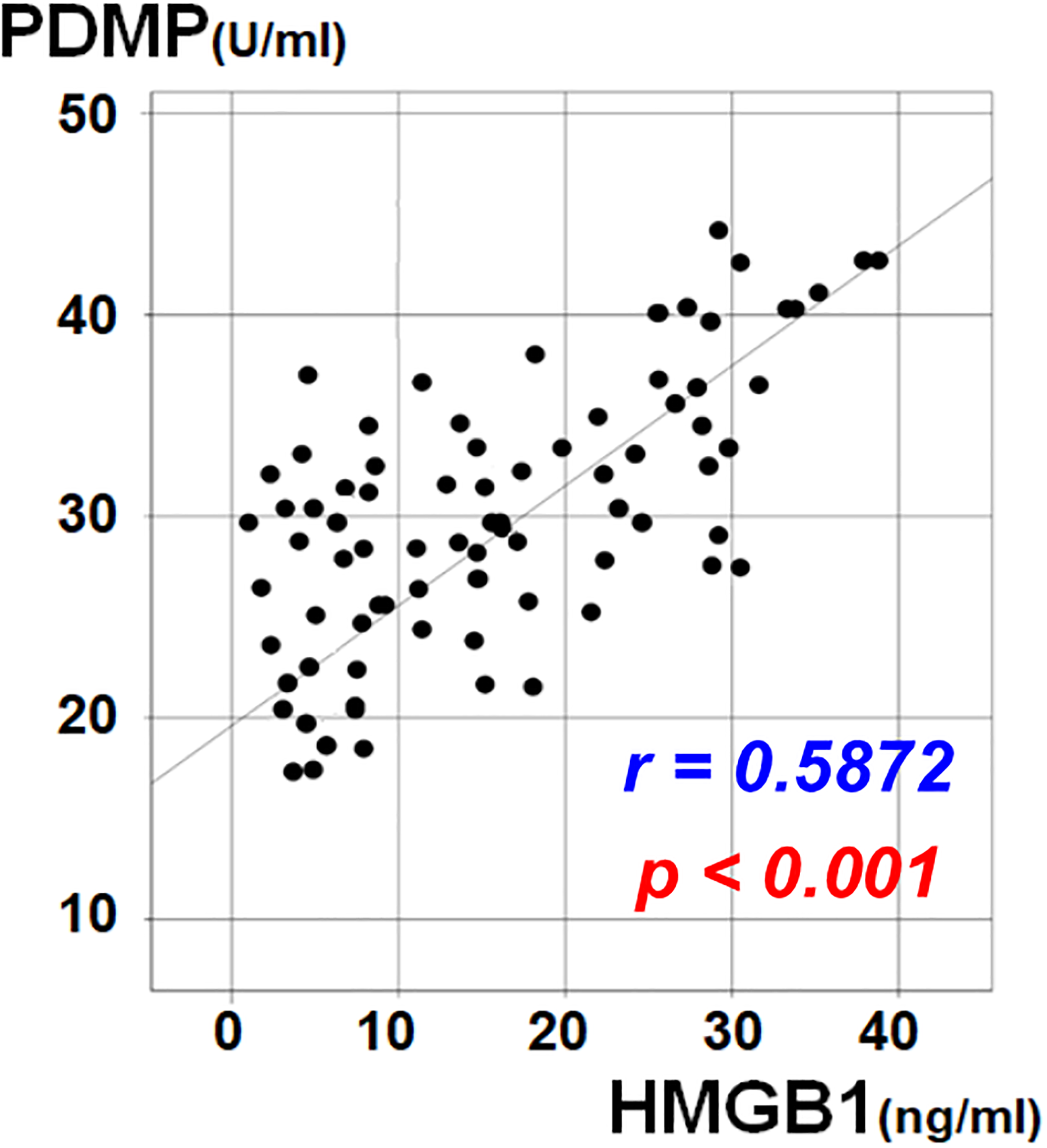
Relationship between HMGB1 and PDMPs in patients with T2DM. PDMP: platelet-derived microparticle; HMGB1: high mobility group box 1. PDMP versus HMGB1; correlation coefficient; r = 0.5872, P < .001.
Multivariate Analysis of HMGB1 in Patients with T2DM.
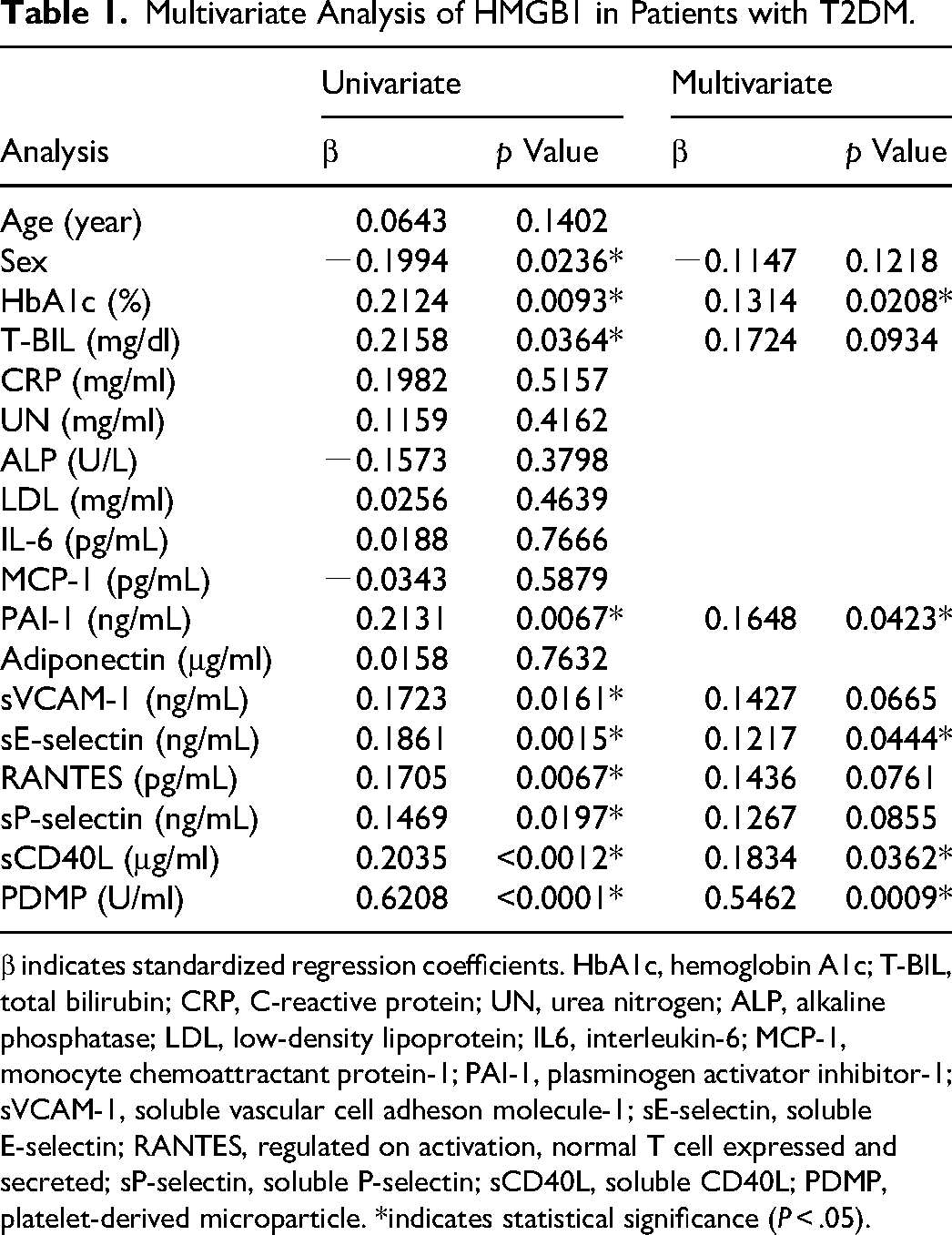
β indicates standardized regression coefficients. HbA1c, hemoglobin A1c; T-BIL, total bilirubin; CRP, C-reactive protein; UN, urea nitrogen; ALP, alkaline phosphatase; LDL, low-density lipoprotein; IL6, interleukin-6; MCP-1, monocyte chemoattractant protein-1; PAI-1, plasminogen activator inhibitor-1; sVCAM-1, soluble vascular cell adheson molecule-1; sE-selectin, soluble E-selectin; RANTES, regulated on activation, normal T cell expressed and secreted; sP-selectin, soluble P-selectin; sCD40L, soluble CD40L; PDMP, platelet-derived microparticle. *indicates statistical significance (P < .05).
Comparison of Levels of Molecular Markers among Patients Stratified by low and High Pretreatment HMGB1 Levels.
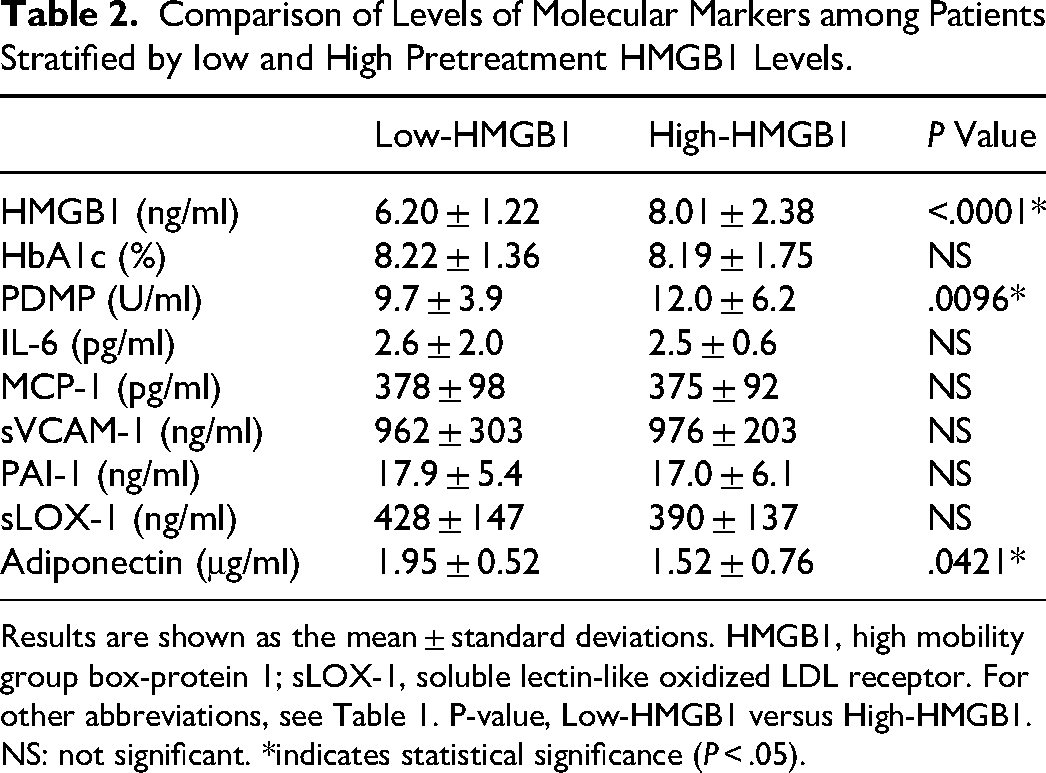
Results are shown as the mean ± standard deviations. HMGB1, high mobility group box-protein 1; sLOX-1, soluble lectin-like oxidized LDL receptor. For other abbreviations, see Table 1. P-value, Low-HMGB1 versus High-HMGB1. NS: not significant. *indicates statistical significance (P < .05).
In an in vitro study using platelet-rich plasma from healthy subjects, PDMP production was observed in an HMGB1 concentration-dependent manner (Figure 2). In particular, a significant increase in PDMP acidity was observed at concentrations above 800 (pg/ml) (Figure 2).
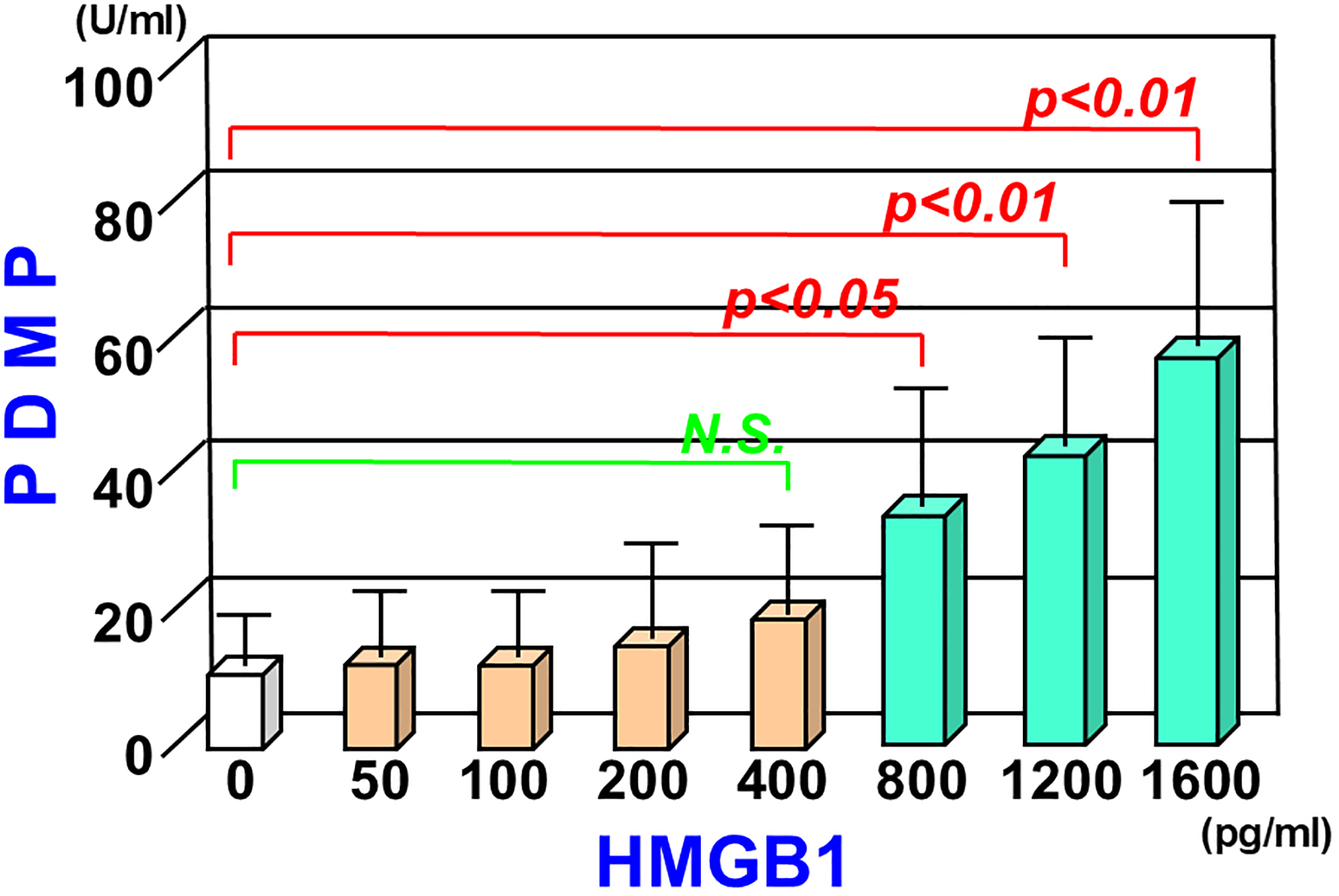
PDMP production under the stimulation of HMGB1 in healthy platelet-rich plasma. Eight repeated measurements were taken using platelets from healthy individuals. Results are shown as the mean ± standard deviation.
As for post-treatment changes in HbA1c, PDMP, and HMGB1 levels, the effects of 12 weeks of monotherapy with TOFO or ANA, as well as combination therapy (24 and 48 weeks), were evaluated by examining the concentrations of each marker. After 12 weeks of TOFO or ANA monotherapy, no significant change was noted in the HbA1c levels (Figure 3A). However, a significant decrease in HbA1c levels was observed with the combination therapy (P < .001) (Figure 3A). Furthermore, 12 weeks of TOFO or ANA monotherapy did not significantly reduce HMGB1 and PDMP levels, whereas the combination therapy significantly reduced HMGB1 and PDMP levels (Figure 3B, C).
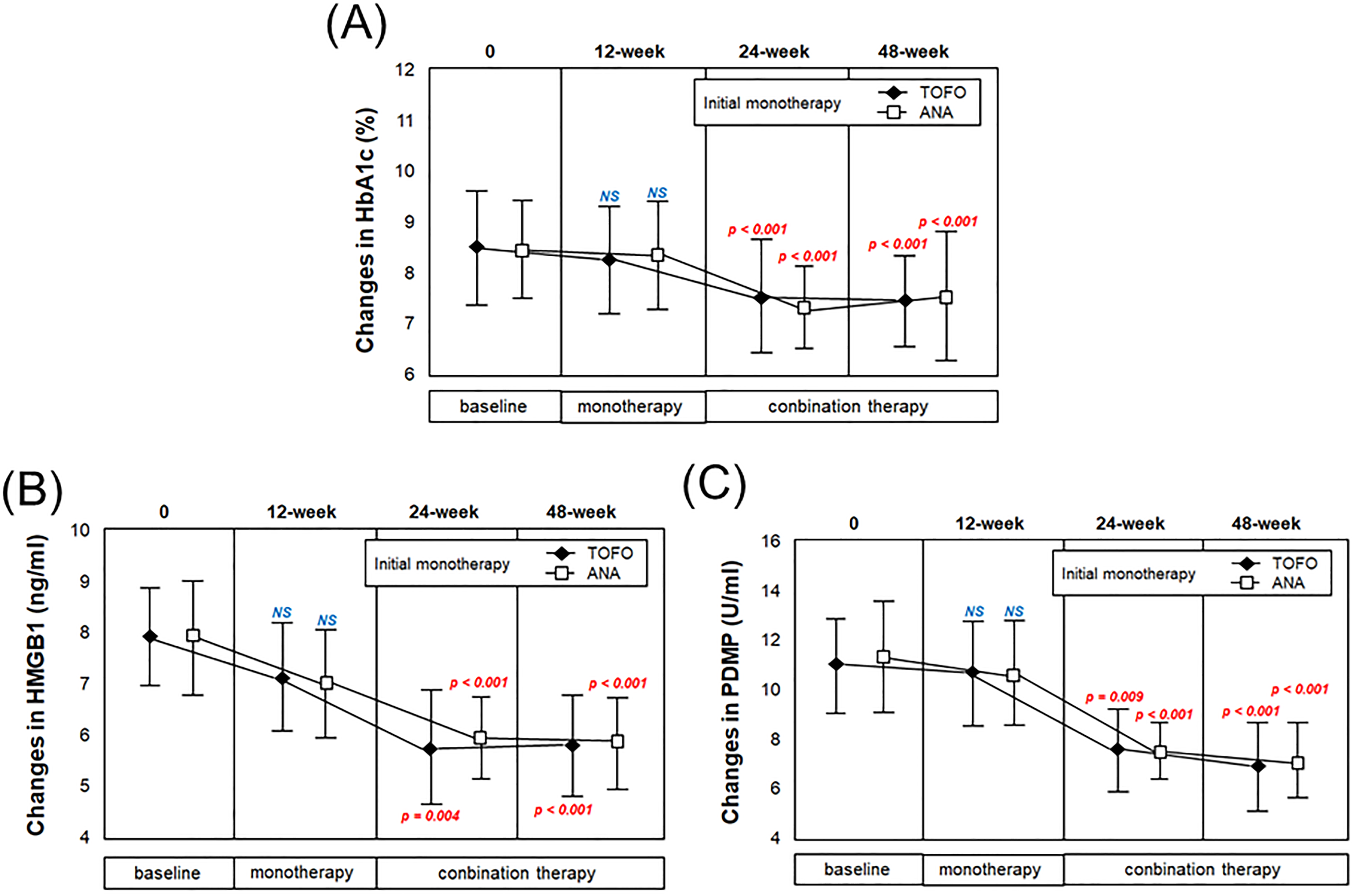
Changes in HbA1c, HMGB1, and PDMP levels following monotherapy and combined therapy. Changes in (A) HbA1c levels, (B) HMGB1 levels, and (C) PDMP levels over time in the TOFO and ANA groups. Assessments were performed at 12- (monotherapy), 24-, and 48-week (combination therapy) using the paired t-test (baseline vs post-treatment at 12-, 24-, and 48-week time points). Red text represents P < .05. NS: not significant.
Discussion
Atherosclerosis is characterized by the accumulation of plaque in the arteries, resulting in stenosis.33–35 Although often asymptomatic in the early stages, it can cause ischemic heart disease, stroke, arteriosclerosis obliterans, and kidney damage in severe cases, depending on the location of the affected artery.35,36 Atherosclerosis generally develops at a young age, and its severity tends to increase with age.33–36 Although the exact etiology of atherosclerosis has not yet been elucidated, several risk factors have been identified, including diet, family history, lipid abnormalities, hypertension, obesity, and smoking.35,36 The simultaneous presence of lifestyle-related diseases, such as diabetes, hypertension, and dyslipidemia, leads to the asymptomatic progression of arteriosclerosis and increases the risk of angina pectoris, myocardial infarction, and cerebral infarction. Several biomarkers related to atherosclerosis have been reported. HMGB1 and procoagulant platelets,37,38 as well as PDMPs, have been studied as biomarkers of atherosclerosis.29–31 In diabetes, ROS produced due to hyperglycemia, excessive production of free fatty acids, and insulin resistance cause oxidative stress, which in turn causes vascular endothelial cell injury, ultimately leading to atherosclerosis.17,21 ROS also plays a major role in the ferroptosis of pancreatic β cells. Ferroptosis, which induces HMGB1 mobilization, has been suggested as a pathogenic mechanism of T2DM (Figure 4).3,14–16
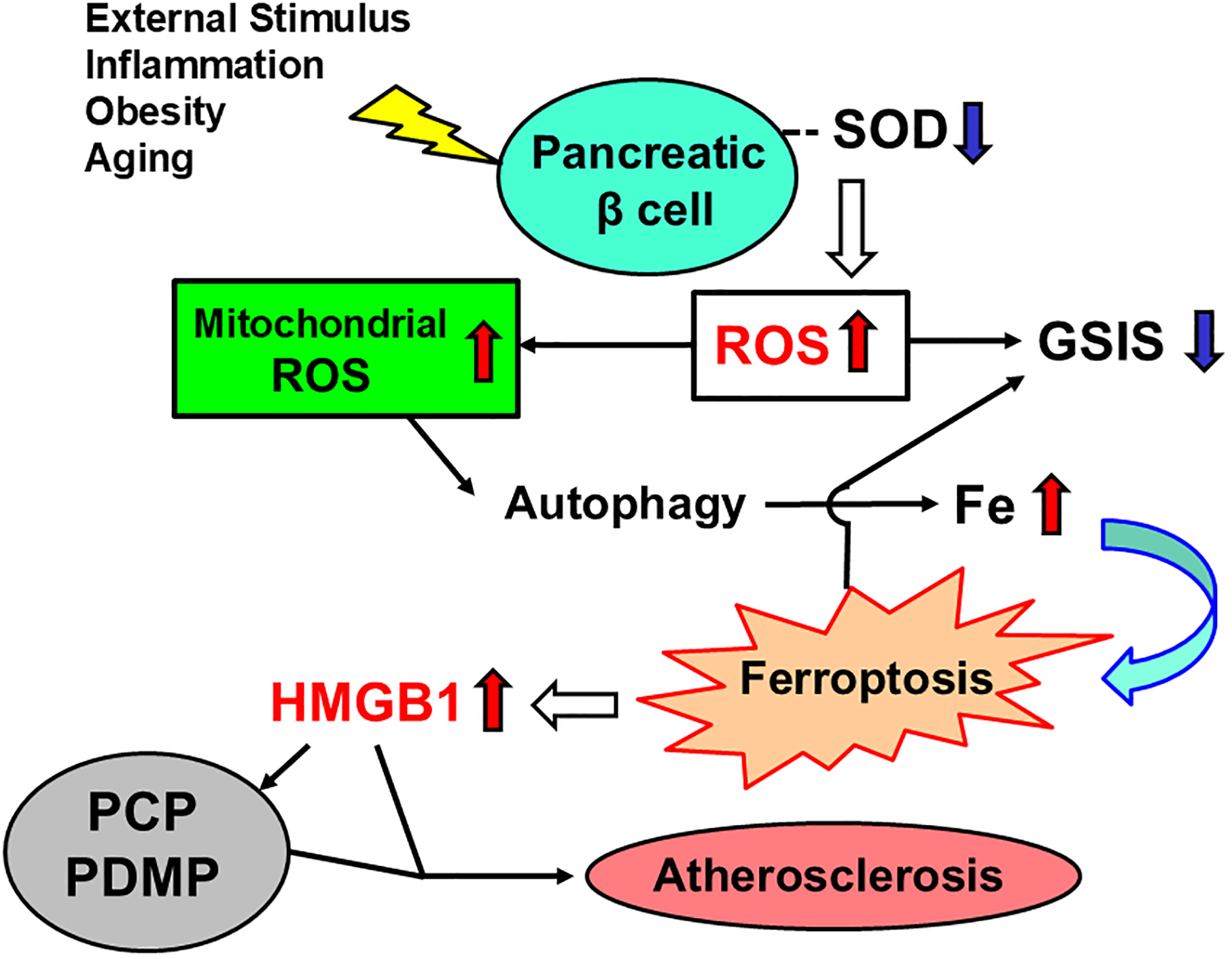
Ferroptosis and HMGB1 production due to ROS in patients with T2DM. SOD, superoxide dismutase; ROS, reactive oxygen species; GCIS, glucose-stimulated insulin secretion; HMGB1, high mobility group box 1; PCP, procoagulant platelet; PDMP, platelet-derived microparticle.
Ferroptosis is caused by the lipid peroxidation of cell membranes, and the mechanism involves iron excess and oxidative stress.12,13 In addition to T2DM, there are various metabolic diseases and illnesses related to ferroptosis, including neurological diseases, renal diseases, and cancer.39–41 Furthermore, the inhibition of ferroptosis is a promising therapeutic strategy for the diseases in which it is involved. 42 In T2DM, the major etiology is decreased insulin secretion due to pancreatic β cell dysfunction, which is also related to why blood glucose levels are difficult to control.43,44 Innovative solutions to preserve pancreatic β cell function are being devised, but it will likely take some time before they materialize in actual clinical practice.45,46 Under the current circumstances, T2DM therapies that inhibit ferroptosis have been demonstrated and are attracting attention as a new approach. 47 We would like to emphasize that the evaluation by several markers related to ferroptosis is important for the success of such an attempt. 48 This may include HMGB1 and PDMP, which are also presented in this study (Figure 4).
In our in vitro study, HMGB1 induced the production of PDMPs from healthy human platelet-rich plasma. In addition, this PDMP production occurred in an HMGB1 concentration-dependent manner. This finding is consistent with the results of the multiple regression analysis of HMGB1 performed on data from untreated T2DM patients in four prior clinical studies. The regression analysis demonstrated a significant correlation between HMGB1 and PDMPs in multivariate analysis (Table 1). Furthermore, the PDMP production was more pronounced in the high HMGB1 group of T2DM patients in the K-CAM-1 study (Table 2). Finally, the significant positive correlation between the HMGB1 level and PDMPs in the 46 T2DM patients from the K-CAM-1 study also supports our concept (Figure 1). Thus, in addition to abnormal glucose metabolism, HMGB1-dependent PDMP production and the resultant atherosclerosis are a cause of concern in patients with T2DM.
The current study also examined the effects of the SGLT2 inhibitor (TOFO) and DPP-4 inhibitor (ANA), the two most popular therapeutic classes for T2DM on HbA1c and atherosclerosis biomarkers. TOFO and ANA did not significantly reduce HbA1c compared to baseline when administered alone. Since the patients in this study those on treatment with once-daily DPP-4 inhibitors were entered, it is possible that some degree of HbA1c improvement was originally observed. This may be the reason TOFO and ANA alone did not lead to a significant decrease in HbA1c. In contrast, the significant reduction in HbA1c levels, as well as the significant reduction in HMGB1 and PDMP levels, after treatment with the combination of TOFO and ANA suggests that this therapy can potentially prevent atherosclerosis and improve abnormal glucose metabolism in T2DM patients. As shown in a previous report, combination therapy with an SGLT2 inhibitor and a DPP-4 inhibitor is likely to be a promising approach in preventing the development of atherosclerosis in diabetic patients,22,27,49 and our results support this hypothesis. For patients with diabetes, fluctuating blood glucose levels reduce quality of life, and long-term blood glucose instability increases the risk of complications. Even considering the psychological burden on the patient, this drug combination may be a useful therapeutic strategy for T2DM.
Conclusion
In addition to abnormal glucose metabolism, HMGB1-dependent PDMP production and the resultant atherosclerosis are a cause for concern in patients with T2DM. While lowering blood glucose and improving HbA1c in T2DM is important, preventing thrombotic complications of this disease is a crucial proposition for the long-term prognosis of patients with T2DM. Therefore, combination therapy with SGLT2 inhibitors and DPP-4 inhibitors for T2DM is likely to prevent atherosclerosis in addition to improving blood glucose levels. Additional clinical data are expected in the future.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Aspects
IRB approval for this investigation was granted by the Medical Ethics Committee of Kansai Medical University (study number 2017009).
