Abstract
Objectives
This study aims to investigate whether PD-1 expressions are abnormal in patients with TAK.
Methods
PD-1 expression was analyzed by flow cytometry. Serum cytokines IL-10, IL-7, IL-2, IL-15, CCL2, CCL3, and CXCL10 were detected using a cytokine cytometric bead array. Immunohistochemistry staining analysis was used to test PD-1 and programmed death-ligand 1 (PD-L1) expression in the aorta of three patients with TAK and three patients with atherosclerosis as controls.
Results
The mean fluorescence intensity of PD-1 in CD4+PD-1+ cells was decreased in patients with TAK and the frequency of CD4+Foxp3−PD-1+ cells among CD4+T cells was also decreased in peripheral blood relative to healthy controls (P < .05). The percentage of CD4+CD25+Foxp3+PD-1+ cells in the CD4+CD25+T cell population was lower in patients with TAK than in healthy control and was lower in active TAK group (P < .05). Comparing PD-1 and PDL-1 expression in aorta tissue showed that patients with TAK tended to have lower levels than patients with atherosclerosis, but the difference was not significant (P > .05). Patients with TAK had higher serum levels of IL-10, IL-7, CCL2, and CCL3 (P < .05).
Conclusions
Abnormal expression of PD-1 in serum and aorta tissue of patients with TAK may contribute to TAK pathogenesis.
Key points
PD-1 expression in both peripheral blood and aorta tissue of TAK patients decreased relative to healthy controls, indicating that PD-1 might be involved in TAK pathogenesis.
Introduction
Takayasu's arteritis (TAK) is a rare type of chronic inflammatory vasculitis that involves large vessels and affects young women of Asian descent with higher frequency. Patients with TAK may present with a variety of symptoms including fever, cervical pain, and tissue ischemia. In severe cases, TAK may be life-threatening due to severe organ ischemia or vessel rupture. 1 The pathogenic mechanism of TAK is unclear, but an imbalance in immune homeostasis may play a crucial role. The activity of regulatory T cells is important for peripheral tolerance and to prevent the development of autoimmune diseases. We previously showed that patients with TAK have decreased numbers of peripheral Treg cells compared to healthy individuals, indicating that abnormal function of Treg cells may be involved in TAK pathogenesis. 2
Treg cell development is regulated by multiple factors. PD-1 is a member of the B7:CD28 coinhibitory signal receptor family and is thought to promote Treg cell differentiation and to inhibit apoptosis as a ligand of PD-L1 or PD-L2. 3 PD-1 dysfunction plays an important role in infection, tumor, and autoimmune diseases including systemic lupus erythematosus, rheumatoid arthritis, Sjogren's syndrome, inflammatory bowel disease, autoimmune hepatitis, Behcet's disease and ankylosing spondylitis. 4 Recent development of anti-PD-1/PD-L1 therapies has produced effective agents to treat various types of tumors. The mechanism of these therapies involves inhibiting the negative regulatory effect of Treg cells in tumor immunity to restore the cytotoxic effect of immune cells toward tumor cells. 5 However, anti-PD-1/PD-L1 therapy can induce autoimmune disease or severe systemic vasculitis, particularly large vessel vasculitis such as giant cell arteritis (GCA). 6 More direct evidence shows that the proportion of CD4+PD-1+ cells in peripheral blood of patients with GCA is decreased, and animal experiments show that blocking PD-1 activity can promote GCA pathogenesis. Together, these results indicate that PD-1 deficiency is likely involved in the pathogenesis of GCA. 7
GCA and TAK share many similarities in terms of clinical manifestations and histopathological changes. These similarities suggest that GCA and TAK represent two phenotypes of the same disease spectrum. 8 In this study we addressed whether patients with TAK have PD-1 deficiency in Treg cells.
Materials and Methods
Study Subjects
A total of 67 patients with TAK presented to Capital Medical University affiliated Anzhen Hospital in China between December 2018 and March 2020. Sixty-four out of 67 TAK patients fulfilled the inclusion criteria for classification of TAK developed by American College of Rheumatology (ACR) in 1990. Three patients were excluded because of combined infections or tumors, or with incomplete clinical data. Thirty-eight age and sex-matched healthy volunteers and three patients with atherosclerosis were enrolled as controls. A flow chart showed the recruitment of study subjects and research contents (Figure 1). The study was approved by the Medical Ethics Committee of Anzhen Hospital.

Sixty-four out of 67 TAK patients fulfilled the inclusion criteria for classification of TAK developed by American College of Rheumatology (ACR) in 1990. Three patients were excluded because of combined infections or tumors, or with incomplete clinical data. Thirty-eight age and sex-matched healthy volunteers and 3 patients with atherosclerosis were enrolled as controls.
Flow Cytometry
Peripheral blood samples were collected and heparinized. The samples were stimulated for 5 h with 20 ng/mL phorbol-12-myristate-13-acetate and 1000 ng/mL ionomycin in the presence of Golgi-Stop (BD Biosciences) and then incubated with anti-human mouse antibodies against CD4-BV510-A (BD Pharmingen), CD25-BV711-A (BD Pharmingen), and CD279-PE-A (BD Pharmingen). After fixation and permeabilization, the samples were incubated with antibodies against Foxp3-Alexa Fluor 647-A (BD Pharmingen). A control antibody having the respective IgG isotype was included in each experiment. Flow assays were carried out on a FACS Calibur flow cytometer (BD, USA) and the data were analyzed with FlowJo v.7.6.4 software (Tree Star).
Cytokine Assay
Sera samples from 49 patients with TAK and 23 healthy controls were collected. Cytokines IL-10, IL-7, IL-2, and IL-15, as well as serum chemokines CCL2, CCL3, and CXCL10 were detected in the samples using a Bio-Plex Pro™ Human Cytokine 27-plex Assay according to the manufacturer's instructions.
Immunohistochemistry Staining Analysis
Aorta specimens from three patients with TAK and three patients with atherosclerosis were fixed in 4% neutral formalin for 24 h. Aortic sections were stained with primary antibodies for PD-1 and PDL-1, and then with secondary antibodies before detection with 3,3′-diaminobenzidine. NIS-Elements BR3.1 software (Nikon) was used for image analysis.
Statistical Analysis
SPSS version 26.0 statistics package (Chicago, IL, USA) was used to analyze the data. Since some data are non-normal distribution, including erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), immunoglobulin(Ig)A, IgG, IgM, complement(C)3, C4, NIH score, mean fluorescence intensity (MFI) of PD-1 in CD4+PD-1+ cells, PD-1, and PDL-1 expression in the aorta, serum levels IL-10, IL-7, CCL2, and CCL3,we described them as median (Q1, Q3). While normal-distributed variables, including age, age of onset, the frequency of CD4+PD−1+ cells, the frequency of CD4+Foxp3−PD-1+ cells, the frequency of CD4+CD25+Foxp3+PD-1+ cells, were described as mean ± SE. Student's t-test or Wilcoxon rank test was used to compare differences between the two groups. P < .05 was considered statistically significant.
Ethics Approval
This study was approved by the Ethics Committee of Beijing Anzhen Hospital, Capital Medical University under ethics approval number 2021077X.
Results
Demographic Characteristics, Laboratory Tests and Numano Type of Patients with TAK
We analyzed the clinical characteristics of samples from 64 patients with TAK. In this study, 92.2% of participants were female and the mean age was 39.32 ± 1.51 years. Most patients were Numano type V (51.6%) (Table 1).
Demographic Characteristics, Laboratory Tests and Numano Type of 64 TAK Patients.

Decreased PD-1 Expression in the Peripheral Blood of Patients with TAK
Flow cytometry of samples from 15 patients with TAK and 18 healthy controls showed that the mean fluorescence intensity of PD-1 in CD4+PD-1+ cells was decreased in the TAK samples [72.2 (45.8, 84.7) versus 363.00 (335, 431), P = .000], but the frequency of CD4+PD-1+ lymphocytes was similar between the two groups (P = .716) (Figure 2).
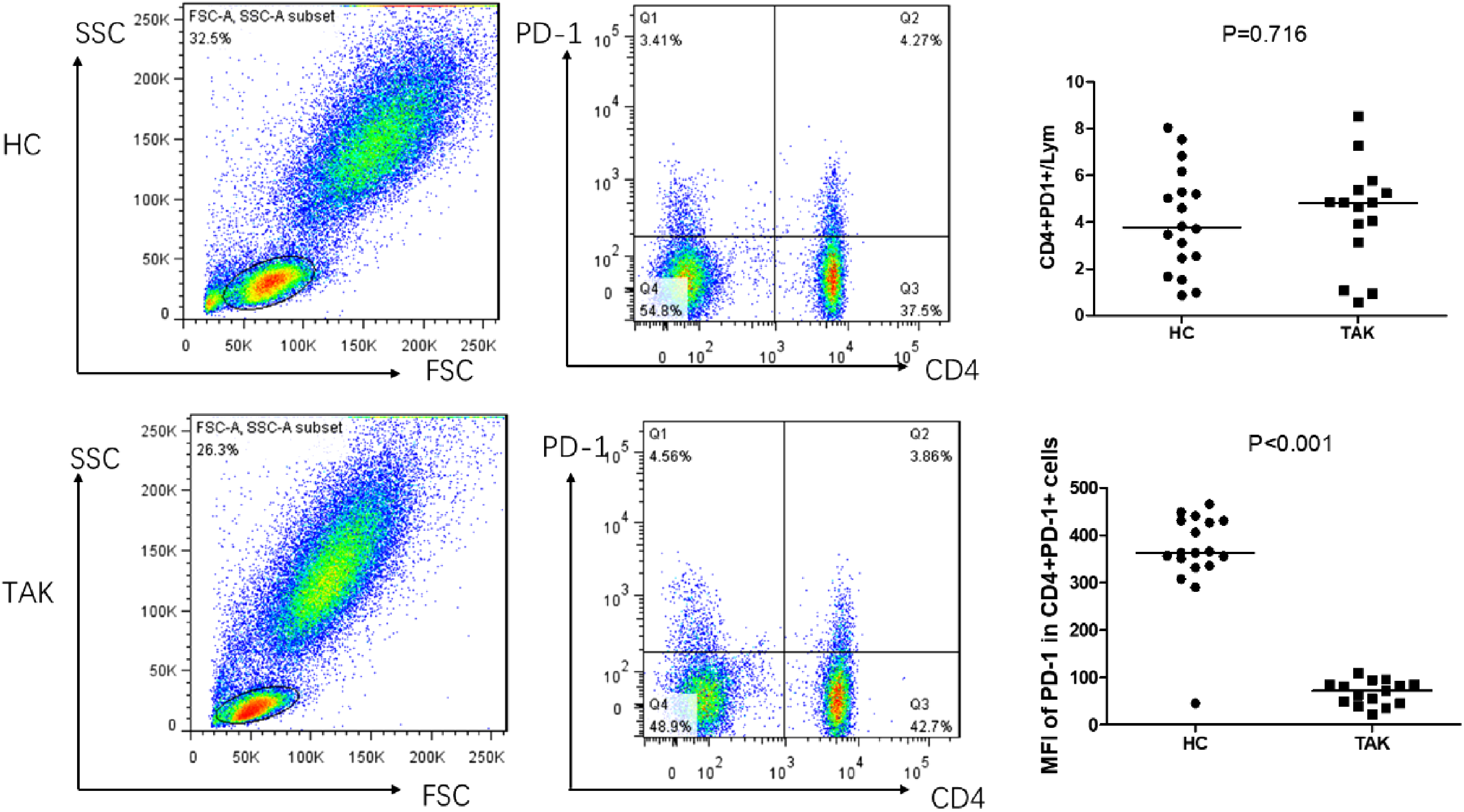
Flow cytometry representation of CD4+PD-1+ cells. CD4+PD-1+/lymphocytes was similar between the two groups (4.04 ± 2.20% vs 4.33 ± 2.22%, P = .716). Mean fluorescence intensity (MFI) of PD-1 in CD4+PD-1+ cells was decreased in the TAK samples [72.2 (45.8, 84.7) versus 363.00 (335, 431), P = .000].
Furthermore, the frequency of CD4+Foxp3−PD-1+ cells in CD4+ T cells was decreased in peripheral blood from patients with TAK (9.05 ± 1.06% vs 12.61 ± 1.17%, P = .034) compared to healthy controls (Figure 3).

Flow cytometry representation of CD4+Foxp3−PD-1+ cells. The frequency of CD4+Foxp3−PD-1+ cells was decreased in TAK patients (9.05 ± 1.06% vs 12.61 ± 1.17%, P = .034) compared to healthy controls.
Peripheral Treg cells in patients with TAK also had lower PD-1 expression as evidenced by the decreased percentage of CD4+CD25+Foxp3+PD-1+ cells among CD4+CD25+T cells (0.40 ± 0.05% vs 0.63 ± 0.09%, P = .047) in the TAK group compared to the healthy control group (Figure 4).

Flow cytometry representation of CD4+CD25+Foxp3+PD-1+ cells. The frequency of CD4+CD25+Foxp3+PD-1+ /CD4+CD25+cells was decreased in TAK patients (0.40 ± 0.05% versus 0.63 ± 0.09%, P = .047) compared to healthy controls.
The Correlation Between PD1 Expression and Clinical Parameters
The correlation between PD1 expression and clinical data including age, age of TAK onset, ESR, CRP, IgA, IgG, IgM, C3, C4, NIH score and Numano types were calculated. We found the percentage of CD4+CD25+Foxp3+PD-1+ cells in CD4+CD25+T cells was lower in active TAK group compared to inactive TAK group [0.305 ± 0.118% versus 0.513 ± 0.207%, P = .026] (Figure 5). The PD1 expression was not correlated to other clinical parameters (P > .05).

The percentage of CD4+CD25+Foxp3+PD-1+ cells was lower in active TAK group than inactive TAK group [0.305 ± 0.118% versus 0.513 ± 0.207%, P = .026]
Tendency for Lower PD-1 and PDL-1 Expression in the Aorta of Patients with TAK
Immunohistochemistry staining of arteries revealed lower PD-1 expression in patients with atherosclerosis than in TAK patients, although the difference is not significant [0.043(0.007, 0.078)% versus 0 .11 (0. 005, 0. 46)%, P = .429] (Figure 6). Analogously, PD-L1 expression in the aorta was not significantly different between the two groups but did tend to be slightly decreased for patients with TAK relative to patients with atherosclerosis [0.08 (0.02, 0.11)% versus 0.10 (0.10, 0.29)%, P = .183] (Figure 7).

Immunohistochemical representation of PD-1 expression in the aortic wall (arrow). PD-1 expression was lower in the aorta of TAK patients than atherosclerosis patients, although the difference was not significant [0.043(0.007, 0.078)% versus 0 .11 (0. 005, 0. 46)%, P = .429].

Immunohistochemical representation of PDL-1 expression in the aortic wall (arrow).PDL-1 expression was lower in the aorta of TAK patients than atherosclerosis patients, although the difference was not significant [0.08 (0.02, 0.11)% versus 0.10 (0.10, 0.29)%, P = .183].
Elevated Serum Levels of Treg-associated Cytokines in Patients with TAK
The Treg-associated cytokines IL-10, IL-7, IL-2, and IL-15 were measured in 49 patients with TAK and 23 healthy controls. Patients with TAK had significantly elevated levels of IL-10 [4.84 (0.07, 409.57)ng/mL versus 3.16 (0, 69.15) ng/mL, P = .004] and IL-7 [10.05 (2.68, 227.2) ng/mL versus 7.4 (2.68, 16.77) ng/mL, P = .014] relative to healthy controls. Meanwhile, there was no significant difference in serum levels of IL-2 [0 (0, 362.58) ng/mL versus 0 (0, 0) ng/mL, P = .054] and IL-15 [0 (0, 360.8) ng/mL versus 0 (0, 0) ng/mL, P = .153] between the TAK and control groups (Figure 8).

Serum levels of Treg-associated cytokines including IL-10 [4.84 (0.07, 409.57)ng/mL versus 3.16 (0, 69.15) ng/mL, P = .004] and IL-7 [10.05 (2.68, 227.2) ng/mL versus 7.4 (2.68, 16.77) ng/mL, P = .014] were increased significantly in TAK patients compared to controls.
Elevated Serum Levels of Treg-associated Chemokines in Patients with TAK
Treg-associated chemokines CCL2, CCL3, and CXCL10 were detected using a cytokine cytometric bead array. Serum levels of both CCL2 [30.64 (0, 686.84)ng/mL versus 10.31 (0, 32.92)ng/mL, P = .000] and CCL3 [3.01 (0.69, 19.24)ng/mL versus 1.84 (0.87, 4.6)ng/mL; P = .000] were increased significantly in TAK patients relative to controls, but the level of CXCL10 was similar in two groups (P = .707) (Figure 9).

Serum levels of Treg-associated chemokines including CCL2 [30.64 (0, 686.84)ng/mL versus 10.31 (0, 32.92)ng/mL, P = .000] and CCL3 [3.01 (0.69, 19.24)ng/mL versus 1.84 (0.87, 4.6)ng/mL; P = .000] were increased significantly in TAK patients compared to controls.
Discussion
Treg cells play an important role in the maintenance of immune tolerance and homeostasis. In recent years, we and others demonstrated that the number of Treg cells in peripheral blood of patients with TAK is reduced relative to healthy controls. This reduction diminishes the inhibitory effect of Treg cells on effector T cell proliferation and differentiation. However, several studies found no difference in the number of Treg cells between patients with TAK and healthy controls. These different results may be associated with the study population, disease severity, or treatment strategy.2,9–11 Consistent with the decrease in the number of Treg cells in peripheral blood from patients with TAK, we further found significant differences in the levels of IL-10, IL-7 and CCL2 and CCL3, which were related to Treg cell development and function.IL-10 is a cytokine that has immunosuppressive function and is secreted by a variety of cells, including macrophages, monocytes, DCs, neutrophils, mast cells, eosinophils, and natural killer cells, as well as by CD4 and CD8 T cells and B cells. 12 Interestingly, the level of IL-10 in serum of TAK patients was increased relative to healthy controls. This increase may be due to negative feedback caused by the decrease in Treg cell numbers in patients with that TAK.
IL-7 is critical for maintenance of T cell subgroups. High levels of IL-7 are associated with autoimmune diseases such as rheumatoid arthritis, ankylosing spondylitis, and inflammatory bowel disease. Maintenance of IL-7R expression in peripheral T cells is dependent on the activity of Foxo1, a transcription factor that plays a role in inhibiting Treg-cell-mediated immune tolerance in mice.13,14
Although immune checkpoint inhibitors (ICIs) including anti-PD-1/PD-L1 therapy have remarkable anti-tumor efficacy, one side effect of such treatments is immune-related adverse events (irAEs).15,16 Patients who experience severe irAEs after anti-PD-1 treatment are also likely to have a significant reduction in the number of effector regulatory T (eTreg) cells.
The activity of eTreg cells is potently suppressed by induced Treg (iTreg) cells that arise from CD4+Foxp3− naive T cells in the periphery. iTregs require prior stimulation for Treg activity and are characterized by high expression levels of CD25, cytotoxic T lymphocyte-associated antigen-4 (CTLA-4), glucocorticoid-induced tumor necrosis factor receptor (GITR), PD-L1 and PD-1.
Although abnormalities in PD-1 activity are reported to play important roles in the pathogenesis of autoimmune diseases, including GCA, a type of large vessel vasculitis,4,7 no role for PD-1 in the pathogenesis of TAK has yet been reported. Here we investigated PD-1 expression in peripheral blood samples from patients with TAK and found that the proportion of CD4+foxp3−PD-1+ T cells and CD4+CD25+Foxp3+PD-1+T cells was decreased relative to healthy controls. These results suggest that PD-1 deficiency may indeed be involved in TAK pathogenesis, and could be associated with an imbalance in Treg cell homeostasis.
PD-1 affects Treg cell homeostasis by regulating Treg cell differentiation. Binding of PD-1 on the surface of CD4+Foxp3−T cell to PD-L1 on the surface of antigen presenting cells (APCs), activates downstream signals that affect Treg differentiation. PD-L1 knockout mouse CD4+T cells from PD-1 knockout mice have diminished capacity to differentiate into Treg cells, resulting in a decrease in the number of Treg cells and severe inflammatory reactions. In vitro experiments also showed that PD-L1 (-/-) APC had weakened ability to induce differentiation of CD4+T cells into Treg cells. Meanwhile, magnetic beads coated with PD-L1 Ig could promote formation of Treg cells, suggesting that PD-1 plays an important role in inducing production of Treg cells. 17 PD-1 is thought to induce development of Treg cells by downregulating expression of protein kinase B(pAkt), mammalian target of rapamycin, mTOR (mTOR), S6 and extracellular regulated protein kinases 2(ERK2), while upregulating expression of phosphatase and tensin homolog deleted on chromosome ten (PTEN). These proteins are important members of signal transduction pathways that govern development of Treg cells. In particular, mTOR plays an important role in the development of autoimmune diseases by affecting Treg differentiation.18,19 Expression of phosphorylated S6 ribosomal protein, a marker of mTOR activity, is decreased in peripheral blood Treg cells from patients with TAK. In the presence of rapamycin, an mTOR inhibitor, the number of Tregs increased, suggesting that mTOR inhibits Treg differentiation in patients with TAK. 20 To clarify changes of PD-1 in CD4+Foxp3−T cells from patients with TAK, we detected the number of CD4+Foxp3−PD-1+ cells in peripheral blood samples. We found that PD-1 expression in CD4+Foxp3− T cells from patients with TAK was significantly decreased compared with the control group. This finding supports the hypothesis that PD-1 deficiency in patients with TAK affects differentiation of CD4+Foxp3−T cells into Treg cells.
The maintenance of the number of Treg cells in peripheral blood is also closely related to Treg cell apoptosis. Blocking PD-1 signaling results in decreased numbers of Treg cells, likely through the promotion of apoptosis. Spleen Treg cells from PD-1(−/−) mice had low expression of the anti-apoptotic molecule Bcl-2 and high expression of the pro-apoptotic molecule FAS that may be associated with significantly more frequent apoptosis of these cells. 21 To clarify changes in PD-1 in Treg cells from patients with TAK, we detected CD4+CD25+Foxp3+PD-1+ cells in peripheral blood. We found that, compared with the control group, the proportion of CD4+CD25+Foxp3+PD-1+ cells in the TAK group was significantly decreased, such that the expression of PD-1 in Treg cells from TAK patients was also decreased, which could indicate that in TAK Treg homeostasis is disrupted by increased apoptosis of Treg cells.
To our knowledge, to date there have been no studies on expression levels of PD-1 and PDL-1 in the aorta of patients with TAK. Our immunohistochemistry analysis of arteries from patients with TAK shows for the first time that PD-1 and PDL-1 expression tended to be lower in patients with TAK compared to those with atherosclerosis, but the differences were not significant. Zhang et al reported that patients with GCA had low expression levels of PD-L1, but they compared patients with GCA patients to healthy controls rather than to patients with atherosclerosis. 7
The limitation of this study is a lack of functional research. Further studies to explore the mechanism of PD-1 deficiency leading to Treg cell imbalance in TAK are warranted.
Conclusion
To our knowledge, this is the first report of abnormal PD-1 expression associated with TAK. In this research, we found decreased expression of PD-1 in CD4+foxp3−T cells and CD4+CD25+Foxp3+T cells in patients with TAK. As previous studies showed Treg abnormalities in TAK patients, additional study of the role of PD-1 deficiency in regulating Treg immune homeostasis should be explored in TAK patients in further study.
Footnotes
Abbreviations
Acknowledgements
Not applicable.
Authors’ Contributions
GN designed the whole experiment and wrote the main manuscript text. LTT performed the histological examination of the aorta tissue. CW and ZLM performed the flow cytometry. CW and ZJH detected the serum cytokines. PLL checked the experiments and revised the manuscript. All authors read and approved the final manuscript.
Availability of Data and Material
All data generated or analyzed during this study are included in this published article and its additional files.
Consent for Publication
All authors have critically reviewed and approved the final manuscript to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National natural science foundation of China, Beijing natural science foundation (grant number 81900448, 7222043).
Ethics Approval and Consent to Participate
This study was approved by the Medical Ethics Committee of Anzhen Hospital. Written informed consent for publication was obtained from the participant.
