Abstract
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by dysregulation of T cells. Programmed death (PD) 1 and programmed death 1 ligand 1 (PD-L1) are cosignaling molecules, and the major role of the PD-1 pathway is the inhibition of self-reactive T cells and to protect against autoimmune diseases. We measured levels of serum soluble PD 1 (sPD-1) and serum soluble PD-L1 (sPD-L1) in 67 patients with ITP (24 newly diagnosed ITP [ndITP], 43 chronic ITP [cITP]) and 21 healthy controls (HCs). We determined decreased serum sPD-1 levels both in patients with ndITP and in patients with cITP when compared to HC. Moreover, there was a positive correlation between sPD-1 levels and platelet counts. The sPD-L1 levels were decreased in patients with ndITP when compared to patients with cITP. This is the first study investigating PD-1 signaling pathway in ITP. Decreased sPD-1 levels may have a role in ITP pathogenesis as without the inhibitory regulation of PD-1, sustained activation of T cells may cause inflammatory responses which is the case in ITP.
Introduction
Immune thrombocytopenia (ITP) is an acquired autoimmune disease which is defined as platelet count <100 × 109/L without the existence of any immune or nonimmune diseases that can cause thrombocytopenia. 1 Immune thrombocytopenia is characterized by peripheral thrombocyte destruction. Similar to the majority of autoimmune diseases, abnormalities in immune cell types, such as antigen-presenting cells, T cells, and B cells, have been shown to play some sort of role in the initiation and/or perpetuation of the disease. 2 The CD4+ T cells and CD8+ cytotoxic T cells with platelet autoreactivity have been found in the peripheral blood of patients with ITP, indicating an impaired peripheral tolerance. 3 In ITP, platelet autoreactive T cells have decreased apoptosis and create a cytokine imbalance that can lead to lower levels and abnormal function of regulatory T cells. 4 There is an imbalance in T-helper 1 (Th1)/Th2 cytokine axis in ITP, leading to autoreactive B cell differentiation. 2
The role of PD-1 in the regulation of T cell immunotolerance was first suggested by the studies carried out in animal models revealing that PD-1 knockout (Pdcd1−/−) mice developed spontaneous autoimmune diseases. 5,6 The PD-1 and its ligands PD ligand 1 (PD-L1) and PD ligand 2 (PD-L2) mainly serve in peripheral tolerance. Self-reactive T cells escaping from central tolerance in the thymus are controlled through peripheral tolerance; otherwise, autoimmunity develops. Both PD-1 and PD-L1 were detected on Treg, which are involved in the regulation of peripheral tolerance by inhibiting effector T cells and suppressing the immune-mediated tissue damage through several mechanisms. 7 The interaction between PD-1 and PD-L1 can influence Treg generation, suppressive Treg properties, and also the interactions between Treg and effector T cells. Studies revealed that conversion of Th1 cells into Treg is a PD-L1-dependent mechanism. 8 A major functional role of the PD-1 signaling pathway is the inhibition of self-reactive T cells and to protect against autoimmune diseases. Elimination of the PD-1 pathway can result in the breakdown of immune tolerance and can eventually lead to the development of pathogenic autoimmunity. 9 To the best of our knowledge, there is no study in the literature demonstrating pathogenic role of PD-1-PD-L1 pathway in ITP. Thus, we decided to conduct this study in order to investigate serum soluble PD-1 (sPD-1) and soluble PD-L1 (sPD-L1) levels in patients with ITP, considering the autoimmune nature of ITP and the pathogenic role of T cells in ITP.
Methods
Sixty-seven patients with primary ITP who were diagnosed and/or followed up in Hematology Department between June 2011 and March 2014 and 21 healthy controls (HCs) were enrolled to this study. All the control participants were matched with patient population in terms of age and sex. The patients were diagnosed according to the criteria of International Working Group (IWG) consensus report, mainly based on excluding other diseases causing thrombocytopenia such as autoimmune diseases. 1 Although patients who have been diagnosed within the 3 last months were accepted as newly diagnosed ITP (ndITP), patients with ITP lasting more than 12 months were accepted as chronic ITP(cITP) according to IWG criteria. 10 In all, 24 patients in the ITP group were ndITP and the remaining 43 patients were patients with cITP. Patients with ITP were managed according to the IWG consensus report. 1 Although 7 of the 24 patients with ndITP and 9 of the 34 patients with cITP were receiving immunosuppressive therapy, the rest of the patients were free of immunosuppressive medication at the time of sampling. Data on sex, age, and complete blood count results were registered at the time of enrollment in patients with ITP and HC. In patients with cITP in addition to this information, data about the treatments they received and whether the patient had splenectomy previously were also registered. Characteristics of patients with ITP and HCs are shown in Table 1. Blood samples were collected from the patients, and the controls and the sera were stored at −80°C. Serum sPD-1 and sPD-L1 levels were determined with commercial enzyme-linked immunosorbent assay kits (Hangzhou Eastbiopharm Co Ltd, China and Cusabio Biotech Co Ltd, China, respectively) according to the manufacturer’s instructions. Concentration of sPD-1 and sPD-L1 was determined using the standard curve (ng/mL and pg/mL, respectively). The study was approved by the Faculty Research Ethical Committee, and informed consents were taken prior to enrollment.
Characteristics of Patients With ITP and HC.
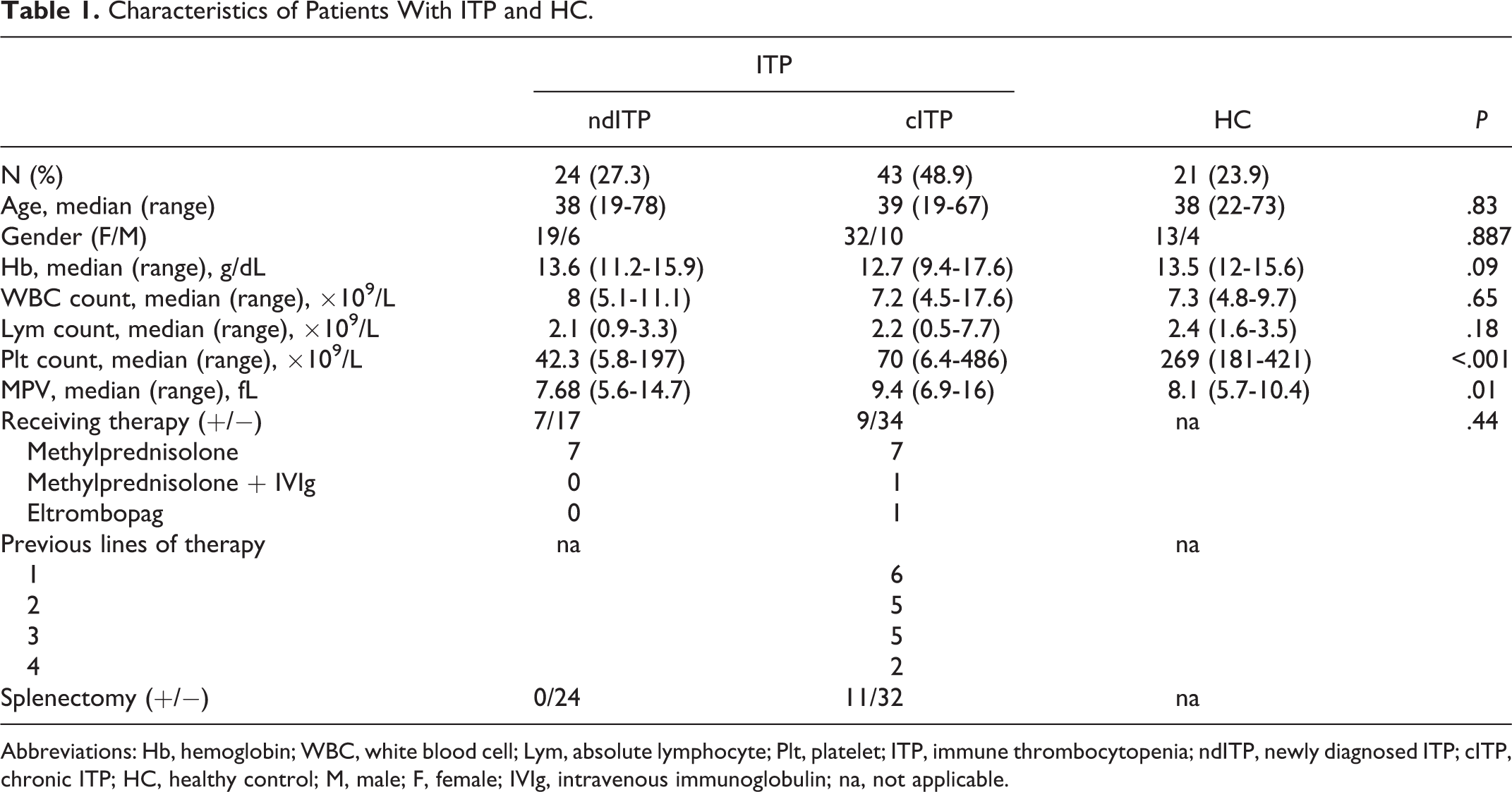
Abbreviations: Hb, hemoglobin; WBC, white blood cell; Lym, absolute lymphocyte; Plt, platelet; ITP, immune thrombocytopenia; ndITP, newly diagnosed ITP; cITP, chronic ITP; HC, healthy control; M, male; F, female; IVIg, intravenous immunoglobulin; na, not applicable.
SPSS 21.0 (SPSS Inc) software was used for statistical analyses. The variables were investigated using visual (histogram and probability plots) and analytical methods (Shapiro-Wilk tests) to determine whether or not they are normally distributed. As the variables were not normally distributed, nonparametric methods were used for analysis. Median values of each parameter were reported with minimum–maximum range. The Kruskal-Wallis test was utilized for multigroup analyses. The Mann-Whitney U test was used to compare nonparametric variables between the 2 groups. While investigating the associations between parameters, correlation coefficients and their significance were calculated using the Spearman test. The chi-square test or Fisher exact test (when chi-square test assumptions do not hold due to low expected cell counts), where appropriate, was used to compare these proportions in different groups. A P value of less than .05 was considered to show a statistically significant result.
Results
There was a statistically significant difference between sPD-1 levels of ndITP, patients with cITP, and HC (P < .001; Figure 1). The sPD-1 levels of patients with ndITP (5.15 [2.07-7.06]) and patients with cITP (5.33 [2.25-32]) were not statistically different (P = .30). However, both sPD-1 levels of patients with ndITP and cITP were significantly decreased when compared to HCs (8.91 [4.83-52.07]; P < .001, P < .001, respectively).
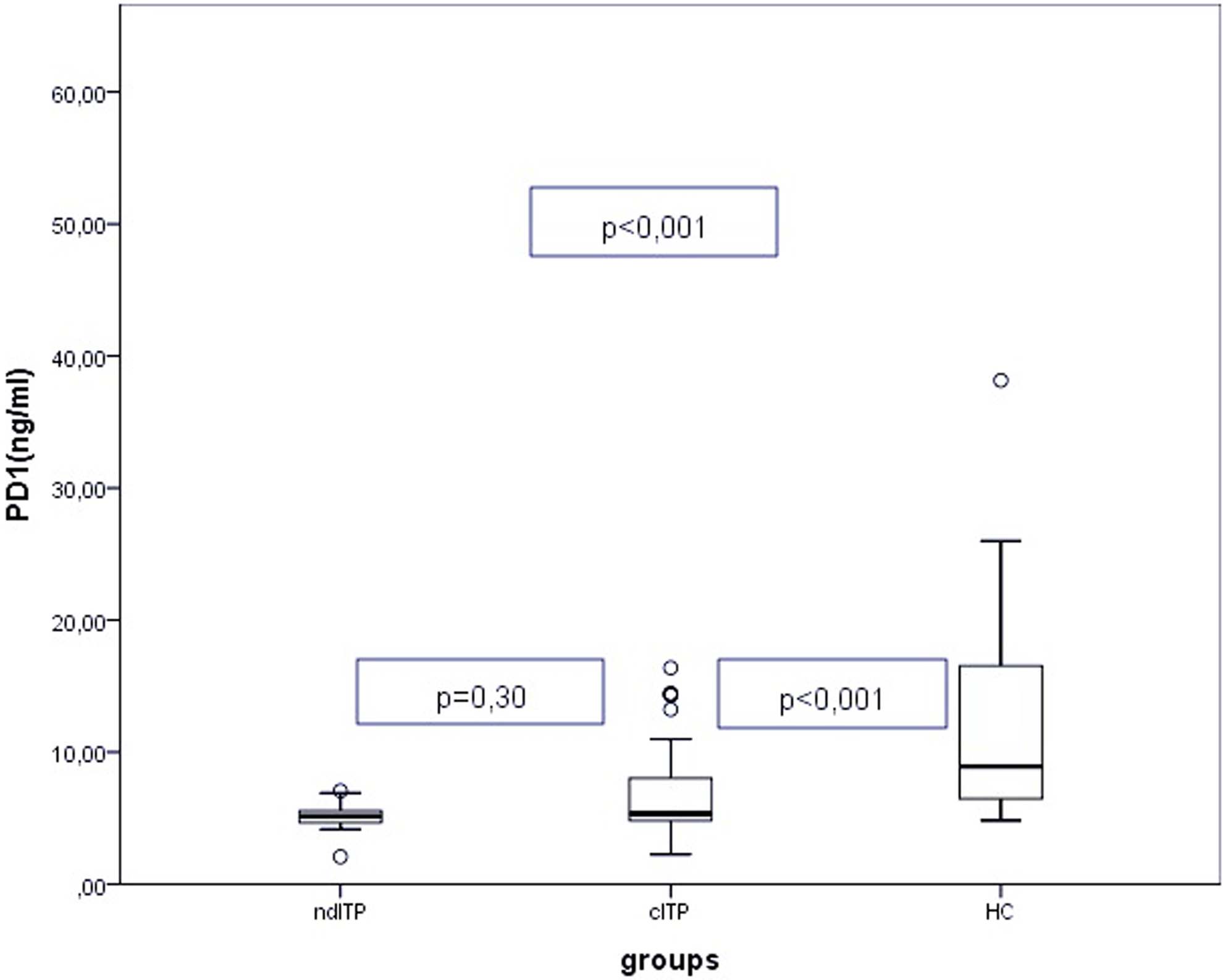
Soluble PD-1 (sPD-1) levels or the study groups, *Mann-Whitney U test.
Serum sPD-L1 levels of patients with ndITP (4.06 [1.22-8.71]), patients with cITP (5.47 [2.53-15.4]), and HCs (4.56 [2.33-10.23]) were not significantly different (P = .06). However, in subgroup analysis, sPD-L1 levels of patients with ndITP were significantly decreased in comparison to patients with cITP (P = .02). No significant difference could be detected between sPD-L1 levels of patients with ndITP or patients with cITP and HC (P = .79, P = .06, respectively).
The sPD-1 or sPD-L1 levels did not differ significantly when the patients were grouped as ones taking immunosuppressive treatment at the time of blood sampling or not and had previously splenectomized or not (P > .05). However, in the ndITP group, sPD-1 levels of patients with platelet counts <30 × 109/L were significantly decreased when compared to patients with platelet counts ≥30 × 109/L (P = .05). Likewise, sPD-L1 levels of patients with ndITP having platelet counts <100 × 109/L sPD-1 were significantly decreased in comparison to patients with platelet counts ≥100 × 109/L.
A positive correlation was detected between serum sPD-1 levels and platelet counts (P = .01, r = .279; Figure 2). However, no correlation could be detected between sPD-1 levels and sPD-L1 levels, age, white blood cell (WBC) counts, and lymphocyte counts (P > .05). Likewise, there were no correlation between sPD-L1 levels and age, platelet counts, WBC counts, and lymphocyte counts (P > .05).
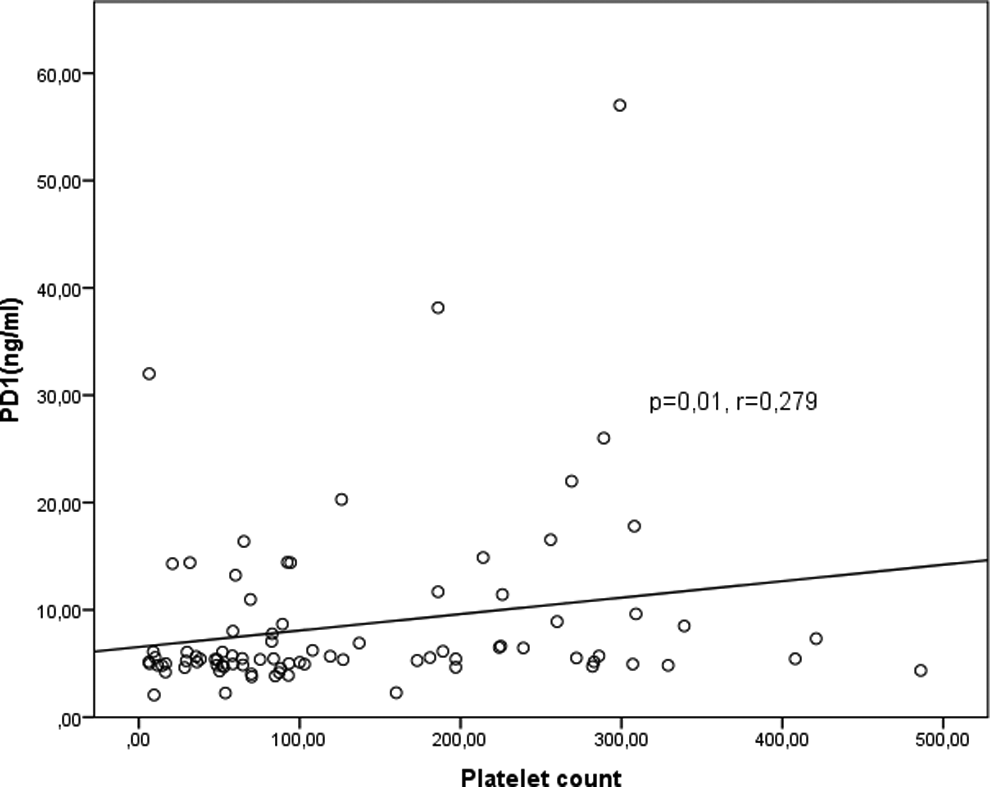
Correlation between soluble PD-1 (sPD-1) levels and platelet counts.
Discussion
Currently, it is clear that PD-1/PD-Ls pathway plays crucial roles in autoimmunity, transplantation immunity, infectious immunity, and tumor immunity. We measured the serum levels of sPD-1 and sPD-L1 in patients with ITP and we determined that sPD-1 levels of patients with ndITP and patients with cITP were significantly decreased when compared to HCs (P < .001, P < .001, respectively). There was a positive correlation between serum sPD-1 levels and platelet counts. The sPD-L1 levels of patients with ndITP were significantly decreased in comparison to patients with cITP (P = .02). Moreover, sPD-L1 levels of patients with ndITP were significantly increased in patients with platelet counts ≥100 × 109/L, describing patients who were in remission when compared to patients with platelet counts <100 × 109/L.
The participation of the PD-1/PD-L1 pathway in autoimmunity was investigated in several autoimmune diseases in animal models and humans. There are several studies investigating PD-1 expression in T cells and serum sPD-1 and sPD-L1 levels. The soluble proteins can play important roles in the regulation of costimulatory signals. 11 In the present study, we measured sPD-1 and sPD-L1 levels in the sera of patients with ITP. In literature, there are no studies investigating PD-1/PD-L1 pathway in ITP. Therefore, we are going to discuss our results with studies conducted in other autoimmune diseases. There are 2 studies in rheumatoid arthritis (RA) with conflicting results. Wan et al found that PD-1 and PD L-1 were overexpressed in synovial T cells and macrophages derived from patients with RA as opposed to controls. Furthermore, the sPD-1 and sPD-L1 occurred at high concentrations in sera and synovial fluid of patients with RA. 12 By contrast, the study conducted by Li et al revealed decreased PD-1 expression in both peripheral CD4+ and CD8+ T cells in RA, and serum levels of sPD-1 were also downregulated in cases with RA. 13 Our results in the present study are in accordance with the study conducted by Li et al. The finding of decreased sPD-1 levels in patients with ITP is perceptible because without the negative regulation of PD-1, sustained activation of T cells may cause inflammatory responses by increasing cytokine production and cytolytic activity as is the case in ITP.
Similar to PD-1, there are no studies in the literature investigating the role of PD-L1 in ITP. There are also conflicting results about serum levels of sPD-L1 in other diseases. However, Frigola et al reported that exposure of CD4+T and CD8+T cells to sPD-L1 induced apoptosis. 14 The decreased PD-L1 levels in patients with ndITP in comparison to patients with cITP and in patients with ndITP who were not in remission in comparison to patients with ndITP who were in remission could suggest for a role of sPD-L1 in the acute phase of ITP, since decreased sPD-L1 might have led to a defect in T cell apoptosis and consequently autoimmunity.
Levels of sPD-1 were not different in patients with ITP who were in remission and who were not. Besides, the level of sPD-1 was not different in patients who were taking immunosuppressive treatment and who were free of immunosuppressive medication. The sPD-1 levels were not affected from the past history of splenectomy. When all these findings were considered, especially sPD-1 can have a role in ITP and its level doesn’t change with immunosuppressive treatment and remission status as long as the patient had been diagnosed as ITP and may be a clue for the relapsing nature of ITP. The study conducted by Gianchecchi et al clearly demonstrated that in humans, single-nucleotide polymorphisms in PDCD1 is associated with a higher risk of developing autoimmune diseases. 15 Hence, a polymorphism in PDCD1 may be speculated in ITP.
This is the first study investigating the serum levels of PD-1 and PD-L1 in patients with ITP, and our results suggest that decreased sPD-L1 levels in ndITP might have role of ITP pathogenesis. However, there are some limitations in the present study. We measured only the serum sPD-1 and sPD-L1 levels, and without evaluating expression of these molecules on the cell surfaces, it is difficult to examine roles of PL-1 pathway in the pathogenesis of ITP. Second, the positive correlation we demonstrated between sPD-1 levels and thrombocyte counts could have been strengthened by serial measurements on the same patients.
Future studies may demonstrate PD-1 and PD-L1 levels with antiplatelet autoantibodies levels. More comprehensive studies with more homogeneous group of patients are required to clarify the exact role of PD-1/PD-L1 pathway in ITP immunopathogenesis and this could lead to new treatment options in ITP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
