Abstract
We aimed to examine the levels of lymphocyte subsets and regulatory T cells in patients with newly diagnosed immune thrombocytopenia (ITP) and their correlation with the course of ITP. The study included 40 pediatric patients with acute ITP and 30 controls. Lymphocytes and regulatory T cells were analyzed by flow cytometry. The percentages of CD19+ and CD8+ cells were significantly increased while that of CD4+ cells and CD4+/CD8+ ratio were significantly decreased. The percentages of CD4+CD25+High and CD4+CD25+High forkhead box protein 3 (Foxp3+) cells and the expression of Foxp3+ in CD4+CD25+High cells were significantly decreased in patients. Age, platelet count, and mean platelet volume (MPV) in patients with brief duration of thrombocytopenia were significantly decreased than in those with prolonged duration. The percentages of CD8+, CD4+CD25+High, and CD4+CD25+High Foxp3+ were significantly increased in patients with brief duration. Age, platelet count and MPV, and CD8+ cells had prognostic significance. CD4+CD25+High Foxp+ T cells may be a helpful prognostic marker in children with acute ITP.
Introduction
Primary immune thrombocytopenia (ITP) is an acquired immune-mediated disorder characterized by isolated thrombocytopenia and increased platelet destruction. 1 –3 It is defined as platelet count <100 × 109/L, normal or increased bone marrow megakaryocytes without any morphological evidence for dysplasia, and no secondary immune or nonimmune diseases that account for patients with thrombocytopenia. 4 Only 3% of children with ITP have clinically significant symptoms such as severe epistaxis or gastrointestinal bleeding. 5,6 Severe bleeding is more likely in children with platelet counts less than 10 × 109/L. The incidence of intracranial hemorrhage in children with ITP is approximately 0.1% to 0.5%. 7 The majority of children with newly diagnosed ITP lack significant bleeding symptoms and may be managed without therapy by raising the platelet count. 8,9
The pathophysiology of ITP is heterogeneous and complex. While the presence of antibodies against platelet glycoproteins has traditionally been considered to play a central role, several abnormalities involving the cellular mechanisms of immune modulation had been described. 10,11
The cellular immune responses of patients with ITP have the characteristic hallmarks of breakdown in their tolerance mechanisms. 12 T helper cells and the cytokines they produce play a key role in this breakdown. 13 The abnormal helper T cells have been suggested to direct autoreactive B cells to secrete autoantibodies. 14 In addition to the involvement of helper T cells in the pathogenesis of ITP, abnormal CD8 responses involving cytotoxic T cell-mediated lysis of autologous platelets in patients with active ITP have been reported. 15 Such abnormal lymphocyte responses in T- and B-cell compartments as well as the reported cytokine imbalance in patients with ITP suggest potential defects in regulatory immune networks. 16
An additional mechanism for peripheral tolerance is active suppression by a functionally distinct subpopulation of CD4+ T cells called regulatory T cells. 17 CD4+ regulatory T cells play an important role in the maintenance of peripheral tolerance and are characterized by the expression of the CD25 surface marker and the transcription factor forkhead box protein 3 (Foxp3), making up 5% to 10% of the normal CD4+ T-cell population. 18 Several studies had described CD4+C25+ regulatory T-cell characteristics in patients with chronic ITP and patients receiving immunosuppressive therapy or thrombopoietic agents. 19 –21 The role of regulatory T cell in newly diagnosed pediatric patients with ITP remains to be elucidated.
The aim of this study is to examine the level of lymphocyte subsets (CD3, CD4, CD8, and CD19) and regulatory T cell in the newly diagnosed pediatric patients with ITP and their correlation with the course of ITP.
Patients and Methods
The study included 40 patients with acute ITP. The patients were recruited from Pediatric Clinical Hematology Unit of Children Hospital, Assiut University. In addition 30 healthy age- and sex-matched children were taken as controls. At diagnosis, all patients and controls were subjected to history and physical examination in addition to the following investigations: full blood picture (Celltac E automated hematology analyzer, Tokyo, Japan), detection of B lymphocytes, T lymphocytes, and regulatory T cells by flow cytometry. The patients were managed according to the grade of severity. 22 According to the treatment response, the patients were then classified into 2 groups: patients with short duration of illness, who recovered before 3 months, and patients with long duration of illness, who did not recovere before 3 months according to the clinical score developed by Edslev et al. 23 The study was approved by the institutional review board and informed consents were obtained from the parents.
Inclusion Criteria
Children aged from 3 months to 16 years, presented with newly diagnosed acute ITP.
Platelet count <20 × 109/L.
Bleeding tendency < grade 4. 22
No prior immunomodulating (intravenous immunoglobulin and corticosteroids) treatment before diagnosis.
Exclusion Criteria
Clinical features that are not compatible with the diagnosis of acute ITP, such as presence of organomegaly, other cytopenias besides thrombocytopenia, other autoimmune phenomena, or features suggestive of infectious disease like hepatitis, Epstein-Barr virus.
Patients with chronic ITP (thrombocytopenia persisting for longer than 6 months from the onset of illness). 24
Immunomodulating treatment within 4 weeks before diagnosis.
Severe or life-threatening bleeding at presentation (grade 4). 22
Flow Cytometric Detection of Regulatory T Cells, B, and T Lymphocytes
CD4+CD25+Foxp3+ regulatory T cells in whole blood samples were enumerated using fluoroisothiocyanate (FITC)-conjugated Foxp3 (e Bioscience, San Diego, California), phycoerythrin (PE)-conjugated CD25 (IQ Product, The Netherland), and peridinium-chlorophyll-protein (Per-CP)-conjugated CD4 (Becton Dickinson Bioscience, San Jose, California). One hundred microliters of blood sample were incubated with 10 µL of CD4 and CD25 for 15 minutes at room temperature in the dark. Following incubation; red blood cell (RBC) lysis, washing with phosphate-buffered saline (PBS), addition of fixed solution to fix the cells, and incubation for 10 minutes were done. After incubation, the cells were washed with PBS, followed by the addition of permeabilizing solution and 10 µL of Foxp3, and incubated for 30 minutes at room temperature. For detection of B and T lymphocytes, 50 µL of blood sample were stained with 10 µL of FITC-conjugated CD4, PE-conjugated CD8, Per-CP-conjugated CD3, and allophycocyanin-conjugated CD19. The last monoclonal antibodies were from Becton Dickinson Biosciences. After incubating for 15 minutes at room temperature in the dark, the RBCs were lysed. After 1 wash, the cells were resuspended in PBS. Flow cytometric analysis was done by fluorescence-activated cell sorter calibur flow cytometry with Cell Quest software (Becton Dickinson Biosciences). An isotype-matched negative control was used for each sample. Forward and side scatter histogram was used to define the lymphocyte population. Then, the percentages of CD19+ (B lymphocytes), CD3+ (T lymphocytes), CD4+ (T-helper cells), and CD8+ (T cytotoxic) were assessed in lymphocytes population (Figure 1).
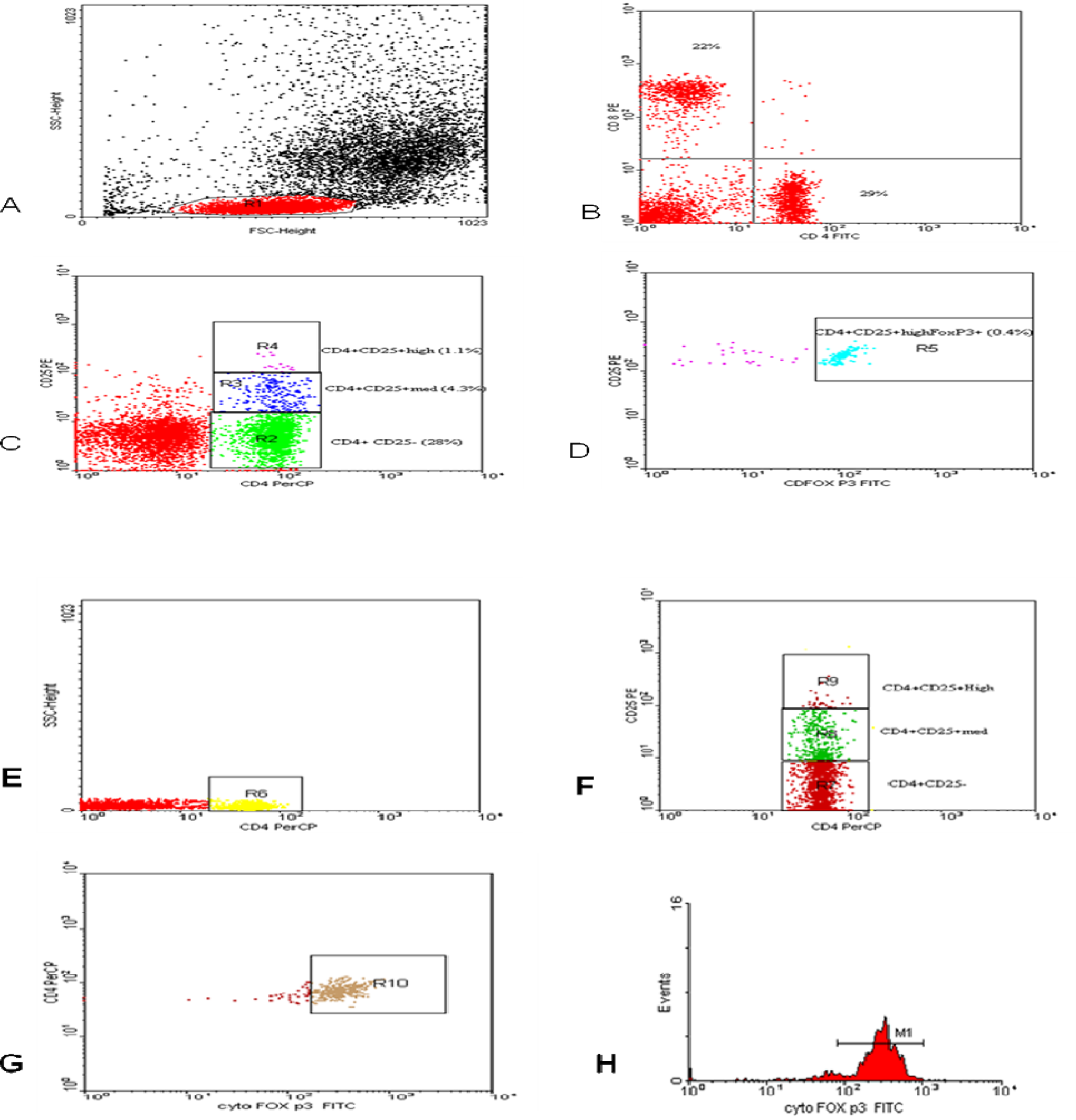
Flow cytometric detection of CD4+CD25+high Foxp3+ regulatory T cells, B lymphocytes, and T lymphocytes. A, Forward and side scatter histograms were used to define the lymphocytes population (R1). (I) B and C, The expression of CD4+ and CD8+ was assessed in lymphocytes population (R1). The expression of CD4 and CD25 in total lymphocytes (R1) was detected, compared with the negative isotype control (not shown), and different gates were drawn to define CD4+ CD25− cells (R2), CD4+CD25+intermediate (med) cells (R3), and CD4+CD25+High cells (R4). D, The percentage of CD4+CD25+high Foxp3+ cells (R5) in total lymphocytes was determined. (II) E, F, and G, The analysis of regulatory T cells in CD4+ cells (R6). CD4+ CD25− cells (R7), CD4+CD25+intermediate (med) cells (R8), and CD4+CD25+High cells (R9). G, The percentage of CD4+CD25+high Foxp3+ cells (R10) in CD4+ cells was determined. H, The expression of Foxp3+ in CD4+CD25+high cells as geometric mean of fluorescent intensity (MFI).
Total CD4+CD25+, CD4+CD25+intermediate, and CD4+CD25+High Foxp3+ regulatory T cells were evaluated both in total lymphocytes and in CD4+ as shown in Figure 1. CD4+CD25+High cells were defined as the population of CD4-positive T cells whose CD25 expression exceeded the level of CD25 positivity seen in the CD4-negative T cells, according to the methods described by Zhu et al and Chi et al. 25,26 The expression of Foxp3+ as geometric mean of fluorescent intensity (MFI) in CD4+CD25+intermediate and CD4+CD25+High was detected.
Statistical Analysis
Statistical package for social sciences, version 16 was used for data analysis. All data were expressed as the mean ± standard error of mean. Because of the small sample size and a propensity for outliers in some of the variables, differences between the groups were examined for statistical significance using the Mann-Whitney analysis. P value ≤.05 denoted the presence of a statistically significant difference.
Results
This study included 21 males and 19 females with a age range of 1 to 12.5 (mean age 5.42 ± 0.51). Cutaneous manifestations were present in all patients in the form of petechial hemorrhage and some bruising. Mild epistaxis was present in 9 patients. In all, 28 (70%) of our patients experienced a preceding febrile illness. None of our patients presented with enlarged liver, spleen, or significant lymphadenopathy. Thirty-two patients (80%) were recovered within 3 months and 8 (20%) patients had prolonged duration of thrombocytopenia more than 3 months. There were no significant difference in the count of white blood cells and hemoglobin concentration between patients with ITP and controls. Platelets count was significantly decreased, and mean platelet volume (MPV) was significantly increased in patients than in controls (Table 1).
Baseline Characteristics of Patients With ITP and Controls.a
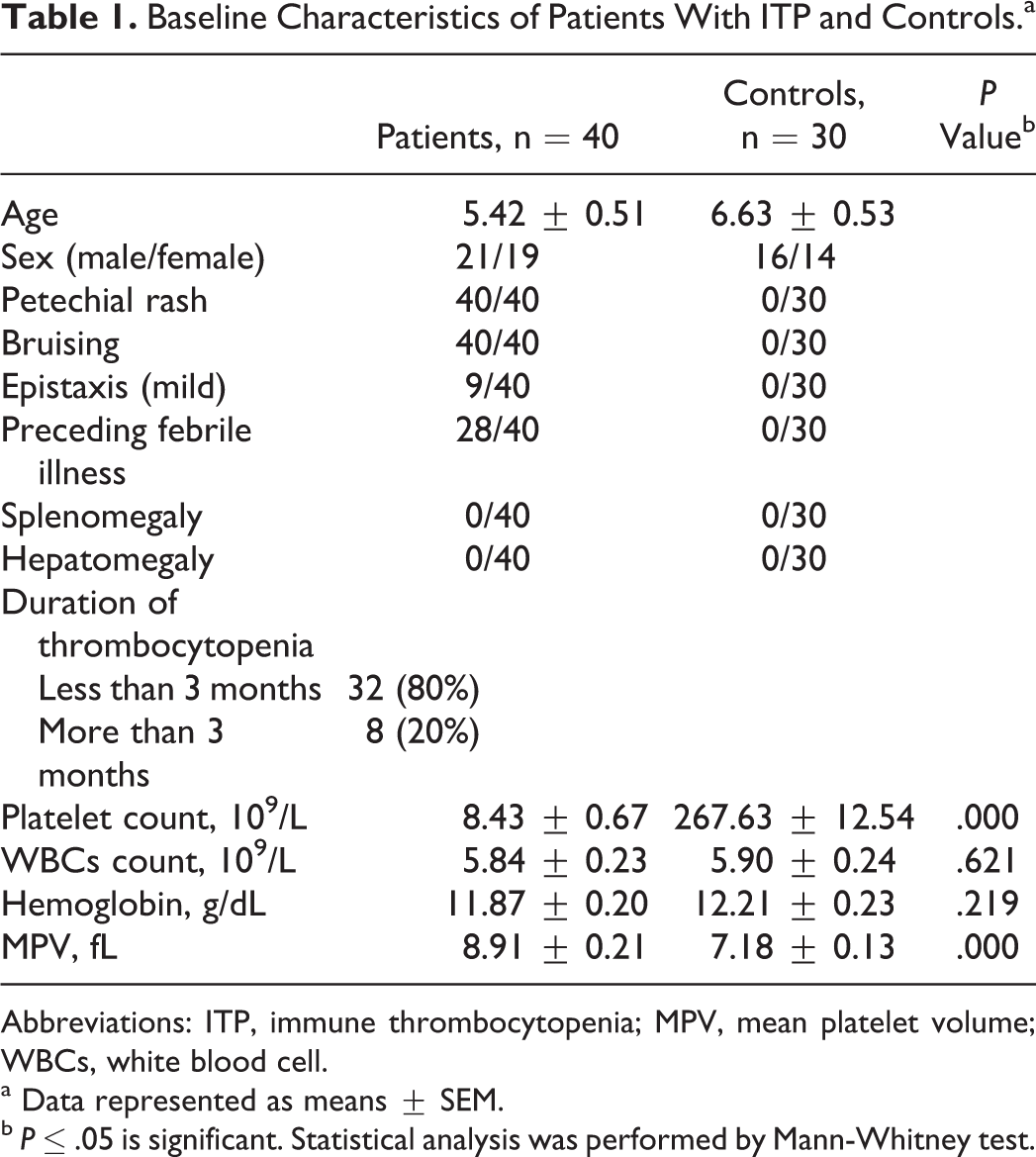
Abbreviations: ITP, immune thrombocytopenia; MPV, mean platelet volume; WBCs, white blood cell.
a Data represented as means ± SEM.
b P ≤ .05 is significant. Statistical analysis was performed by Mann-Whitney test.
Regarding the subset of lymphocytes, in our study, the percentage of T lymphocytes (CD3+) was not significantly different in patients and controls. The percentages of B lymphocytes (CD19+) and T cytotoxic cells (CD8+) were significantly increased in patients than controls. The percentage of T helper cells (CD4+) was significantly decreased in patients than in controls. The CD4+/CD8+ ratio was significantly decreased in patients than controls (Table 2).
Lymphocytes Subsets in Patients With Acute ITP and Controls.a
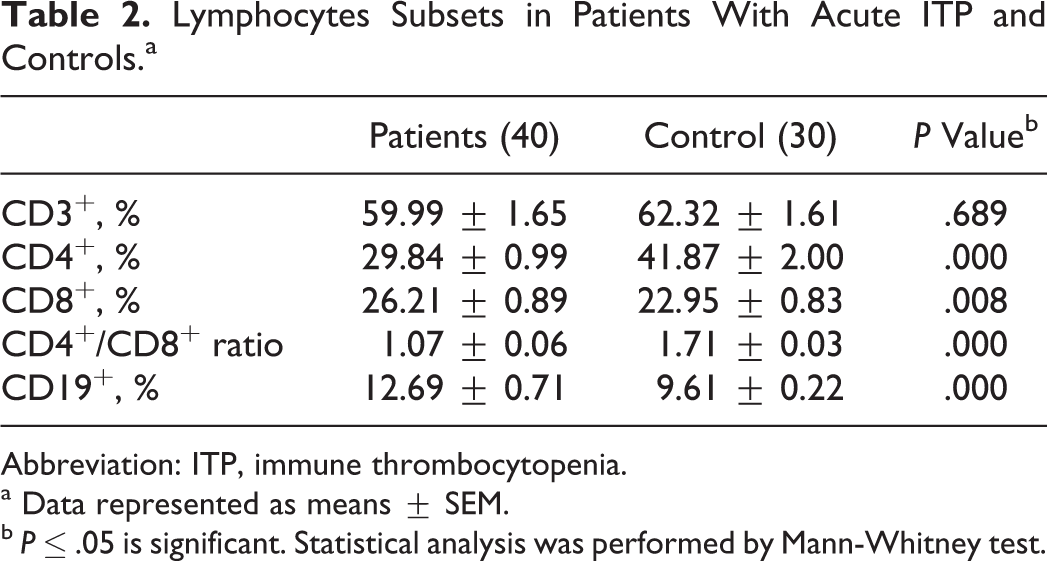
Abbreviation: ITP, immune thrombocytopenia.
a Data represented as means ± SEM.
b P ≤ .05 is significant. Statistical analysis was performed by Mann-Whitney test.
The percentages of total CD4+CD25+ and CD4+CD25+Intermediate both in total lymphocytes and in CD4+ cells were not significantly different between patients and controls. The percentages of CD4+CD25+High and CD4+CD25+High Foxp3+ both in total lymphocytes and in CD4+ cells were significantly decreased in patients than in controls.
The MFI of Foxp3+expression in CD4+CD25+High cells was significantly decreased in patients than in controls, while its expression in CD4+CD25+Intermediate cells was not significantly different between patients and controls (Table 3).
Regulatory T Cells in Patients With Acute ITP and Controls.a
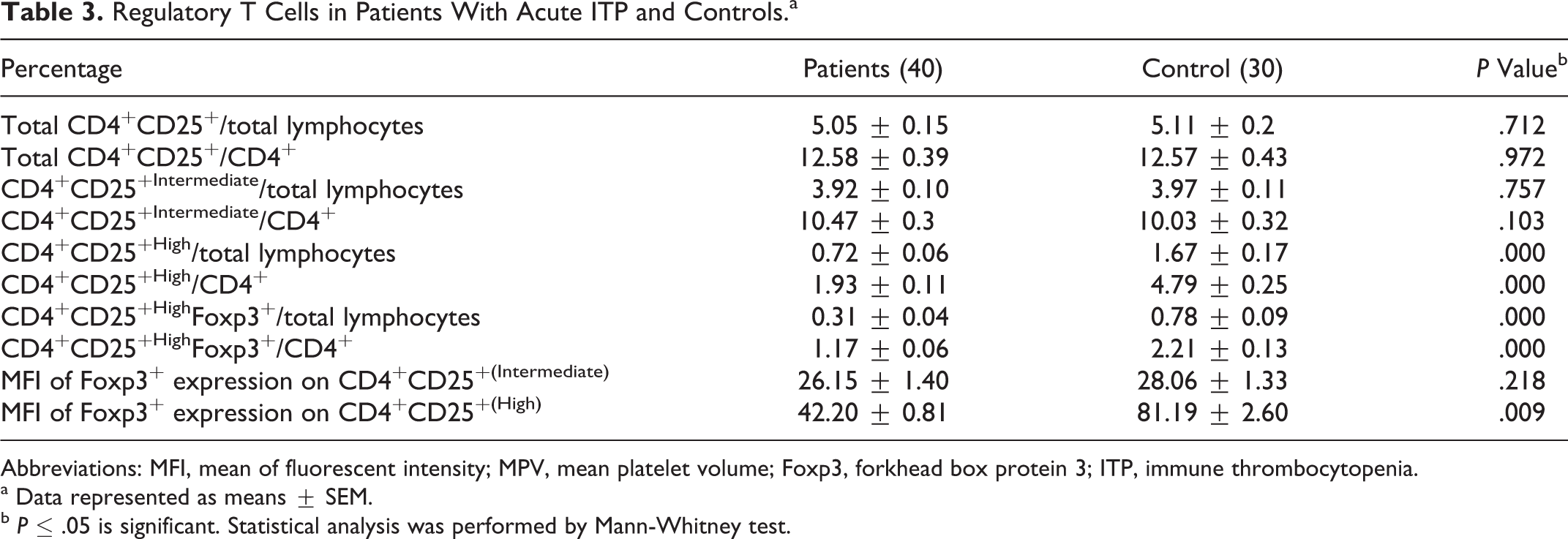
Abbreviations: MFI, mean of fluorescent intensity; MPV, mean platelet volume; Foxp3, forkhead box protein 3; ITP, immune thrombocytopenia.
a Data represented as means ± SEM.
b P ≤ .05 is significant. Statistical analysis was performed by Mann-Whitney test.
The age of patients with brief duration of thrombocytopenia was significantly decreased than that in patients with prolonged duration. Platelet count and MPV in patients having thrombocytopenia for a brief duration were significantly decreased than in those having thrombocytopenia for a prolonged duration. The percentage of CD8+ cells was significantly increased in patients with brief duration of thrombocytopenia. The percentages of CD4+CD25+High and CD4+CD25+High Foxp3+ both in total lymphocytes and in CD4+ cells were significantly increased in patients having thrombocytopenia for abrief duration than in those with prolonged duration of thrombocytopenia (Table 4).
Clinical and Laboratory Data in Patients With Brief Duration and Patients With Prolonged Duration of Thrombocytopenia.a
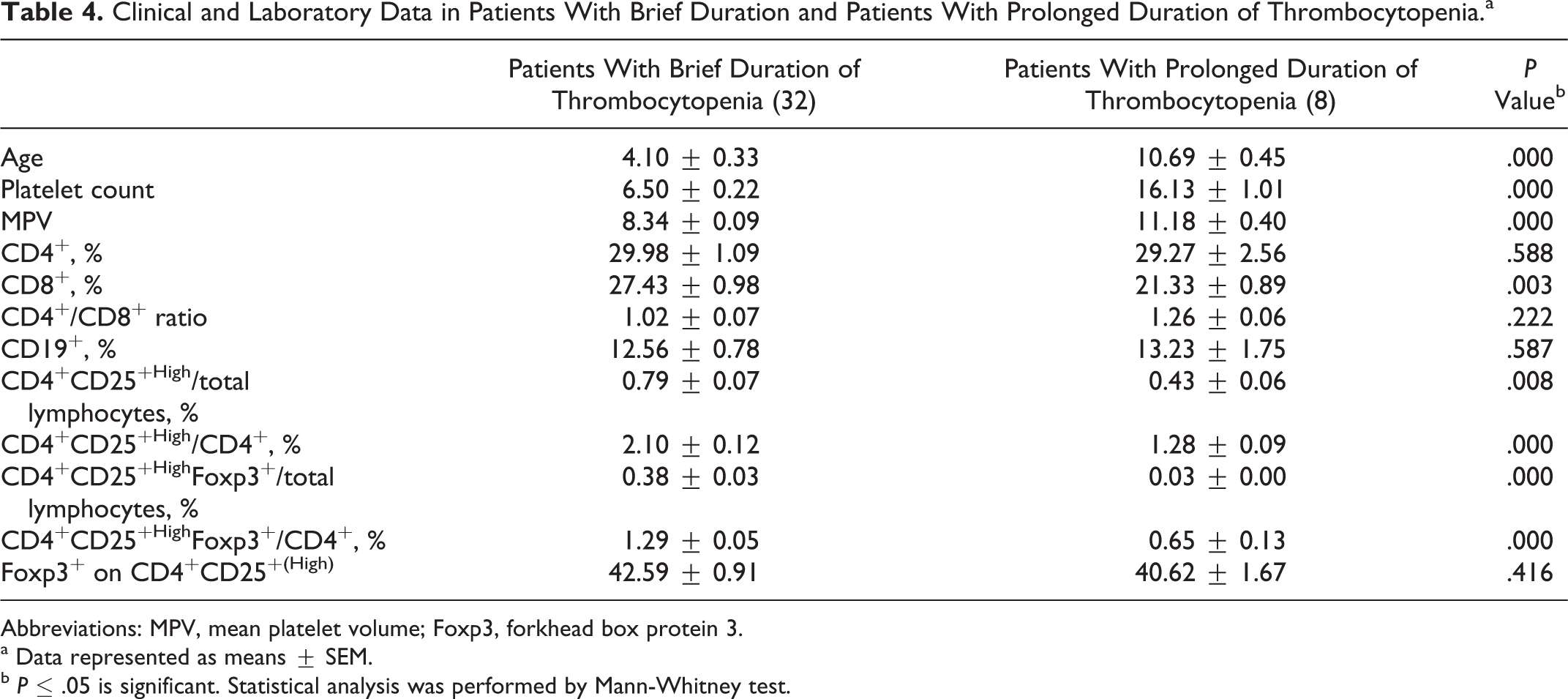
Abbreviations: MPV, mean platelet volume; Foxp3, forkhead box protein 3.
a Data represented as means ± SEM.
b P ≤ .05 is significant. Statistical analysis was performed by Mann-Whitney test.
Discussion
Patients having thrombocytopenia for a brief duration had lower age and platelet count than those with prolonged duration. This is in agreement with that of Edslev et al. 23 At the onset of the disease, the MPV was significantly lower in those having thrombocytopenia for a brief duration than those with prolonged duration. This is in agreement with Ahmed et al 27 who reported that patients with acute ITP with low MPV (<8) achieved a durable remission within 6 months. Also Sakha and Rezamands 28 reported good prognosis for patients with low and normal MPV.
In patients with ITP, the maintenance of self-tolerance and the effective immune response seems to be altered. In ITP, circulating antibodies and/or immune complexes adsorb to the platelet, resulting in early destruction by macrophages. Impaired platelet production and T cell-mediated platelet destruction play significant roles in more than 1 step of the immune regulation in ITP. 29,30
In the present study, the percentage of B cells (CD19+) was significantly higher in patients than in controls. This may be because the B cells are responsible for secreting antiplatelet antibodies, as patients with ITP often have elevated titers of antiplatelet antibodies. 31 Previous studies reported increased frequency of anti-glycoprotein IIb-IIIa antibody-producing B cells, which is similar with our finding. 32 –34
In our patients, we found disequilibrium in proportion of T lymphocyte subsets with relatively insufficient T helper cells and excessive T cytotoxic cells. Our results are consistent with that of Chen et al 33 and Wang et al. 35 The percentage of cytotoxic CD8+ cells was significantly increased in patients having thrombocytopenia for a broef duration than in those having thrombocytopenia for a prolonged duration. This may be explained by the fact that cytotoxic CD8+ cells are increased in patients with acute ITP as a response to viral infection. This is supported by the fact that 70% of our patients had preceding febrile illness. Immune thrombocytopenia has been attributed to thr production of platelets expressing viral antigens, 36 which induce cytotoxic CD8+ response as cytotoxic T cells destroy virally infected cells. 37 Evidence suggests a direct cytotoxic effect of T cells on platelets. The CD3+ T cells from patients with ITP were found to have an increased expression of genes involved in cell-mediated cytotoxicity, and the presence of cytotoxic T cells against autologous platelets was shown in patients with active ITP. 15,38,39 In patients with prolonged duration, the other immunological factors of thrombocytopenia as elevated titers of antiplatelet antibodies may be responsible for its pathogenesis.
The emergence of antiplatelet autoantibodies and antiplatelet cytotoxic T cells is a consequence of a loss of the immunological tolerance for self-antigens. 10 One of the several mechanisms that play a role in maintaining peripheral self-tolerance is the existence of naturally occurring regulatory T cells that actively prevent both the activation and the effector function of autoreactive T cells that have escaped different mechanisms of tolerance. 40,41
In this study, the frequency of total CD4+CD25+ and CD4+CD25+Intermediate both in total lymphocytes and in CD4+ cells was not significantly different in patients with ITP and controls, while the frequency of CD4+CD25+High and CD4+CD25+High Foxp3+ both in total lymphocytes and in CD4+ cells was significantly decreased in patients compared to controls. The present study confirms previous reports, indicating that in ITP patients with active disease, the number of regulatory T cells are reduced. 42 –45 The decrease in frequency of CD4+CD25+High and CD4+ CD25+High Foxp+ T cells in our patients may implies that a deficiency of regulatory T cells is associated with ITP pathogenesis. We consider CD4+CD25+High and CD4+CD25+High Foxp3+ as regulatory T cells, because the suppressive capacity of regulatory T cells in humans seems to be confined to CD4+CD25+ cells with the highest expression of CD25 (CD4+CD25High), whereas CD4+ T cell with intermediate expression of CD25 might also contain recently activated T cells and effector T cells without regulatory function. 46 –48 The expression of Foxp3 is considered an optimal marker for regulatory T cells. 49,50 The suppressive phenotype and the development of regulatory function depend on the expression of Foxp3. 51 –53
As the percentages of CD4+CD25+High and CD4+CD25+High Foxp3+ both in total lymphocytes and in CD4+ cells were significantly decreased in patients than controls, and they were significantly increased in patients having thrombocytopenia for a brief duration than in those having thrombocytopenia for aprolonged duration; so the estimation of CD4+ CD25+High and CD4+ CD25+High Foxp+ T cells may be a helpful prognostic marker in children with acute ITP.
The Foxp3 is a critical molecular switch for the genetic programming of natural regulatory T cell development and function. 52 –54 In this study, 42.20% ± 0.81% and 81.19% ± 2.60% of CD4+CD25High cells in patients and controls, respectively, expressed Foxp3, a hallmark of regulatory T cells, 49 while only 26.15% ± 1.40% and 28.06% ± 1.33% of CD4+CD25itermediate cells in patients and controls, respectively, expressed Foxp3. These results are consistent with those of Qian et al. 55 The expression of Foxp3+ in CD4+CD25+HighFoxp3 cells was significantly decreased in patients compared to controls. Ling et al 44 and Guo et al 56 studied the expression of Foxp3 messenger RNA (mRNA) by real-time quantitative reverse transcriptase–polymerase chain reaction in patients with ITP and found that the expression of Foxp3 mRNA in patients with ITP was lower than that of healthy controls.
In conclusion, in addition to young age, lower platelet count, and preceding febrile illness, lower MPV at presentation, and higher percentage of CD8 cytotoxic T cell may be added as a prognostic significance. Estimation of CD4+ CD25+High Foxp+ T cells may be a helpful prognostic marker in children with acute ITP. Therapeutic use of regulatory T cells in patients with ITP should be investigated in further research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
