Abstract
Objective
Deep vein thrombosis (DVT) is a common peripheral disease. This study aimed to elucidate the diagnostic biomarker of lncRNA nuclear-enriched abundant transcript 1 (NEAT1) in the DVT, and explore possible mechanisms in Human umbilical vein endothelial cells (HUVECs).
Methods
101 patients with lower extremity DVT and 82 healthy controls were enrolled. RT-qPCR was designed to resolve the mRNA levels of NEAT1, miR-218-5p, and GAB2. ROC was applied for the diagnosis of DVT. Systemic inflammation (IL-1β, IL-6, and TNF-α) and adhesion factor (SELP, VCAM-1, and ICAM-1) were examined by the ELISA. And cell proliferation, migration, and apoptosis were conducted by the CCK-8, Transwell, flow cytometry assay. The targeting relationship was validated by Dual luciferase reporter and RIP analysis.
Results
NEAT1 and GAB2 were upregulated in patients with DVT, while miR-218-5p was decreased (P < .01). Serum NEAT1 can identify DVT patients from healthy individuals. NEAT1 was positively correalted with fibrinolysis factors, coagulation factors, and vasoconstrictors. NEAT1 inhibited the proliferation, migration, and promoted apoptosis as well as inflammation and adhesion factors secretion of HUVECs (P < .05), but all were impaired by overexpression of miR-218-5p (P < .05). NEAT1 promoted GAB2 expression in DVT by acting as a sponge for miR-218-5p.
Conclusion
Elevated NEAT1 is a possible DVT diagnostic biomarker, and is implicated in vascular endothelial cell dysfunction via miR-218-5p/GAB2 axis.
Introduction
Deep vein thrombosis (DVT) is a cardiovascular illness in which a thrombus develops in the lumen of a deep vein, impairing blood flow. 1 DVT, is the world's third most common blood illness, with up to 50 000 new cases per year in the United States and up to 12 million in China.2,3 DVT misdiagnosis is common due to its gradual development and the absence of clinical signs in the early stage. 4 Furthermore, the currently utilized diagnostic biomarker D-dimer is significantly expressed in non-thrombotic illnesses such as stroke, infection, and diffuse vascular coagulation, discounting its specificity. 5 Although spontaneous breakdown is uncommon, some individuals continue to lose out on effective diagnosis and therapy, resulting in impaired lower extremity function, ulceration, 20%–50% post-thrombotic syndrome, and even 10% fatal pulmonary embolism. 6 Anticoagulation, thrombolysis, and thrombectomy have all been tried to prevent and cure DVT, although they have not always been effective. 1 As a result, prompt and precise diagnosis, as well as effective therapies, are especially critical for DVT patients.
Long non-coding RNA (lncRNA) is a member of the non-coding RNA family, which is characterized as longer than 200 base pairs. Abundant LncRNAs are engaged in the onset and development of DVT, highlighting their potential as biomarkers and therapeutic tools. Disorders of lncRNA ANRIL, 7 GUSBP5-AS, 8 and MALAT1 9 have been proven to be associated with the development of DVT. LncRNA nuclear-enriched abundant transcript 1 (NEAT1), also known as LINC00084, has been identified as a regulator of para-nucleosome stability on human chromosome 11q13.1. 10 Damage to vascular endothelial cells causes the cells to lose their usual anticoagulation, increasing thrombosis. In hemangioma endothelial cells, NEAT1 silencing reduces proliferation and promotes apoptosis. 11 NEAT1 suppression alleviates ox-LDL-induced damage in human umbilical vein endothelial cells (HUVECs) through miR-30c-5p. 12 Furthermore, atherosclerotic (AS) disease produces lumen constriction and increased blood viscosity, both of which can lead to thrombosis. 3 NEAT1 levels are higher in those with coronary artery disease or carotid AS plaques, and silencing it reduces vascular endothelial cell inflammation. 13 Dou et al. discovered that NEAT1 is abnormally high in the serum of individuals with pulmonary hypertension, a typical consequence of DVT. 14 Unfortunately, the understanding of NEAT1 in DVT pathophysiology is limited.
Therefore, we conducted the present investigation to explore the clinical actions and potential mechanisms of NEAT1 in DVT, in an attempt to contribute new insights and approaches to the pathogenesis and management of DVT.
Materials and Methods
Ethical Approval
The Affiliated Hospital of PanZhiHua University Ethics review board accepted the clinical protocol (approval number: 2018012). Before the experiment, the subjects provided written informed consent. And the investigation was carried out following the Helsinki Declaration.
Participants in This Research
Patients aged 18–60 years with idiopathic lower extremity DVT first diagnosed from January 2018 to February 2020 in Affiliated Hospital of PanZhiHua University were enrolled. Patients were diagnosed with DVT by color Doppler ultrasound and lower extremity angiography and were not treated with anticoagulants or surgery before treatment. Additionally, patients with diabetes, neurological disorder, coagulation disorders, cancer, NAFLD, liver fibrosis, autoimmune system diseases, and moderate infectious diseases, and women during pregnancy were excluded. Finally, 101 patients with DVT were included in this study. Besides, 82 healthy individuals who were examined at the same time and matched for age and gender were collected as controls, which had normal Doppler ultrasound tests and no combined chronic diseases, and no statistical differences in age, sex, and smoking demographic characteristics between individuals. Biochemical indicators and demographic characteristics of patients were recorded in Table 1. After fasting 6 h, peripheral venous blood was collected from the patient's median elbow vein. The blood specimens were allowed to rest at room temperature, then centrifuged at 3000 rpm for 10 min before collecting the serum which was subsequently stored at −80°C.
Baseline Characteristics of the Subjects.

Abbreviations: BMI, body mass index; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; TC, total cholesterol; SBP, systolic blood pressure, DBP, diastolic blood pressure.
Cell Culture and Transfection
HUVECs (cat No. BNCC342438) were acquired from the BeNa Culture Collection (Beijing, China). And maintained in DMEM (D6046, Sigma-Aldrich, USA) containing 10% fetal bovine serum (16000044, Gibco, Thermo Scientific), 10% endothelial cell growth supplement (356006, BD biosciences, USA), and cultured at 37°C in humidified 5% CO2 incubator. NEAT1 small interfering (si) RNA (si-NEAT1, C02004) and its negative control siRNA (si-NC, C03002), and overexpression plasmid of pcDNA3.1-NEAT1 and pcDNA3.1 were acquired from GenePharma Co. miR-218-5p mimic (miR10000275-1-5), miR-218-5p inhibitor (miR20000275-1-5), mimic NC (miR1N0000001-1-5), and inhibitor NC (miR2N0000001-1-5) were all from the RIBOBIO Co. Transfection was performed when HUVECs grew to logarithmic growth phase with a confluence of 80%. The above vector was mixed with Lipofectamine 3000 (L3000015, Invitrogen, USA) in DMEM without FBS and then dropped into the cells. Continue to culture for 5 h followed by a new medium.
RNA Extraction and Real-Time Quantitative Reverse Transcription PCR (RT-qPCR)
Total RNA from subjects’serum and HUVEC cells were isolated and extracted by Trizol LS reagent (10296010, Invitrogen, USA). Fastking gDNA Dispelling RT SuperMix Kit (KR118-02) and miRcute Plus miRNA First Stand cDNA Kit (KR211, TIANGEN, Beijing, China) reverse-transcribed RNA after purity identification (OD260/280 = 1.8-2.2) into cDNA. The amplified reaction solution was prepared according to the instruction of SuperReal PreMix Plus (SYBR Green, FP205-02) or miRcute Plus miRNA qPCR Kit (SYBR Green, FP401, TIANGEN, Beijing, China), which contains forward primers, reverse primers, kit reagents, and template DNA. The system was supplemented with ddH2O to a final volume of 20 μl. The PCR amplification reactions were performed on ABI PRISM 7300 (ABI). GAPDH served as an internal control of NEAT1 and GAB2, U6 was applicated as an internal control for miR-218-5p, and the 2−ΔΔCt method calculated the relative expression levels. All experiments were repeated three times.
Cell Proliferation and Apoptosis Assays
For cell proliferation, 1 × 104 HUVCEs were inoculated into 96-well plates. After every 24 h, DMEM and cell counting kit-8 (CCK-8, CK04, Dojindo, Japan) in a ratio of 9:1 followed by incubation for an additional 2 h. The absorbance at 450 nm was measured. The apoptosis was examined using the Annexin V-FITC/PI apoptosis detection kit (556547, BD Biosciences, USA). Specifically, transfected cells were re-suspended in a binding buffer containing PI and FITC, protected from light, and subsequently analyzed on a flow cytometer.
Cell Migration Assay
Transwell (3422, Corning, USA) was utilized to explore migration. Briefly, cells were resuspended in DMEM containing 1% FBS and inoculated into the upper chamber of the Transwell. The lower chamber was filled with 500 μl of DMEM containing 10% FBS and incubation was continued for 24 h in the incubator. After removing the cells in the upper chamber, the cells that migrated to the lower part of the chamber were fixed with methanol for 10 min and stained with crystal violet for 20 min. Following a water rinse, cell counting was performed under a light microscope.
Enzyme-Linked Immunosorbent Assay (ELISA)
Human serum samples and supernatant culture of HUVECs were collected and protein expression levels of tumor necrosis factor (TNF)-α (ab181421), IL (interleukin) −6 (IL-6, ab178013), and IL-1β (ab229384), and intercellular adhesion molecule 1 (ICAM1, ab174445), Vascular cell adhesion molecule 1(VCAM-1, ab223591) and p-selection (SELP, ab272202, Abcam, USA) were measured in triplicate using commercial human ELISA kits. The levels of fibrinogen (ab241383), endothelin-1 (ET-1, ab133030), platelet-activating factor (PAF, ab187801), tissue-type plasminogen activator (t-PA, ab108914) and D-dimer (ab229437, Abcam, USA) were also examined by the ELAIS.
Dual Luciferase Reporter (DLR) Assay
ENOCRI predicted the target relation for NEAT1 and miR-218-5p, and TargetScan 7.2 for the GAB2 and miR-218-5p. Sequences or the mutated sequences of the respective 3′- UTRs of NEAT1 and GAB2 which contain miR-218-5p were subcloned into the pmirGLO luciferase vector (Promega). The wild-type (WT) plasmids of NEAT1-WT and GAB2-WT and mutant (MUT) plasmids of NEAT1-MUT and GAB2-MUT were constituted, respectively. Lipofectamine 3000 was used to co-transfected their plasmid with miR-218-5p mimic or inhibitor. After 48 h, cells were harvested and luciferase activity was detected by DLR (E1960, Promega, USA).
Nuclear-Cytoplasmic Fractionation
The PARISkit (AM1921, Invitrogen, USA) was utilized to segregate the cytoplasm or nucleus of NEAT1. Briefly, HUVECs were lysed on ice, subsequently collected and vortexed for 15 s to ensure complete suspension of cell particles, and hatched on ice for 10 min. Pre-chilled CERII from the kit was then added. The cytoplasmic extract was obtained by centrifugation at 1200 rpm for 5 min, followed by the collection of the precipitate and addition to the NER reagent, which was left on ice for 10 min. After centrifugation, the cell nuclear extract (supernatant) was removed for RT-qPCR quantification of NEAT1 expression in both cytoplasm and nucleus using U6 and GAPDH as nuclear and cytoplasmic controls.
RNA Immunoprecipitation (RIP) Assay
Magna RNA immunoprecipitation (RIP) kit (17-701, Millipore, USA) was utilized to investigate the binging of miR-218-5p to NEAT1 or GAB2. HUVECs were lyse using kit buffer and cell extracts were incubated with magnetic beads conjugated to human anti-Ago2 antibodies or control IgG. The samples were collected and treated with protease K to digest the protein and the total RNA was used to measure the expression of NEAT1, miR-218-5p, and GAB2 by RT-qPCR analysis.
Statistical Analysis
The above experiments were conducted in triplicate and the results were reported as mean ± SD. Data analysis and charting were performed using GraphPad Prism 6.0 and SPSS 23.0 with statistical analysis including Student's t-test (for two groups) and ANOVA followed by Tukey (for multiple groups) were examined for statistical analysis. Spearman correlation was used to explore the relationship between miRNA and LncRNA or gene. ROC curve was employed to assess the diagnostic value for DVT. P < .05 was indicated as statistically significant.
Results
Demographic and Clinical Characteristics of the Subjects
The baseline characteristics of the subjects are summarized in Table 1. Age, gender, smoking, alcohol, and other demographic and clinical characteristics were statistically insignificant in both groups (P < .05). However, fibrinogen, ET-1, PAF, TAT III, t-PA, D-dimer, hs-sensitivity C-reactive protein (hs-CRP), IL-6, and TNF-α in DVT patients were significantly higher in patients with DVT compared to healthy controls (P < .05, Table 2).
Biomarkers of Venous Thromboembolism.

Abbreviations: ET-1, endothelin-1; t-PA, tissue type plasminogen activator; PAF, platelet activating factor; hs-CRP, high-sensitivity C-reactive protein; TAT III, thrombin-an-thrombin III; IL-6, interleukin 6; TNF-α, tumor necrosis factor α
Elevated NEAT1 Positivity Correlates with Thromboembolic Biomarkers and has Diagnostic Potential for DVT
To elucidate the function of NEAT1 in DVT, RT-qPCR was performed. As illustrated in Figure 1A, serum NEAT1 was statistically elevated in patients with DVT (1.83 ± 0.54) than in healthy controls (1.00 ± 0.423, P < .05). In particular, elevated ENAT1 expression was positively correlated with venous thromboembolic markers in patients with DVT (P < .05, Table 3). ROC curves were conducted to evaluate the diagnostic abilities of NEAT1. The AUC was 0.895 (95%CI, 0.850–0.940), and at a cut-off value of 1.405, NEAT1 showed a high diagnostic potential with a sensitivity of 86.6% and specificity of 81.2% in identifying DVT patients from healthy individuals (P < .05, Figure 1B).

Serum NEAT1 expression in DVT patients and its diagnostic value.
Correlation of NEAT1 Expression and Biomarkers of Venous Thromboembolism.

Abbreviations: ET-1, endothelin-1; t-PA, tissue type plasminogen activator; PAF, platelet activating factor; hs-CRP, high-sensitivity C-reactive protein; TAT III, thrombin-an-thrombin III; IL-6, interleukin 6; TNF-α, tumor necrosis factor α
NEAT1 Inhibits Cell Proliferation and Migration, Promotes Apoptosis, and Stimulates the Secretion of Inflammation and Adhesion Molecule
To explore whether NEAT1 plays a role in DVT occurrence, we transfected si-NEAT1 and pcDNA3.1-NEAT1 plasmids into HUVECs. As illustrated in Figure 2A, the expression of NEAT1 was typically inhibited by si-NEAT1 (0.41 ± 0.17) or promoted by pcDNA3.1-NEAT1 (2.27 ± 0.32) than the control (1.00 ± 0.09, P < .05). In addition, overexpression of NEAT1 typically suppressed cell proliferation (0.54 fold) and migration (0.50 fold), while promoting apoptosis (0.46 fold) in HUVECs. Conversely, si-NEAT1 enhanced cell proliferation (1.46 fold) and migration (2.30 fold), while suppressing apoptosis (0.46 fold) compared to controls (P < .05, Figure 2B-D). Elevated NEAT1 levels also significantly increased concentrations of pro-inflammatory factors TNF-α (1.61 fold), IL-1β (1.90 fold), and IL-6 (1.68 fold) compared to controls, whereas si-NEAT1 led to the opposite result (P < .05, Figure 2E). Overexpression of NEAT1 significantly reduced the expression levels of adhesion factors SELP (0.47 fold), ICMA-1 (0.72 fold), and VCAM-1 (0.53 fold), while si-NEAT1 typically elevated their expression levels compared to controls (P < .05, Figure 2F).
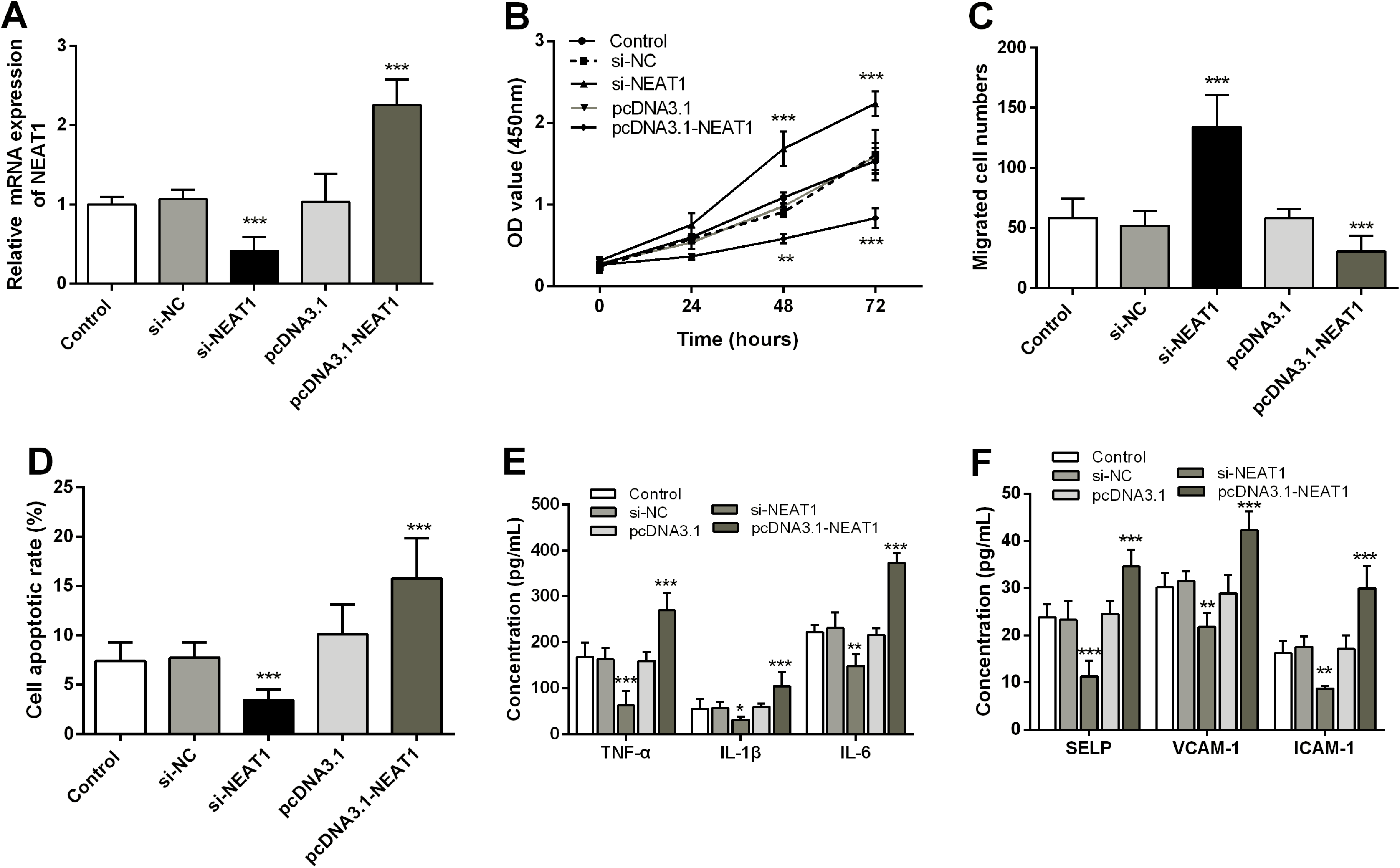
NEAT1 inhibits cell proliferation and migration, promotes apoptosis, and regulates levels of inflammatory and adhesion factors. A. RT-qPCR was applied to resolve NEAT1 levels in HUVECs transfected with si-NEAT1 and pcDNA3.1-NEAT1 plasmids. CCK-8 (
NEAT1 as ceRNA Competitively Binds miR-218-5p in DVT
To investigated the potential mechanisms of NEAT1 in DVT, we analyzed its subcellular localization and found that it is predominantly located in the cytoplasm (81.62%) of HUVECs (Figure 3A). We then utilized biological information software to identify miRNAs that interacted with NEAT1, and discovered a target binding sequence for miR-218-5p (Figure 3B). DLR assay demonstrated that transfection with a miR-218-5p mimic resulted in over a 0.5-fold decreased in luciferase activity of NEAT1-WT group (P < .05), but had insignificant effect on luciferase activity of NEAT1-MUT group (P > .05, Figure 3C). RIP assay confirmed that both miR-218-5p (4.27 fold) and NEAT1 (4.48 fold) were enriched in the Ago-IP fraction (P < .05, Figure 3D). Next, serum miR-218-5p was found to be decrased 0.52 fold in the patients with DVT compared to the HC group (P < .05, Figure 3E). With the increase of NEAT1, there was a significant downregulation in the level of miR-218-indicating a negative correlation between them (r = −0.699, P < .05, Figure 3F). Finally, miR-218-5p was typically by 2.06 fold increased in HUVECs that interfere with NEAT1, while overexpression of NEAT1 significantly by 0.27 fold eliminated miR-218-5p levels compared with controls (P < .05, Figure 3G).
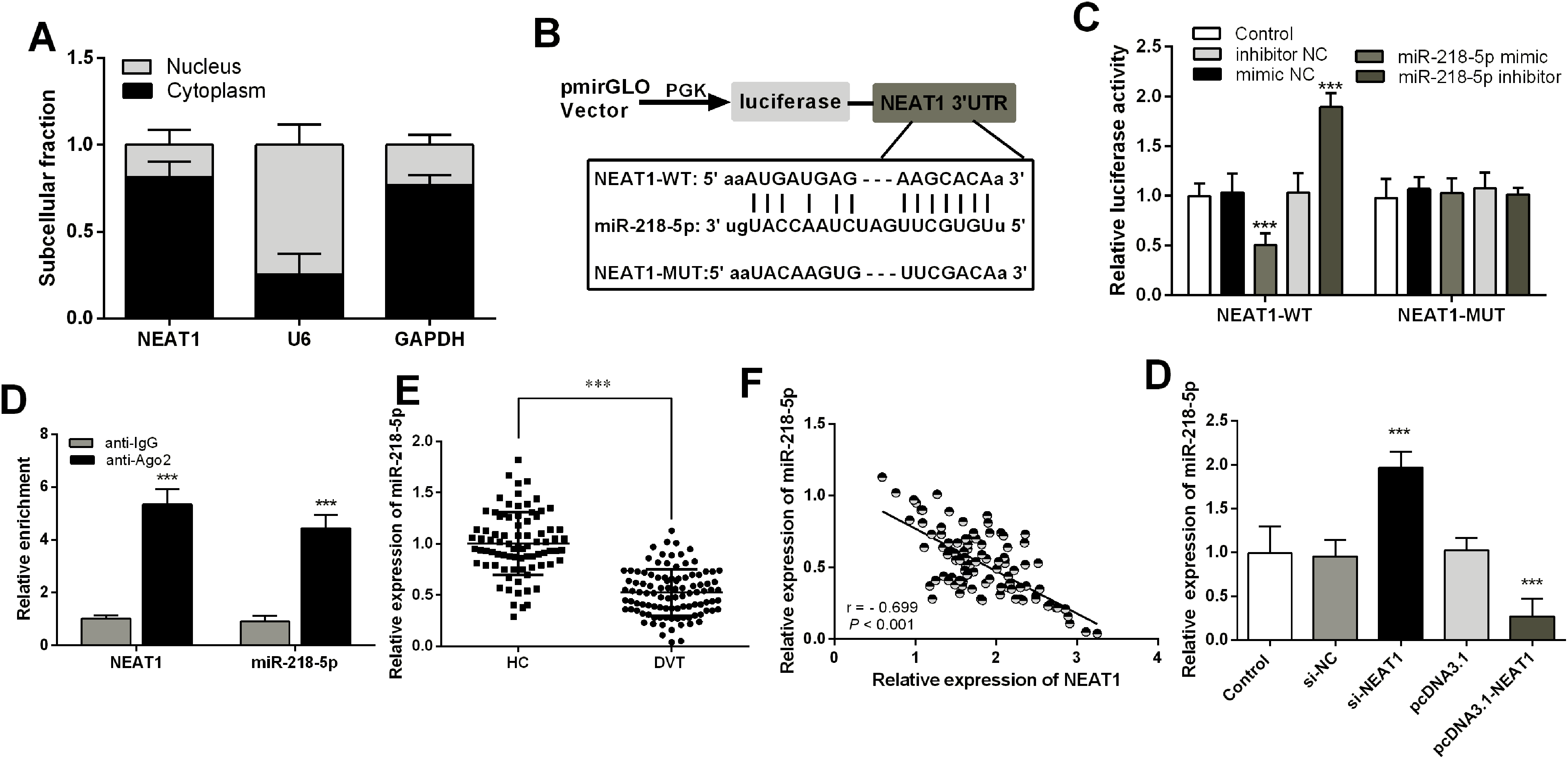
NEAT1 as ceRNA competitively binds miR-218-5p in DVT.
NEAT1 Affects the Biological Function of HUVECs Cells in DVT by Regulating miR-218-5p
To investigate the regulatory role of NEAT1 in DVT and its dependence on miR-218-5p, we transfected HUVCEs overexpressing NEAT1 with a miR-218-5p mimic. Our results observed a significant increase in miR-218-5p levels (2.98 fold, P < .05, Figure 4A). Furthermore, elevated NEAT1 inhibited cell proliferation and migration compared to control; however, this inhibition was significantly reversed by miR-218-5p (P < .05, Figure 4B-C). Additionally, promotion of apoptosis in HUVECs by NEAT1 was significantly reduced (0.64 fold) by miR-218-5p (P < .05, Figure 4D). What's more, NEAT1 significantly increased the overproduction of inflammatory factors TNF-α (1.50 fold), IL-1β (1.53 fold) and IL-6 (1.46 fold) and adhesion factors SELP (1.89 fold), ICMA-1 (1.54 fold) and VCAM-1 (1.51 fold), but this increase was typically attenuated by miR-218-5p (P < .05, Figure 4E-F).

NEAT1 affects the biological function of HUVECs cells in DVT by regulating mir-218-5p. A. RT-qPCR was applied to resolve miR-218-5p levels in HUVECs transfected with pcDNA3.1-NEAT1 and miR-218-5p mimic plasmids. CCK-8 (
NEAT1 Regulates GAB2 Expression Through Competitive Binding of miR-218-5p
To identify the target of miR-218-5p, TargetScan 7.2 predicted a putative binding site for GAB2 to miR-218-5p (Figure 5A). DLR analysis confirmed that the relative luciferase activity of GAB2-WT was typically (0.51 fold) reduced by a miR-218-5p mimic in HUVECs, whereas there was no significant difference in luciferase activity for NEAT1-MUT (P < .05, Figure 5B). RIP assay confirmed significant enrichment of NEAT1 (4.13 fold), miR-218-5p (6.04 fold), and GAB2 (8.10 fold) in Ago within HUVECs (P < .05, Figure 5C). Further experiments revealed that serum GAB2 was upregulated by in 2.10-fold in the patients with DVT (P < .05, Figure 5D) and exhibited a negative correlation with miR-218-5p (r = −0.627, P < .05, Figure 5E) as well as a positive correlation with NEAT1 (r = 0.601, P < .05, Figure 5F). Additionally, DLR also found that the reduction (0.38 fold) of GAB2-WT luciferase activity by miR-218-5p mimic was elevated (2.30 fold) by overexpression of NEAT1 (P < .05, Figure 5G). miR-218-5p was (2.16 fold) elevated in NEAT1 overexpressing HUVECs, but this elevation was typically (0.68 fold) abolished by miR-218-5p (P < .05, Figure 5H).

NEAT1 regulates GAB2 expression through competitive binding of miR-218-5p.
Discussion
DVT is a prevalent arteriovenous thromboembolic disease. Due to its insidious onsent rapid progression, and partially asymptomatic characteristics, numerous laboratory biomarkers have been explored for reliable diagnosis of DVT. Although D-dimer is currently the only and best diagnostic biomarker for DVT, its ground specificity discounts its diagnostic value as it is also highly expressed in non-thrombotic diseases such as disseminated vascular coagulation, stroke, and infection. 15 Additionally, while anticoagulation (heparin anticoagulation, vitamin K anticoagulation), vena cava filters, thrombolysis (urease, streptokinase, tissue fibrinogen activator), and thrombus extraction are utilized in the treatment of DVT treatment, 16 their efficacy is always guaranteed.
In recent years, numerous studies have highlighted the irreplaceable role of ncRNAs, particularly LncRNAs and miRNAs in the diagnosis and treatment of cardiovascular diseases, especially DVT, such as LncRNA TUG1, 17 MALAT1, 18 sirT1-AS1. 19 NEAT1 has been implicated in various pathologies. For example, NEAT1 is upregulated in the blood of patients with coronary artery disease and human coronary artery endothelial cells, 20 as well as elevated in the peripheral blood of patients with acute ischemic stroke, serving as a prognostic biomarker. 21 Hypertension and diabetes can lead to vascular endothelial cell damage and thrombosis formation due to metabolic disorders. 3 Additionally, NEAT1 has been shown to participate in the alleviation of diabetic retinopathy through crocetin treatment. 22 High glucose induces a time-dependently upregulates of NEAT1 expression, which is elevated in diabetic nephropathy and accelerates proliferation, oxidative stress, and inflammation. 23 Additionally, the increase in NEAT1 activates autophagy and exacerbates myocardial ischemia injury in diabetic rats. 24 Furthermore, AS leads to luminal narrowing of the blood vessel and increased blood viscosity, both of which can contribute to thrombosis. Trimethylamine N-oxide (TMAO) induced endothelial cell dysfunction and increased NEAT1 expression in atherosclerotic (AS) patients when it enhanced the thickened inner and outer vessel walls as well as membrane plaques. 25 Given the potential function of NEAT1 in the above thrombosis-related disease, we hypothesized that it may also play a significant role in DVT. Our findings confirmed our hypothesis as NEAT1 expression was significantly elevated in DVT patients and showed a positive correlation with conventional thrombus biomarkers. These results suggest that serum NEAT1 has diagnostic value for distinguishing DVT patients from healthy individuals. Thrombosis is the formation of an occlusive blood clot within a blood vessel, resulting inobstruction of blood flow and reduced delivery of nutrients and oxygen to distant tissues. Ultimately, thrombosis may lead to tissue and organ necrosis. 26 Previous studies have confirmed that vessel wall damage, changes in blood flow, and abnormal blood composition are the three most significant factors contributing to thrombosis.4,27 And the thrombotic process involves vascular endothelial cells, coagulation/anticoagulation system, fibrinolytic/antifibrin system, platelets, inflammation, and other factors. 28 Among them, vascular wall damage is caused by endothelial damage of blood vessels. Endothelial cells act as a barrier separating the blood from surrounding tissues and play an indispensable role in the hemostatic system. Normal endothelial cells can express anticoagulant molecules that inhibit platelet aggregation and fibrin formation. However, when endothelial cell damage or apoptosis impairscells damage or apoptosis impairs their normal anticoagulant effect, promoting thrombosis, and affecting intravascular reactive substance levels to aggravate thrombosis.3,29 The importance of vascular endothelial cells necessitates investigation into mechanisms in vitro. Mechanistically, we found that NEAT1 significantly inhibited HUVECs proliferation and migration while increasing apoptosis. A significant positive correlation was found between NEAT1 and vasoconstrictors ET-1, which suggested that blood vessel walls were damaged. Additionally, d-dimer is a distinctive product resulting from the crosslinking of birdbrain monomers by activating factors and subsequent hydrolysis by plasminase. It serves as a molecular marker that reflects the fibrinolytic activation of coagulation and indicates the formation of thrombosis. A significant positive correlation between NEAT1 and d-dimer was also found in our study. Additionally, both PAF and TAT III were also positively correlated with NEAT1, suggesting the relevance of NEAT1 to coagulation and fibrinolytic activation in thrombus in the pathological progression of DVT.
Furthermore, the DVT process is closely associated with inflammatory factors. Elevated levels of IL-6 have been reported in patients with DVT, and persistently dysregulated IL-6 can lead to tissue damage and plays an important role in the inflammatory injury of vascular endothelium. 30 TNF-α has been found to generate free radicals that harm vascular endothelial cells, cause vasoconstriction, exacerbate clotting conditions, and promote thrombosis. this can result in cerebral ischemia, hypoxia, and even more severe consequences. 31 It is well known that ICAM-1, VCAM-1, and SELP, as adhesion molecules, are markers of vascular function. 30 In our study, the NEAT1 overexpression group exhibited significantly elevated levels of inflammatory and adhesion factors. Therefore, it can be concluded that NEAT1 may contribute to vascular endothelial dysfunction by promoting the apoptosis of vascular endothelial cells and the secretion of inflammatory and adhesion factors.
In general, lncRNAs can function as competing endogenous RNAs (ceRNAs) for microRNAs (miRNAs) to regulate gene expression and thus participate in disease progression. Therefore, we investigated the potential miRNA and mRNA of NEAT1 in DVT. miR-218-5p has been identified to have potential binding sites with NEAT1. miR-218-5p is down-regulated in atherosclerosis of endothelial dysfunction 32 and is a biological marker for its early clinical diagnosis. 33 It was decreased in glucose-induced human retinal pigment epithelial cells 34 and up-regulated to relieve diabetic nephropathy. 35 miR-218-5p is also implicated in the common complication of DVT, chronic obstructive pulmonary disease. 36 Endothelial progenitor cells (EPCs) are precursors to vascular endothelial precursor cells, and previous studies have shown that EPCs are recruited into thrombi to accelerate their lysis. 37 miR-218-5p has been reported to play a role in regulating EPCs function. 38 More importantly, Zhang et al. conducted a study on numerous miRNAs that were differentially expressed in rat DVT. In this study, miR-218-5p was identified and its targeting relationship with NEAT1 was demonstrated in breast cancer. 6 And the targeting relationship between miR-218-5p and NEAT1 has been demonstrated in breast cancer. 39 As previously demonstrated, our findings confirm the targeting between the NEAT1 ad miR-218-5p, which is down-regulated in patients with DVT Additionally, we observed that miR-218-5p also weakened the promoting effect of NEAT1 on the dysfunction of HUVECs cells. miR-218-5p targets GAB2 in cerebral ischemia-reperfusion injury 40 and malignant transformation of esophageal carcinoma epithelial cells. 41 Additionally, GAB2 has been implicated in the formation of DVT and regulation of thrombus inflammation. 42 Our study also confirms miR-218-5p's targeting ofGAB2, revealing a negative correlation between them while also identifying a positive correlation between GAB2 and NEAT1. Serum levels of GAB2 were found to be up-regulated in patients with DVT.
It is undeniable that although we have confirmed the potential of NEAT1 as a biomarker for patients with DVT, it should be noted that NEAT1 is frequently overexpressed in human tumors and its expression has been linked to reduced survival rates among cancer patients. NEAT1 has been identified as a novel biomarker for different types of cancer and also promotes the progression of non-alcoholic fatty liver disease (NAFLD), liver fibrosis, and so on. However, upon implementing stringent inclusion and exclusion criteria, we have discovered that its expression levels can significantly distinguish patients with DVT. While its clinical application remains distant, our study has laid the groundwork for future investigations. Furthermore, based on our current results we found that NEAT1 may be involved in various pathological states of DVT. Firstly, it affected the proliferation, apoptosis, and inflammatory damage of HUVECs, suggesting that NEAT1 may be involved in endothelial damage. Also, t-PA as a product of endothelial injury and fibrinolytic system activation confirms this view. Furthermore, the association of NEAT1 with Fibrinogen and D-dimer suggests its involvement in the hypercoagulable state as a procoagulant in DVT. However, the mechanism of the specific impact still needs further in-depth study, which will be the focus of our next studies.
In general, elevated NEAT1 is a potential diagnostic biomarker for DVT and is involved in vascular endothelial cell dysfunction in DVT through the miR-218-5p/GAB2 axis. Our study suggests new perspectives and possibilities for the treatment and diagnosis of DVT.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The clinical program was endorsed by the Affiliated Hospital of PanZhiHua University Ethics review board, written informed consent was secured, and the investigation was carried out under the Declaration of Helsinki.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
Informed Consent
Written informed consent was obtained from the patient(s).
