Abstract
Activated partial thromboplastin time (aPTT) or prothrombin time (PT) is often detected after rivaroxaban administration, and it is known that aPTT or PT can be prolonged or normal. However, the clinical risk factors and outcomes of prolongation were unknown. In a single-center, retrospective case-control study, adult inpatients who had aPTT/PT tested before and after administration of rivaroxaban during a designated 12-month period were eligible for inclusion. Depending on whether their aPTT/PT was prolonged, patients were allocated to the prolonged case or normal control group. Demographics, rivaroxaban indications and daily dose, and laboratory values were compared. Multivariate logistic regression was used to identify independent risk factors. The changes in laboratory values and clinical outcomes were analyzed. A total of 155 patients were included in the study among which 54 (34.84%) were reported to have either or both aPTT/PT prolonged. The average prolongation time of PT was between 0.8 and 0.9 s, and 1.8 and 3.5 s for aPTT. Multivariable regression modeling showed that height (odds ratio [OR] 1.12, 95% confidence interval [CI] 1.02-1.22, P = .02) and human albumin replacement (OR 3.19, 95% CI 1.05-9.74, P = .04) were independent risk factors for aPTT/PT prolongation. The incidence of bleeding events and changes in laboratory values were similar between the groups. Patient height and receiving human albumin replacement were independent risk factors for aPTT/PT mild prolongation caused by rivaroxaban. The prolongation was not associated with an increased occurrence of bleeding events.
Keywords
Introduction
As an effective alternative to warfarin and low molecular weight heparin (LMWH), direct oral anticoagulants (DOACs) require neither invasive administration nor frequent laboratory monitoring, and their excellent safety, efficacy, and patient compliance are widely favored by patients and physicians, making them an increasingly popular choice for the prevention and treatment of thromboembolic diseases. It was estimated that the proportion of US nursing home residents with atrial fibrillation treated with DOACs increased from 8.6% in 2011 to 47.8% in 2016, and 35.8% of those on DOACs were receiving rivaroxaban. 1 Despite the wide therapeutic window of rivaroxaban, the most common adverse effect similar to other anticoagulants, is bleeding, especially for specific populations with high risks, for example, elderly patients, those with hepatic or renal insufficiency, and those receiving concomitant treatment with strong inhibitors of both CYP3A4 and glycoprotein (P-gp). 2
Laboratory assays to monitor DOAC concentrations can help to improve the effectiveness and safety of using these medications. The International Council for Standardization in Hematology recommends using liquid chromatography with tandem mass spectrometry to determine the concentration of rivaroxaban as the gold standard for drug concentration monitoring. 3 The chromogenic antifactor Xa assay is another widely recognized method for which sensitivity and specificity have been proven in several studies.4–6 However, both laboratory tests are time consuming and require comparatively expensive equipment thus have limited availability. On the other hand, routine coagulation tests, such as prothrombin time (PT)/international normalized ratio and activated partial thromboplastin time (aPTT), do not accurately reflect the drug concentration of rivaroxaban. There was an unstable linear correlation between PT or aPTT values and rivaroxaban concentrations, and test results were significantly influenced by assay reagents and laboratories.7–15 Nevertheless, a prolonged PT and/or aPTT in a patient with known DOACs exposure should be expected and is likely to be drug related.16–18 In the past, studies have explored the influence of DOACs on routine coagulation assays, or the relationship between drug concentration and coagulation test results.19,20 However, little is known about the clinical characteristics of patients with prolonged PT and/or aPTT while receiving the standard pharmacotherapy of DOACs such as rivaroxaban. Most importantly, the clinical relevance of these abnormal coagulation test results is not clear.
Therefore, for the present retrospective case-control study, we analyzed the clinical characteristics of patients who were taking rivaroxaban with prolonged PT and/or aPTT, and explored the association of these characteristics with aPTT/PT prolongation. In addition, we evaluated the changes in laboratory values related to hemorrhage and subsequent clinical outcome of those patients with aPTT/PT prolongation receiving rivaroxaban administration.
Method
Study Design
This single-center, retrospective case-control study was conducted in a large tertiary care teaching hospital in China. This study was approved by the Institutional Review Board at the First Affiliated Hospital of Chongqing Medical University (2020-371). The requirement for informed consent was waived because of the anonymous nature and retrospective analysis of the data. Patient medical orders of rivaroxaban prescribed from June 30, 2020 to June 30, 2021 were obtained from the pharmacy system. Patients with the following conditions were included in the study: (1) hospitalized patients aged 18 years and older who were treated with rivaroxaban and with at least one reported aPTT and PT testing results during rivaroxaban administration and (2) patients with normal aPTT and PT testing results reported within 7 days before initiation of rivaroxaban. Patients with the following conditions were excluded from the study: (1) shortened PT or aPTT during rivaroxaban administration; (2) missing important clinical data such as body mass index (BMI) and some laboratory parameters; and (3) non-rivaroxaban-related death.
Data Collection
We reviewed the PT and aPTT assays from 7 days before the administration of rivaroxaban, and the most recent test results within 7 days prior to rivaroxaban administration were adopted. All PT and aPTT test results during rivaroxaban administration were reviewed, and by comparison with coagulation testing normal range of the institutional laboratory, patients with at least one prolongation of PT and/or aPTT were allocated to the prolonged case (PC) group, and their last abnormal coagulation test results were recorded if there were several. Patients with normal PT and aPTT test results were allocated to the normal control (NC) group, and their last coagulation test results were recorded if there were several.
The following data of both groups were collected and comparisons were made: (1) demographic data such as age, gender, height, weight, BMI, blood pressure, surgery, the presence of comorbidities (hypertension, diabetes mellitus, coronary artery disease, cerebrovascular disease, chronic liver disease, and chronic renal disease), other medications used during hospitalization (parenteral anticoagulants, antiplatelet agents, nonsteroidal anti-inflammatory drugs [NSAIDs], antibacterial drugs, insulin, steroid, proton pump inhibitors [PPIs], loop diuretics, iron supplements, erythropoietin, and/or thrombin preparations, and human albumin replacement); (2) indications for rivaroxaban and its daily dosage; (3) laboratory tests prior to and during the administration of rivaroxaban: hemoglobin (HB), red blood cell count (RBCC), platelet count (PLT), albumin, total protein, fibrinogen (FIB), d-dimer (DD), serum creatinine, and creatinine clearance (CrCl) calculated by the Cockcroft-Gault formula.
At the end of the study, the clinical outcomes of both groups were investigated. The primary outcome was clinical bleeding events. Because of the heterogeneity of bleeding definitions across clinical trials, criteria from Thrombolysis in Myocardial Infarction were used in this study. 21 Bleeding events were stratified into 2 types: major bleeding events (if they involved intracranial hemorrhage or cardiac tamponade or were associated with a decrease in HB concentration of ≥50 g/L) and minor bleeding events (if the patient had a decrease in HB of more than 30 g/L but did not exceeded 50 g/L with an identified bleeding site or if the patient had spontaneous gross hematuria, hemoptysis, hematemesis, or bleeding that required intervention including blood transfusion(s) and/or anticoagulation therapy discontinuation or interruption specifically due to bleeding). 21 The secondary outcome was changes in laboratory values that could potentially be affected by bleeding events, such as platelet count, HB level, and serum albumin level. The formula Nadler 22 which was originally used to calculate arthroplasty perioperative blood loss 23 was applied here to calculate the estimated blood volume (EBV) and the estimated blood loss. All data were collected from electronic medical records.
Statistics
For coagulation characteristics, the fraction of PT and aPTT above the upper limit of normal in the PC group is depicted in median and interquartile range (IQR) and box and whisker plot. Descriptive statistics were used to summarize the demographic and clinical characteristics of the study patients. Continuous variables were evaluated from normal distribution and reported as the mean and standard deviation (mean ± SD) or as the median and IQR. Categorical variables were reported as numbers and percentages. The characteristics of the 2 groups were compared using independent sample t-tests or Mann-Whitney U tests for the continuous variables and chi-square tests or Fisher's exact tests for the categorical variables. The comparison of ordered rank variables used the Mann-Whitney U test. The P value < .05 was considered statistically significant. Multivariable logistic regression analyses for prolonged aPTT/PT after rivaroxaban intake were performed. Regression models included variables that reached significance in the univariate analysis, 7 clinical factors had P < .05 were eligible for inclusion into the regression model. The multicollinearity between the variables was also tested, with variance inflation factor (VIF) values >5 considered as a potential correlation between the 2 variables, and the stepwise regression analysis was used to screen out the best regression sub-sets from the relevant variables, and the independent variables to be included in the final regression equation were determined. Otherwise, the ENTER method will be used for multivariate logistic regression analysis. Model fit was assessed using the Hosmer-Lemeshow test. Statistical significance was set as a 2-tailed P value of <.05. All statistical analyses were performed with IBM SPSS Statistics Version 22 (IBM Corp, Armonk, NY, USA).
Results
Study Population
A total of 472 adult patients who took rivaroxaban during their hospital stay and had at least one aPTT/PT tested while on rivaroxaban were reviewed (Figure 1). After excluding 238 patients with unknown (n = 94) or abnormal (n = 144) aPTT/PT prior to rivaroxaban administration, 10 with shortened aPTT/PT during rivaroxaban administration, 66 with important clinical data missing, and 3 with non-rivaroxaban-related deaths, a total of 155 patients were ultimately included in the study. Based on whether the aPTT/PT during rivaroxaban administration was prolonged by comparison with the upper limit of coagulation testing normal range of the institutional laboratory, 101 patients with normal aPTT/PT were allocated to the control group (NC group) and 54 patients with prolonged aPTT/PT were allocated to the case group (PC group).

Flow chart of included patients. Abbreviations: aPTT, activated partial thromboplastin time; BMI, body mass index; NC, normal control; PC, prolonged case; PT, prothrombin time.
Coagulation Characteristics: Prolonged Values
In the present study, we found that 54 out of 155 patients (34.84%) had prolonged aPTT or PT results and were allocated to the PC group. The PC group was composed of the following three subgroups of patients: with prolonged PT only (n = 29), with prolonged aPTT only (n = 14), with both PT and aPTT prolonged (n = 11). By comparison with the upper limit of the normal range of the institutional laboratory: 14.50 s for PT, and 44.00 s for aPTT, the prolongation values for the three subgroups were: 0.80 [0.20,1.40] seconds-median[IQR] for the group with prolonged PT only, 1.80 [0.43,4.40] seconds for group with prolonged aPTT only, and 0.90[0.30,4.40] seconds for PT and 3.50 [2.60,7.60] seconds for aPTT in the group with both values prolongated (Figure 2).

Box and whisker plot of aPTT/PT prolonged values in PC group. Abbreviations: aPTT, activated partial thromboplastin time; PT, prothrombin time; s, seconds.
Baseline Characteristics of the Study Population
Demographic characteristics were similar between the 2 groups with respect to age, weight, BMI, comorbidities, blood pressure level, and the proportion of surgical patients (Table 1). The mean height of the PC group was 1.65 ± 0.08 m, which was higher than that of the NC group of which the average height was 1.60 ± 0.08 m (P < .01). Additionally, a higher percentage of males was found in the PC group (66.67% for the PC group vs 48.51% for the NC group, P = .03). The percentage of patients who received parenteral anticoagulants (unfractionated heparin and LMWH) before rivaroxaban initiation was significantly higher in the PC group (62.96% vs 37.62%, P < .05). Notably, the subgroup analysis showed that both types of parenteral anticoagulants and daily dosages were similar between the 2 groups (P > .05). Also, a higher percentage of patients in the PC group than in the NC group received human albumin replacement (27.78% vs 7.92%, P < .01). It was also found that there was a higher percentage of patients in the NC group using NSAIDs (70.30% vs 44.44%, P < .01) and PPIs (67.33% vs 42.59%, P < .01). However, the differences among indications as well as daily dosages of rivaroxaban were not significant (Table 2).
Baseline Characteristics of the Study Population.

The use of parenteral anticoagulants was collected prior to the administration of rivaroxaban, as there was no concomitant use of parenteral anticoagulants with rivaroxaban in all patients included in this study.
LMWH, including enoxaparin, nadroparin, and dalteparin.
The use of antiplatelet agents and EPO and/or thrombin preparations were collected for the entire duration of the patients’ hospitalization, considering the long-term effects of the medications.
Abbreviations: BMI, body mass index; CrCl, creatinine clearance; DP, diastolic blood pressure; EPO, erythropoietin; LMWH, low molecular weight heparin; NC, normal control; NSAIDs, nonsteroidal anti-inflammatory drugs; PC, prolonged case; PPIs, proton pump inhibitors; SP, systolic blood pressure; UFH, unfractionated heparin.
Comparison of Indication and Daily Dosage of Rivaroxaban.
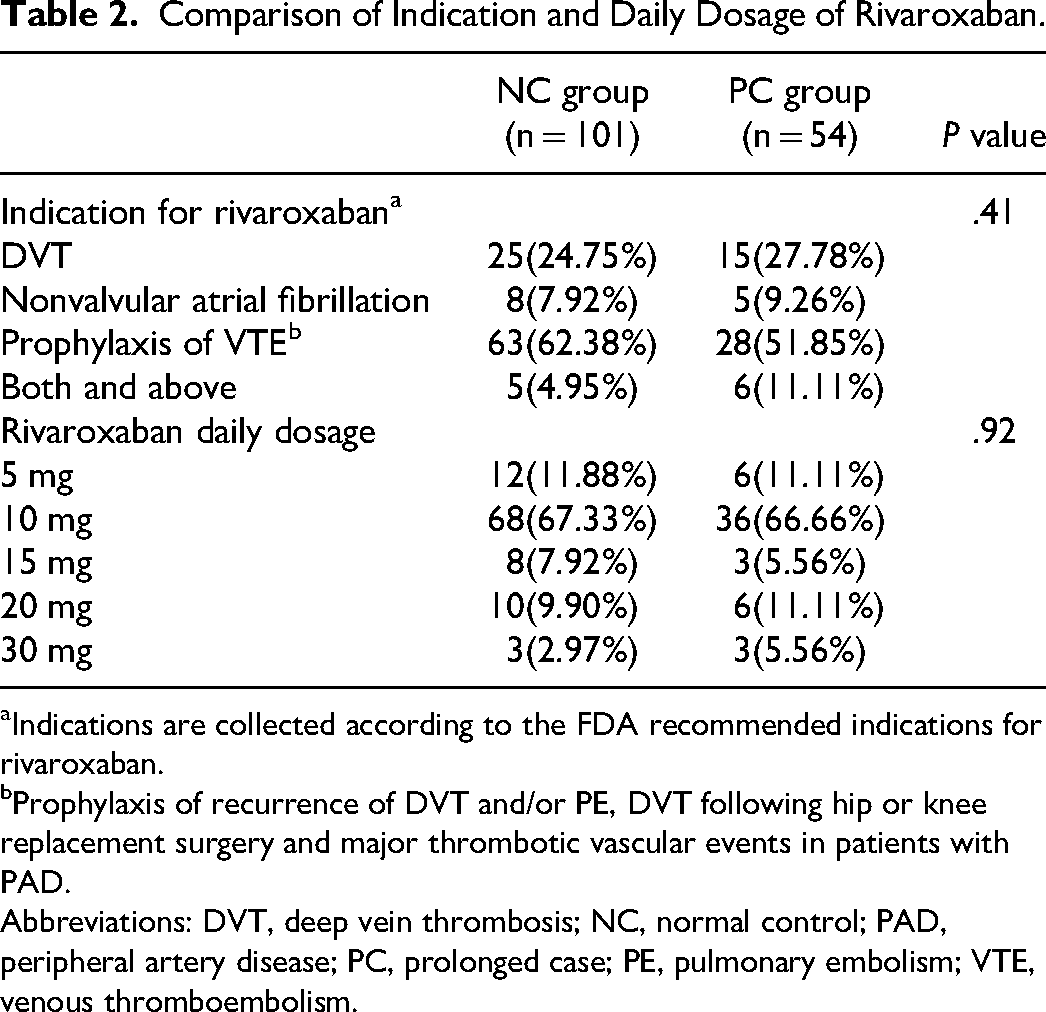
Indications are collected according to the FDA recommended indications for rivaroxaban.
Prophylaxis of recurrence of DVT and/or PE, DVT following hip or knee replacement surgery and major thrombotic vascular events in patients with PAD.
Abbreviations: DVT, deep vein thrombosis; NC, normal control; PAD, peripheral artery disease; PC, prolonged case; PE, pulmonary embolism; VTE, venous thromboembolism.
Comparison of Laboratory Values
Before rivaroxaban was administrated, patients from the PC group had a higher EBV than those from the NC group (4.41 [3.65,4.72] vs 3.84 [3.24,4.31] L, P < .01), but similar serum levels of HB, PLT, RBCC, albumin, total protein, FIB, DD, and creatinine (Table 3). Even though the differences were not significant, the PC group had a lower serum albumin level (36.04 ± 4.01 vs 37.02 ± 4.38 g/L) and a higher serum creatinine level (0.77[0.62,0.97] vs 0.72[0.58,0.90] mg/dL) than the NC group. In addition, there was no significant difference in CrCl between the 2 groups (NC 86.83[61.29,104.81] vs PC 80.97[59.05,119.03] mL/min, P = .93).
Comparison of Laboratory Values Prior to Rivaroxaban Administration.

The formula of Nadler 22 :
EBV (L) = A × H0.725 × G0.425−B
where EBV = estimated blood volume (L); H = height (cm); G = weight (kg); A = 0.0236 for men; A = 0.0248 for women; B = 1.229 for men; and B = 1.954 for women.
Abbreviations: HB, hemoglobin; NC, normal control; PC, prolonged case; PLT, platelet count; RBCC, red blood cell count.
Risk Factors for Prolonged aPTT/PT
Seven variables including gender, height, parenteral anticoagulant use, NSAIDs use, PPIs use, human albumin replacement, and EBV were eligible for inclusion into the multivariable model. All 7 variables had VIF values <5, and the ENTER method was used for logistic regression analysis. A multivariable logistic regression analysis was then performed, showing that height (odds ratio [OR] 1.12, 95% confidence interval [CI] 1.02-1.22, P = .02) and human albumin replacement (OR 3.19, 95% CI 1.04-9.74, P = .04) were independent risk factors for aPTT/PT prolongation. In contrast, gender and administration of medications such as parenteral anticoagulants, NSAIDs, PPIs and EBV were not identified as independent risk factors for aPTT/PT prolongation (Figure 3).

Independent risk factors for aPTT/PT prolongation. Abbreviations: aPTT, activated partial thromboplastin time; CI, confidence interval; EBV, estimated blood volume; NSAIDs, nonsteroidal anti-inflammatory drugs; OR, odds ratio; PPI, proton pump inhibitor; PT, prothrombin time.
Clinical Bleeding Events and Changes in Bleeding-Associated Laboratory Parameters
To explore the possible clinical impact of aPTT/PT prolongation caused by rivaroxaban administration, clinical bleeding events from both groups were collected. Three patients (5.56%) in the PC group were found to have minor bleeding events: 2 patients received blood transfusion, and 1 experienced spontaneous hematemesis. Four patients (3.96%) in the NC group experienced minor bleeding events: 2 patients exhibited HB drops of 30 to <50 g/L and the other 2 patients experienced spontaneous hematuria and hematemesis, respectively. There were no major bleeding events recorded. Nevertheless, there was no significant difference in bleeding events between the 2 groups (P = .70).
In addition to clinical bleeding events, we also calculated the changes in the laboratory parameters that may be associated with bleeding (values tested before rivaroxaban administration minus values tested after rivaroxaban administration) such as HB, RBCC, and PLT. The differences of changes in HB, RBCC, and PLT before and after rivaroxaban exposure were not significant between the 2 groups: patients in the PC group showed a decrease in HB of 7.50 [−0.25,13.25] g/L, RBCC of 0.20 [−0.07,0.48] × 106/μL, and an increase in PLT of 0.50 [53.00,−36.25] × 103/μL, these changes were similar to those of the NC group (Table 4). Likewise, changes in the levels of serum albumin, total protein, DD, fibrinogen, and serum creatinine were similar between the 2 groups. In addition to the directly detectable parameters, estimated blood loss appeared to be greater in the PC group than in the NC group (0.21 ± 0.46 vs 0.14 ± 0.40 L), even though the difference was not significant (P = .32). The above results indicated that for rivaroxaban recipients exhibiting a prolonged aPTT/PT, estimated blood loss as well as changes in other laboratory parameters associated with bleeding were similar to those recipients with a normal aPTT/PT.
Clinical Bleeding Events and Changes in Laboratory Values Related to Hemorrhage.

Translate: D- “X”, difference of “X” = “X”(pre-administration of rivaroxaban) -“X”(postadministration of rivaroxaban); negative values represent an increase in “X” after rivaroxaban administration.
EBL(L) = EBV × ln (HCT-pre/HCT-post)
where EBL = estimated blood loss (L); EBV = estimated blood volume (L); HCT-pre = haematocrit pre-administration of rivaroxaban; HCT-post = haematocrit postadministration of rivaroxaban.
Abbreviations: DD, d-dimer; FIB, fibrinogen; HB, hemoglobin; NC, normal control; PC, prolonged case; PLT, platelet count; RBCC, red blood cell count; Scr, serum creatinine.
Discussion
Although PT and aPTT are often normal in patients on therapeutic doses of rivaroxaban, it is known that patients taking rivaroxaban occasionally have prolonged aPTT/PT. However, it was not clear whether these changes were associated with a higher risk of bleeding events or what are the possible clinical risk factors were associated with the prolongation. In the present study, we found that 54 out of 155 patients (34.84%) had mildly prolonged aPTT or PT results. However, there were no differences in clinical bleeding events or changes in bleeding-related laboratory parameters between the groups of patients with normal and prolonged aPTT/PT after rivaroxaban administration. Also, height and human albumin replacement were possibly independent risk factors for rivaroxaban induced aPTT/PT prolongation.
Human albumin replacement is widely used for the correction of hypoalbuminemia. Past studies have shown that hypoalbuminemia was a positive predictor of bleeding risk after taking rivaroxaban. With an increase in albumin concentration of 10 g/L, the risk of bleeding could drop by 75%. 2 Drugs exhibiting high protein binding have potential increased action in patients with hypoalbuminemia. Rivaroxaban is 92% to 95% protein bound, 16 which may explain why the risk of aPTT/PT prolongation increased for patients who require human albumin replacement during hospitalization. Also, based on the odds ratio calculation from this study, the probability for aPTT/PT prolongation may increase by approximately 12% with each centimeter increase in height after taking rivaroxaban. To our knowledge, no previous studies have described the influence of height on bleeding risk induced by anticoagulation. Our previous study showed that patient BMI (OR 2.513, 95% CI 1.929-3.011, P < .001) was an independent risk factor for limb ecchymosis in rivaroxaban users after total knee arthroplasty. 24 In contrast, BMI was not found to be an independent risk factor for aPTT/PT prolongation.
Our findings showed that a higher percentage of patients were found to be premedicated with parenteral anticoagulants, primarily LMWH in the group with prolonged aPTT/PT during rivaroxaban administration. As all enrolled patients had normal aPTT/PT before rivaroxaban initiation, and most of them had switched to rivaroxaban immediately after discontinuation of LMWH, we speculate that the residual anticoagulant effect of LMWH may play a role. It has been revealed by other studies that several factors can lead to increased residual anticoagulant effects of parenteral anticoagulants, such as renal insufficiency, age, and obesity.25–27 In our study, among the group of patients with prolonged aPTT/PT who received LMWH before switching to rivaroxaban, 78.13% of them (25 of 32) had at least one of the following risk factors: age ≥ 65 years, BMI ≥24 kg/m2, and chronic kidney disease. These factors may contribute to the residual effect of LMWH thus causing mild prolongation of aPTT/PT when rivaroxaban is initiated. This also explains why premedication with parenteral anticoagulant has not been defined as an independent risk factor for aPTT/PT prolongation by regression analysis, as its impact was probably confounded by other factors.
Despite the fact that rivaroxaban plasma levels may impact aPTT/PT results, the present study fails to show influences of rivaroxaban daily dosages (from 5 to 30 mg) on aPTT/PT testing results. As shown by other investigations, both the average peak and trough plasma concentrations of rivaroxaban increased greatly when daily dosage increased from 10 to 20 mg (peak: 249 ng/mL for 20 mg/day vs 125 ng/mL for 10 mg/day; trough: 44 ng/mL for 20 mg/day vs 9 ng/mL for 10 mg/day).3,28 However, the linear correlation between rivaroxaban plasma concentration and PT or aPTT was poor. 29 Due to the lack of sensitivity, these assays are of limited value in detecting drug plasma levels, especially for low on therapy levels. 13 Our findings coincide with these studies that mild prolongation of rivaroxaban seems to be irrelevant with different rivaroxaban therapeutic doses.
Finally, we do not recommend adjusting or even discontinuing anticoagulation therapy in patients with mild aPTT/PT prolongation. In our study population, there was no significant difference in outcomes of bleeding events and bleeding-associated laboratory values between groups with mildly prolonged and normal aPTT/PT after taking rivaroxaban. In the RECORD30–33 series RCTs, the incidence of major bleeding after rivaroxaban administration was 0.1% to 0.7% and nonmajor bleeding was 4.3% to 10.2%. Probably due to the small sample size, major bleeding events were not observed in our study. The incidence of nonmajor bleeding events was almost comparable to that of RECORD series, with a lower incidence found in the NC group than in the PC group (3.96% vs 5.56%, P = .70), although the difference was not statistically significant. In addition, no deaths related to rivaroxaban use in our study. Those 3 patients who dropped out of the study all died from other causes (2 from severe pneumonia and 1 from renal failure) at least 2 weeks after discontinuation of rivaroxaban. Therefore, the current data does not support discontinuing rivaroxaban due to mild aPTT/PT prolongation. Our findings are in line with the general understanding that PT and aPTT are not indicators to predict the therapeutic or supratherapeutic effects of rivaroxaban. 18 Therefore, rivaroxaban therapy cannot be adjusted based on aPTT/PT results alone. In contrast, abrupt interruption of anticoagulation therapy carries a great risk of recurrent thromboembolic events and even leads to a fatal outcome. 34
Limitation
First, as this was a retrospective study, due to the absence of important information, some patients were excluded from the study, resulting in a small sample size. Second, aPTT/PT were tested at different time points with different frequencies after rivaroxaban initiation during hospitalization, which may result in heterogenicity of the data. Moreover, as the medical records were reviewed retrospectively, it is possible that some minor bleeding events especially minor bleeding events were not recorded in the patient's electronic medical records, resulting in underestimation of the incidence of bleeding and incomplete collection of these events. Hence, future prospective studies with a larger sample size are needed to further verify the findings of the present study.
Conclusion
In this real-life study, rivaroxaban administration can cause mild aPTT/PT prolongation, and patients’ height as well as human albumin replacement use were independent risk factors. As such, we recommend enhanced monitoring of patients taking rivaroxaban with hypoproteinemia. As no increase of bleeding events was found with mild aPTT/PT prolongation, discontinuation or dosage adjustment of rivaroxaban may not be necessary under such circumstances.
Footnotes
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This study was approved by the Institutional Review Board at the First Affiliated Hospital of Chongqing Medical University (2020-371). The requirement for informed consent was waived because of the anonymous nature and retrospective analysis of the data.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
