Abstract
This study evaluated the prothrombin time (PT) assay for the measurement of plasma concentrations of rivaroxaban using calibrators and controls. The intra- and interlaboratory precision of the measurement was investigated in a field trial involving 21 laboratories. Each laboratory was provided with rivaroxaban calibrators and control plasma samples containing different concentrations of rivaroxaban, and PT reagents. The evaluation was carried out over 2 consecutive weeks using centrally provided and local PT reagents. A calibration curve was produced each day (for inter-run precision), and day-to-day precision was evaluated by testing 3 control plasma samples. A large interlaboratory variation (in seconds) was observed with local PT reagents. The results were less variable when expressed as rivaroxaban concentrations (ng/mL) or when central PT reagent was used (STA Neoplastine CI Plus). The widely available PT assay, in conjunction with rivaroxaban calibrators, may be useful for the measurement of peak plasma levels of rivaroxaban.
Introduction
New oral anticoagulants that target individual components of the coagulation cascade have been developed in recent years, and some of the agents have already shown promise in phase III clinical studies. Rivaroxaban is an oral, direct factor Xa inhibitor that has been approved in more than 100 countries worldwide, including the European Union, for the prevention of venous thromboembolism (VTE) in adult patients after elective hip or knee replacement surgery. It is currently undergoing phase III studies for the prevention of VTE in acutely ill medical patients, treatment and secondary prevention of VTE (including deep vein thrombosis and pulmonary embolism), prevention of stroke and systemic embolism (noncentral nervous system) in patients with nonvalvular atrial fibrillation, and prevention of cardiovascular death, myocardial infarction, or stroke in patients with a recent acute coronary syndrome.
Rivaroxaban inhibits free and clot-bound factor Xa as well as prothrombinase-bound factor Xa1,2 and has been found to have predictable pharmacokinetics and pharmacodynamics in humans.3,4 Although it is used at a fixed dose without routine coagulation monitoring, a quantitative determination of rivaroxaban might be of assistance in several clinical settings, such as in patients with low body weight, obese patients, those with deteriorating renal or hepatic function, those who accidentally or deliberately overdose, or to measure compliance.
Emerging evidence suggests that routinely used clotting assays are not suitable for measuring the anticoagulant effect of rivaroxaban (as with some other new oral anticoagulants). The prothrombin time (PT) is prolonged with rivaroxaban, but the slope of the concentration–effect relationship varies depending on the thromboplastin reagent used, as shown in several studies.5–7 The reason for this variability is that thromboplastin reagents used in the test have varying sensitivities (ie, different International Sensitivity Index [ISI] values) to anticoagulants that directly inhibit factor Xa, including rivaroxaban.7,8 The international normalized ratio (INR) system cannot be applied to rivaroxaban 8 because it was specifically developed for interlaboratory standardization for monitoring the vitamin K antagonists.
Measuring plasma drug concentrations is an established standard for the therapeutic monitoring of many drugs, and measuring the plasma concentrations of rivaroxaban may be the optimal method for its quantitation, if required. Although high-performance liquid chromatography–tandem mass spectrometry (HPLC-MS/MS) allows sensitive and specific quantification of rivaroxaban, 9 this method cannot be practically implemented in routine clinical laboratories. In addition, the HPLC-MS/MS method is unsuitable for use in emergency situations (such as in suspected overdose) because it takes approximately 2 to 4 days to obtain the results. Although other assays (such as anti-Factor Xa assays) are commercially available for clinical use, they are performed mainly in specialized clinical laboratories. The PT assay, however, is a frequently used clotting assay and is widely available at any time in almost every routine clinical laboratory worldwide. In addition, studies have shown that prolongation of the PT correlated closely with rivaroxaban plasma concentrations.4,10,11 For these reasons, PT was chosen in the present study to validate rivaroxaban calibrators and controls. The multicenter PT field trial assessed the intra- and interlaboratory precision of the measurements of rivaroxaban plasma concentrations (expressed as ng/mL) when using the PT assay together with rivaroxaban calibrators and controls.
Materials and Methods
Rivaroxaban Calibrators
Rivaroxaban (Bayer HealthCare, Berlin, Germany) was dissolved in 100% dimethyl sulfoxide (DMSO; 20 mg rivaroxaban in 56 mL DMSO), which was then diluted with water to obtain a stock solution of 25 µg/mL (in 7% DMSO). Aliquots were then added to apheresis citrated pooled plasma originating from transfusion blood banks (thawed at 37°C), with additives (lactose, saccharose, and a N-2-hydroxyethylpiperazine-N’-2-ethanesulfonic acid [HEPES] buffer) to obtain rivaroxaban calibrators with absolute weight values of 0, 50, 250, and 500 ng/mL. The final concentration of DMSO was 0.15%. A control was tested in each assay run with DMSO alone. The rivaroxaban-containing plasmas were then freeze-dried and stored at 2°C to 8°C. The actual rivaroxaban concentration of the freeze-dried calibrators (lot number SD08311) obtained by HPLC-MS/MS 9 was 0, 41, 219, and 430 ng/mL, respectively. The accuracy of the PT prolongation by the calibrators after freeze-drying was tested by reconstituting each plasma vial with 1 mL distilled water, which was then left for 30 minutes at room temperature (18°C to 22°C) before testing. Their effect on the PT was compared with that of fresh pooled plasma.
In order to determine the influence of the freeze-drying process, a concentration response of the rivaroxaban calibrators on the PT was determined using STA Neoplastine CI Plus (Diagnostica Stago, Asnières-sur-Seine, France) under different conditions. A concentration response for the PT was established with normal fresh pooled plasma spiked with rivaroxaban and compared with that using frozen apheresis pooled plasma spiked with rivaroxaban. Because the PT was prolonged in frozen apheresis plasma compared with that in fresh plasma, a predetermined quantity of recombinant procoagulant factor VIIa was added to each plasma calibrator before freeze-drying (patent pending) to adjust the clotting times. The quantity of factor VIIa added to each plasma calibrator was calculated in order to obtain PT values similar to those obtained from fresh plasma spiked with rivaroxaban.
Stability and Reproducibility of the Calibrators
The stability of the rivaroxaban calibrators during the 4 hours after reconstitution at room temperature was assessed by testing the effects of the calibrators on the PT (expressed in seconds), using STA Neoplastine CI Plus. Freeze-dried calibrators were reconstituted with 1 mL of distilled water and were then left at room temperature (18°C−22°C) for a period of 30 minutes (time 0) or 4.5 hours (time 4) before starting the assay. Three vials of each calibrator were tested in triplicate and the mean values were calculated (n = 9). The relative change in the PT over baseline was expressed as percentage changes ([mean PT at time 4 – mean PT at time 0]/mean PT at time 0 × 100).
The reproducibility within lot of the calibrators was tested by performing PT tests (in seconds; using STA Neoplastine CI Plus) with each calibrator on the same day. Fifteen vials (5 vials taken at the beginning, in the middle, and at the end of the aliquot distribution before freeze-drying, respectively) of respective freeze-dried calibrator were tested in duplicate, and the mean values, standard deviation, and coefficient of variation (CV) for each test were calculated (n = 15) for each calibrator.
The PT Field Trial
Twenty-one laboratories in Europe and North America received supplies required for participation in the PT field trial, and 20 laboratories returned their study results. Of these, 1 laboratory used a different method for the PT measurement (Owren method) and was, therefore, excluded from the analysis. The final analyses were performed for 18 sites for the local PT reagents and for 19 sites for the centrally provided STA Neoplastine CI Plus reagent.
Each laboratory was supplied with a detailed protocol, sets of lyophilized rivaroxaban calibrators (0, 41, 219, and 430 ng/mL), control lyophilized pooled human plasma samples (control plasma samples) containing 3 different concentrations of rivaroxaban: 19, 160, and 643 ng/mL (which were unknown to the participating laboratories), and PT reagents: STA Neoplastine CI Plus and STA Coag Control (Diagnostica Stago; control plasma, normal, and abnormal levels).
The intralaboratory variation was determined by testing the control plasma samples (containing 19, 160, and 643 ng/mL rivaroxaban) over 2 consecutive weeks (10 days) by each laboratory. A calibration curve was produced each day using the calibrators containing rivaroxaban 0, 41, 219, and 430 ng/mL (which were freshly reconstituted with both STA Neoplastine CI Plus and local PT reagents). The day-to-day precision was evaluated by testing in duplicate the 3 rivaroxaban-containing control plasma samples. The rivaroxaban-containing pooled plasma sample was diluted 2-fold with the 0 ng/mL rivaroxaban calibrator and retested if the rivaroxaban level was above the highest concentration of the calibration curve. The local PT reagents and the different instruments used by the participating laboratories for analytical evaluation are listed in Table 1. The local PT reagents were as follows: 6 used STA Neoplastine CI Plus, 2 used STA Neoplastine CI, 4 used RecombiPlasTin 2G (Instrumentation Laboratory, Bedford, Massacheusetts), 3 used Thromborel S (Siemens/Dade Behring Inc.), and 3 used Innovin (Siemens/Dade Behring Inc.). The different instruments used by the participating laboratories were as follows: 6 used STA R (Diagnostica Stago), 3 used STA Compact (Diagnostica Stago), 2 used KC 10 (Heinrich Amelung, Lemgo, Germany), 1 used ACL 1000 (Instrumentation Laboratory), 2 used ACL TOP (Instrumentation Laboratory), and 4 used BCS (Siemans/Dade Behring Inc). All reagents and instruments were used in accordance with the manufacturers’ instructions.
List of Local PT Reagents and Instruments Used by the Participating Laboratories
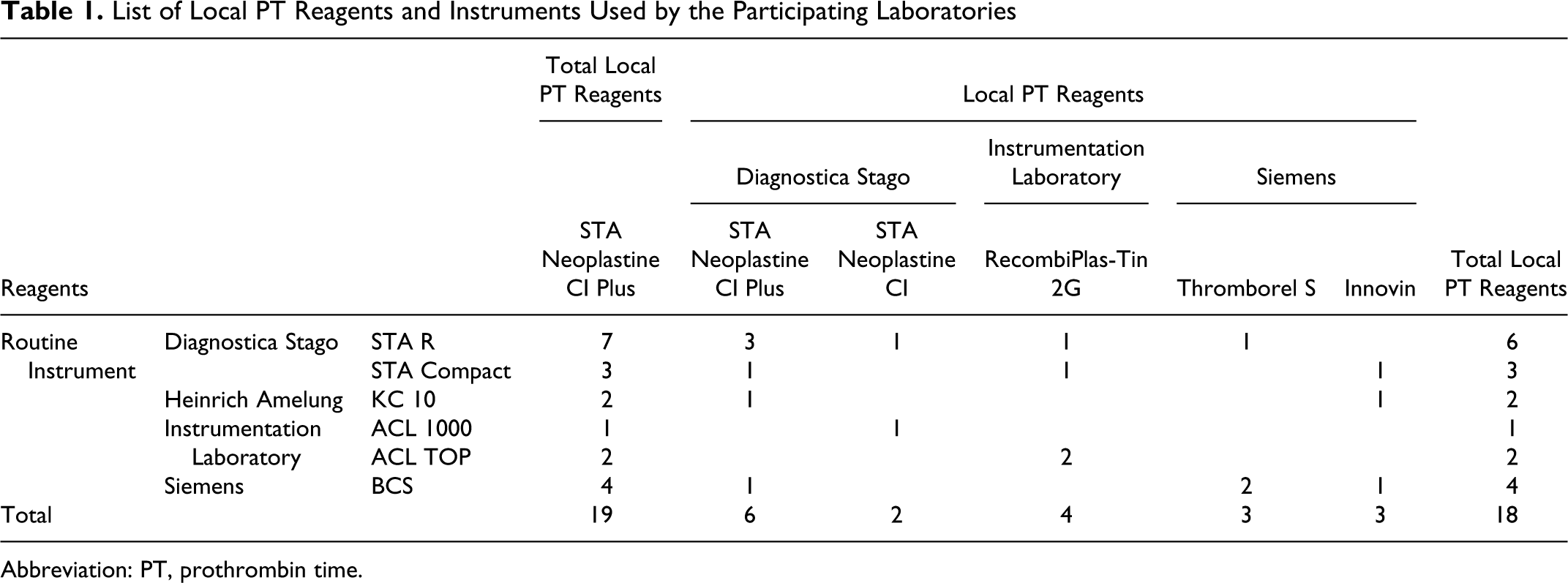
Abbreviation: PT, prothrombin time.
Data Analysis
All raw and calculated results were entered by each participating laboratory into a centrally provided Microsoft Office Excel 95 worksheet. The mean values, standard deviation, and the CV of duplicates during 10 days (n = 10) were calculated for each laboratory and for each calibrator and control. The intra- and interlaboratory variation determined by mean values, standard deviation, and the CV of the 19 sites using the central PT reagent and of the 18 sites using the local PT reagents were calculated for each calibrator and control. Microsoft Excel audit procedures were used to verify the mathematical formulae. For the PT field trial, the results were expressed in seconds, as a ratio (vs calibrator containing 0 ng/mL rivaroxaban), and in rivaroxaban concentrations (ng/mL). The rivaroxaban concentrations in the 3 control plasma samples were calculated from linear calibration curves. Both absolute (weighed) and measured (by HPLC-MS/MS) rivaroxaban concentrations in the control samples and calibrators are presented. Statistical analysis of differences between the data obtained using local and central PT reagents was performed using 1-way analysis of variance. Comparison of mean values obtained with local PT reagents versus central PT reagent was statistically analyzed using the nonparametric Wilcoxon signed rank test (used as a paired test; Tables 2 and 3). The statistical analysis of the influence of the different instruments used (in Table 3) was performed by comparing the mean values obtained with central PT reagent and the instrument of each site versus that obtained by the first site (using STA R instrument and central PT reagent). A P value of < .05 was considered significant. W (Wilcoxon signed ranked statistic) values are also presented (W < 100: nonsignificant; greater W value indicates greater significant difference).
Interlaboratory Variations: Comparison of Results Obtained With Local PT Reagents Versus Central PT Reagent a
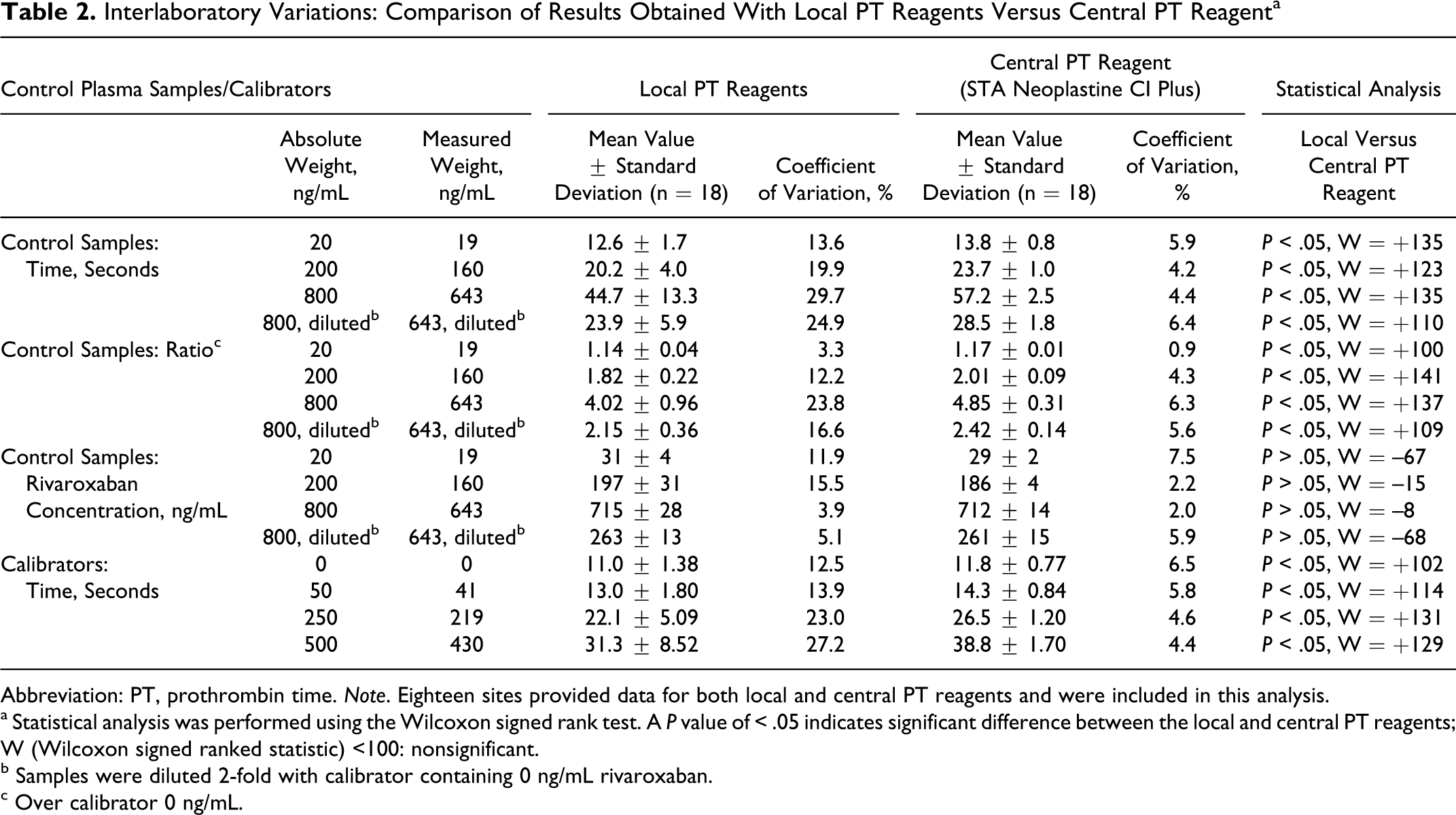
Abbreviation: PT, prothrombin time. Note. Eighteen sites provided data for both local and central PT reagents and were included in this analysis.
a Statistical analysis was performed using the Wilcoxon signed rank test. A P value of < .05 indicates significant difference between the local and central PT reagents; W (Wilcoxon signed ranked statistic) <100: nonsignificant.
b Samples were diluted 2-fold with calibrator containing 0 ng/mL rivaroxaban.
c Over calibrator 0 ng/mL.
Intralaboratory Precision Obtained With Central and Specific Local PT Reagents by Each Participating Laboratory and the Influence of Different Instruments a
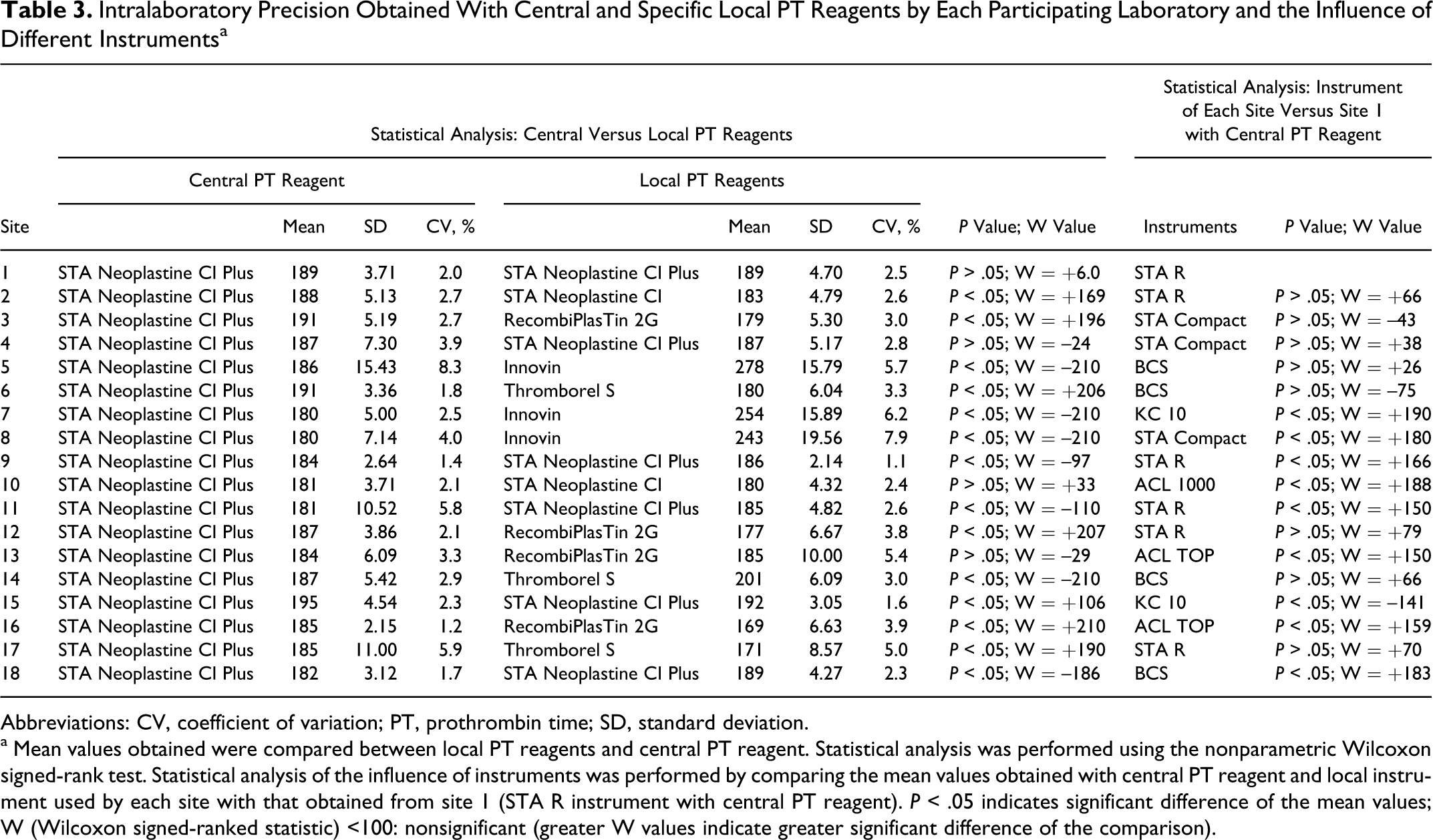
Abbreviations: CV, coefficient of variation; PT, prothrombin time; SD, standard deviation.
a Mean values obtained were compared between local PT reagents and central PT reagent. Statistical analysis was performed using the nonparametric Wilcoxon signed-rank test. Statistical analysis of the influence of instruments was performed by comparing the mean values obtained with central PT reagent and local instrument used by each site with that obtained from site 1 (STA R instrument with central PT reagent). P < .05 indicates significant difference of the mean values; W (Wilcoxon signed-ranked statistic) <100: nonsignificant (greater W values indicate greater significant difference of the comparison).
Results
Manufacture and Initial Validation of Rivaroxaban Calibrators Using the PT in Different Plasma Preparations
The PT was prolonged by 1.5, 1.7, 3.8, and 6.4 seconds for calibrators containing 0, 41, 219, and 430 ng/mL rivaroxaban, respectively, compared with that observed with fresh plasma. Dimethyl sulfoxide did not significantly influence the clotting time of various coagulation assays including the PT (data not shown). The prolongation increased with increasing rivaroxaban concentrations and was corrected in each case by adding the procoagulant factor VIIa—the concentration of which was predetermined for each calibrator and each control. After freeze-drying, the PT increased by 0.4, 0.2, 0.9, and 4.6 seconds for calibrators with 0, 41, 219, and 430 ng/mL rivaroxaban, respectively, compared with that obtained with fresh plasma. Similar results were obtained with 2 further batches of rivaroxaban calibrators (lot numbers SD09106 and SD10011; data not shown).
Stability and Reproducibility of Rivaroxaban Calibrators
The rivaroxaban calibrators prolonged the PT in a concentration-dependent manner and seemed stable at room temperature during the 4 hours after reconstitution. The change in clotting time was 0.1 to 0.2 seconds compared with baseline; the relative changes ranged from 0.4% to 0.8% (data not shown). When the reproducibility within lot of the rivaroxaban calibrators was tested, the mean PT was 12.1, 14.4, 25.9, and 37.9 seconds, for calibrators with 0, 41, 219, and 430 ng/mL rivaroxaban, respectively, with the mean CV ranging from 0.3% to 1.0% (data not shown).
The PT Field Trial
Local PT Reagents
The instruments and PT reagents used in the participating laboratories are presented in Table 1. The interlaboratory mean PT, standard deviation, and CV of all the participating laboratories, calculated for each calibrator and control sample, are presented in Table 2. The results are expressed in seconds, as a ratio (over a calibrator containing 0 ng/mL rivaroxaban and 0.15% DSMO), in rivaroxaban concentration (ng/mL) for the control plasma samples, and in seconds for the calibrators (Table 2). The intralaboratory precision of the measurements within each laboratory calculated for the control sample containing 200 ng/mL rivaroxaban (absolute weighed value) is presented in Table 3.
As expected, there was a large, concentration-dependent interlaboratory variation in the PT (in seconds) for the control plasma samples, with the CV ranging from 13.6% to 29.7%. The results were less variable when expressed as a ratio over calibrator containing 0 ng/mL rivaroxaban (CV: 3.3% to 23.8%). A lower interlaboratory variation in the PT was obtained when the results were expressed as rivaroxaban concentration (CV: 3.9% to 15.5%; Table 2). The mean rivaroxaban concentrations in the control plasma samples obtained from all participating laboratories were similar to the absolute weighed values but were higher than the actual (by HPLC-MS/MS) rivaroxaban concentrations (Table 2 and Figure 1). The overestimation was 63%, 23%, and 11% for the 19, 160, and 643 ng/mL rivaroxaban samples, respectively (actual/measured ng/mL: 19/31, 160/197, and 643/715, respectively).
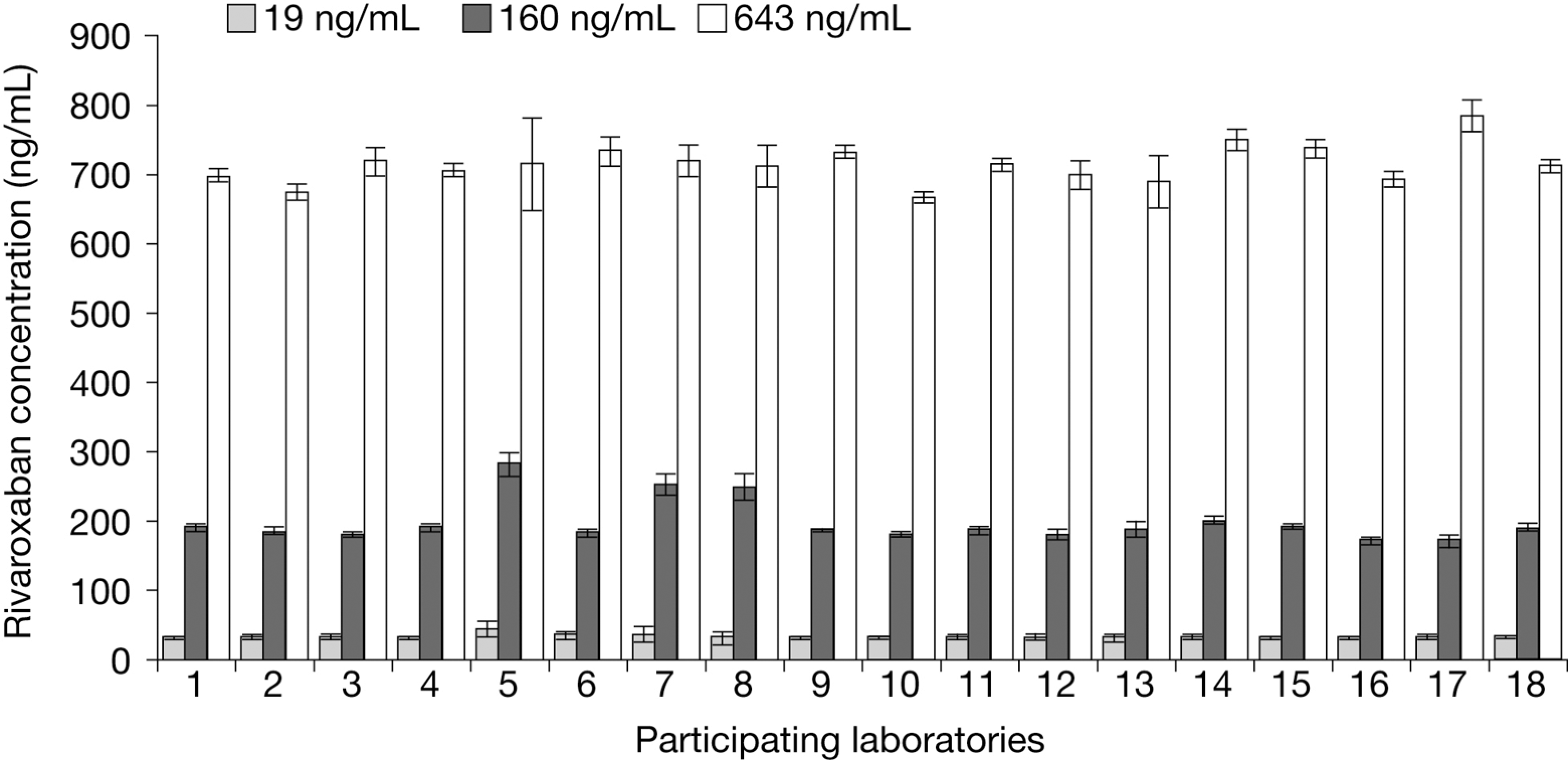
Rivaroxaban concentrations in control plasma samples reported from individual laboratories using local prothrombin time reagents. Results are presented as median values ± standard deviation (n = 18). Intralaboratory coefficients of variation were 2.7% to 34.1% (for 19 ng/mL), 1.1% to 7.9% (160 ng/mL), and 1.1% to 9.6% (643 ng/mL). One site used a different method for the PT measurement (Owren method) and was, therefore, excluded from the analysis.
The intralaboratory variations in the PT measurements were mainly dependent on the local PT reagents used. The variations were higher with 2 reagents (Innovin and RecombiPlasTin 2G), for which the clotting times were shorter, regardless of the type of instruments used (CV: 5.7% to 7.9% with Innovin; 3.0% to 5.4% with RecombiPlasTin 2G compared with 1.1% to 2.8% with STA Neoplastine CI Plus; Table 3).
Central PT Reagent
The interlaboratory mean PT, standard deviation, and CV of the participating laboratories (using STA Neoplastine CI Plus) calculated for each calibrator and control are presented in Table 2. The intralaboratory variations of the PT measurements of each laboratory calculated for the sample containing 200 ng/mL rivaroxaban (absolute weighed value) are presented in Table 3.
There was less interlaboratory variation in the PT among the different laboratories when using the centrally provided PT reagent compared with using local PT reagents. In undiluted control samples, the CV ranged from 4.2% to 5.9% (in seconds, vs 13.6% to 29.7% using local PT reagents) and 0.9% to 6.3% (as a ratio over the calibrator containing 0 ng/mL rivaroxaban, vs 3.3% to 23.8% using local PT reagents). When the results were expressed in seconds or as a ratio, the difference in the mean PT value was significant (P < .05) compared with that obtained using local PT reagents. In contrast, when the results were expressed in rivaroxaban concentrations (ng/mL), there was no significant difference (P > .05) in the interlaboratory variations of the PT measurement (mean values) between the local and central PT reagents, although smaller CVs in undiluted samples were found when the central PT reagent was used. The CV ranged from 2.0% for the highest rivaroxaban plasma concentration to 7.5% for the lowest (Table 2). However, as with the local PT reagents, the measured rivaroxaban concentrations were close to the absolute weighed values but were higher than the actual concentrations in the samples determined by HPLC-MS/MS. The overestimation was 53%, 16%, and 11% for the 19, 160, and 643 ng/mL rivaroxaban samples, respectively (Table 2 and Figure 2). The actual/measured values (ng/mL) were 19/29, 160/186, and 643/712, respectively. Moreover, dilution of the sample (2-fold) reduced the accuracy of the test as indicated by the underestimation of 19% of the rivaroxaban concentration compared with the actual value (Table 2). The CV for the calibrators was 6.5% (0 ng/mL), 5.8% (41 ng/mL), 4.6% (219 ng/mL), and 4.4% (430 ng/mL) with the central PT reagent compared with 12.5%, 13.9%, 23.0%, and 27.2%, respectively, when the local PT reagents were used (Table 2).
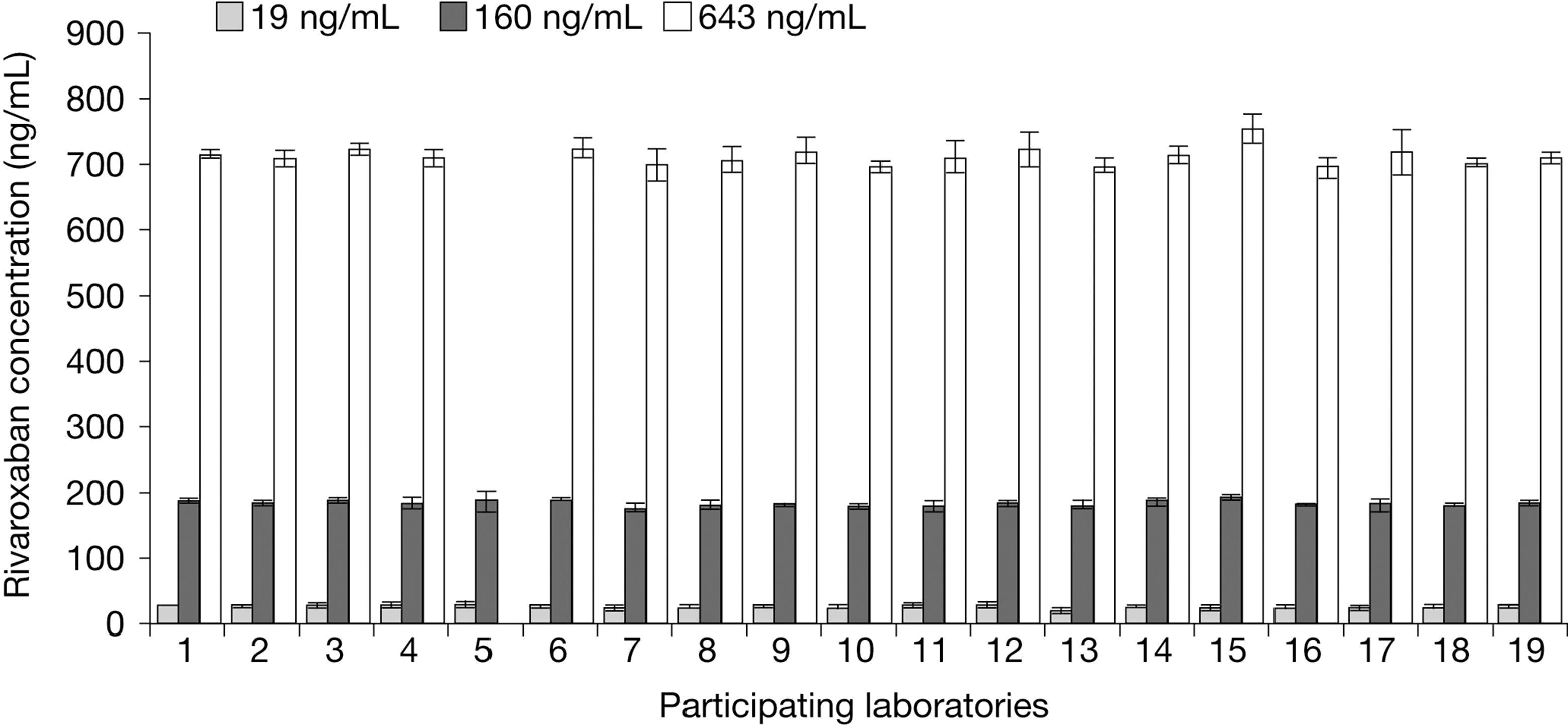
Rivaroxaban concentrations in control plasma samples reported from individual laboratories using centrally provided prothrombin time reagent (STA Neoplastine CI Plus). Results are presented as median values ± standard deviation (n = 19). Intralaboratory coefficients of variation were 4.5% to 19.3% (for 19 ng/mL), 1.2% to 8.3% (160 ng/mL), and 0.9% to 5.0% (643 ng/mL).
When the plasma sample containing 160 ng/mL rivaroxaban (actual value determined by HPLC-MS/MS) was tested by each laboratory, the CV was 1.1% to 7.9% for local PT reagents and 1.2% to 8.3% for centrally provided PT reagent (Table 3). The mean PT measured using local PT reagents was significantly different compared with that obtained with the central PT reagent in 14 of the 18 sites (P < .05; Table 3). In addition, the instruments used by each site seemed to influence the interlaboratory variation as well. When the combination of local instrument/central PT reagent of each site was compared with that used by site 1 (STA Neoplastine CI Plus/STA R), a significant difference was observed in 9 of the 17 sites (P < .05; Table 3).
Discussion
New oral anticoagulants that target a specific step or coagulation factor have been developed in an attempt to overcome the drawbacks of traditional agents. Although routine coagulation monitoring is not required with the new oral anticoagulants, recent consensus suggests that measuring the concentration of the drug or its activity might be useful in certain patient populations and clinical situations.12,13
The direct factor Xa inhibitor rivaroxaban has been shown to have predictable pharmacokinetics, with no significant variation in plasma concentrations with age, body weight, or renal function (creatinine clearance ≥30 mL/min) after a fixed-dose (10 mg) administration, 5 a wide therapeutic window, few clinically relevant drug–drug interactions, and predictable pharmacodynamics.14–19 Rivaroxaban prolongs the PT in a dose-dependent manner (eg, when the test is performed with STA Neoplastine CI Plus).4,10 A major advantage of the PT assay is that it is widely available in routine clinical laboratories. However, variation in the response sensitivity of different PT reagents to rivaroxaban has been described previously, and the conventional INR correction of results does not overcome this variability,7,8 although a recent report suggested the possibility of using a modified INR specific for rivaroxaban to normalize the PT results. 20 The use of rivaroxaban calibrators and controls is a more suitable method for measuring rivaroxaban blood concentrations because the modified INR may lead to confusion with the existing INR method used for monitoring vitamin K antagonist therapy.
The development of commercially available calibrators and controls (the results of which are based on measurements of the PT) is hampered by 2 types of problem. First, the plasma used for manufacturing the calibrators differs substantially from a freshly prepared laboratory plasma sample, particularly with regard to the clotting time. Because it is prepared from blood transfusion banks and frozen after apheresis, the anticoagulant (citrate) concentration is substantially lower than that used in the patient sampling tubes provided by the laboratory (citrate 12 to 20 mmol/L in apheresis plasma according to the apheresis treatment of plasma compared with 21 to 26 mmol/L in patient plasma samples; personal observation). Moreover, blood from transfusion banks may contain adjuvants, such as dextrose, depending on the apheresis bag manufacturer. Second, the modifications resulting from freezing, thawing, adding excipients, filtering, and freeze-drying plasma lead to prolongation of the PT compared with that of fresh plasma. Adding an appropriate quantity of procoagulant factor VIIa (0 to 0.3 IU/mL) to plasma supplemented with rivaroxaban corrected the observed PT prolongation (compared with fresh plasma containing the same concentration of rivaroxaban). Factor VIIa is a constituent of normal plasma, although it is present at a very low concentration (approximately 1% of Factor VII) 21 and its addition to rivaroxaban calibrators and controls shortens the clotting time and allows better comparability to normal plasma. Dimethyl sulfoxide was used in very low concentrations and was shown not to affect the clotting time, as reported previously for the anti-factor Xa assay. 22
The results of this field trial showed that the intralaboratory variations in the PT were mainly dependent on the sensitivity of the local PT reagents, with the largest variations observed with Innovin and RecombiPlasTin 2G irrespective of which instruments were used. In a previous study, we found that Innovin was less sensitive to rivaroxaban compared with other reagents tested. 7 The different instruments used by the participating laboratories contributed to a lesser extent to the variations observed. When the results of undiluted control plasmas were expressed as rivaroxaban concentrations (ng/mL), a lower interlaboratory variation in the PT was obtained and the differences between local PT reagents and central PT reagent were not significant. Overall, the results suggest that plasma concentrations of rivaroxaban may be measured using a rivaroxaban calibration curve, particularly using a single (central) PT reagent. For example, for the control plasma sample containing 160 ng/mL rivaroxaban (actual measured value by HPLC-MS/MS), the interlaboratory CV was 2.2% when using the central PT reagent. The results from this field trial indicate that satisfactory interlaboratory precision can be achieved using freeze-dried rivaroxaban calibration plasma, with results that are equivalent to or better than those obtained with the INR system used for the monitoring of coumarin. 23 Dilution of plasma samples is not recommended; the reason for the nonlinearity of the results after sample dilution is not known: it might be related to the high protein binding of rivaroxaban in plasma. The rivaroxaban concentrations measured in the pooled plasma samples, although close to absolute weight, were higher than the actual values, particularly at lower rivaroxaban concentrations. This overestimation of rivaroxaban concentrations in the manufactured pooled plasma controls is questionable, which may be related to either the addition of different concentrations of factor VIIa to the calibrators and controls for the adjustment of clotting times during the manufacturing process or an underestimation of concentrations determined by HPLC-MS/MS due to the extraction step as expected.
In healthy participants, rivaroxaban reaches its peak level 2 to 3 hours after administration, and the terminal half-life is 5 to 9 hours in young individuals or 11 to 13 hours in elderly individuals.11,24 The maximum plasma concentration (Cmax) ranged from 112 to 184 ng/mL after a 10-mg dose and 111 to 294 ng/mL after a 20-mg dose, 10 and the mean Cmax was 318 ng/mL after taking 20 mg twice daily. 11 In phase II studies for the prevention of VTE, the mean concentration range (minimum plasma concentration [Ctrough] to Cmax) for a total daily rivaroxaban dose of 5 to 20 mg was 12 to 152 ng/mL in the hip study and 19 to 201 ng/mL in the knee study. 4 Therefore, the results from the current study suggest that this PT-based method may be able to measure the peak concentrations of rivaroxaban that would be expected after the administration of a 10-mg dose (for the prevention of VTE), a 20-mg dose (the once-daily dose used in the phase III trials for the treatment of VTE and for the prevention of stroke in patients with atrial fibrillation), or to confirm a suspected overdose, providing that the patient’s baseline PT is not abnormal.
The PT assay is a global clotting test and baseline levels may differ in specific patient populations, being shortened in some and prolonged, or even markedly prolonged, in others. Baseline variability in the PT also limits its utility for the measurement of low rivaroxaban blood or plasma concentrations. Furthermore, the PT assay is not specific for anti-factor Xa activity. However, there are advantages to the PT method, such as its wide availability in every routine clinical laboratory at any time and its low cost. The PT method also provides rapid results in emergency situations, whereas the HPLC-MS/MS method would not be appropriate in such situations because of its turnaround time (2 to 4 days) and limited availability. Using rivaroxaban calibrators, plasma concentrations of rivaroxaban can be measured and expressed in ng/mL, which reduces the interreagent variations (although the results of this study do not indicate that the method could be used to assess compliance or to determine the minimum plasma level of rivaroxaban).
In conclusion, the results of this field trial suggest that it is feasible to measure rivaroxaban plasma concentrations (expressed in ng/mL) using the PT combined with rivaroxaban calibrators and controls. Similar results were obtained for control samples sent to the individual laboratories. Nonetheless, because of the interlaboratory variability observed, in particular at low rivaroxaban plasma concentrations, alternative methods that are more specific and more sensitive are needed for the measurement of a wider range of rivaroxaban plasma concentrations, if required.
Addendum
Participants and Laboratories
D. M. Adcock, Esoterix Colorado Coagulation, Englewood, CO, USA; J. P. Cambus, Rangueil Hospital, Toulouse, France; I. Elalamy, Tenon Hospital, Paris, France; G. Gerotziafas, Tenon Hospital, Paris, France; I. Gouin-Thibault, Charles Foix–Jean Rostand Hospital, lvry-sur-Seine, France; J. Harenberg, Medical Faculty of Mannheim, Ruprecht-Karls University of Heidelberg, Mannheim, Germany; M-H. Horellou, Hôtel-Dieu University Hospital, Paris, France; M. Jacquemin, Catholic University Hospital Leuven, Leuven, Belgium; K. Jochmans, Brussels University Hospital, Brussels, Belgium; C. Le Courvoisier-Flaujac, Hôtel-Dieu Hospital, Paris, France; C. Legnani, St. Orsola-Malpighi University Hospital, Bologna, Italy; E. Lindhoff-Last, Johann Wolfgang Goethe University, Frankfurt, Germany; J. L. Martinoli, Diagnostica Stago, Gennevilliers, France; T. L. Ortel, Duke Clinical Coagulation and Platelet Immunology Laboratories, Durham, NC, USA; G. Palaretti, St. Orsola-Malpighi University Hospital, Bologna, Italy; D. Peetz, Johann Gutenberg University, Mainz, Germany; MMS, Biomnis, Ivry-sur-Seine, France; A. Schade, Cleveland Clinic, Cleveland, OH, USA; S. Testa, Haemostasis & Thrombosis Center, General Hospital, Cremona, Italy; A. Tripodi, Department of Internal Medicine, University of Milan, Milan, Italy; A. M. H. P. van den Besselaar, Hemostasis and Thrombosis Research Center, Leiden, The Netherlands; J. Weitz, Henderson Research Centre, Hamilton, Ontario, Canada; W. Wijns, Erasme Hospital, Brussels, Belgium.
Footnotes
Acknowledgments
The authors would like to acknowledge Yong-Ling Liu who provided editorial support with funding from Bayer HealthCare Pharmaceuticals and Johnson & Johnson Pharmaceutical Research & Development, L.L.C.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MMS is an employee of Biomnis and Hôtel-Dieu University Hospital, is a consultant for Bayer, sanofi-aventis, Eli Lilly, and Daiichi, and is a member of an advisory committee of Johnson & Johnson and Pfizer. TES is an employee of Bayer HealthCare Pharmaceuticals Inc. EP is an employee of and owns stock and options and patents and royalties with Bayer. CG and LLF are employees of Biomnis. YG and GC are employees of Diagnostica Stago. JLM is a consultant for Diagnostica Stago. The authors acknowledge Yong-Ling Liu who provided editorial support with funding from Bayer HealthCare Pharmaceuticals and Johnson & Johnson Pharmaceutical Research & Development, L.L.C.
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: supported by Bayer HealthCare Pharmaceuticals and Johnson & Johnson Pharmaceutical Research & Development, L.L.C. This study has been previously presented at the 54th Congress of the Gesellschaft für Thrombose und Hämostaseforschung (GTH) and Nederlandse Vereniging voor Trombose en Hemostase (NVTH), Nürnberg, Germany, 24-27 February 2010; and the 21st International Congress on Thrombosis (ICT), Milan, Italy, 6-9 July 2010.
