Abstract
Background:
We frequently encounter high levels of activated partial thromboplastin time (aPTT) during heparin anticoagulation. The purpose of this study is, first, to investigate the rate of achieving and maintaining therapeutic aPTT in patients treated with heparin anticoagulation and second, to assess the adequacy the current nomogram.
Methods:
We included 197 patients who underwent anticoagulation with unfractionated heparin (UFH) according to the standard nomogram between September 2008 and May 2010. The primary endpoints were the rate of achieving a therapeutic range (TR) at the first sample, 24 hours, or 48 hours. We also compared heparin nomograms according to age (<70 years vs ≥70years).
Results:
Of the 197 patients, 131 had heparin loading. In the heparin loading group, there were 19.1% (n=25), 69.5% (n=91), and 90.1% (n=18) achieving TR at the first aPTT, 24 hours, and 48 hours, respectively. The therapeutic aPTT proportion was 39.2%, and the rate of peak level above 90 seconds was 93.1%. Peak levels of aPTT were higher in the older age group than in the younger age group (202.3 ± 124.2 versus 152.0 ± 78.9, p=0.007).
Conclusion:
Our results indicate a high rate of achieving therapeutic aPTT at 24hous and 48hours, but a low success rate for maintenance within the TR. Most patients had supratherapeutic aPTT of more than 90 seconds. Therefore, the TR of aPTT that matches heparin levels of 0.3 to 0.7 IU/mL measured by antifactor Xa assay should be determined. If not, we should consider adopting a new heparin dosing nomogram.
Introduction
Intravenous unfractionated heparin (UFH) remains a leading therapeutic option for various thromboembolic disorders and atrial fibrillation, despite drawbacks of its use, particularly with respect to dosing and monitoring and heparin-induced thrombocytopenia. 1 The wide variability in patient response to UFH necessitates continuous monitoring and adjustment. Heparin dosing nomograms began to appear in the literature as early as 1985, 2 and weight-based dosing protocols started to become the standard after 1993. 3 Since that time, many dosing nomograms have been examined and have maintained the same basic structure of a weight-based, bolus dose and infusion rate with periodic monitoring utilizing activated partial thromboplastin time (aPTT). A fixed therapeutic range (TR) for aPTT of 1.5 to 2.5 times the control value has become widely accepted, though other relevant ranges have been applied. 4 A good nomogram helps to achieve a therapeutic aPTT rapidly and maintains the TR for prolonged periods while avoiding periods of overanticoagulation or underanticoagulation, and standardization of a nomogram minimizes adverse events related to interphysician variability, ordering different boluses and rates, and safety is improved with this standardization. There was rare data regarding aPTT maintenance in the Asian population. Therefore, we examined how well aPTT is maintained within clinically relevant ranges in the Samsung Medical Center (SMC).
Methods
We retrospectively analyzed 197 patients who underwent anticoagulation with UFH, according to a standard nomogram between September 2008 and May 2010. A total of 131 patients received a bolus UFH dose of 60 units/kg intravenously, followed by an intravenous infusion of 12 units/kg/h based on actual body weight, with no maximum dosage. The aPTT levels were measured every 6 hours after the infusion was initiated, and the infusion rates were changed until TR of aPTT was achieved. The “TR” was defined as aPTT within 60 to 85 seconds. Our hospital nomogram is summarized in Table 1. The SMC nomogram uses a TR of aPTT referenced by studies, though loading and maintenance dosages are different. 5,6
Nomogram of Unfractionated Heparin (UFH).
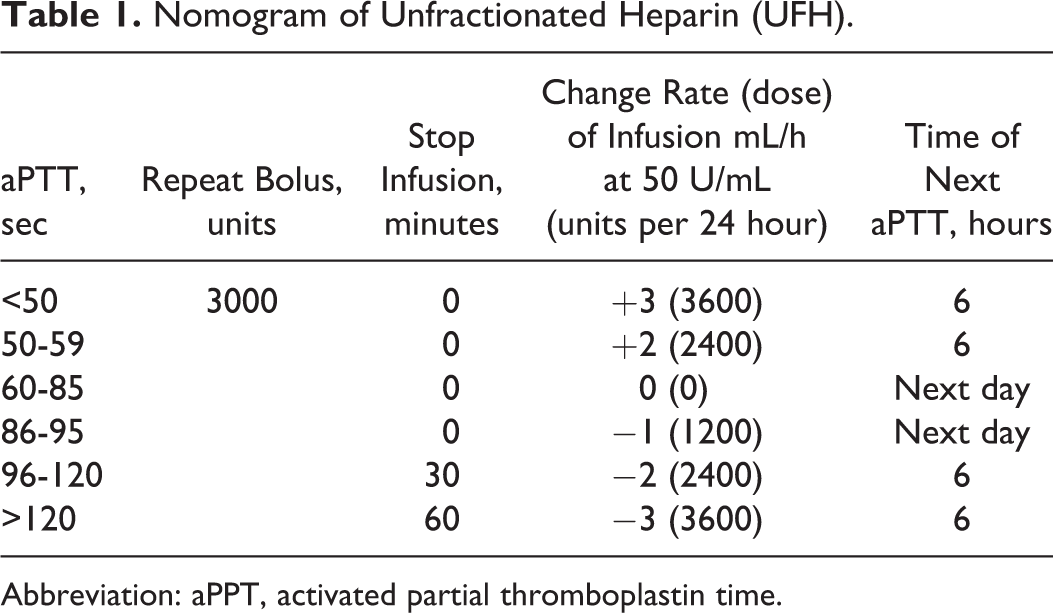
Abbreviation: aPPT, activated partial thromboplastin time.
The STA-PTT Automate 5 (Diagnostica Stago, Asnieres, France) was used for aPTT tests on the STA-R Evolution coagulation analyzer (Diagnostica Stago). The reference range of aPTT was established as 29.1 to 41.9 seconds, according to the Clinical and Laboratory Standards Institute guidelines. The TR of aPTT for monitoring UFH therapy in our center was developed to be 60 to 85 seconds that corresponds to 0.3 to 0.7 IU/mL antifactor Xa activity.
Indications for heparinization included atrial fibrillation with or without ischemic stroke, deep vein thrombosis, and/or pulmonary thromboembolism. We excluded perioperative bridging therapy for patients who take oral anticoagulation therapy, have indications of venous thromboembolism, atrial fibrillation, and mechanical heart valve. Another exclusion criteria were patients who received thrombolytic therapy due to listed indications and patients with acute coronary syndrome because most heparinization in acute coronary syndrome is short-term, bridging therapy before revascularization. Primary end points were the achievement of the TR at the first sample, 24 hours, and 48 hours. Secondary end points were the time to the first TR, the time to first exceed therapeutic level (TL), therapeutic aPTT proportion (therapeutic aPTT results/ the total number of aPTT determinations), peak level and rate of peak level above 90 seconds, and major bleeding and recurrent thromboembolism within 3 months of heparinization initiation. The “TR” was defined as aPTT within 60 to 85 seconds. To exceed, “TL” was defined as aPTT exceeding the upper TR for 85 seconds. We compared aPTT data between the heparin loading group and the no heparin loading group. We also analyzed the heparin nomogram according to age (<70 years vs ≥ 70years) and compared it between the 2 groups.
We used PASS program to evaluate power calculation and performed “inequality tests for one proportion” in PASS program. It was estimated that 130 patients would be required to provide 80% power to detect 0.85 “success rate” that is presented in Table 2. The success rate is the proportion of patients who reached the “ therapeutic threshold” within 24 hours of treatment. We gained 0.85 by calculating the average percentage from 4 data that presented success rate in Table 2. Comparison of data between the 2 groups was assessed by a paired t test for continuous variables, and Pearson chi-square test for categorical variables. A P value <.05 was considered significant for all statistical analyses. Statistical analyses were performed using SPSS version 12 (SPSS, Inc, Chicago, Illinois).
Comparison of Weight-based Nomograms Including SMC Data.
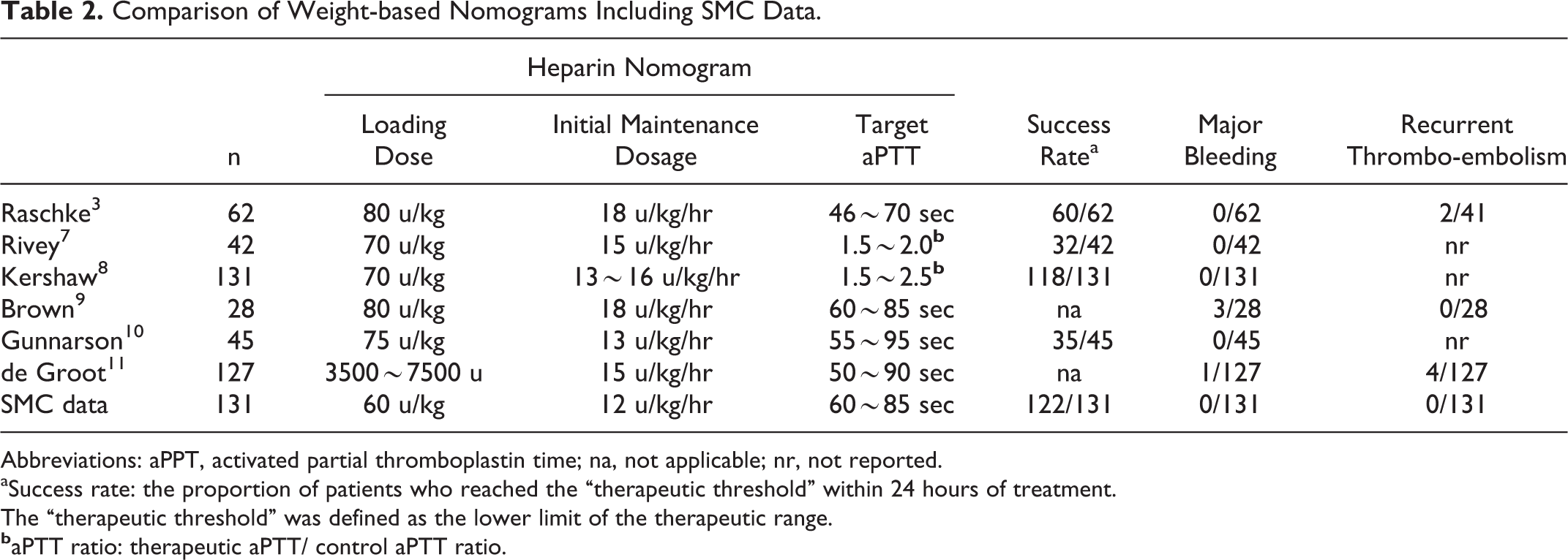
Abbreviations: aPPT, activated partial thromboplastin time; na, not applicable; nr, not reported.
aSuccess rate: the proportion of patients who reached the “therapeutic threshold” within 24 hours of treatment.
The “therapeutic threshold” was defined as the lower limit of the therapeutic range.
Results
Of the 197 patients that were reviewed, 131 had heparin loading. And 158 patients received a heparin infusion because of atrial fibrillation and 39 patients were infused with heparin for venous thromboembolism and/or pulmonary thromboembolism. The mean age of enrolled patients was 66 years. Over 50% of the patients had prior antithrombotic medication use (15.2% aspirin and 38.1% warfarin). There were no significant differences between the loading and no loading groups in terms of the baseline clinical characteristics except for warfarin use and baseline aPTT (Table 3).
Patient Baseline Characteristics.
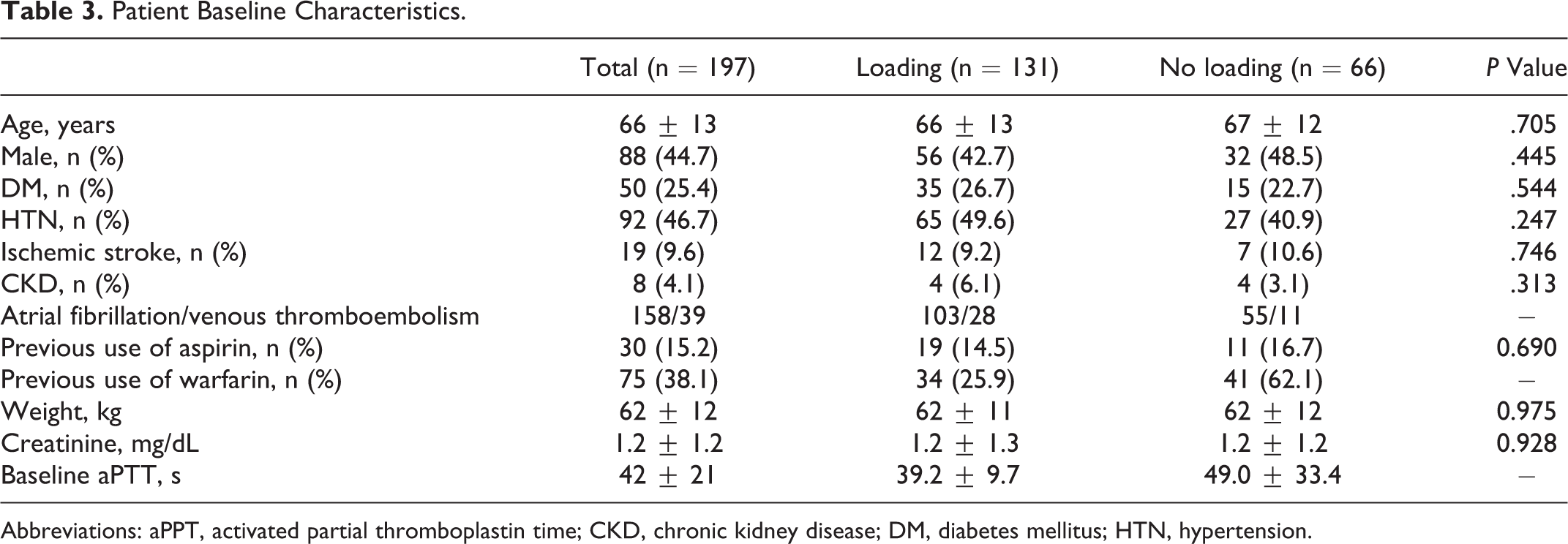
Abbreviations: aPPT, activated partial thromboplastin time; CKD, chronic kidney disease; DM, diabetes mellitus; HTN, hypertension.
In the heparin loading group, TR was achieved at the first aPTT, 24 hours, and 48 hours by 25 (19.1%) patients, 91 (69.5%) patients , and 118 (90.1%) patients, respectively. The mean time to the first TR was 25.6 hours, and the mean time to first exceed the therapeutic threshold was 17.6 hours.
The “therapeutic aPTT proportion” was 39.2% and the peak level was 176 ± 105 seconds; 122 (93.1%) patients had a rate of peak level above 90 seconds. The percentage of patients achieving TR in the loading group was significantly different from the no loading group at the first aPTT but was not significantly different at 24 hours and 48 hours (Table 4).
Comparison of Nomogram Data Between the Heparin Loading and No-loading Groups.
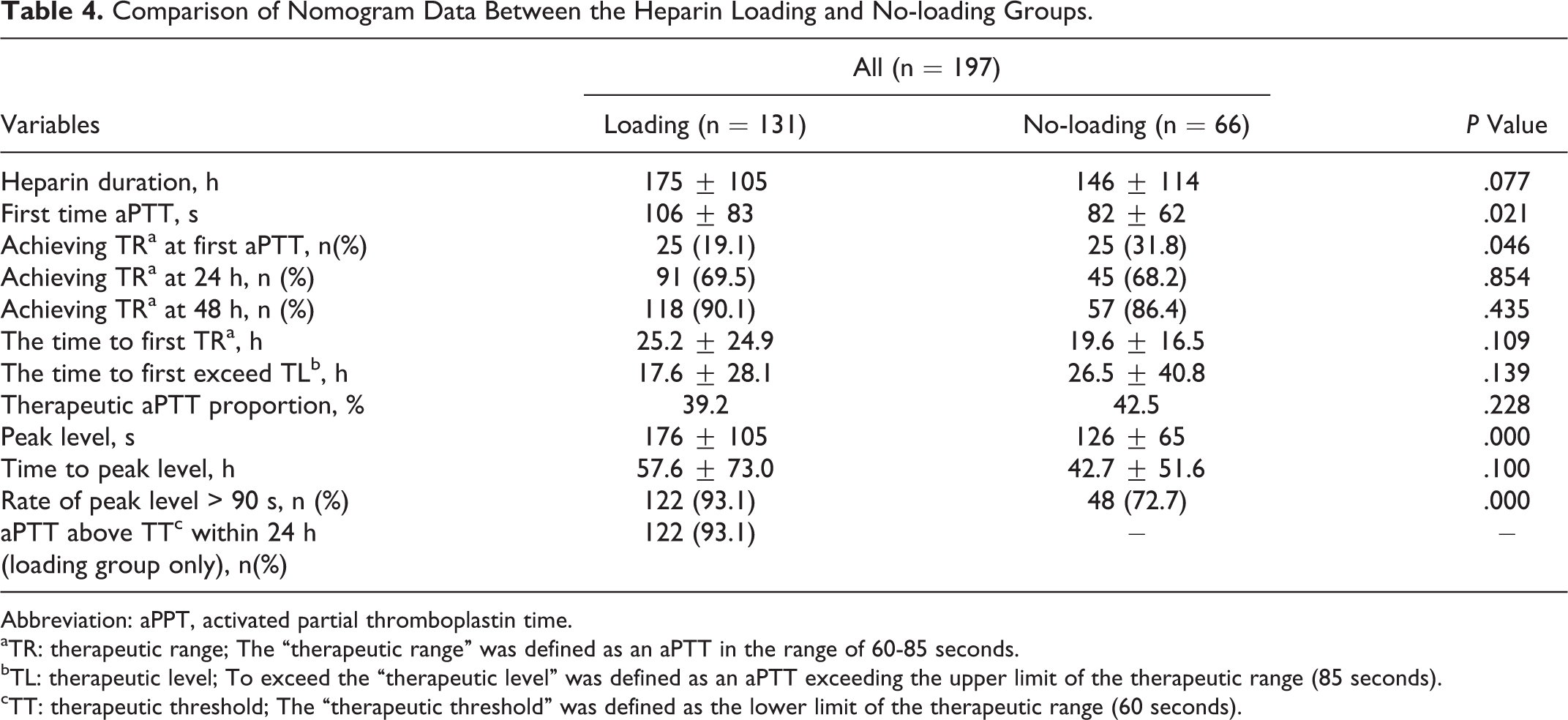
Abbreviation: aPPT, activated partial thromboplastin time.
aTR: therapeutic range; The “therapeutic range” was defined as an aPTT in the range of 60-85 seconds.
bTL: therapeutic level; To exceed the “therapeutic level” was defined as an aPTT exceeding the upper limit of the therapeutic range (85 seconds).
cTT: therapeutic threshold; The “therapeutic threshold” was defined as the lower limit of the therapeutic range (60 seconds).
After dividing the loading group into 2 subgroups according to age, we found that some differences existed between the groups. First, the peak level of aPTT was higher in the older age group (202.3 ± 124.2 vs 152.0 ± 78.9, P = .007). Second, the first time aPTT (125 ± 105 vs 89 ± 53 seconds, P = 0.018
Comparison of Nomogram Data According to Age (<70 years vs ≥70 Years).
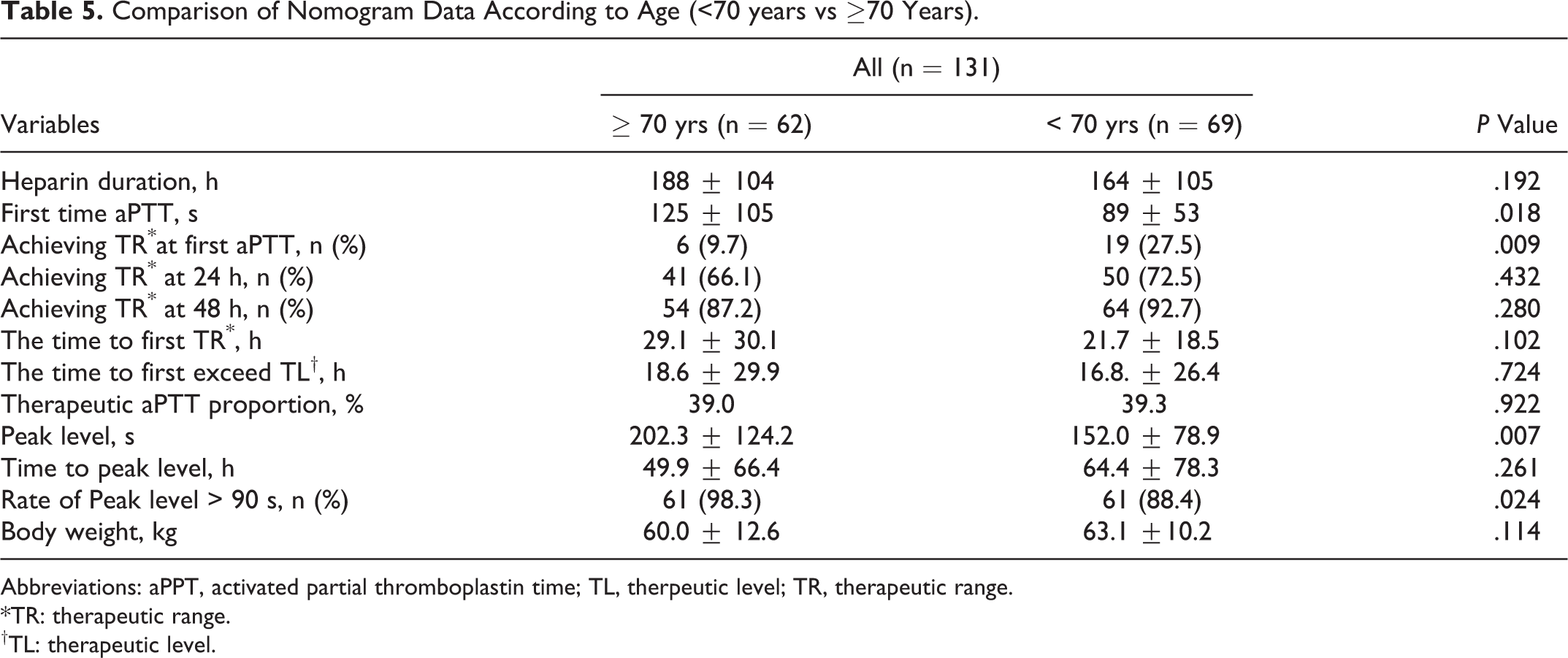
Abbreviations: aPPT, activated partial thromboplastin time; TL, therpeutic level; TR, therapeutic range. *TR: therapeutic range. †TL: therapeutic level.
Discussion
The most widely used laboratory assay for monitoring UFH therapy is aPTT. A TR of 1.5 to 2.5 times the mean control value was widely accepted in the past; however, it was challenged by the demonstration of potential risk of subtherapeutic dosing due to different sensitivities of aPTT reagents and even lot–lot variations. 12 Indeed, the sensitivity of the test to heparin is highly reagent and instrument dependent. In an attempt to resolve this issue, the College of American Pathologists (CAP) has published consensus guidelines for laboratory monitoring of UFH using aPTT, which mandates individual laboratories to develop their own TR of aPTT to correspond to 0.3 to 0.7 IU/mL antifactor Xa activity or 0.2 to 0.4 USP U/mL by protamine titration. 13,14 The clinical laboratory at our institution is a CAP-accredited laboratory and has implemented the aPTT TR according to the guidelines.
It is important to maintain an aPTT level within the TR in patients treated with UFH, because previous studies have reported an increased risk of recurrent thrombosis in patients who do not achieve the therapeutic aPTT level within 24 or 48 hours of commencing UFH. 3,15,16 According to the studies by Hull et al, failure to achieve a therapeutic aPTT by 24 hours was associated with a 23.3% frequency of recurrent venous thromboembolism, compared with 4% to 6% for those whose aPTT exceeded the therapeutic threshold by 24 hours. 16 There have also been reports of a relationship between UFH concentration and bleeding, 17,18 although the data used heparin levels, not aPTT, and the association between overanticoagulation and bleeding were less accurate. Therefore, the success of a heparin dosing nomogram depends on the percentage of patients achieving a target laboratory value within a certain time frame.
The results of this study show that we achieved the desired target ranges in 69.5% of the patients within 24 hours and in 90.1% of the patients within 48 hours. These findings are not inferior to previously published data based on the weight-based heparin nomogram developed by Raschke et al (Table 6). 3 Our study results are similar to those of studies of weight-based nomograms if the primary end point is changed to the proportion of patients who reached the therapeutic threshold within 24 hours of treatment, not the proportion of the TR.” 3,7 –11 We found relatively high levels of peak aPTT and a high rate of peak level over 90 seconds in the loading group, especially remarkable in the older age group. These data have clinical implications because a higher aPTT may be a potential risk of bleeding complications even though we were unable to show an association between laboratory data and bleeding events.
Proportion of Activated partial Thromboplastin Time Values Within Clinically Relevant Ranges Over Time: A Comparison of SMC Data With Western Published Data.
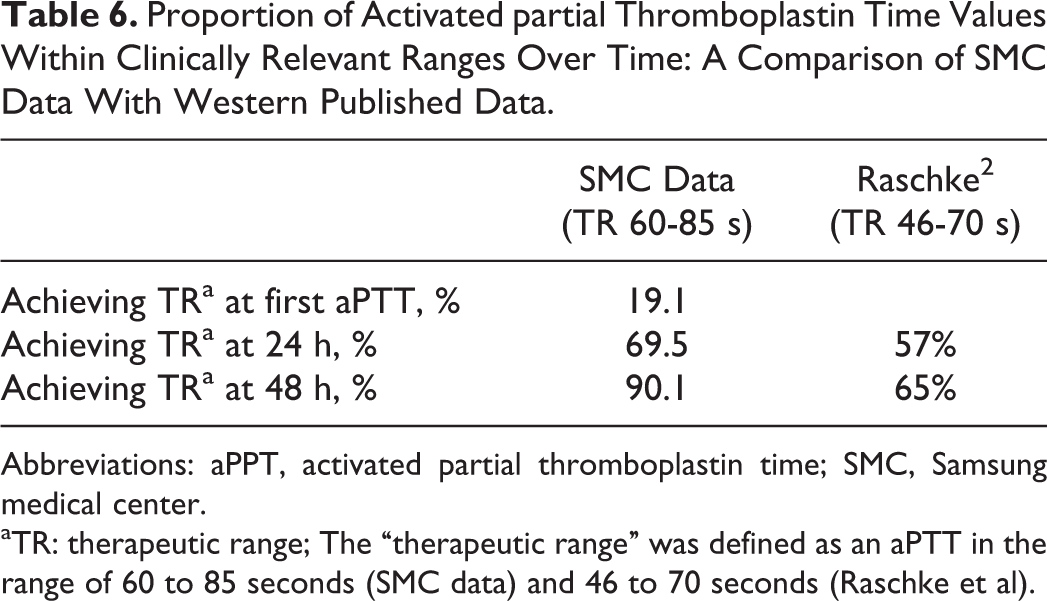
Abbreviations: aPPT, activated partial thromboplastin time; SMC, Samsung medical center.
aTR: therapeutic range; The “therapeutic range” was defined as an aPTT in the range of 60 to 85 seconds (SMC data) and 46 to 70 seconds (Raschke et al).
Aging is a risk factor for heparin-related bleeding treated with anticoagulation. 19 Campbell et al reported that aging was associated with increased bleeding episodes, increased heparin levels, a tendency for an increase in aPTT, and lower heparin dose requirements. 19
We used a lower loading and maintenance dose compared to similar published data, 3,7 –11,20 but gained a relatively higher peak aPTT level and rate of aPTT over 90 seconds, which can be partially explained by racial differences (Table 2). There was some report about genetic polymorphism associated with aPTT. We think these kinds of genetic polymorphism associated with aPTT may affect variable aPTT response to UFH. 21
Recently, the antifactor Xa assay 22,23 was introduced as an alternative monitoring strategy after the drawbacks of using aPTT levels to monitor heparin were identified: poor correlation with blood heparin concentration, varying response to aPTT reagents, and the responsiveness of aPTT to biological factors independent of heparin activities. 24 –27 Unlike aPTT, the antifactor Xa assay is unaffected by the concomitant use of warfarin or by varying concentrations of sodium citrate in collection tubes and is only subject to minimal interference from the presence of lupus anticoagulants, elevated factor VIII activity, liver disease, and other factors. 28 Recent reports suggest that monitoring UFH infusion with antifactor Xa assay, compared with conventional nomogram, achieves therapeutic anticoagulation more rapidly, maintains the values within the goal range for a longer time, and requires fewer adjustments in dosage and repeated tests. 22,23 As explained in method, the TR of aPTT for monitoring UFH therapy in our center was developed to be 60 to 85 seconds, which corresponds to 0.3 to 0.7 IU/mL antifactor Xa activity. Whenever there is a lot change of aPTT reagent, we perform comparison tests to confirm that the results from the reagent with a new lot are acceptable. Although we used heparin nomogram based on antifactor Xa activity, our weight-adjusted heparin nomogram was not equal to antifactor Xa monitoring method. So far, antifactor Xa assay is better than conventional nomogram. But most centers used conventional weight-adjusted heparin nomogram, and so more frequent next aPTT monitoring even when we achieve the TR initially, which is another alternative strategy for improving nomogram accuracy.
Study Limitations
This study has a few limitations. First, our data were based on short-term UFH anticoagulation before changing with oral anticoagulation. There was no major bleeding event or thromboembolic event during hospitalization. Unfortunately, we cannot explain whether the bleeding and embolic event happen or not, because we did not keep track of event outside the hospital, for example, by telephone contact with the patients. Although the bleeding and thromboembolic events may not be common if we think intuitively, the delayed event can happen. We could not prove the existence of a meaningful association between laboratory tests and clinical events.
Second, we did not check platelet count regularly. Platelet count monitoring is important after UFH anticoagulation, because heparin-induced thrombocytopenia as well as high aPTT level may be associated with bleeding event. Third, we analyzed a small number of patients; thus, it is difficult to discern whether age really has an impact on the aPTT value. A larger sample is required to prove the existence of a relationship between a laboratory test and a clinical event and the impact of age on the aPTT value.
Conclusion
Our current heparin nomogram using aPTT achieved a therapeutic aPTT within 24 hours and 48 hours, but the peak aPTT level and rate of aPTT over the therapeutic value were high, especially in the older age group. This suggests that the aPTT nomogram protocol should be revised, loading dose and frequency of aPTT monitoring, especially in the elderly patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article .
