Abstract
Chronic thromboembolic pulmonary hypertension (CTEPH) is characterized by thrombotic obstruction of the pulmonary arteries, and right ventricular (RV) dysfunction is a major cause of death. Cardiac magnetic resonance (CMR) is the gold standard for assessing heart wall deformation; therefore, we aimed to determine the prognostic value of CMR strain in patients with CTEPH. Strain derived by CMR was measured at the time of diagnosis in 45 patients with CTEPH, and the relationship between RV strain and prognosis was determined through follow-up. The value of RV strain in the prognostic model was compared with that of pulmonary arterial hypertension (PAH) risk stratification. The RV global peak longitudinal strain (GLS) and global peak circumferential strain (GCS) in CTEPH patients were lower than the normal references of RV strain in the control group. GLS and longitudinal strain in the basal segment were independent risk factors for adverse events (P < .050). Adding CMR parameters to PAH risk stratification improved its predictive power in patients with CTEPH. GLS and GCS scores were impaired in patients with chronic RV overload. RV strain derived by CMR imaging is a promising noninvasive tool for the follow-up of patients with CTEPH.
Keywords
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) is caused by obstruction of the pulmonary vasculature, leading to the redistribution of blood flow and secondary remodeling of the pulmonary microvascular bed.1–3 Right ventricular (RV) function is a major determinant of prognosis in patients with various forms of pulmonary hypertension (PH),4–6 and most patients with CTEPH die from RV failure.7,8 Thus, the assessment of RV function plays an important role in managing patients with CTEPH.
Cardiac magnetic resonance (CMR) imaging is the gold standard for measuring RV function because of its high spatial resolution, high sensitivity, and reproducibility. 9 Most studies on CTEPH have focused on estimating the RV ejection fraction (RVEF), RV stroke volume (SV), and other parameters that emphasize right ventricle-pulmonary artery coupling.10–12 However, changes in these parameters may occur in the late stage of impaired systolic/diastolic function and have limited sensitivity for identifying regional myocardial impairment. Measurements of cardiac contractility and the assessment of myocardial deformation by strain analyses are emerging, and promising tools to characterize impaired cardiac function13,14 have been demonstrated to be feasible and sensitive quantitative techniques for assessing RV contractile function in various cardiac diseases.15–17 RV strain has previously been shown to be related to cardiovascular events and mortality in pulmonary arterial hypertension (PAH).15,18,19 However, little is known about the relationship between RV strain and long-term clinical outcomes in patients with CTEPH. Consequently, this study investigated the CMR-derived strain patterns in patients with CTEPH to assess their prognostic implications.
Materials and Methods
Ethics Statement
This study was conducted following the principles of the Declaration of Helsinki. The study protocol was approved by the Ethics Committee of the Beijing Chao-Yang Hospital, and all participants provided written informed consent before the initiation of the study.
Study Population and Data Collection
This was a single-center, retrospective, observational study of patients admitted to Chao-Yang Hospital between December 2016 and September 2020 who were diagnosed with CTEPH according to the criteria of 2022 European Society of Cardiology (ESC)/European Respiratory Society (ERS) guidelines. 20 CTEPH diagnosis is based on mean pulmonary artery pressure (mPAP) ≥ 25 mmHg with a pulmonary arterial wedge pressure ≤ 15 mmHg, mismatched perfusion defects on the lung scan, and specific diagnostic signs for CTEPH observed by multidetector computed tomography angiography, magnetic resonance (MR), imaging or conventional pulmonary cineangiography after at least 3 months of effective anticoagulation to discriminate this condition from “subacute PE,” 21 as well as baseline CMR. The exclusion criteria were: (a) existing PH from other causes, (b) ongoing pneumonia, (c) malignant tumor or lobectomy, and (d) loss to follow-up CMR imaging. The control group consisted of 20 patients without PH, pulmonary, embolism, or other diseases that seriously affected the function of the right heart and met the same exclusion criteria. The patients in the control group were matched in terms of age and gender distribution for comparison with those in CTEPH group.
The following data were collected: (a) demographics; (b) clinical evaluation including the assessment of World Health Organization (WHO) functional class and 6-min walk distance (6WMD); and (3) right heart catheterization (RHC) parameters, CMR parameters, and laboratory tests (N-terminal pro-brain natriuretic peptide [NT-proBNP]) were performed to evaluate cardiac function. Then, according to PAH risk stratification in ESC/ERS guidelines, the population was divided into low-risk and mediate-high-risk. 20 Using CMR parameters for reclassification, the risk level increased if the conditions were met; otherwise, the risk level decreased.
MR Acquisition and Analysis
MR images were acquired at 3.0T (Prisma, Siemens Healthcare, Germany) with an 8-channel cardiac coil and retrospective electrocardiogram gating, breath-hold technique. After scout imaging, CMR cine images were acquired continuously in a short-axis plane to cover the whole ventricle using Ture fast imaging with steady-state precession sequence (repetition time 3.0-3.2 ms, echo time 1.4 ms, flip angle 70°, field of view 320 mm × 360 mm, matrix size 256 mm × 256 mm, slice thickness 8 mm, and phases per cardiac cycle 25), and LV long-axis plane including images of 2-, 3-, and 4-chamber views were acquired as well.
Image analysis was performed offline by 2 experienced observers (B.W. Zhang and X.J. Guo with 3 and 10 years of cardiovascular MR imaging [MRI] experience, respectively) using commercial software (CMR42 version CVI 5.11.3, Circle Cardiovascular Imaging, Calgary, Alberta, Canada). The endocardial contours were automatically drawn on end-systolic images and end-diastolic images with manual correction if necessary. And then, myocardial epicardial contours were semiautomatically tracked on end-systolic images and end-diastolic images with manual correction if necessary. The trabeculae and papillary muscles were excluded from the cavity to derive the endocardial contours. Biventricular volumes (end-diastolic volume index [EDVi], SV index, end-systolic volume index [ESVi]), cardiac output, and ejection fraction (EF) were also performed on the same software based on the short-axis cine images.
Myocardial strain analysis was performed using a tissue tracking model of offline software (CVI 5.13.5) (Figure 1), by importing all short-axis and long 4-chamber plane images. The RV global and regional (apical, mid-ventricular, and basal) feature tracking parameters were derived automatically, including myocardial longitudinal and circumferential peak strain (PS). PS was defined as the maximum absolute value of the strain in the entire cardiac cycle. 22 Manual border readjustment and the propagation algorithm were reapplied immediately when the investigator found poor tracking. Radial strain parameters were not performed as it is reported that the quantification of radial deformation using MR-FT was less reliable. 23
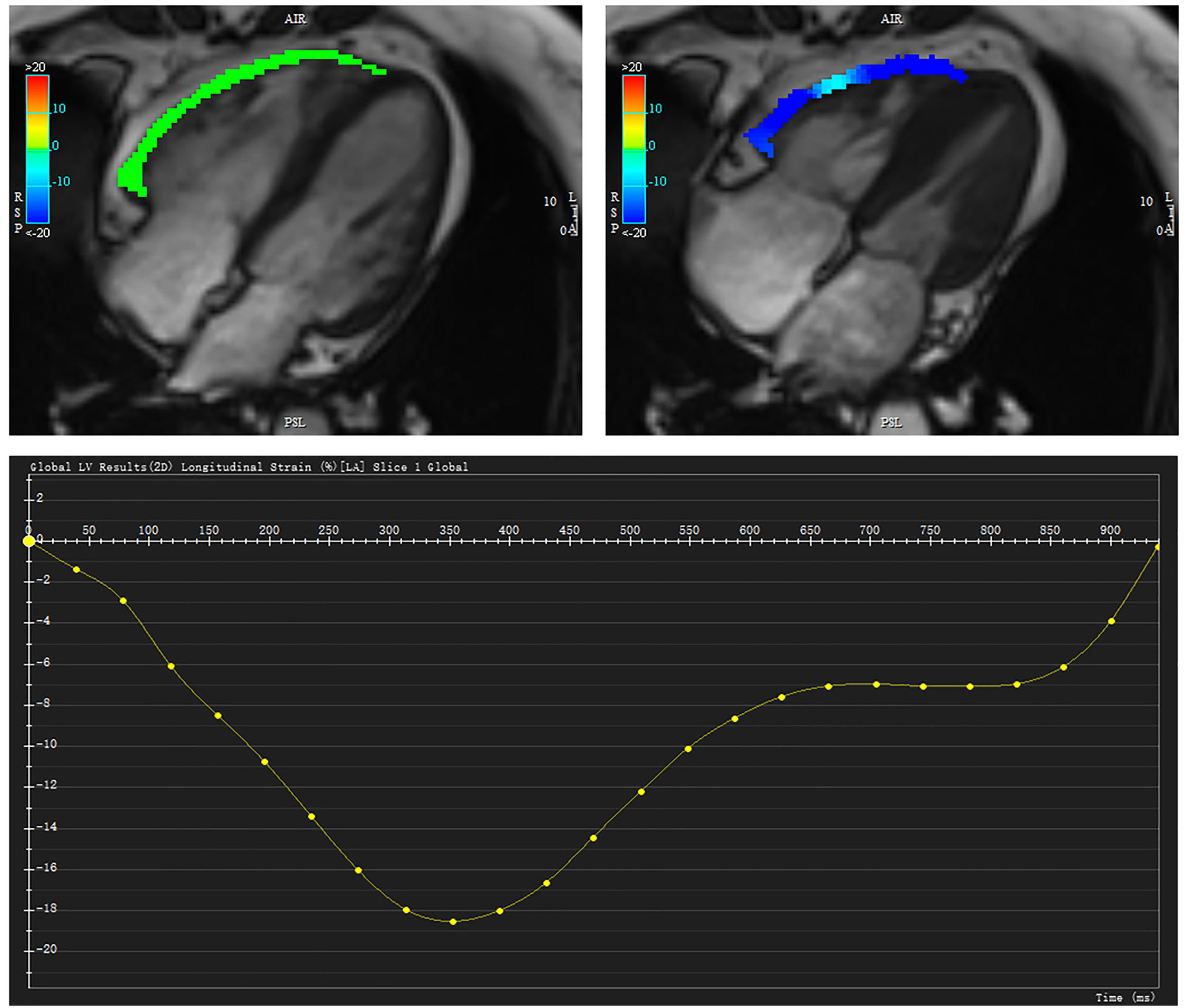
Right ventricular (RV) longitudinal strain analysis obtained from a 4-chamber view.
Follow-up and Endpoint
All patients were followed up until January 1, 2022, by telephone or review of hospital medical records in the event of return visits to the emergency room or hospital admission. The primary endpoints were all-cause death and hospitalization due to worsening heart failure.
Reproducibility
Intraobserver and interobserver variability of the RV longitudinal and circumferential strain parameters was analyzed in a group of 20 randomly selected subjects by 2 investigators (B.W. Zhang. and X.J. Guo, with 3 and 10 years of cardiovascular MRI experience, respectively) who were blinded to each other's results.
Statistical Analysis
IBM SPSS Statistic Software Version 26 (International Business Machines, Armonk, New York, USA) and RStudio (version 1.4.1103; RStudio, Inc.) were used for all statistical calculations. Categorical data are presented in absolute numbers and percentages. Continuous data are reported as median with interquartile range (IQR) or mean ± SD. Independent samples student t-tests, one-way analysis of variance, and Kruskal-Wallis tests were used to compare continuous variables between groups, and chi-square test or Fisher exact test, where appropriate, were used for categorical variables. For the predictors of adverse events, the best cut-off value according to the Youden index was calculated. Then the patient population was categorized according to the cut-off value. And the 2 patient groups’ associations of strain variables with time to events were assessed using Kaplan-Meier curves and the log-rank test. Associations between variables and prognosis were assessed using Cox proportional hazards models. Only variables associated with outcome (P < .100) in univariable analysis were entered into multivariable models. For model comparison, the concordance (C-index) and Akaike information criterion (AIC) were performed. The C-index of 1.0 indicates the best predictive power, while the C-index of 0.5 indicates no predictive power at all. AIC was used to describe the accuracy of the prognostic model, and a lower AIC value indicates a higher model accuracy. We used the net reclassification improvement (NRI) to assess the incremental value of adding the value of strain on top of the PAH risk stratification. All tests were 2-sided and P < 0.050 was considered statistically significant.
Results
Baseline Characteristics
Baseline CMR and RHC data were available for 51 patients with CTEPH; however, 5 (9.8%) patients who either did not reach or refused to participate and 1 (2.0%) patient with an atrial septal defect were excluded. In total, 45 patients were included in this study, and their baseline characteristics associated with the occurrence of events (death and heart failure) are displayed in Table 1. The study population was 57 ± 12 years old and predominantly male.
General Characteristics of the Entire Study Group and Patients With and Without Events.

Abbreviations: BSA, body surface area; BMI, body mass index; HR, heart rate; SAP, systolic arterial pressure; WHO, World Health Organization; 6WMD, 6-min walk distance; NT-proBNP, N-terminal pro-brain natriuretic peptide; mPAP, mean pulmonary artery pressure; PVR, pulmonary vascular resistance; RAP, right atrial pressure; CI, cardiac index; SvO2, mixed venous oxygen saturation; EF, ejection fraction; EDVi, end-diastolic volume index; LVEDVi, left ventricular EDVi; RVEDVi, right ventricular EDVi; ESVi, end-systolic volume index; LVESVi, left ventricular ESVi; RVESVi, right ventricular ESVi; LVEF; left ventricular ejection fraction; RVEF, right ventricular ejection fraction.
During a median follow-up time of 40 months and IQR of 29 to 53 months, 5 (11.1%) patients died, and 5 (11.1%) patients were hospitalized for heart failure symptoms. These patients were classified as event patients, whereas those classified as survivors were considered no-event patients. The no-event survival rates were 86.7% at 1 year and 76.9% at 5 years.
Tissue Tracking CMR and Events
A comparison of CTEPH patients with the control group showed that the absolute value of the strain in longitudinal and circumferential axes of CTEPH patients was lower than that of the control group (Table 2).
Comparison of RV Strain Between CTEPH and Control Groups.

Abbreviations: CTEPH, chronic thromboembolic pulmonary hypertension; RV, right ventricular; GCS, global peak circumferential strain; GCS-a, GCS for apical; GCS-b, GCS for basal; GCS-m, GCS for mid; GCS D′, GCS diastolic strain rate; GCS S′, GCS systolic strain rate; GLS, global peak longitudinal strain; GLS-a, GLS for apical; GLS-b, GLS for basal; GLS-m, GLS for mid; GLS D′, GLS diastolic strain rate; GLS S′, GLS systolic strain rate.
Table 3 summarizes the parameters of RV strain by CMR for all patients and the differences between the strains in patients with and without events. The RV global peak longitudinal strain (RVGLS) of the event group was significantly lower than that of the no-event group; however, there were no statistically significant differences in other parameters. Subsequently, Youden index was applied to determine the best cut-off point of −7.665%.
Comparison of RV Strain Between the Events Group and No Events Group of CTEPH Patients.

Abbreviations: CTEPH, chronic thromboembolic pulmonary hypertension; GCS, global peak circumferential strain; GCS-a, GCS for apical; GCS-b, GCS for basal; GCS-m, GCS for mid; GCS D′, GCS diastolic strain rate; GCS S′, GCS systolic strain rate; GLS, global peak longitudinal strain; GLS-a, GLS for apical; GLS-b, GLS for basal; GLS-m, GLS for mid; GLS D′, GLS diastolic strain rate; GLS S′, GLS systolic strain rate.
Eleven patients (24.4%) were considered to have low-risk CTEPH and 34 patients (75.56%) had mediate-high-risk CTEPH. Ten patients (22.2%) had RVGLS ≥ −7.665 and 35 (77.8%) had RVGLS < −7.665 (Figure 2). According to the cut-off RVGLS, 40% of patients were reclassified, with 17 from the mediate-high-risk group moving to the global peak longitudinal strain (GLS)-defined low-risk group and 1 from the low-risk group to the GLS-defined mediate-high-risk group (Figure 3). Analysis of all parameters revealed that RVGLS ≥ −7.665 was associated with a lower 6MWD (231 ± 120 vs 372 ± 99, P = .003), higher mPAP (60 ± 14 vs 47 ± 12, P = .008), and lower mixed venous oxygen saturation (SvO2) (48.2 ± 9.0 vs 59.2 ± 8.4, P = .004), but not NT-proBNP, pulmonary vascular resistance (PVR) (P = .081), cardiac index (CI) (P = .955), or RV SV index for body surface area (RVSV index) (P = .530) (Figure 4). Patients with RVGLS ≥ −7.665 had a higher event rate than patients with RVGLS < −7.665 (Figure 5), but this difference was only observed in the mediate-high-risk group (Figures 2 and 6). NRI was 8.6% (P < .001), significantly increasing the predictive value of adding RVGLS to the ESC model.

Bar chart of the observed events by risk categories from a prognostic algorithm with GLS in the study population.

Reclassification across risk categories between ESC model and ESC model with GLS in the study population. If GLS ≥ −7.665, the risk is increased; otherwise, reduce the risk level. (a) ESC model and (b) ESC model with GLS.
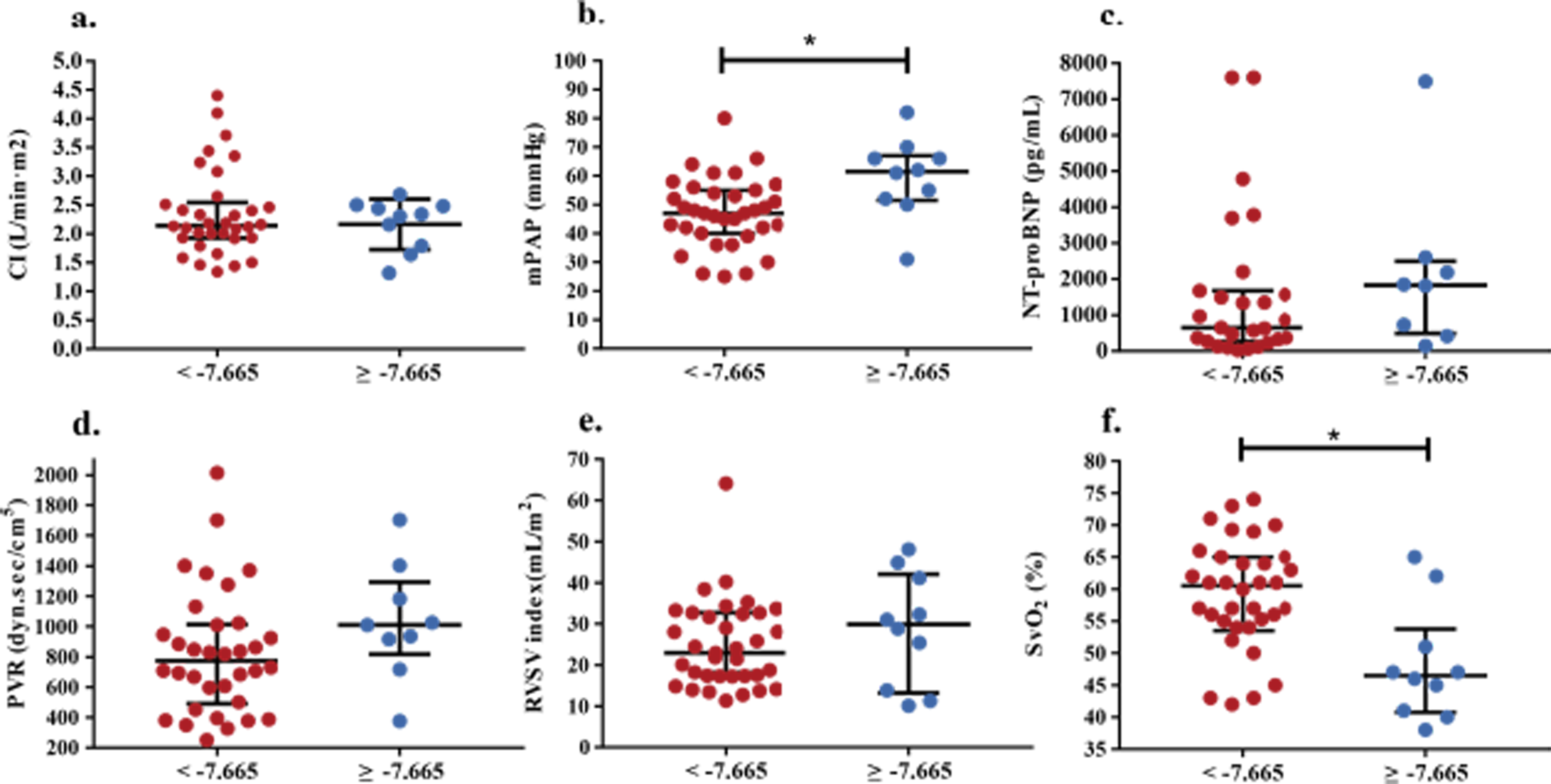
Association of right ventricular global longitudinal strain with clinical, right-sided heart catheterization, echocardiographic parameters. (a) CI. (b) mPAP. (c) NT-proBNP. (d) PVR. (e) RVSV index. (f) SvO2. *P < .05.

Kaplan-Meier estimates event-free survival of all CTEPH patients.
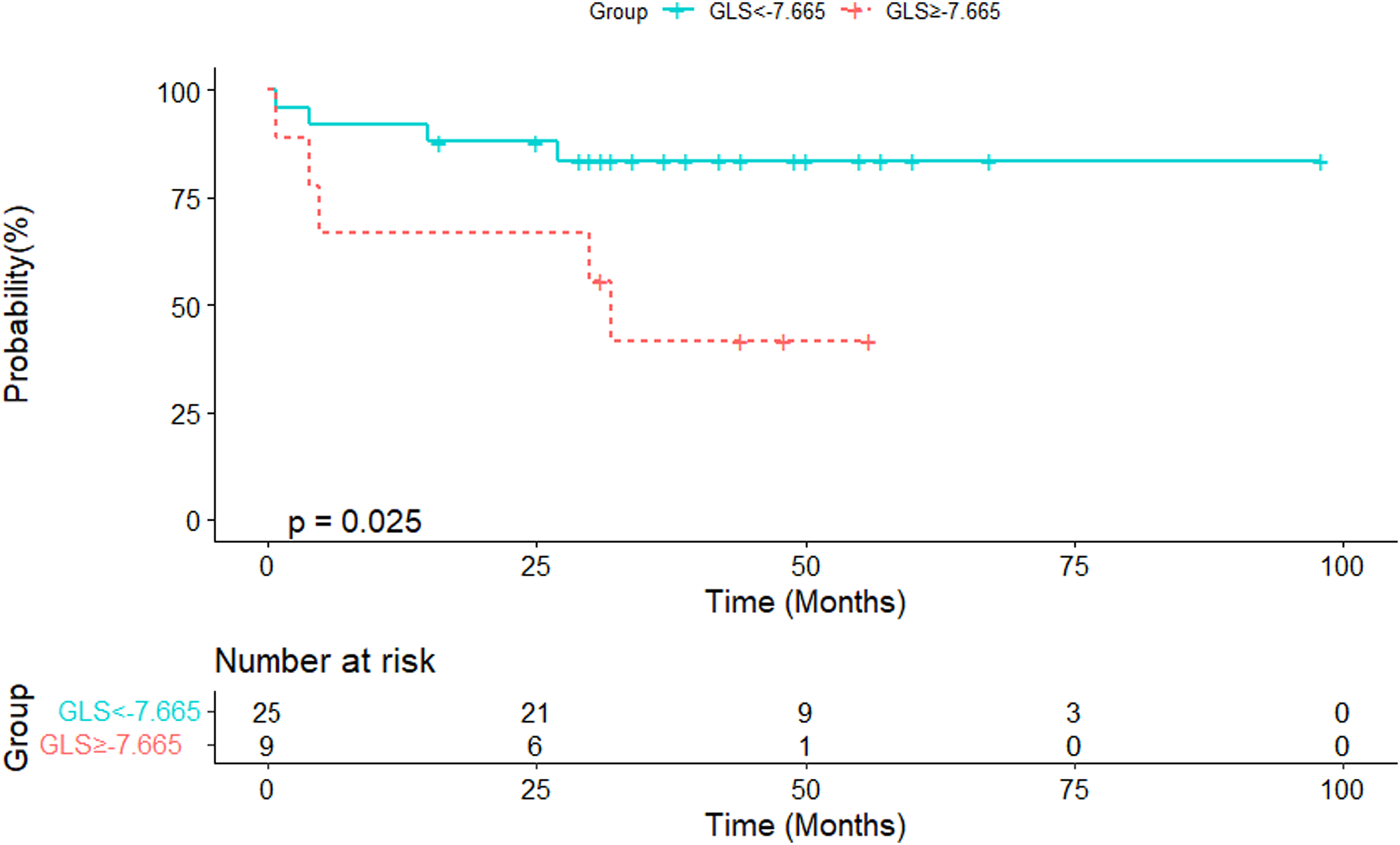
Kaplan-Meier estimates event-free survival of mediate-high risk patients.
Survival and Prognostic Factors
Univariate Cox regression analysis was performed to determine the individual predictive values of baseline parameters and strain (Table 4). Accordingly, sex, WHO functional class, 6WMD, mPAP, PVR, CI, SvO2, GLS, GLS for basal (GLS-b), and GLS for apical (GLS-a) were associated with the risk of events.
Predictors of Events: Univariable Study.

Abbreviations: HR, heart rate; SAP, systolic arterial pressure; WHO, World Health Organization; 6WMD, 6-min walk distance; NT-proBNP, N-terminal pro-brain natriuretic peptide; mPAP, mean pulmonary artery pressure; PVR, pulmonary vascular resistance; RAP, right atrial pressure; CI, cardiac index; SvO2, mixed venous oxygen saturation; RV, right ventricular; GCS, global peak circumferential strain; GCS-a, GCS for apical; GCS-b, GCS for basal; GCS-m, GCS for mid; GLS, global peak longitudinal strain; GLS-a, GLS for apical; GLS-b, GLS for basal; GLS-m, GLS for mid.
Multivariate Cox regression analysis was performed to compare the additional CMR parameter values (Table 5). First, we determined the baseline characteristic model using Cox regression analyses with sex, WHO functional class, 6MWD, mPAP, and PVR. Model 1 with only sex, WHO functional class, and 6MWD reached statistical significance with an AIC of 51.96 and a C-index of 0.92. Model 2, with the addition of GLS, had an AIC of 62.02 and a C-index of 0.87. Similarly, model 3, comprising sex, WHO functional class, 6MWD, and GLS-b, had an AIC of 49.45 and a C-index of 0.94. When GLS-a was added to model 1, GLS-a (P = .249) was not statistically significant; therefore, GLS-a was excluded from the final model.
Predictors of Events: Multivariable Study.

Abbreviations: WHO, World Health Organization; 6WMD, 6-min walk distance; GLS, global peak longitudinal strain; GLS-b, GLS for basal; AIC, Akaike information criterion.
Data Reproducibility
Both intraobserver and interobserver reproducibility were excellent for RV global longitudinal and circumferential strain parameters and basal and mid-ventricular segmental strain parameters (Supplemental Figure 1).
Discussion
The present study compared the prognostic value of baseline characteristics and RV strain by CMR after CTEPH diagnosis, revealing that CTEPH patients who experience events have impaired RVGLS compared with no-event patients; RVGLS by CMR has a prognostic value after CTEPH diagnosis with a prognostic model combining sex, WHO functional class, 6WMD, and GLS-b being the best. The GLS cut-off value at the first CMR helps distinguish populations who are prone to adverse events, and adding this value to PAH risk stratification is more applicable to the risk stratification of CTEPH.
RV Strain in CTEPH Patients
CMR, as the gold standard for assessing deformation of the heart wall, can provide high-quality cross-sectional images of the heart across the cardiac cycle.24–26 Despite the ability of echocardiography to assess myocardial strain at lower costs and in less time than CMR, 27 it is strongly dependent on the operator's experience and patients’ echogenic windows, whereas its reproducibility for strain measures may not be optimal at the segmental level. 28 In contrast, in healthcare facilities with appropriate CMR scanners and experience, the evaluation of RV regional wall abnormalities is presumably more robust because of better-quality images. 9 Evidence shows that RVEF measured by CMR is often used as a reference for RV function in clinical practice. Indeed, several studies have revealed that myocardial strain has a significant clinical value, especially for early detection of cardiac function or structural abnormalities before reduction.14,29,30 However, few studies have investigated the predictive function of strain at the time of diagnosis in CTEPH patients. Herein, we demonstrated that CTEPH patients had significantly lower global peak circumferential strain (GCS), peak circumferential systolic strain rate (GCS S′), GCS for basal (GCS-b), GCS for mid (GCS-m), GCS for apical (GCS-a), GLS, and GLS for mid (GLS-m) than patients without PH. In addition, the strain values of the control group in the non-PH population were consistent with those reported by Liu et al. 31
RVGLS ≥−7.665% in CTEPH patients identified a cohort of patients with marked RV dysfunction and a marked predisposition to subsequent right-sided heart failure, higher pulmonary artery pressure, clinical deterioration, and a high risk of poor events. This view is consistent with the research of Sachdev et al 15 on patients with PAH.
Prognostic Value of CMR at Initial Diagnosis
The WHO functional class, 6WMD, right atrial pressure (RAP), and PVR at diagnosis are independent risk factors for CTEPH,32,33 but only 3 risk factors were identified in the current study, including WHO functional class, 6WMD, and gender. This difference may be due to these indicators showing significant differences when the patient's RV function was decompensated; consequently, they cannot be used to evaluate the prognosis of patients in the early stage of the disease. In line with previous reports, the female gender is a risk factor for inferior hemodynamic improvement after pulmonary thromboendarterectomy.34,35 This study indicates that the female gender is an independent risk factor for adverse events, although this effect is slight. The reasons for this poor prognosis related to gender are unclear, as male CTEPH patients may have better cardiopulmonary functions than female patients, such as ventilation and gas exchange efficiency 36 ; however, it may be the effect of estrogen on endothelium-triggered vasoconstriction in the small pulmonary vessels.35,37 Our study also proved that when a myocardial injury occurs in the longitudinal plane, it is more likely to lead to a reduced deformation ability of RV myocardium and decompensation of the myocardium in the case of CTEPH, resulting in a poor prognosis. RV geometry is complex, but our results are consistent with the main direction of muscle fibers in RV in the longitudinal plane,38–41 and the measurement of circumferential function cannot directly reflect the main components of RV contraction function.42,43
RVGLS-b, in combination with WHO functional class, 6WMD, and gender, provides the best prognostic information and confirms that the reduced RVGLS-b is linked to poor prognosis since RVGLS-b declined more in those who experienced adverse events. Sunbul et al 41 also stated that significantly lower basal RV lateral strains were observed in patients with shorter 6MWD, and it may reflect a poorer prognosis. This may be because segmental shortening is nonuniform in the longitudinal plane, and contraction at the basal level contributes most to shortening in the longitudinal direction of RV myocardium. We hypothesized that when chronic PH decompensates RV myocardium, it may damage the deformability of the myocardium at GLS-b. Furthermore, once the deformability of the basal plane is damaged, the systolic function of the longitudinal plane is decompensated, which affects the prognosis. This finding is in agreement with those of previous animal studies,44,45 indicating that the maximum contraction was across the basal level.
Changes in Follow-Up Protocols for CTEPH Patients
Since there is currently no risk stratification for CTEPH patients, we used the risk stratification for PAH in the ESC guidelines. First, NRI significantly increased when the GLS cut-off value was added to the ESC model. Some patients were reclassified across risk categories, which may provide the basis for follow-up and treatment changes in CTEPH patients. For example, when low-risk CTEPH patients have abnormal GLS, they should be closely followed up. RHC parameters were not independent risk factors in our study and were unsuitable for follow-up, considering that RHC is an invasive method. Therefore, we recommend that a series of noninvasive methods, such as CMR combined with 6WMD, be used for routine follow-up of CTEPH patients in the future. This finding is consistent with Bruggen et al's 46 research results in PAH but must be validated using a larger sample size.
Our study has several limitations. First, this was a single-center study with a relatively small sample size. Second, we only evaluated variables at baseline to establish and validate the model and did not utilize data from follow-up visits. This was a retrospective study; therefore, the association between strain parameters and clinical outcomes in CTEPH must be confirmed in a prospective, long-term follow-up study. Third, we excluded healthy controls to compare strain parameters with those of CTEPH patients; rather, we used previously reported strain parameters for the Chinese population for comparison. Fourth, we only focused on the RV-free wall, which mainly contributes to RV contractile function, for simpler evaluation and more convenient clinical application. 47 Finally, the inconsistent follow-up period may have resulted in underestimating adverse events.
Supplemental Material
sj-jpg-1-cat-10.1177_10760296231176253 - Supplemental material for The Role of Strain by Cardiac Magnetic Resonance Imaging in Predicting the Prognosis of Patients with Chronic Thromboembolic Pulmonary Hypertension
Supplemental material, sj-jpg-1-cat-10.1177_10760296231176253 for The Role of Strain by Cardiac Magnetic Resonance Imaging in Predicting the Prognosis of Patients with Chronic Thromboembolic Pulmonary Hypertension by Xuefei Zhang, MS, Xiaojuan Guo, MD, Bowen Zhang, MS, Qi Yang,MD, Juanni Gong, MD, Suqiao Yang, MD, Jifeng Li, MD, Tuguang Kuang, MD, Ran Miao, PhD, and Yuanhua Yang, MD in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We thank all patients who voluntarily participated in this study.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors without any undue reservation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Beijing Chao-Yang Hospital (protocol code: NCT04326777).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chinese National Scientific Research Foundation (81300044).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
