Abstract
This study aimed to identify the risk factors of deep vein thrombosis (DVT) in adults with acute compartment syndrome (ACS) following lower extremity fractures. We collected data on adults with ACS following lower extremity fractures in our hospital from November 2013 to January 2021. Patients were divided into the DVT group and the non-DVT group according to whether they had DVT or not. The demographics, comorbidities, and admission laboratory examinations were computed by univariate analysis, logistic regression analysis, and receiver operating characteristic (ROC) curve analysis. In our study, the rate of DVT (26 of 110) was 23.6%. Univariate analysis showed that numerous factors were associated with the formation of DVT. Logistic regression analysis showed that patients with multiple fractures (P = .015, OR = 5.688), patients with a history of hypertension (P = .011, OR = 16.673), and patients with a higher BMI (P = .008, OR = 1.950) and FDP (P = .013, OR = 1.031) were relevant predictors of DVT. ROC curve analysis indicated 24.73 kg/m2 and 28.33 μg/mL were the cutoff values of BMI and FDP to predict the DVT, respectively. Furthermore, the combination of BMI and FDP had the highest diagnostic accuracy. Our findings identified multiple fractures, BMI, and FDP as independent risk factors for DVT in patients with ACS following lower extremity fractures and determined the cutoff values of BMI and FDP, helping us individualize the assessment of the risk of DVT to manage early targeted interventions.
Keywords
Introduction
Acute compartment syndrome (ACS) is a serious orthopedic emergency that can occur in 1-10% of patients who have suffered lower extremity fractures. 1 It is typically diagnosed based on clinical symptoms such as pain that is out of proportion to the injury, pain with passive stretching, and swelling. Delayed or incorrect treatment of ACS can lead to severe consequences, including loss of function or even death. 2 The pathophysiological mechanism of ACS is that postprocedural reperfusion causes edema, which can lead to increased pressure in the compartment and tissue necrosis. 3 ACS can result in various clinical symptoms, such as pain, swelling, blistering, or deep venous thrombosis (DVT).
One of the most common complications in hospitalized patients following lower extremity fractures is DVT, which can cause disability and fatal pulmonary embolism (PE). 4 If not prevented or treated promptly, DVT may lead to chronic pain, secondary varicose veins, ulcers, and pulmonary embolism. The long-term effects of DVT can cause significant disability, such as post-thrombotic syndrome and chronic thromboembolic pulmonary hypertension. Researchers have investigated the occurrence and predictors of DVT in patients following orthopedic surgeries, with previous studies finding an incidence of DVT ranging from 8 to 34.9%. 5 Previous studies have found that the incidence of DVT increased to 54% as the period between the injury and admission increased by 2 days. 6 A meta-analysis found that advanced age, prolonged time from injury to admission, lengthy time from injury to surgery, female patients, high-energy injuries, patients with a history of coronary heart disease, liver and kidney diseases, pulmonary disease, smoking, fibrinogen, C-reactive protein, and < 35 g/l albumins were associated with DVT in patients who were admitted.7,8 As we all know, early diagnosis of DVT is critical because determining predictors after hospital admission is beneficial to DVT prevention.
Several prior studies have reported the predictors of DVT after lower extremity fractures. Zhu et al suggested that gender, hypertension, open fracture, D-dimer > 0.5 mg/L, sodium concentration < 135 mmol/L, and alkaline phosphatase > 100 u/L were related predictors. 9 However, as far as we know, limited studies have focused on DVT after ACS following lower extremity fractures. Because ACS patients are at increased risk of serious complications, identifying risk factors for DVT in these patients is critical. We conducted a retrospective review of patients with ACS following lower extremity fractures to identify risk factors associated with the formation of DVT.
Materials and Methods
Ethics Statement
For this study, we examined the electronic medical records of all ACS adult patients treated at our hospital between November 2013 and January 2021. The institutional review board of our hospital gave their permission to this study (NCT04529330, S2020-022-1), which was based on the 1964 Declaration of Helsinki's ethical standards. Since this was a retrospective study, informed consent papers from patients were not required.
Patients
This retrospective study was carried out in the Third Hospital of Hebei Medical University, a tertiary hospital with a level I trauma center. Data from 110 patients’ electronic medical records were extracted following the inclusion and exclusion criteria. The inclusion criteria were as follows : (1) traumatic ACS patients; (2) patients with ACS with closed fractures of lower limbs; and (3) patients older than 18 years. The exclusion criteria were as follows: (1) incomplete data; (2) patients with nontraumatic ACS; and (3) patients with a history of DVT (Figure 1).

Flow diagram of included patients.
A total of 110 patients (99 men and 11 women) were enrolled in this research. Depending on whether these individuals had DVT or not, we separated them into the DVT group and the non-DVT group. DVT has been categorized into 3 types, each distinct in its respective characteristics: proximal DVT, distal DVT, and mixed type. Proximal DVT specifically refers to the development of thrombosis in or proximal to the popliteal vein, such as the iliac, superficial femoral, femoral, or popliteal veins. Conversely, distal DVT pertains to the occurrence of thrombosis that develops below the knee, which includes the tibial or peroneal veins. The mixed type, as its name implies, involves the co-existence of both proximal and distal thrombosis. In our study, all DVT occurred in the same leg as the fracture, and there were 2 patients who developed DVT in both legs. During admission and hospitalization, all patients received chemoprophylactic DVT prophylaxis. Low-molecular-weight heparin sodium (LMWH) injections were used to achieve chemotherapy prophylaxis. From hospitalization to 12 h prior to surgery and 12 h after surgery to discharge, LMWH was commonly administered (4250 IU once daily). Anticoagulation therapy (LMWH sodium, 4250 IU every 12 h) was chosen to be given to DVT patients. During hospitalization, mechanical prophylaxis was carried out intermittently by a pneumatic compression device. It is important to note that the use of mechanical prophylaxis should be carefully monitored in patients with ACS, as excessive pressure on the affected area could potentially exacerbate the condition. A total of 2 patients received mechanical prophylaxis due to delayed diagnosis caused by mild early symptoms of their ACS. Our medical staff has meticulously followed the instructions for applying mechanical prophylaxis, and they have adjusted the duration and intensity of the intervention to each patient's specific requirements. Consequently, no patient has become worse as a result of mechanical prophylaxis. Besides, all patients underwent color Doppler ultrasound for the purpose of detecting DVT in both lower limbs upon admission. Two or more ultrasound physicians made the DVT diagnosis. In our study, no patients were found to have symptoms related to PE, and no severe thrombosis was detected; therefore, no patient underwent CTPA examination or inferior vena cava filter treatment.
The demographics, comorbidities, and admission laboratory examinations of patients were reviewed in this study. The demographic data included age, gender, BMI, fracture types, ASA (American Society of Anesthesiologists), smoking, alcohol, injury mechanism (car crash injury, fall injury, crush injury, and hurt by a crashing object), time from injury to admission, emergency fasciotomy or not, number of debridements, and whether or not a dehydrating agent was used. The ASA score was divided into 2 categories: grades 1-2 and grades 3-4. Comorbidities included hypertension, diabetes, arrhythmia, coronary heart disease, cerebral infarction, and anemia. We also studied several laboratory indicators covering basophil (BAS), eosinophil (EOS), hematocrit (HCT), hemoglobin (HGB), immature (IMM), lymphocyte (LYM), mean corpuscular hemoglobin concentration (MCHC), monocyte (MON), mean platelet volume (MPV), neutrophil (NEU), platelet (PLT), red blood cell (RBC), white blood cell (WBC), activated partial thromboplastin time (APTT), antithrombin III (AT), D-dimer, fibrin degradation products (FDP), fibrinogen (FIB), thrombin time (TT), albumin (ALB), alkaline phosphatase (ALP), aspartate aminotransferase (AST), alanine transaminase (ALT), Calcium (Ca), K, Na, Mg, P, Cl, globulin (GLOB), cholinesterase (CHE), creatine kinase (CK), creatinine (CREA), direct bilirubin (DBIL), glucose (GLU), lactic dehydrogenase (LDH), triglyceride (TG), total cholesterol (TC), total carbon dioxide (TCO2), ureophil (UREA), and uric acid (UA). We gathered all these laboratory indicators at the time of admission.
Statistics
In this study, we utilized SPSS software (version 25.0, SPSS Inc., New York, USA) and considered P < .05 to be statistically significant. For continuous variables, the Shapiro-Wilk test was used to check for normality. When normality is satisfied, these variables would be expressed as mean ± SD (standard deviation) using the t-test. Still, if not, the Mann-Whitney U test was used to perform statistical analysis between these 2 groups. We compared the between-group difference using the Chi-square and Fisher's exact tests for classified variables, which were expressed as a number and its percentage. In addition, to identify the best predictors of DVT, we utilized binary logistic regression analysis to detect independent predictors of DVT in patients with ACS following lower extremity fractures.
When the Youden index (sensitivity + specificity-1) reached its maximum, we routinely used ROC (receiver operating characteristic) analysis to determine the optimal cutoff values for continuous variables, such as BMI. These indexes were classified as low versus high risk because of the cutoff value. The area under the ROC curve (AUC) was used to determine the diagnostic ability, which ranged from 0% to 100%, with more area meaning better ability.
Result
This study included 110 patients, with 99 men and 11 women participating. Among these 110 patients, 88 underwent emergency fasciotomy and 22 did not. The rate of DVT following lower extremity fractures was 23.6%, with 26 patients having DVT and 84 not having it. In addition, out of the patients observed, 5 individuals (19.2%) were diagnosed with proximal DVT, while 15 patients (57.7%) had distal DVT, and 6 patients (23.1%) were diagnosed with mixed DVT.
According to Table 1, there were significant differences between the DVT group and non-DVT group in age (P = .001), BMI (P = .02), and patients with multiple fractures (P = .011). From this table, it could be observed that the age and BMI levels were higher in the DVT group compared to the non-DVT group. In terms of multiple fractures, the DVT group had a higher proportion than the non-DVT group. Between these 2 groups, there were no significant differences in gender, fracture type, injury mechanism, time from injury to admission, emergency fasciotomy or not, time from injury to fasciotomy, number of debridements, use of a dehydrating agent or not, ASA classification, smoking, or drinking (all P > .05).
Demographic Data of Patients with and Without DVT.

Abbreviations: BMI, body mass index; ASA, American Society of Anesthesiologists.
Values are presented as the number (%) or the median (interquartile range). *P < .05, statistical significance.
Table 2 compares the data on comorbidities between the 2 groups. Patients with a history of hypertension were found to have an increased risk of DVT (P = .014). No significant differences were found between the 2 groups in terms of the presence or absence of other comorbidities, including arrhythmia, coronary heart disease, diabetes, cerebral infarction, hypoproteinemia, and anemia (all P > .05). The laboratory results between the DVT group and the non-DVT group are summarized in Table 3. The levels of FDP (P = .037) and glucose (P = .001) were significantly higher, but the levels of calcium (P = .027), ALB (P = .043), and AT (P = .001) were markedly lower in the DVT group than in the non-DVT group. However, these 2 groups had no significant differences in other laboratory results (all P > .05).
Comorbidity Data of Patients with and Without DVT.

Values are presented as the number (%) or the median (interquartile range). *P < .05, statistical significance.
Laboratory Results of Patients with and Without DVT.

Abbreviations: FDP, fibrin degradation products; ATIII, antithrombinIII; BAS, basophil; EOS, eosinophil; HCT, hematocrit; HGB, hemoglobin; IMM, immature; LYM, lymphocyte; MCHC, mean corpuscular hemoglobin concentration; MON, monocyte; MPV, mean platelet volume; NEU, neutrophil; PLT , platelet; RBC , red blood cell; WBC, white blood cell; APTT, activated partial thromboplastin time; FIB, fibrinogen; TT , thrombin time, ALB , albumin; ALP, alkaline phosphatase; AST, aspartate aminotransferase; ALT, alanine transaminase; GLOB, globulin; CHE, cholinesterase; CK, creatine kinase; CREA, creatinine; DBIL, direct bilirubin; GLU, glucose; LDH, lactic dehydrogenase; TG, triglyceride; TC, total cholesterol; TCO2, total carbon dioxide; UREA, ureophil; UA, uric acid.
Values are presented as the number (%) or the median (interquartile range). *P < .05, statistical significance.
According to logistic regression analysis, patients with multiple fractures (P = .015, OR = 5.688, 95% CI (1.406,23.013)), a history of hypertension (P = .011, OR = 16.673, 95% CI (1.927, 144.295)), and higher BMI (P = .008, OR = 1.950, 95% CI (1.195 to 3.182)) and FDP (P = .013, OR = 1.031, 95% CI (1.006 to 1.056)) were closely related to DVT in patients with ACS following lower extremity fractures. In addition, higher AT (P = .001, OR = 0.927, 95% CI (0.886 to 0.971)) represented a protective factor for the formation of DVT in these patients. (Table 4).
Binary Logistic Regression Analysis of Variables Associated with DVT.

Abbreviations: BMI, body mass index; FDP, fibrin degradation products; ATIII, antithrombin III.
*P < .05, statistical significance.
The risk factors revealed by the ROC curve analysis are presented in Figure 2, which shows that the optimal cutoff points for BMI and FDP were 24.73 kg/m2 and 28.33 μg/mL, respectively. Figure 2 also depicts the AUC for these 2 indexes, BMI alone (P = .010, AUC = 0.650, 95% CI (0.053,0.739)), FDP alone (P = .049, AUC = 0.635, 95% CI (0.538,0.725)), and BMI + FDP (P = .001, AUC = 0.684, 95% CI (0.589,0.769)). After combining different factors, the AUC increased significantly, which represented a better diagnostic value. The diagnostic value of BMI + FDP was highest when the AUC was 0.684 (Figure 3).
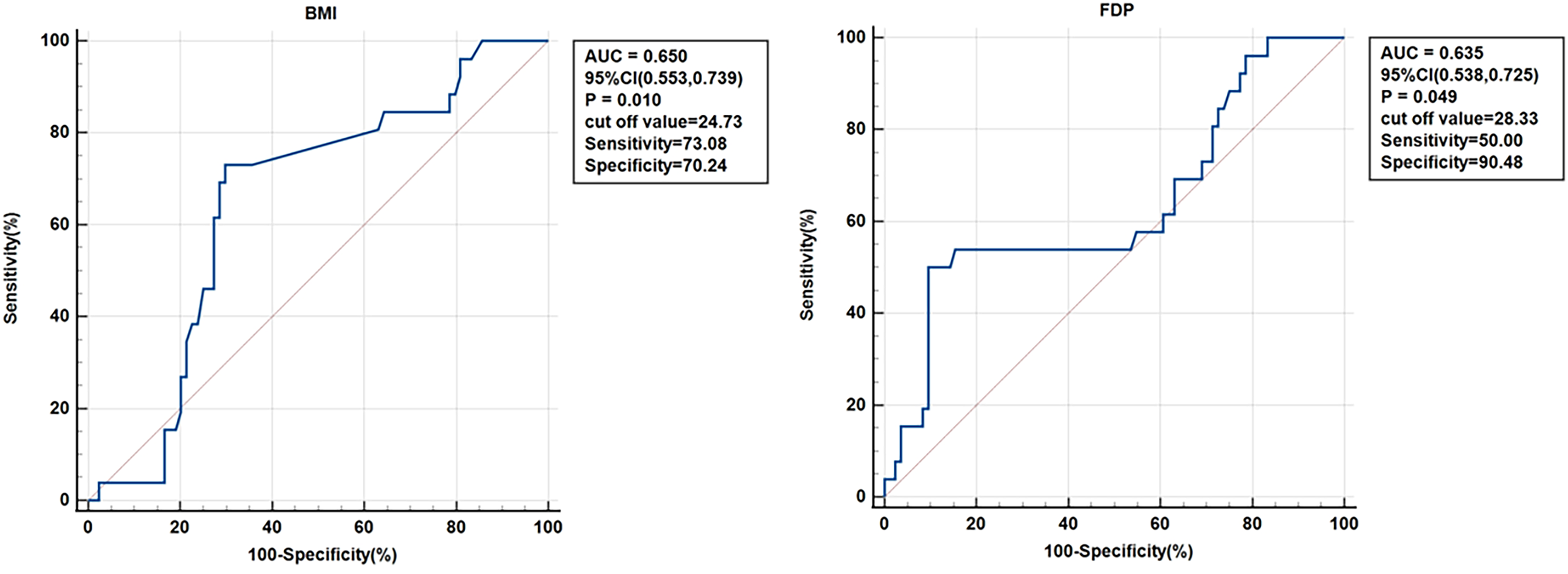
ROC curves for BMI and FDP.

ROC curve for BMI + FDP.
Discussion
ACS is a serious complication that can arise after lower extremity fractures, and it is crucial to promptly detect and treat this condition to avoid severe consequences.2,10 One effective treatment for ACS is surgical fasciotomy, but it may result in prolonged periods of immobilization, which increases the risk of DVT.11,12 DVT is one of the most common complications in hospitalized patients, especially those with lower extremity fractures, and it can cause disability and even death. 2 Researchers investigated the prevalence of DVT in patients undergoing orthopedic surgery and found that it varied from 8.9% to 34.9%, 5 which was in line with our findings. Due to DVT's high incidence and poor prognosis, we believe it is necessary to analyze its predictors and take measures to prevent its occurrence. Although ongoing research has focused on investigating DVT after lower extremity fractures, 9 less attention has been paid to its occurrence in patients with ACS following lower extremity fractures. Thus, in this study, we aimed to identify the risk factors associated with the formation of DVT in patients with ACS following lower extremity fractures.
In the present study, we retrospectively reviewed patients with ACS following lower extremity fractures and found that the rate of DVT was high (23.6%). Several predictors were identified to be associated with DVT by using univariate analysis, including age, BMI, FDP, glucose, calcium, albumin, AT, patients with multiple fractures, and patients with a history of hypertension. According to logistic regression analysis, we found that patients with multiple fractures, a history of hypertension, and higher BMI and FDP were at an increased risk of developing DVT. ROC curve analysis revealed that the cutoff values for BMI and FDP to predict DVT were 24.73 kg/m2 and 28.33 μg/mL, respectively. Furthermore, the combination of BMI and FDP had the highest diagnostic accuracy. These findings suggest that identifying risk factors for DVT is crucial in patients with ACS following lower extremity fractures to prevent its occurrence and manage its associated complications.
Our study found an increased incidence of DVT in patients with ACS after multiple fractures. We also discovered that, compared with patients with a single fracture, those with multiple fractures had a 5.688-fold increase in the risk of DVT. DVT and PE are both types of venous thromboembolism (VTE), and DVT is the preceding stage of PE. Jakub Godzik et al reported that patients with multiple fractures have a significantly increased risk of PE (OR = 1.89), which often happens when a fragment of a DVT separates and moves to the pulmonary circulation. 13 Stawicki et al reported that patients who sustained complex injuries involving both femur and pelvic fractures had a higher incidence of thrombosis. 14 Wang et al also found that patients with complex acetabular fractures had a higher incidence of DVT than patients with simple acetabular fractures. 15 All of the above studies are consistent with our results. The similarity of these studies’ research results also suggests that multiple fractures are a significant risk factor for DVT. These findings can be interpreted by the severity of the injury, which is associated with injury to or stasis of adjacent vessels because of displacement of bony fragments or increased bleeding. These results also have some clinical implications. For patients with ACS accompanied by multiple fractures, in addition to active decompression, it is also necessary to intervene in the anticoagulation of multiple fractures. In some ways, this study explained that the location of multiple fractures did not affect lower extremity thrombosis. However, it must remain highly vigilant for signs of vascular injury.
The level of BMI was significantly higher in the DVT group compared with the non-DVT group. Previous research has identified the level of BMI as a significant risk factor, which was consistent with our findings.16–18 The formation of DVT is commonly multifactorial, with many risk factors interacting with each other. A higher BMI level may act as an amplifier for other potential risk factors and may contribute to the development of DVT in a variety of high-risk situations, such as surgery. An earlier observational study found that the incidence of DVT in patients with a higher level of BMI was 2.5 times higher than that in the general population,19,20 which was different from our results to some extent (OR = 1.9). This difference could be due to previous studies not measuring relevant risk factors such as physical ability or activity, smoking habits, or other underlying conditions that need to be adequately captured. There may be several underlying causes for the association between DVT and BMI. First, DVT development may be influenced by mechanical factors such as venous stasis. A higher level of BMI could indeed affect the venous return and raise the risk of chronic venous insufficiency and DVT. 21 Besides, the altered liver metabolism in patients with a higher BMI may result in increased levels of procoagulant factors and inhibition of the fibrinolytic system. The latter is mediated by increased plasminogen activator inhibitor synthesis.22,23 Surprisingly, the trend toward increased DVT diagnosis 24 coincided with the prevalence of elevated BMI levels, 20 providing direct evidence of an association between DVT and BMI. Moreover, curbing BMI levels through public health interventions may lower the prevalence of DVT.
Our study found that FDP was an independent risk factor for DVT in patients with ACS after lower extremity fractures. Nozomi et al 25 discovered in a previous study that the level of FDP could be used to diagnose thrombotic diseases. Bai et al found that the FDP and D-dimer levels in patients with thrombosis were significantly higher than the usual ranges. 26 The above findings were consistent with our conclusions. FDP is the byproduct of plasmin's decomposition of fibrinogen and fibrin. The level of FDP can reflect the degree of fibrinolytic activity in vivo. FDP can inhibit fibrin formation, act as antithrombin, and inhibit platelet adhesion, aggregation, and release. Given the importance of FDP, we should pay much more attention to these patients’ FDP levels in our clinical work. AT is a coagulation function indicator. 27 Our findings suggested that AT was a significant protective factor in patients with ACS after lower extremity fractures, which was consistent with previous findings by He et al. 28 When the body's coagulation function is hypercoagulable, thrombin activity increases while plasma antithrombin activity decreases, resulting in the downregulation of plasma. Once plasma AT levels are reduced, platelets become activated and aggregate, making thrombosis more likely. 29
In addition to the relevant factors mentioned above, hyperglycemia and low protein levels also had an effect on the occurrence of thrombosis. In patients with lower limb fractures, Liu et al. 30 and Paul et al. 31 discovered a link between DVT and hyperglycemia, which was consistent with the results of our study. This association may be due to the fact that hyperglycemia is typically associated with chronic inflammation, which increases the risk of thrombosis. 32 Complement activation, which is implicated in platelet activation and thrombosis, is brought on by chronic inflammation. We found that the level of ALB was a risk factor for predicting DVT in lower limb fractures, which was consistent with the findings of former researchers.33–35 The potential association between DVT and decreasing ALB levels could be explained by hypofibrinogenemia caused by low protein and platelet aggregability. 36 As a result, quick and efficient treatment of low protein and hyperglycemia can play an essential role in DVT prevention.
Advanced age was frequently regarded as the risk factor for DVT. 15 In the univariate analysis of this study, the difference in age between the DVT and non-DVT groups was statistically significant, but the multivariate analysis failed to show age as a DVT predictor. This might be because of the small sample size and the fact that ACS patients were much younger than participants in earlier studies. Pini et al's findings 37 demonstrated that hypertension conferred a hypercoagulable condition by completing 3 aspects of Virchow's triad for thrombogenesis. In the univariate analysis and logistic regression of our study, there was a statistically significant difference between the 2 groups regarding whether or not the patients had hypertension. However, the AUC was less than 0.5, representing a relatively poor diagnostic capability. This may be related to the fact that our patients are relatively young and, therefore, less likely to suffer from hypertension. The D-dimer was commonly used to assist clinicians in estimating the occurrence of DVT. However, D-dimer levels did not differ statistically between the DVT and non-DVT groups in our study, which was likely due to the small number of patients with ACS after lower extremity fractures in our hospital over the 10-year period. Another reason was that D-dimer was affected by a variety of factors, including age, surgery, inflammation, hospitalization, and other acute illnesses,38–41 making it less convincing.
Although this study contains several novel findings, some limitations should be noted. First, this was a single-center study. A large sample size, multicenter, and clinical study were required. Furthermore, because this was a retrospective study, some potential variables that may be associated with the risks of DVT, such as previous surgical history, were only partially available. Third, as with any other multivariate analysis, we could not include all confounding factors, and residual confounding remains a problem.
In summary, we discovered in univariate analysis that age, BMI, calcium, glucose, FDP levels, multiple fractures, hypertension, low protein, and hyperglycemia were related to DVT and that BMI and FDP levels and multiple fractures were independent predictors of DVT after logistic regression analysis. We determined that the cutoff values for BMI and FDP in the ROC curve to predict DVT are 24.73 kg/m2 and 28.33 μg/mL, respectively. AT was also discovered to be a significant protective factor for developing DVT in these patients. Our findings provided an individualized assessment of the risk of DVT in patients with ACS following lower extremity fractures, allowing for early targeted interventions.
Footnotes
Authors’ Contributions
SY and TW were responsible for the study concept and writing the article. JFG and TW were responsible for screening the abstracts and reviewing the article. ZYH and YBL were responsible for reviewing and writing the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Before data collection, this retrospective research was approved by the Institutional Review Board of our hospital (NCT04529330, S2020-022-1). There is no need to write informed consent forms from patients because this is a retrospective study.
Funding
The research was supported by the Science and Technology Project of the Hebei Education Department (SLRC2019046); the Government-funded Clinical Medicine Outstanding Talent Training Project (2019); and the Natural Science Foundation of Hebei (H2020206193 and H2021206054). The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
