Abstract
Thromboprophylaxis is not well defined after liver transplantation (LT). The aim of this study was to evaluate the incidence of splanchnic vein thrombosis (SVT) and nonsplanchnic vein thrombosis (NSVT) after LT. Liver transplantations performed between 2009 and 2013 in our institution were reviewed. Demographic, intraoperative, and postoperative data were recorded. Low-molecular-weight heparin was only administered postoperatively if intraoperative thrombectomy was performed or in patients preoperatively anticoagulated. Of a total of 328 patients, 72% were male with a median age of 56 years, score of model for end-stage liver disease 18 (11-23), and 88% had liver cirrhosis. The incidence of postoperative venous thrombotic events was 4.6%: 8 (2.4%) patients had SVT and 7 (2.1%) patients had NSVT. After logistic regression analysis, intraoperative thrombectomy and Child A classification emerged as risk factors for SVT (odds ratio [OR]: 77, 95% confidence interval [95% CI]: 14-421) and NSVT (OR: 20, 95% CI: 3-170), respectively. The incidence of SVT in patients who undergo intraoperative thrombectomy was 33%, whereas the incidence of NSVT in patients grouped as Child A was 7.5%. Our results suggest that thromboprophylaxis should be considered after LT in patients with cirrhosis grouped as Child A and in patients who undergo intraoperative thrombectomy.
Introduction
Traditionally, perioperative bleeding complications were a major concern during liver transplantation (LT); however, the improvement in surgical techniques and critical support that led to a dramatic reduction in transfusion requirements together with the new paradigm of “rebalanced hemostasis” in patients with cirrhotic liver disease contributed to the 1,2 increasing awareness toward thrombotic complications.
Clinical and laboratory evidence suggests a role for the hemostatic system in thrombotic complications occurring during and after orthotopic LT, which involves primary (high levels of the platelet-adhesive protein von Willebrand factor with low levels of its regulator ADAMTS13) and secondary (increased potential to generate thrombin) hemostasis as well as hypofibrinolysis. 3 Moreover, an abnormal hemostatic function 1 year after orthotopic LT, attributed to endothelial cell activation, has been recently described as a potential reason for the increased risk for vascular disease seen in LT recipients. 4 These findings suggest that antihemostatic therapy in prevention or treatment of thrombotic complications after LT may be relevant.
Although the benefit of thromboprophylactic anticoagulation in postoperative general surgical patients has been well established with regard to the prevention of thrombotic complications (25% deep vein thrombosis without any prophylaxis and 7%-8% with prophylaxis) and its standard of care, 5 anticoagulation after LT has not been established, mainly because of the lack of studies of the incidence of venous thrombotic events, 6,7 along with the reasonable fear of anticoagulation in this clinical scenario. However, prophylactic anticoagulation in venous thromboembolism (VTE) reported in hospitalized patients with cirrhosis is not associated with high rates of gastrointestinal bleeding 8 –10 ; and the use of heparin in patients with cirrhosis who developed portal thrombosis does not significantly increase bleeding risk. 11,12 Therefore, it is conceivable that thromboprophylaxis in LT recipients could be improved. The aim of this study is to evaluate the incidence and the clinical factors associated with venous thrombosis after LT.
Materials and Methods
We retrospectively evaluated 360 consecutive adult LT performed at the Hospital Clinic of Barcelona between January 2009 and December 2013. We excluded 22 liver–kidney, 2 liver–heart combined transplants, 2 patients with Budd-Chiari syndrome, and patients who undergo early retransplant (<30 days after the first one) because of primary nonfunction or arterial thrombosis (n = 6). Finally, a total of 328 patients were included in the study. Prior to LT, preoperative thrombophilia screening was done in those patients with SVT and/or in those with antecedents of other thrombotic events. The model for end-stage liver disease (MELD) score was weighted for hepatocarcinoma according to Sharma and colleagues 13 (range 6-40). General anesthesia was administered as previously described, 14 blood products were transfused according to our protocol that we started from 2007. 15 Patients under coumarin drugs were preoperatively reversed with prothrombin complex concentrate (Octaplex; Octapharma, Canada) until the international normalized ratio < 2. Thromboprophylaxis with low-molecular-weight heparin (LMWH, enoxaparin) was not routinely given after LT, unless intraoperative thrombectomy was performed or the patient was under anticoagulant treatment before LT. Besides, thromboprophylaxis was avoided if the platelet count was under 30 × 109/L. If the thrombophilic state remained unsolved despite LT (ie, antiphospholipid syndrome), anticoagulation was resumed when the patient’s conditions allow to do so. If thrombosis occurred, anticoagulation was given with LMWH (enoxaparin) 1.5 mg/kg/d or heparin infusion targeting the aPTT values below 2, the upper normal limit for 72 hours, if no bleeding complications occur. Doppler ultrasound was systematically performed within the first postoperative day to assess the liver vascular permeability. Splanchnic vein thrombosis (SVT) was defined as the absence of flow in part or whole of the lumen of the portal vein trunk, portal vein branches, splenic vein, or superior mesenteric vein associated with the presence of solid material within the vein; it was defined as early or late according to its appearance before or after 30 days post LT; venous thrombotic event other than splanchnic thrombosis (nonsplanchnic vein thrombosis or NSVT) was defined as verified presence of clinical signs and symptoms of venous thrombosis combined with objective confirmation tests by Doppler ultrasound. Intraoperative thrombectomy was performed by removal of the thrombus together with the innermost layer of the vessel (thromboendovenectomy). Medical records were reviewed during hospitalization and until 6 months after surgery, death, or retransplantation, due to the later than usual appearance of postoperative thrombotic events reported in this setting. 16
Acute liver failure, familial amyloidotic polyneuropathy, and polycystic disease were grouped as no cirrhotic etiologies, and α1-antitrypsin deficiency, congenital hepatic fibrosis, and Wilson disease were grouped as other etiologies. The ethics and research committee of the Hospital Clinic of Barcelona approved the study (reg. HCB/2004/0980).
Statistical Methods
Continuous variables were expressed as median and interquartile range. The χ2 test or the Fisher exact test was used for qualitative or dichotomized variables, and the Mann-Whitney test was used for the continuous variables. A 2-tailed P value of <.05 was considered significant. Logistic regression analysis was used to find the predictive factors of postoperative venous thrombotic event. All analyses were performed with SPSS software version 18 (SPSS, Inc, Chicago, Illinois).
Results
Baseline data characteristics of the 328 LTs at the time of surgery are summarized in Table 1. Seventy-two percent of patients were men, with a median age of 56 (48-62) years, and a MELD score of 18 (11-23); hepatitis C virus (HCV; 51%) positive was the most prevalent etiology. Overall, the incidence of postoperative venous thrombotic events was 4.5% (15 of the 328 patients); 8 (2.4%) patients had SVT and 7 (2.1%) patients had NSVT. Details of thrombotic events are shown in Table 2.
Univariate Analysis of the Demographic Data and Characteristics of LT in Patients With No Postoperative Vein Thrombosis, SVT, and NSVT.a
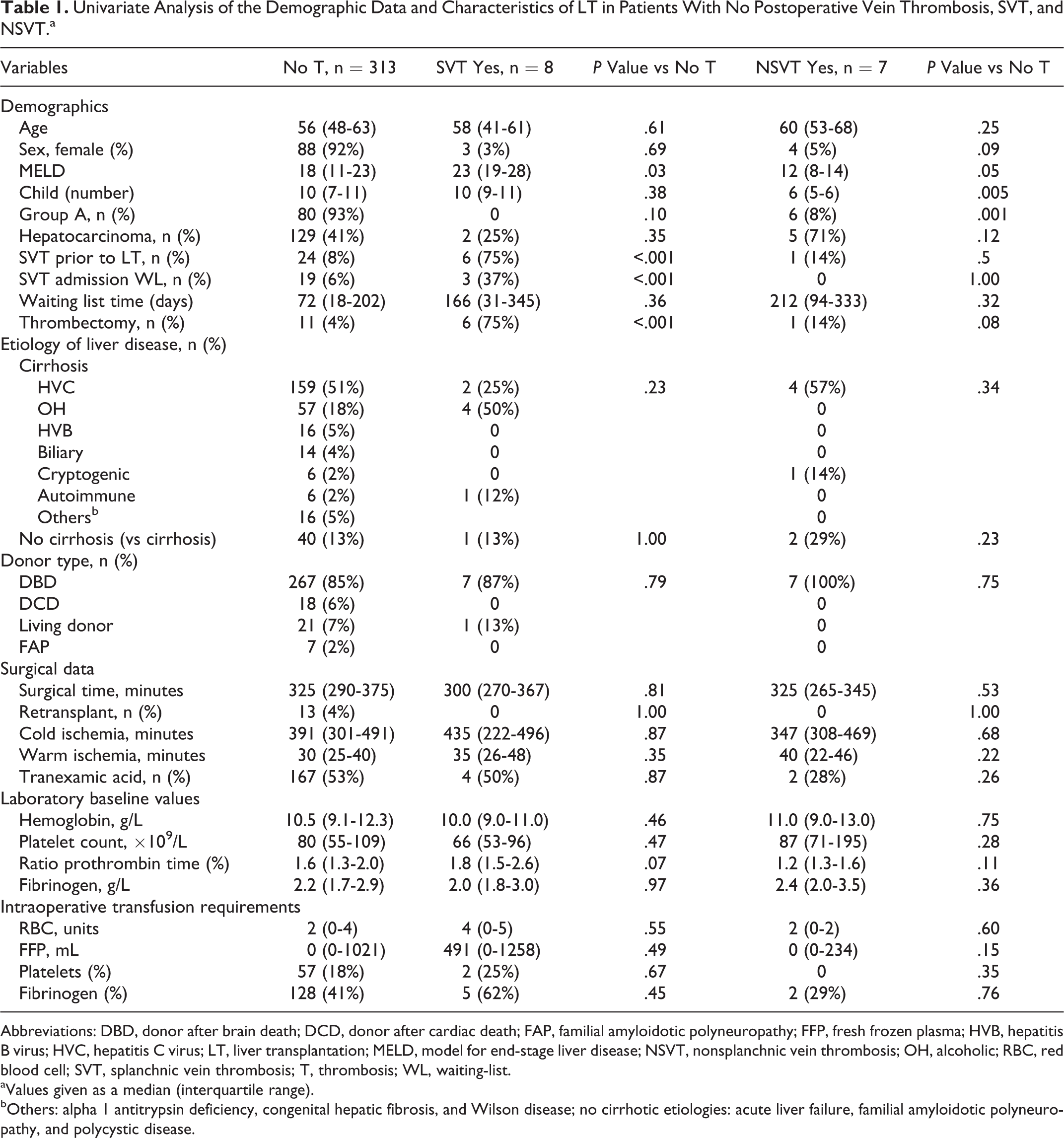
Abbreviations: DBD, donor after brain death; DCD, donor after cardiac death; FAP, familial amyloidotic polyneuropathy; FFP, fresh frozen plasma; HVB, hepatitis B virus; HVC, hepatitis C virus; LT, liver transplantation; MELD, model for end-stage liver disease; NSVT, nonsplanchnic vein thrombosis; OH, alcoholic; RBC, red blood cell; SVT, splanchnic vein thrombosis; T, thrombosis; WL, waiting-list.
aValues given as a median (interquartile range).
bOthers: alpha 1 antitrypsin deficiency, congenital hepatic fibrosis, and Wilson disease; no cirrhotic etiologies: acute liver failure, familial amyloidotic polyneuropathy, and polycystic disease.
Characteristics of All Thrombotic Events.
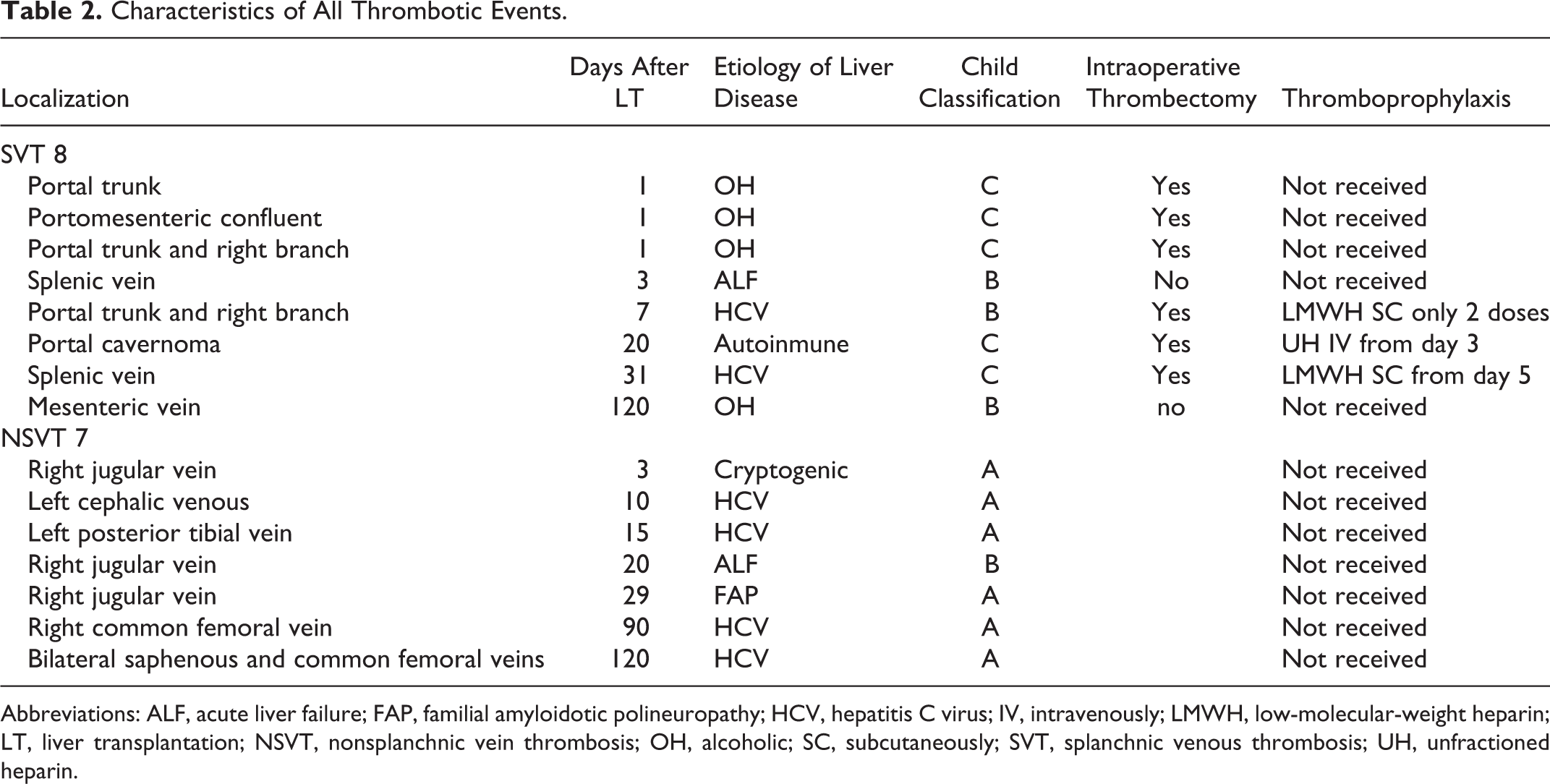
Abbreviations: ALF, acute liver failure; FAP, familial amyloidotic polineuropathy; HCV, hepatitis C virus; IV, intravenously; LMWH, low-molecular-weight heparin; LT, liver transplantation; NSVT, nonsplanchnic vein thrombosis; OH, alcoholic; SC, subcutaneously; SVT, splanchnic venous thrombosis; UH, unfractioned heparin.
Preoperative SVT involved portal vein with/without minimal extension to superior mesenteric vein. Of the 8 patients with SVT, 6 underwent intraoperative thrombectomy, in which 5 of them experienced early SVT (Figure 1); in 3 of them it happened 24 hours after LT, and in 1 patient it happened on the seventh day after receiving only 2 doses of LMWH because of severe thrombocytopenia; the remaining patient had very low portal flow intraoperatively and experienced SVT 20 days after surgery despite early postoperative heparin infusion. Thromboprophylaxis was initiated 3 (1-4) days after surgery in patients who underwent intraoperative thrombectomy and they did not developed postoperative SVT. The only patient who developed early SVT without undergoing intraoperative thrombectomy was due to multiorganic failure. Late SVT occurred in 2 patients: 1 patient who underwent intraoperative thrombectomy and received thromboprophylaxis developed SVT due to acute pancreatitis; in the other, SVT appeared without previous thrombectomy, due to chronic liver rejection. Splanchnic vein thrombosis was occlusive in 1 patient (portal trunk) who needed reintervention 24 hours after LT. The remaining could be managed medically with anticoagulation. None of the patients needed retransplantation nor was reintroduced in the waiting-list because of SVT.
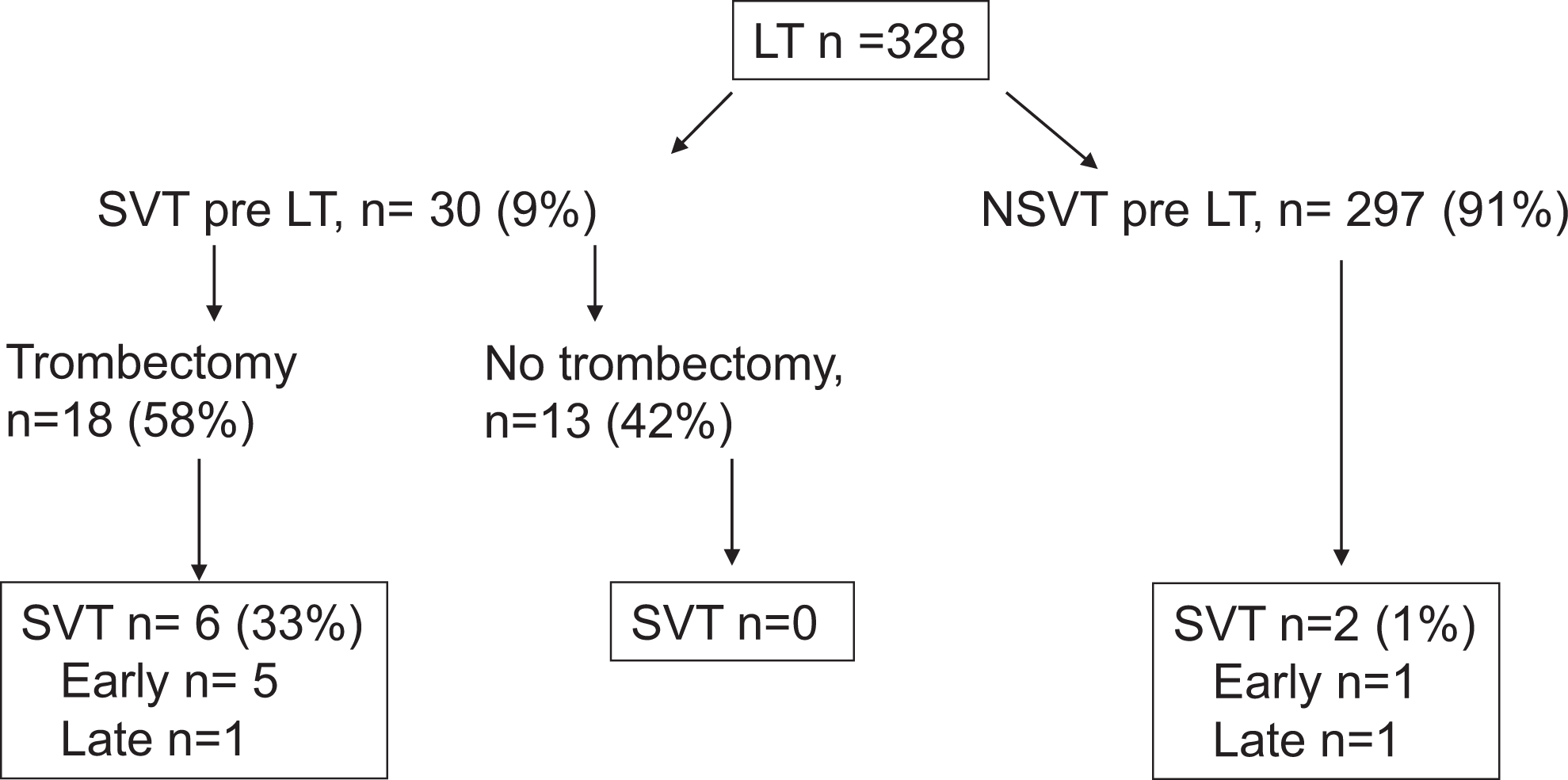
Occurrence of thrombotic events after LT. LT indicates liver transplantation; NSVT, nonsplanchnic vein thrombosis; SVT, splanchnic vein thrombosis.
Among the patients with NSVT, 4 were catheter related and 3 were related to deep venous thrombosis; the median of appearance was 90 days in the last group. All of them were placed under anticoagulation for at least 6 months after NSVT diagnosis. Notably, all patients with chronic liver disease who developed NSVT were grouped as Child A, whereas all those who developed SVT were grouped as Child B/C (Table 2). Patients with cirrhosis and noncirrhotic patients had no statistically different incidence of postoperative thrombotic events (4.2% vs 7.0%; P = .42).
Thrombophilic study was done preoperatively in 29 patients (9%), being positive in 5 of them (17%): 3 with antiphospholipid antibodies and 2 with heterozygous factor V Leiden. No postoperative thrombotic events were seen in patients with positive thrombophilic screening.
Splanchnic vein thrombosis prior to LT and intraoperative thrombectomy were associated with postoperative venous thrombotic event after LT, in the univariate analysis. Patients with intraoperative thrombectomy had an incidence of postoperative SVT of 33%, whereas it was of 0% in patients with SVT pre-LT who did not undergo thrombectomy (P < .001). Child A condition was associated with NSVT in the univariate analysis (Table 1); it occurred in 7 (5%) of Child A versus 0 (4%) of Child B/C.
In logistic regression analysis, intraoperative thrombectomy emerged as a risk factor for SVT (OR 77, 95% CI 14-421, P < .001). On the other hand, Child A emerged as a risk factor for NSVT (OR 20, 95% CI 3-170, P = .006).
Discussion
In the present series of 328 LT recipients, postoperative thrombotic venous event occurred in 15 patients (4.5%): 8 (2.4%) were SVT and 7 (2.1%) were NSVT; intraoperative thrombectomy and Child A classification were identified as a risk conditions for SVT and NSVT, respectively. The incidence of SVT in patients who undergo intraoperative thrombectomy was 33%, whereas the incidence of NSVT in patients grouped as Child A was 7.5%, which suggest that thromboprophylaxis should be considered in both groups.
The prevalence of SVT and NSVT reported in this study is in accordance with the previously reported data when postoperative pharmacologic thromboprophylaxis is not commonly administered. 16,17 Because of the fear of postoperative bleeding, thromboprophylaxis is not outright recommended after LT. However, this study evidences that a high-risk subset of patients, that is, grouped as Child A and those who undergo intraoperative thrombectomy, might have benefited from this recommendation, considering that the current guidelines of American College of Chest Physicians (which exclude cirrhotic patients) recommend thromboprophylaxis in patients at moderate risk for VTE (3%). 18 The incidence of major bleeding after LT is highly variable, the intraoperative blood loss being the main determinant of postoperative transfusion and/or surgical reintervention. Although postoperative bleeding doesn’t seem to be related with the use of thromboprophylaxis, 19 –21 and LMWH has been proven safe in patients with cirrhosis having moderate coagulation impairment, 8,9,22 it could be reasonable to delay the onset of pharmacological thromboprophylaxis until hemostasis is achieved, 23,24 particularly when the patient has significant blood loss intraoperatively.
Splanchnic vein thrombosis after LT is a relatively rare event (2%). 25 However, SVT in patients with preoperative SVT is less uncommon, ranging from 36% of pioneering experiences to 2% to 3% of some recent one 26,27 ; rethrombosis is more frequent in the early (65%) postoperative period. 17,25 The association between preoperative SVT and SVT after LT is quite variable as well as the thromboprophylaxis policy reported from different centers. 25,28
Information about the intraoperative approach of SVT is not always given; different from previous studies, 29 we look for the performance of intraoperative thrombectomy in patients with previous SVT, finding that intraoperative thrombectomy (but nor previous SVT) was actually the main risk factor for SVT after LT. Three of the 4 patients who undergo thrombectomy developed SVT 24 hours after LT, and the fourth patient developed SVT on the seventh day after LT, suggesting that the immediate postoperative period is the most risky for lapse of thrombosis: intimal damage during thrombectomy may trigger early recurrence of thrombosis. None of the aforementioned patients could receive thromboprophylaxis due to thrombocytopenia. Those patients with intraoperative thrombectomy who do not experience postoperative SVT were started on LMWH 3 days (1-5) after surgery; thromboprophylaxis could play a role in avoiding the thrombotic complication in them. Thrombectomy was performed in 2 patients who displayed late SVT: in 1 patient, SVT was developed due to acute pancreatitis, and in the other SVT was developed 120 days after LT due to liver rejection. Therefore, it seems that thrombectomy is implicated in the occurrence of early SVT post-LT, whereas other factors could be implicated in the occurrence of late SVT.
Child A classification was a risk factor for NSVT. The incidence was 7.5% versus 0.4% in patients who do not meet this condition, which suggests that this group could obtain clear benefit of pharmacological thromboprophylaxis. Thrombin generation, which is normal in platelets-free plasma from patients with cirrhosis, 1 has been shown to correlate with platelet count. 23 The relatively preserved platelet number in patients grouped as Child A could confer a superior capacity for thrombin generation, making them at highest risk of thrombotic events in relation to patients with more advanced cirrhosis. However, hepatocellular carcinoma is the most common indication of liver transplant in patients grouped as Child A (99%); the prothrombotic state inherent to this condition could be the main possible explanation for this finding. In fact, patients who develop postoperative NSVT were grouped as Child A in 86%, whereas patients with postoperative SVT were not grouped as Child A, pointing to the different physiopathology of both thrombotic events. Namely, the best hemostatic competence of patients grouped as Child A favors NSVT in them; conversely, SVT was linked to advanced stage of liver disease (Child B/C), with preoperative SVT. Different from Child classification, MELD score failed to be significant in the multivariate analysis; probably, Child score reflect a better assessment than the MELD score by combination of clinical assessment and laboratory values.
The present study has several pitfalls. Aside from its retrospective nature, patients who undergo intraoperative thrombectomy were considered to receive anticoagulation thromboprophylaxis therapy that obviously entails a bias in order to evaluate its ability to predict the appearance of SVT. Although thromboprophylaxis was initiated at third postoperative day in those patients with intraoperative thrombectomy and who did not develop SVT, we cannot assure that it was the factor that prevented the postoperative thrombotic event. However, even with this policy in place, intraoperative thrombectomy has emerged as an important risk factor for the development of SVT. A necessary balance between the risk of bleeding against the risk of developing an SVT has to be considered in these patients. To highlight that conventional coagulation tests do not the ability to assess the perioperative thrombotic risk, we don’t have any parameter pointing to the need of thromboprophylaxis other than the identification of patients at risk.
Finally, besides revealing intraoperative thrombectomy as one of the main risk factors for vein thrombosis after LT, this study evidences the difficulties to provide suitable postoperative anticoagulation policy in this patient population due to its coagulative status. It could be speculated that if thrombectomy is required, small doses (0.5 mg/kg) of unfractioned heparin given intraoperatively could cover the period of maximum risks and reduce the incidence of rethrombosis; it is not being practiced and only a clinical trial could prove this hypothesis.
In conclusion, our results reveal Child A classification and intraoperative thombectomy as independent risk factors for postoperative venous thrombotic event after LT. Early thromboprophylaxis could be crucial in order to avoid postoperative SVT when intraoperative thrombectomy was needed. However, in many cases after thrombectomy, thromboprophylaxis has to be delayed due to postoperative coagulopathy.
Footnotes
Authors’ Note
The authors want to point out that part of the patient sample reported in the present article have been also analyzed in another manuscript recently submitted to Universal Surgery of Transplant addressing the intraoperative portocaval shunt contribution to early portal vein thrombosis after liver transplantation. Annabel Blasi contributed to the analysis and interpretation of data and drafting the article. Virginia Hernandez, Javier Fernandez, Jordi Colmenero, Joan Beltran, and Juan Carlos Garcia-Valdecasas were involved with data acquisition and revising the article critically for important intellectual content. Joan Carles Reverter were involved with analysis and interpretation of data, drafting the article, and revising it critically for important intellectual content. All authors gave final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
