Abstract
Atrial fibrillation (Afib) can contribute to a significant increase in mortality and morbidity in critically ill patients. Thus, our study aims to investigate the incidence and clinical outcomes associated with the new-onset Afib in critically ill patients with COVID-19. A multicenter, retrospective cohort study includes critically ill adult patients with COVID-19 admitted to the intensive care units (ICUs) from March, 2020 to July, 2021. Patients were categorized into two groups (new-onset Afib vs control). The primary outcome was the in-hospital mortality. Other outcomes were secondary, such as mechanical ventilation (MV) duration, 30-day mortality, ICU length of stay (LOS), hospital LOS, and complications during stay. After propensity score matching (3:1 ratio), 400 patients were included in the final analysis. Patients who developed new-onset Afib had higher odds of in-hospital mortality (OR 2.76; 95% CI: 1.49-5.11, P = .001). However, there was no significant differences in the 30-day mortality. The MV duration, ICU LOS, and hospital LOS were longer in patients who developed new-onset Afib (beta coefficient 0.52; 95% CI: 0.28-0.77; P < .0001,beta coefficient 0.29; 95% CI: 0.12-0.46; P < .001, and beta coefficient 0.35; 95% CI: 0.18-0.52; P < .0001; respectively). Moreover, the control group had significantly lower odds of major bleeding, liver injury, and respiratory failure that required MV. New-onset Afib is a common complication among critically ill patients with COVID-19 that might be associated with poor clinical outcomes; further studies are needed to confirm these findings.
Keywords
Introduction
Atrial fibrillation (Afib) is a common type of arrhythmia affecting critically ill patients.1,2 Afib can contribute to a significant increase in mortality and morbidity in critically ill patients.3,4 The mortality rate of critically ill septic patients associated with new-onset Afib was reported to be 44% compared to 22% in non-Afib critically ill patients. 5 Several factors could increase the risk of developing new-onset Afib during hospitalization. During intensive care units (ICUs) admission, Afib can be triggered by receiving vasopressor agents, electrolyte disturbances, inflammatory states, infections, hemodynamic instabilities, and severe illnesses. Infections and inflammation are known to increase cardiac structural and electrical abnormalities contributing to the occurrence of Afib during critical illness. 6 A study reported that 35% of new-onset Afib occurring during hospitalization was attributed to acute infection. 7
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a highly contagious virus causing coronavirus disease 2019 (COVID-19). 8 The major complications of COVID-19 are mostly related to the respiratory system. Nonetheless, SARS-CoV-2 can trigger a systemic inflammatory response which can also lead to cardiovascular complications, ranging from vascular damage, cardiac injury, and arrhythmias to thrombosis. 9 In critically ill patients with COVID-19, the incidence of new-onset arrhythmia reached 25%. The most common type of these arrhythmias was atrial fibrillation or flutter at 95%. 10 Hospitalized critically ill patients with COVID-19 have been associated with new-onset Afib. The incidence of new-onset Afib among hospitalized patients with COVID-19 ranges from 5.4% to 15%. 11 Moreover, new-onset Afib was more frequent and associated with worse outcomes in older patients with preexisting cardiovascular complications such as hypertension and coronary artery disease.12–14
The occurrence of new-onset Afib in critically ill patients has been associated with poor clinical outcomes, including a significant increase in hospital and 90-day mortality when compared to patients with chronic Afib. 15 Several studies have identified associations between new-onset Afib and increased risk of death during sepsis and critical illness.5,15–17 On the other hand, a multicenter American registry including over 30 000 patients with COVID-19 reported no significant association between new-onset Afib and mortality. 11 However, the study included both ICU and non-ICU patients. 11 Since the included patient population and clinical outcomes varied in the aforementioned studies, the incidence and clinical outcomes of critically ill patients with COVID-19 after new-onset Afib remain unconfirmed. Therefore, our study aimed to investigate the incidence and clinical outcomes associated with new-onset Afib in critically ill patients with COVID-19 in a matched patient population.
Methods
Study Design
A multicenter cohort study that retrospectively included all critically ill adult patients admitted to the ICUs from March 1, 2020, until July 31, 2021, with confirmed COVID-19. Patients were diagnosed with COVID-19 using Reverse Transcriptase–Polymerase Chain Reaction nasopharyngeal or throat swabs. Patients were categorized into two groups based on developing Afib during ICU stay (control vs. new-onset Afib.). New-onset Afib was defined as new onset or a first detectable episode of atrial fibrillation (any type), whether symptomatic or not. All patients were followed until they were discharged from the hospital or died during the in-hospital stay. The study was approved by King Abdullah International Medical Research Center (KAIMRC) in June 2021 (Ref.# NRC21R.197.04). The IRB committee waived informed consent from the study patients due to the retrospective observational nature of the study. All methods were performed following relevant guidelines and regulations.
Study Participants
All adult patients (age ≥ 18 years) admitted to the ICUs with confirmed COVID-19 at included five centers were screened for eligibility. Patients were excluded if they had a history of venous thromboembolism or myocardial infarction (MI), was known to have chronic Afib, had an unknown medical history, if the ICU length of stay (LOS) ≤ one day, died or were labeled as “Do-Not-Resuscitate” or “No Code” within the first 24 h of ICU admission (Figure 1).

Flow diagram showing critically ill patients with COVID-19. COVID-19, coronavirus disease; ICU, intensive care unit; LOS, length of stay.
Study Setting
This multicenter study was conducted at five hospitals in Saudi Arabia: King Abdulaziz Medical City (Riyadh & Jeddah), King Abdulaziz University Hospital (Jeddah), King Abdullah bin Abdulaziz University Hospital (KAAUH) (Riyadh), and King Salman Specialist Hospital (Hail). The primary center for this study was King Abdulaziz Medical City (Riyadh), an academic tertiary care center. 29
Data Collection
Each participant's data were collected and controlled using KAIMRC's Research Electronic Data Capture (REDCap®) software. We collected patients’ demographic data, comorbidities, vital signs and laboratory tests, baseline severity scores (ie, Acute Physiology and Chronic Health Evaluation II (APACHE II), Sequential Organ Failure Assessment (SOFA), Nutrition Risk in Critically ill (NUTRIC), and Multiple Organ Dysfunction Scores [MODS]), Glasgow Coma Score (GCS), acute kidney injury (AKI) status, use of prone positioning, receipt of mechanical ventilation (MV), MV parameters (eg, PaO2/FiO2 ratio, FiO2 requirement) within 24 h of ICU admission. Moreover, renal profile, liver function tests, coagulation profile (ie, INR, aPTT, fibrinogen, D-dimer), and inflammatory markers were collected within 24 h of ICU admission. Lastly, new-onset atrial fibrillation, and the use of tocilizumab and corticosteroids therapies during ICU stay were assessed and recorded for all eligible patients.
Outcomes
The primary outcome was in-hospital mortality. The secondary outcomes were all thrombosis cases, bleeding, 30-day mortality, hospital LOS, ICU LOS, ventilator-free days (VFDs) at 30 days, multiple organ dysfunction (MOD) score onday #3, liver injury, AKI, and respiratory failure requiring MV.
Outcome Definition(s)
In-hospital mortality was defined as the percentage of patients with COVID-19 who died in the hospital.
All thrombosis cases were defined and identified using the International Statistical Classification of Diseases (ICD)10-CM code, chart review, and radiology finding (ie, MI, ischemic stroke, pulmonary embolism, deep vein thrombosis). 23
Major bleeding was defined according to the International Society on Thrombosis and Hemostasis (ISTH) definition. Any bleeding not fulfilling the criteria of major or clinically significant bleeding was identified as a minor bleed. 24
The 30-day mortality was defined as a death occurring for any cause within 30 days of the admission date during hospital stay; patients who were discharged from the hospital alive before 30 days were presumed to be survivors.
VFDs at 30 days were calculated as the following: if the patients die within 30 days of MV, then the VFDs = 0, VFDs = 30 - days after MV initiation, if patient survived and was successfully liberated from MV, and VFDs = 0 if the patient is on MV for >30 days.
Multiple Organ Dysfunction Score is used to measure the severity of the multiple organ dysfunction syndromes in six organ systems: (1) the respiratory system (PO2/FIO2 ratio); (2) the renal system (serum creatinine concentration); (3) the hepatic system (serum bilirubin concentration); (4) the hematologic system (platelet count); (5) the central nervous system (Glasgow Coma Scale), and (6) the pressure-adjusted heart rate (heart rate multiplied with the ratio of central venous pressure to mean arterial pressure). 25
AKI was defined as a sudden decrease of renal function within 48 h, defined by an increase in absolute serum creatinine of at least 26.5 μmol/L (0.3 mg/dL) or by a percentage increase in serum creatinine ≥ 50% (1.5× baseline value) during ICU stay. 26
Acute liver injury was defined as alanine aminotransferase (ALT) exceeding 3 times the upper limit of normal or double in patients with elevated baseline ALT during the ICU stay. 27
Respiratory failure was defined as either hypoxemic respiratory failure (PaO2 < 60 mm Hg with low or normal PaCO2) or hypercapnic respiratory failure (PaCO2 > 50 mm Hg) that requires MV. 28
Statistical Analysis
Continuous variables were presented as mean with standard deviation (SD), or median with lower and upper quartile (Q1, Q3), and categorical variables as a number with a percentage as appropriate. All continuous variables’ normality assumptions were assessed using a statistical test (ie, Shapiro-Wilk test) and graphical representation (ie, histograms and Q-Q plots).
Baseline characteristics and outcome variables were compared between the twostudy groups. Chi-square or Fisher exact test was used for categorical variables. Normally distributed continuous variables were compared using the Student t test and Mann-Whitney U test for other non-normally distributed variables. Multivariable regression analysis and negative binomial regression were used for the outcomes considered in this study as appropriate. Regression analysis was done by considering the PS score as one of the covariates in the model. Model fit was assessed using the Hosmer-Lemeshow goodness-of-fit test. The odds ratios (OR) or estimates with 95% confidence intervals (CIs) were reported as appropriate. No imputation was made for missing data as the cohort of patients in our study was not derived from random selection.
Patients were matched using the Proc PS match procedure (SAS) with a ratio of 3:1 based on the patient's age, gender, chronic kidney disease, APACHE II score, MV status, AKI, and early use of dexamethasone within 24 h of ICU admission. A greedy nearest neighbor matching method was utilized, in which one patient developed a new-onset Afib during ICU stay (active group) paired with three patients who did not (control), which eventually produced the smallest within-pair difference among all available pairs with treated patients. The difference in the logits of the propensity scores for pairs of patients from the two groups was matched if it was less than or equal to 0.1 times the pooled estimate of the SD. We considered a P value of <.05 statistically significant and used SAS version 9.4 for all statistical analyses.
Results
A total of 1598 critically ill patients were screened; 1256 were included in our analysis based on the eligibility criteria. Among them, 135 (10.7%) patients developed new-onset Afib during their ICU stay. After propensity score (PS) matching (3:1 ratio), 400 patients were included in PS analysis based on the predefined criteria.
Demographic and Clinical Characteristics
Prior to the propensity score matching, most of the patients in both arms were male (62.3%), with a mean age of 61 years (SD 14.89 years). The most common underlying comorbidities were diabetes mellitus (59.4%), followed by hypertension (55.5%), and dyslipidemia (20.5%). Patients who developed new-onset Afib were more likely to need MV and inotropes/vasopressors support within 24 h of ICU admission than those who did not. Furthermore, patients with new-onset Afib had higher APACHE II score and AKI within 24 h of ICU admission. Also, received more anticoagulation treatment doses and aspirin compared to those who did not develop new-onset Afib. On the other hand, patients who did not develop new-onset Afib had higher CRP and CPK levels at baseline compared to those who developed new-onset Afib (Additional file 1). Following the PS matching, most of the differences in the baseline and demographic characteristics were similar between the two groups, as presented in Table 1.
Baseline Characteristics of Patients Admitted to the ICU.

Wilcoxon rank sum test is used to calculate P value.
Chi-square test.
Fisher exact test is used to calculate P value.
T test.
30-Day and In-Hospital Mortality
After propensity score matching, the 30-day mortality was insignificantly higher in those who developed new-onset Afib compared to those who did not (OR 1.55; 95% CI: 0.91-2.63; P = .10). On the other hand, patients who developed new-onset Afib had significantly higher odds of in-hospital mortality compared to the control group (OR 2.76; 95% CI: 1.49-5.11, P = .001) (Table 2).
The Outcomes of Critically Ill Patients With COVID-19 After Propensity Score Matching.
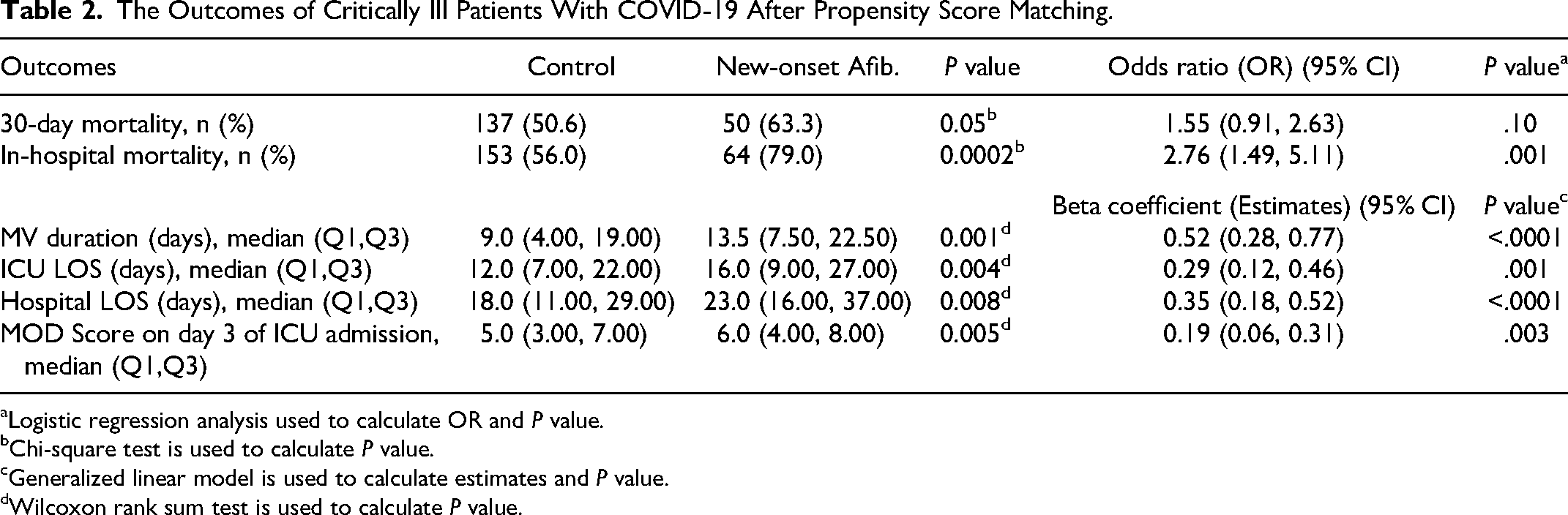
Logistic regression analysis used to calculate OR and P value.
Chi-square test is used to calculate P value.
Generalized linear model is used to calculate estimates and P value.
Wilcoxon rank sum test is used to calculate P value.
MV Duration and LOS
During ICU stay, the MV duration was longer in patients who developed new-onset Afib with a median duration of 13.5 days (7.5-22.5) compared to 9.0 days (4.0-19.0) in the control group (beta coefficient 0.52; 95% CI: 0.28-0.77; P < .0001). Moreover, both ICU and hospital LOS were significantly longer in patients who developed new-onset Afib during ICU than the control group (beta coefficient 0.29; 95% CI: 0.12-0.46; P < .001), and (beta coefficient 0.35; 95% CI: 0.18-0.52; P < .0001), respectively (Table 2).
Complications During ICU Stay
The major bleeding events were significantly higher in patients who developed new-onset Afib than those who did not (14.4% vs. 4.2%). Moreover, the logistic regression analysis results demonstrated a higher odds of major bleeding in patients who developed new-onset Afib by close to 4-fold (OR 3.74; 95% CI: 1.66-8.44; P = .002). While the odds of minor bleeding were not statistically significant across the two groups (OR 1.34; 95% CI: 0.58-3.11; P = .49) (Table 3). However, the use of anticoagulation treatment, as well as aspirin during ICU stay, was higher in patients who developed new-onset Afib (Table 1).
Complications During ICU Stay.

Logistic regression is used to calculate the OR and P value.
Denominator of the percentage is the non-MV patients.
Denominator of the percentage is the total number of patients.
Chi-square test is used to calculate P value.
Patients who did not develop new-onset Afib had significantly lower MODS on day 3 of ICU stay, with a median score of 5.0 (3.0-7.0) compared to 6.0 (4.0-8.0) days for patients who developed new-onset Afib (beta coefficient of 0.19; 95% CI: 0.06-0.31; P = .003) (Table 2). Among the non-mechanically ventilated patients within 24 h of ICU admission, a respiratory failure that required MV during ICU stay was higher in patients who developed new-onset Afib (OR 3.02; 95% CI: 1.08-8.46; P = .04). In addition, liver injury occurred more frequently in patients who developed new-onset Afib (OR 2.42; 95% CI: 1.31-4.49; P = .005). In terms of AKI and all thrombosis cases, there were no statistically significant differences between the two groups (Table 3).
Discussion
In this multicenter, retrospective propensity score-matched study, we evaluated the incidence and clinical outcomes associated with the new-onset Afib in critically ill patients with COVID-19. In this cohort, we found that the incidence of new-onset Afib was 10.7%. In comparison to the control group, patients who developed new-onset Afib had worse outcomes and significantly higher in-hospital mortality. In addition, this study found a significant increase in the duration of MV, hospital, and ICU LOS in the new-onset Afib group. Further, the occurrence of new-onset Afib in critically ill patients with COVID-19 during hospitalization was associated with less favorable outcomes in terms of MODS, major bleeding, liver injury, and hospital-acquired infection.
The overall incidence of new-onset Afib in our cohorts was within the range that was reported in previous studies, with a higher incidence in critically ill patients (5%-14%).11,14,18–20 Different risk factors have been identified in previous studies as potential causes for developing new-onset Afib in COVID-19 patients, which are mostly consistent with our findings.11,14,19
In the present study, we found that patients with new-onset Afib were older, had a higher APACHE II and MODS, developed AKI, and required more MV and inotropes/vasopressors within 24 hours of ICU admission. In addition, new-onset Afib was more frequent in patients with a history of hypertension, chronic kidney disease, and dyslipidemia. However, the history of heart failure, cancer, and chronic obstructive pulmonary disease (COPD) was not detected as risk factors in our study.
The pathophysiological mechanism of Afib development in COVID-19 patients remains not well understood. However, several proposed mechanisms have been suggested, including systemic inflammatory response and cytokine storm, direct viral damage to the myocardial cell, hypoxemia, increase in catecholamines, electrolytes and acid-base disturbances.18,21 Moreover, preexisting cardiovascular diseases may potentiate the arrhythmogenesis risk in COVID-19 patients. 20
Our study confirms the findings from previous studies that showed an association between the new-onset Afib and worse clinical outcomes in COVID-19 patients.11,12,14,19–21 In the present study, there was a significant increase in hospital mortality and respiratory failure requiring MV in patients with a new-onset Afib compared to patients without a new-onset Afib. These findings are comparable to results from a retrospective study that reported a higher rate of in-hospital death (53.3% vs 13.4%, P ≤ .001) and an increase in the requirement for invasive ventilation (56.7% vs 13.9%, P ≤ .001) in critically ill COVID-19 patients with Afib. 12 Ergün et al also found a significant increase in hospital mortality in patients with COVID-19 who developed new-onset Afib during their ICU stay (87% vs 67%, P = .019). 14 The results from Rosenblatt et al study suggest that new-onset Afib among patients with COVID-19 is considered a marker of disease severity rather than an independent risk factor of mortality. 11
Patients with new-onset Afib experienced worse in-hospital outcomes than COVID-19 patients with preexisting Afib, including mortality, acute respiratory distress syndrome (ARDS), sepsis, multiple organ dysfunction, hemorrhage, and stroke. 22 These findings might add to the hypothesis that hemodynamic instability driven on by cardiac damage and systemic inflammation results in the development of new-onset Afib in COVID-19 patients.Since the non-Afib group's CRP level was much higher and troponin level, a direct indicator of myocardial damage, was not measured in this study, we were unable to confirm this hypothesis with our data.
An increase in the incidence of embolic events, thrombosis, and bleeding was previously reported in COVID-19 patients with new-onset Afib.11,19,22 However, our study did not find a significant increase in thrombosis or infarction in the new-onset Afib group compared to the control group. Further, the bleeding events were more frequently reported in the new-onset Afib group. These variations in our results from previous studies with regard to the incidence of thrombosis might be attributed to the higher use of therapeutic anticoagulation and aspirin in the new-onset Afib group in our cohort.
Although this study is one of the largest that evaluated the consequences of new-onset Afib in critically ill patients with COVID-19, several limitations have been observed. First, the retrospective design of this study makes it prone to documentation bias and residual confounding risk despite the use of propensity score matching. Second, data about cardiac function biomarkers such as troponin and B-type natriuretic peptide were lacking. Therefore, we could not assess the level of cardiac damage in our population. Lastly, this study did not assess long-term complications that could happen after hospital discharge.
Conclusion
New-onset Afib is a common complication in critically ill patients with COVID-19 that may be associated with poor clinical outcomes and higher in-hospital mortality. To validate our findings, further studies are warranted to asses the long-term clinical outcomes.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231156178 - Supplemental material for Incidence and Clinical Outcomes of New-Onset Atrial Fibrillation in Critically Ill Patients with COVID-19: A Multicenter Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296231156178 for Incidence and Clinical Outcomes of New-Onset Atrial Fibrillation in Critically Ill Patients with COVID-19: A Multicenter Cohort Study by Raed Kensara, Ohoud Aljuhani, Ghazwa B Korayem, Hadeel Alkofide, Sumaya N Almohareb, Yousef S Alosaimi and Ali F Altebainawi, Khalid Bin saleh, Norah Al Andas, Shmeylan Al Harbi, Abdullah F Al Harthi, Uhood Ashkan, Rema Alghamdi, Hisham A Badreldin, Awatif Hafiz, Mashael AlFaifi, Rahaf A Alqahtani, Ramesh Vishwakarma, Abeer A Alenazi, Mai Alalawi, Reem mahboob, Renad A Alfouzan, Layan B Al Tuhayni, Nouf Al Qahtani, Khalid Al Sulaiman in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
We want to thank Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R78), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. The authors also would like to thank all the investigators who participated in this project from the Saudi critical care pharmacy research (SCAPE) platform.
Authors’ Note
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request. The study was approved in June 2021 by King Abdullah International Medical Research Center Institutional Review Board, Riyadh, Saudi Arabia (Ref.# NRC21R.197.04). King Abdullah International Medical Research Center Institutional Review Board waived the informed consent due to the retrospective nature of the study. Participants’ confidentiality was strictly observed throughout the study by using anonymous unique serial numbers for each subject and restricting data only to the investigators. All methods were performed following relevant guidelines and regulations.
Authors’ Contribution
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas; they took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R78), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
