Abstract
Background:
Several reports linked the use of repurposed drugs such as hydroxychloroquine (HCQ), azithromycin, lopinavir/ritonavir, and favipiravir with QT interval prolongation in patients with SARS-CoV2 infection. Little is known about the risk factors for QT interval prolongation in this population. We sought to describe the prevalence and identify the main risk factors associated with clinically significant corrected QT (QTc) prolongation in this population.
Methods:
We conducted a retrospective analysis of critically ill patients who were admitted to our intensive care unit (ICU), had at least one electrocardiogram performed during their ICU stay, and tested positive for SARs-CoV-2. Clinically significant QTc interval prolongation was defined as QTc >500 milliseconds (ms).
Results:
Out of the 111 critically ill patients with SARS-CoV-2 infection, QTc was significantly prolonged in 47 cases (42.3%). Patients with a clinically significant QTc prolongation had significantly higher proportions of history of cardiac diseases/surgery (22 [46.8%] vs. 10 [15.6%],
Conclusion:
The prevalence of clinically significant QTc prolongation in critically ill patients with SARS-CoV-2 infection was high and independent of drugs used. Larger prospective observational studies are warranted to elucidate independent risk factors associated with clinically significant QTc prolongation in this study population.
Introduction
The SARS-CoV-2 pandemic has significantly impacted many countries around with an influx of critically ill patients secondary to acute respiratory distress syndrome. 1 -4 Data has shown that elderly patients and those with underlying comorbid conditions such as diabetes mellitus, hypertension, and cardiovascular disease are more prone to develop severe SARS-CoV-2 infection. 1 -4 Several studies have linked the use of certain repurposed drugs such as hydroxychloroquine (HCQ), azithromycin, lopinavir/ritonavir, and favipiravir with QT interval prolongation in this population. 5 -10
QT interval prolongation is a disorder of myocardial repolarization identified by a prolonged QT interval on the electrocardiogram (ECG). This syndrome is associated with an increased risk of the polymorphic ventricular tachycardia torsades de pointes (TdP) which is a life-threatening condition. 6,11 QT interval prolongation is also associated with palpitations, syncope, seizures, and sudden cardiac death. These clinical manifestations may be difficult to detect in critically ill patients who may be mechanically ventilated with pharmacological-induced sedation and neuromuscular blockade. 6,11,12
Corrected QT (QTc) prolongation can be either congenital or acquired. Congenital long QT syndrome is an inherited cardiac condition with a predisposition for abnormal myocardial repolarization. 11,13 Conversely, acquired QT prolongation usually results from drug therapy. Electrolyte derangements, including hypokalemia, hypomagnesemia, and hypocalcemia. Also, bradycardia can increase the risk of drug-induced QT interval prolongation. Other risk factors such as impaired kidney and/or liver function, underlying heart failure, myocardial infarction, left ventricular hypertrophy, and advanced age can also contribute. 11,13 Several pharmacologic agents are associated with QT interval prolongation such as fluoroquinolones, macrolides, hydroxychloroquine, favipiravir, and azole antifungals in addition to antiemetics such as metoclopramide and ondansetron. Many of these agents carry the United States FDA boxed warnings for QTc prolongation and require close monitoring when used. 11,13
Experts suggest that QTc values of >450 milliseconds (ms) in adult males, and >460 ms in adult females are considered prolonged. 11 -15 Clinically significant QTc interval prolongation >500 ms increases the risk of life-threatening arrhythmias. 11 -15 We observed several critically ill patients with SARS-CoV-2 infection with significant QTc prolongation. The primary objective was to describe the prevalence of clinically significant QTc interval prolongation in critically ill patients with the SARS-CoV-2 infection. The secondary objective of the study was to identify the independent risk factors associated with clinically significant QTc interval prolongation in these patients.
Methods
This was a retrospective observational study conducted at our quaternary care hospital between March 1, 2020, and June 30, 2020. Data collection began after approval from the Research Ethics Committee. Patient demographics and baseline characteristics were collected. Pertinent data on use of medications potentially associated with QTc prolongation was collected. Our primary objective was to examine the prevalence of clinically significant QTc prolongation in patients with a positive SARS-CoV-2 infection. Our secondary objective was to identify the independent risk factors associated with clinically significant QTc prolongation in these patients.
Patient Selection
Patients were included if they were adults (≥18 years of age), admitted to our intensive care unit (ICU) and tested positive for SARS-CoV-2 by the polymerase chain reaction method to detect the virus using a nasopharyngeal swab. Each patient had at least one ECG performed during the hospital admission. The longest QTc ECG measurement during each patient’s hospital admission was included in the analysis. All associated laboratory findings (either on the same day of ECG or within 24 hours prior to the ECG reading) and all potential drugs (prior to the ECG reading) were included in this analysis. Due to the prolonged half-life of HCQ (40 days), we included patients who received this medication at any time during the same admission prior to the QTc measurement. All data were collected from the electronic medical records system.
Definitions of Variables
QTc prolongation was defined as ECG with QTc >450/460 ms in males and females, respectively. Additionally, our primary analysis was conducted for patients who had clinically significant QTc prolongation defined as QTc >500 ms. All computer-generated QT readings were further measured manually by a cardiac electrophysiologist who calculated QTc intervals using the Bazett’s formula.
16
In case of any significant discrepancy in the measurements, the physician reading was used for analysis. There was a substantial agreement between the cardiac electrophysiologist reading and the machine reading (kappa-statistic: 0.69 ± 0.09,
Data Analysis
Normality of data distribution was assessed using the Shapiro-Wilk test and by visual inspection of each variable’s distribution (histogram). Data are expressed as mean ± SD when normally distributed or as median (IQR) when non-normally distributed. Proportions were used as descriptive statistics for categorical variables. Comparisons of values between independent groups were performed by the two-tailed Student’s
Independent risk factors of clinically significant QTc prolongation were assessed using multivariable logistic regression analysis. All variables with a
A two-side
Results
From March 1 to May 29, 2020, 111 adult patients who were admitted to the ICU with acute respiratory failure caused by SARS-CoV-2 infection and had an ECG performed during the study period were included in the analysis. The QTc interval was prolonged in 86 patients (77.5%) with QTc >450/460 (males/females, respectively). Clinically significant QTc prolongation (QTc > 500 ms) was observed in 47 patients (42.3%).
Table 1 represents the demographics and baseline characteristics of patients with clinically significant QTc prolongation compared to those with no significant QTc prolongation. Patients with clinically significant QTc prolongation had higher rates of cardiac history (22 [46.8%] vs. 10 [15.6%],
Comparisons of Demographics and Baseline Characteristics Between QTc ≤500 ms and QTc >500 ms Groups.a

Abbreviations: AKI, acute kidney injury; BMI, body mass index; CHF, congestive heart failure; CKD, chronic kidney disease; ECG, electrocardiogram; EP, electrophysiologist; HR, heart rate; ms, milliseconds; RRT, renal replacement therapies (IHD and CVVH).
a Data are shown as mean ± SD, median (1st-3rd), and count (%).
b Quetiapine, haloperidol, fluoroquinolones, ondansetron, amiodarone, or metoclopramide.
In the multivariable logistic regression analysis after including heart rate, sex, body mass index, age, comorbidities (including any cardiac disease), renal function, and electrolytes, only a history of cardiac disease was the only risk factor independently associated with clinically significant QTc prolongation (Table 2).
Multivariate Logistic Regression Analysis (QTc > 500 ms).a
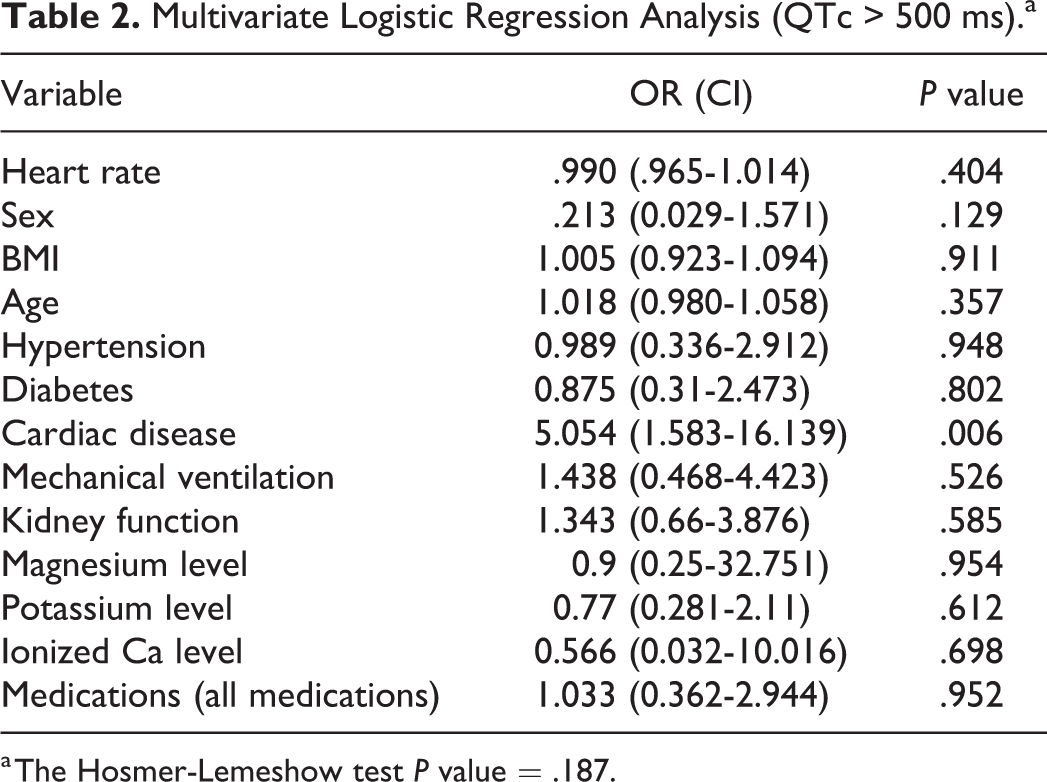
a The Hosmer-Lemeshow test
Additional analyses were performed on patients who had QTc prolongation >450/460 ms (Table 1S, supplementary file). No risk factors were found to be independently associated with prolonged QTc (>450/460 ms) in the multivariable logistic regression analysis (Table 2S, supplementary file).
Discussion
Our study’s findings demonstrated a relatively high prevalence (42.3%) of clinically significant QTc interval prolongation in our sample population. A history of cardiac disease was the sole risk factor that was independently associated with clinically significant prolonged QTc intervals.
More than 200 medications are known to cause QTc prolongation. The incidence of QTc prolongation in critically ill patients can reach 0.07% over 2 months period.11 Moreover, the incidence of cardiac arrest caused by TdP can reach up to 6%. 11
Hydroxychloroquine and azithromycin were widely used following the onset of the pandemic to treat patients with the SARS-CoV-2 infection. These medications are known to prolong the QTc interval and have been associated with TdP. 5 -10 The incidence of QTc interval prolongation associated with HCQ use was reported as approximately 10% with an increase following the addition of azithromycin to the treatment regimen. 11 Similarly, lopinavir/ritonavir and favipiravir were reported to prolong the QTc interval when combined with HCQ. 6 -10 A retrospective cohort study of 90 patients with the SARS-CoV-2 infection who received HCQ for at least one day, or HCQ with azithromycin, demonstrated significant QTc interval prolongation compared to baseline (19% of patients receiving HCQ and 21% of patients receiving HCQ and azithromycin).5 Similarly, Chorin et al retrospectively followed the ECG measurements of 251 patients diagnosed with SARS-CoV-2 infection and treated with HCQ and azithromycin and observed clinically significant QTc prolongation >500 ms in 23% of the patients. 6 Additionally, previous separate studies have corroborated the association of the medication combination of HCQ and azithromycin with QTc interval prolongation. 7,9,17 Further, Russo et al retrospectively investigated the effect of the triple combination of HCQ, azithromycin, and lopinavir/ritonavir on QTc interval prolongation in 87 patients infected with SARS-CoV-2 infection. The authors found that 23% of patients showed clinically significant QTc prolongation and concluded that close monitoring of patients receiving those medications is necessary. 9 Additionally, Çap et al retrospectively compared the QTc intervals in 189 patients with the SARS-CoV-2 infection who received HCQ, HCQ plus favipiravir, or favipiravir alone. 10 The authors found that HCQ, but not favipiravir, was independently associated with QTc interval duration in the multivariable linear regression analysis. Finally, Changal and colleagues, in a retrospective observational study, observed QT interval prolongation in 69 out of 279 hospitalized patients with SARS-CoV-2 infection. The authors determined that underlying cardiovascular disease, elderly, CKD, and drugs such as hydroxychloroquine/azithromycin could have been associated with QT prolongation. 17
Our study demonstrated a higher rate of clinically significant QTc prolongation in our sample compared to previous studies. 5 -10,17 The discrepancy may be explained by differences in the populations studied. Indeed, we focused on patients with life-threatening SARS-CoV-2 disease with acute respiratory failure requiring ICU admission while previous reports included patients with less severe manifestations. We did not find an independent association between the different medications (HCQ, favipiravir, azithromycin, and lopinavir/ritonavir) and clinically significant QTc prolongation in our population. It is noteworthy that, unlike our analyses, the investigators of the aforementioned studies 5 -10,17 did not compare patients with QTc prolongation with those who did not to ascertain whether the interval prolongation was associated with medication administration or the underlying disease state.
Several risk factors may contribute to QTc prolongation such as female sex; advanced age; acute myocardial infarction; electrolytes imbalance; heart failure; concomitant administration of QTc prolonging medications; and inadequate drug dose adjustments in patients with compromised renal or hepatic function. 11 -15 Additionally, genetic predisposition comprises around 30% of patients who develop drug-induced QTc prolongation. 11,15 Our findings showed no association of any of the previously mentioned factors with clinically significant QTc prolongation (Table 2) with the exception of a history of cardiac disease. This suggests a possible correlation between direct effects of the SARS-CoV-2 virus and QTc interval prolongation in patients with underlying cardiac disease. This suggestion is in compliance with the findings of Brauninger and colleagues where the investigators advocated that the viral infection can destruct the cardiomyocytes. 18,19 Future studies are warranted to investigate a potential pathophysiological mechanism of QTc interval prolongation in patients infected by SARS-CoV-2.
Our study has some limitations. This was a single-center retrospective study at a quaternary care facility in the Middle East region. Also, the majority of our patients was male (88.3%). This might be explained by the high proportion of male in the UAE population, which is around 72%. Thus, our findings may not be generalizable to other facilities and populations. Despite multivariable analysis, we cannot rule out selection bias. Our analysis did not include the occurrence of any documented cardiac arrhythmias in those who had significantly prolonged QTc. However, the in-hospital mortality did not differ between patients who had clinically significant QTc prolongation and those who did not. Also, we can be sure that symptomatic arrhythmias did not occur as these arrhythmias would have been documented and would not have been missed. There was a lack in information regarding patient’s genetic history with respect to congenital long QT syndrome in our sample. Finally, we do not have the pre-SARS-CoV-2 incidence of QTc prolongation at our institution.
Conclusion
Critically ill patients with SARS-CoV-2 infection can exhibit high prevalence of clinically significant QTc prolongation irrespective of medications used. QTc prolongation in this population should be closely monitored. Larger prospective observational studies are required to find the independent factors associated with QTc prolongation in this population and also in vaccinated individuals.
Supplemental Material
Supplemental Material, sj-docx-1-cpt-10.1177_10742484211069479 - QT Prolongation in Critically Ill Patients With SARS-CoV-2 Infection
Supplemental Material, sj-docx-1-cpt-10.1177_10742484211069479 for QT Prolongation in Critically Ill Patients With SARS-CoV-2 Infection by Wasim S. El Nekidy, Khalid Almuti, Hazem ElRefaei, Bassam Atallah, Lana M. Mohammad, Wael AlMahmeed, Mohamed Badr, Khaled Abdallah, Fadi Hamed and Jihad Mallat in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
This work was approved by the institutional Research Ethics Committee.
Author Contributions
All authors have contributed significantly to all parts of this paper and all authors agree with the content of the manuscript. Authors WE, KL, HE, BA, WA, FH, KA, MB, and JM contributed to design; authors WE, LM, BA, and HE contributed to data collection; authors WE and JM contributed in the data analysis. All authors contributed to writing, interpretation, and reviewing the final manuscript. All authors contributed to the writing of the final manuscript. All members of the team contributed to the management or administration of the trial. All authors contributed significantly to the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
