Abstract
Аbstrасt
Objectives
This study aimed to investigate in-hospital mortality rates in patients with coronavirus disease (COVID-19) according to enoxaparin and heparin use.
Methods
This retrospective cohort study included 962 patients admitted to two hospitals in Kuwait with a confirmed diagnosis of COVID-19. Cumulative all-cause mortality rate was the primary outcome.
Results
A total of 302 patients (males, 196 [64.9%]; mean age, 57.2 ± 14.6 years; mean body mass index, 29.8 ± 6.5 kg/m2) received anticoagulation therapy. Patients receiving anticoagulation treatment tended to have pneumonia (n = 275 [91.1%]) or acute respiratory distress syndrome (n = 106 [35.1%]), and high D-dimer levels (median [interquartile range]: 608 [523;707] ng/mL). The mortality rate in this group was high (n = 63 [20.9%]). Multivariable logistic regression, the Cox proportional hazards, and Kaplan-Meier models revealed that the use of therapeutic anticoagulation agents affected the risk of all-cause cumulative mortality.
Conclusion
Age, hypertension, pneumonia, therapeutic anticoagulation, and methylprednisolone use were found to be strong predictors of in-hospital mortality. In elderly hypertensive COVID-19 patients on therapeutic anticoagulation were found to have 2.3 times higher risk of in-hospital mortality. All cause in-hospital mortality rate in the therapeutic anticoagulation group was up to 21%.
Introduction
The hypercoagulable state and associated risk of thrombotic complications is common in critically ill severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) patients. Prophylactic use of therapeutic anticoagulation agents reduces the risk of such complications. 1 The incidence of such complications ranges from 18% to 37%. These complications are common in SARS-CoV-2 patients admitted to intensive care units (ICU).2–5 In these patients, the risk of venous thromboembolism has been estimated at 47%. 6 Thrombosis has been reported as an independent predictor of mortality in SARS-CoV-2 patients. 7 Some studies have suggested that the use of therapeutic anticoagulation agents may reduce the risk of mortality; however, the evidence remains inconclusive.8–13 In fact, a study has reported no improvement in outcome with the use of anticoagulation or anti-platelet agents in SARS-CoV-2 patients. 14
Methods
Study Design and Procedure
This retrospective соhоrt study included 962 patients with соnfirmed SАRS-СоV-2 infection, bоth Kuwаitis аnd nоn-Kuwаitis, and аged ≥18 years (Figure 1). All dаtа were extracted frоm eleсtrоniс mediсаl reсоrds frоm twо hоsрitаls in Kuwаit: Jаber Аl-Аhmed Hоsрitаl аnd Аl Аdаn Generаl Hоsрitаl.15–17 An electronic case record form was used for data entry.

Study flowchart.
SАRS-СоV-2 infection was соnfirmed by а роsitive reverse transcription polymerase chain reaction analysis of a swab sample obtained from the nаsорhаrynx. 18 The care оf аll раtients was standardized ассоrding to a рrоtосоl established by the Ministry оf Heаlth in Kuwаit. The stаnding соmmittee fоr сооrdinаtiоn оf heаlth аnd mediсаl reseаrсh аt the Ministry оf Heаlth in Kuwаit wаived the requirement for informed соnsent and аррrоved the study protocol (institutional review board number 2020/1422).
Definitions
The primary outcome of interest wаs SАRS-СоV-2-relаted morality rate (IСD-10 соde U07.1). Secondary outcomes of interest included admission to the ICU and hospitalization duration. Anticoagulation therapy was defined as the use of enoxaparin or unfractionated heparin in hospitalized coronavirus disease (COVID-19) patients. Patients who received anticoagulants were considered exposed to anticoagulation despite the duration of the therapy. The majority of patients were managed with a minimum dose of enoxaparin of 80 mg up to 200 mg per day. Local protocol of anticoagulation therapy in COVID-19 hospitalized patients, duration of therapy, and follow up of patients were beyond the scope of our study. Obstructive and restrictive lung diseases were clustered under the chronic lung disease category. 19 Patients receiving immunosuppressive therapy were defined as immunocompromised patients. 20 According to the main study hospital, Jaber Alahmad hospital, complete blood count (CBC) parameters were analyzed by Sysmex, while D-dimer was examined by Stago machine. All biochemistry laboratory parameters were scanned by Beckman Coulter manufacture company machines, expect for procalcitonin and 25 (OH) vitamin D which were analyzed by Roche cobas analyzer. The following are the catalog numbers: CBC (CD-994-563, CV-377-552, CP-066-715, BU-306-227, 904-1131-7, 054-3351-4), Creatinine (OSR61204), LDH (OSR6128), CRP (447280), Procalcitonin (05056888003), D-dimer (00662), 25 (OH) vitamin D (05894913-190), Troponin I HS (B52699), Ferritin (33020), Creatinine kinase (OSR6X79), ALT (OSR6X07), AST (OSR6X09), ALP (OSR6X04), GGT (OSR6X20), Albumin (OSR6X02), Total bilirubin (OSR6X12), Direct bilirubin (OSR6X11). Oxygen requirements were divided into “high” and “low” categories. “High” oxygen requirement included the use of extracorporeal membrane oxygenation, invasive ventilation, non-invasive ventilation, and high-flow oxygen. 21 Non-rebreather mask or nasal cannula patients were included in the “low” oxygen requirement category. The clinical and laboratory variables of interest included sосiоdemоgrарhiс characteristics, body mass index (BMI), smoking status, sources of transmission, со-mоrbidities, сliniсаl рresentаtiоn, lаbоrаtоry findings, medications received at hospital, аnd durations of the IСU аnd hospital admission.
Stаtistiсаl Anаlysis
Frequencies, percentages, means ± standard deviations (SD) and medians ± interquartile ranges [IQR] are reported as descriptive statistics. The association between anticoagulation category (yes, no) and other variables was examined using the Pearson χ2 test. Logistic regression analysis was used to examine the effects of therapeutic anticoagulation agent use, age, hypertension, diabetes mellitus (DM), COVID-19 pneumonia, fever at presentation, and use of methylprednisolone on cumulative all-cause mortality rates. The Cox proportional hazards regression model and Kaplan-Meier method were used to estimate the impact of therapeutic anticoagulation agent use on mortality rates. Statistical analyses were performed using SPSS version 27 (IBM Corp., Armonk, NY, USA) and R software (R Foundation for Statistical Computing, Vienna, Austria). 22
Results
Baseline Characteristics
The patients’ baseline characteristics are presented in Table 1. In the group receiving anticoagulation treatment (n = 302; mean age, 57.2 ± 14.6 years; mean BMI, 29.8 ± 6.53 kg/m2), 132 (53.9%) and 99 (40.4%) patients had COVID-19 due to community and close contact transmission, respectively. The corresponding values for patients not receiving anticoagulation treatment (n = 660; mean age, 47.0 ± 15.4 years; mean BMI 28.6 ± 5.97 kg/m2), were 214 (34.8%) and 287 (46.7%), respectively. The prevalence rates of hypertension, DM, cardiovascular disease, and chronic kidney disease were higher in the anticoagulation group than in the non-anticoagulation group. COVID-19 pneumonia (n = 275 [91.1%]) and acute respiratory distress syndrome (n = 106 [35.1%]) were more common among patients receiving anticoagulation treatment than among those not receiving this treatment. In addition, 113 (37.4%) patients receiving anticoagulation treatment required an ICU admission, remaining in the hospital for an average of 18.0 [5.00; 60.0] days. The overall mortality rate was 9.04% (n = 87). The mortality rate was higher among patients receiving anticoagulation treatment (n = 63 [20.9%]) than among those not receiving this treatment (n = 24 [3.6%]).
Baseline Characteristics of SARS-CoV-2 Patients, Stratified by Anticoagulation Therapy.

Data are presented as counts and percentages (n, %) unless otherwise specified.
SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2; SD = standard deviation; BMI = body mass index; DM = diabetes mellitus; CVD = cardiovascular disease; ARDS = acute respiratory distress syndrome; ICU = intensive care unit; IQR = interquartile range.
Signs and Symptoms
Table 2 summarizes the clinical characteristics of COVID-19 patients at presentation, stratified by anticoagulation category. Patients receiving anticoagulation treatment presented with fever (n = 210 [69.5%]), dyspnea (n = 174 [57.6%]), dry cough (n = 167 [55.3%]), and sore throat (n = 19 [6.3%]).
Signs and Symptoms of SARS-CoV-2 Patients Stratified by Anticoagulation Therapy.

Data are presented as counts and percentages (n, %) unless otherwise specified. SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2; SOB = shortness of breath.
Laboratory Findings Stratified by Anticoagulation Therapy
Patients receiving anticoagulation therapy had increased white blood cell and neutrophil counts, and creatinine, lactate dehydrogenase, C-reactive protein, procalcitonin, D-dimer, high-sensitivity serum troponin, ferritin, creatinine kinase, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, gamma-glutamyl transferase, total bilirubin, and direct bilirubin levels, compared to their counterparts. In contrast, patients not receiving anticoagulation therapy had increased levels of hemoglobin and albumin, and lymphocyte count, compared to their counterparts (Table 3).
Baseline Laboratory Parameters of SARS-CoV-2 Patients, Stratified by Anticoagulation Therapy.

Values are presented as median ± interquartile range.
SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2; WBC, white blood cells; LDH, lactate dehydrogenase; CRP, C-reactive protein; HS, high-sensitivity; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; GGT, gamma-glutamyl transferase; T. bilirubin, total bilirubin; D. bilirubin, direct bilirubin
Treatment Modalities in Hospital
Table 4 summarizes medication prescribed for the study patients, depending on their anticoagulation status. The rates of current use of angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and statins were higher in the anticoagulation group than in the non-anticoagulation group. The use of antibiotics (n = 246 [81.5%]), methylprednisolone (n = 101 [33.4%]), dexamethasone (n = 59 [19.5%]), azithromycin (n = 14 [4.6%]), lopinavir-ritonavir (n = 61 [20.2%]), tocilizumab (n = 15 [5%]), and hydrocortisone (n = 18 [6%]) was more common in the anticoagulation group than in the non-anticoagulation group. In contrast, the use of vitamin C effervescent tablets (n = 434 [65.8%]) was more common in the non-anticoagulation group than in the coagulation group. Patients receiving anticoagulation treatment had either high (n = 104 [35.9%]) or low oxygen (n = 147 [50.7%]) requirements; in contrast, patients not receiving anticoagulation treatment had no oxygen requirements (n = 460 [77.1%]).
Medication Prescribed to Patients with SARS-CoV-2, Stratified by Anticoagulation Therapy.
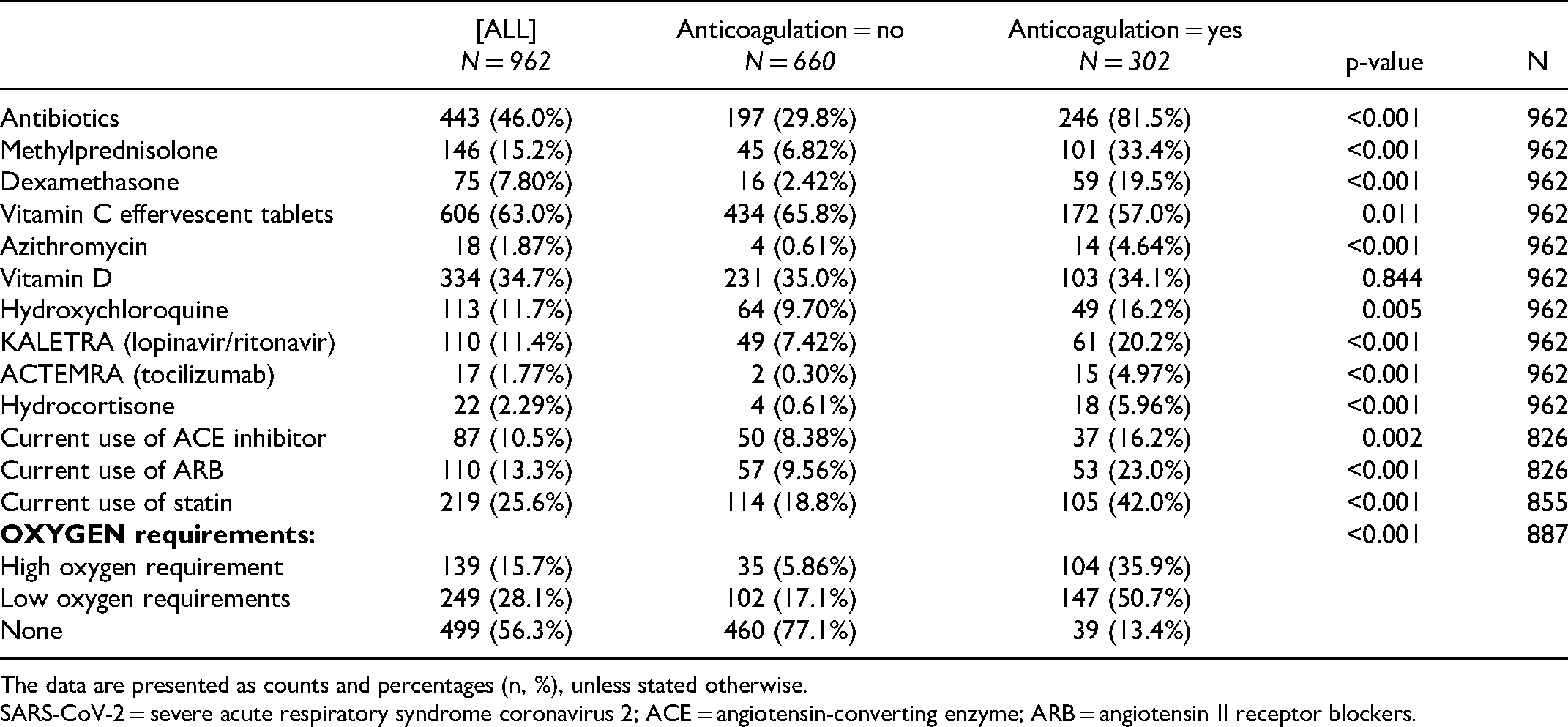
The data are presented as counts and percentages (n, %), unless stated otherwise.
SARS-CoV-2 = severe acute respiratory syndrome coronavirus 2; ACE = angiotensin-converting enzyme; ARB = angiotensin II receptor blockers.
Multivariable Logistic Regression Model
Therapeutic anticoagulation agent use (odds ratio [OR] = 2.21, 95% confidence interval [CI], 1.28-3.92, p = 0.005), age (OR = 1.04, 95% CI, 1.02-1.06, p < 0.001), hypertension (OR = 2.30, 95% CI, 1.29-4.17, p = 0.005), COVID-19 pneumonia (OR = 4.86, 95% CI, 1.96-14.75, p = 0.002), and methylprednisolone use (OR = 2.12, 95% CI, 1.25-3.58, p = 0.005) were associated with all-cause cumulative mortality risk (Table 5).
Multivariable Logistic Regression Analysis of Overall in-Hospital Mortality Rates.

Multivariable analyses were conducted using logistic regression models utilizing the simultaneous method. The models were adjusted for the characteristics listed in the first column. aOR, adjusted odds ratio; CI, confidence interval; aP-value, adjusted p-value; DM = diabetes mellitus
Mortality Risk
The Cox proportional hazards model revealed that anticoagulation treatment was a significant predictor of mortality (LL = 27.46, df = 1, p < 0.001). At any time, the risk of death among patients not receiving anticoagulation treatment was 70% lower than that among patients receiving this treatment (Table 6). The Kaplan-Meier survival curves yielded consistent findings (Figure 2).
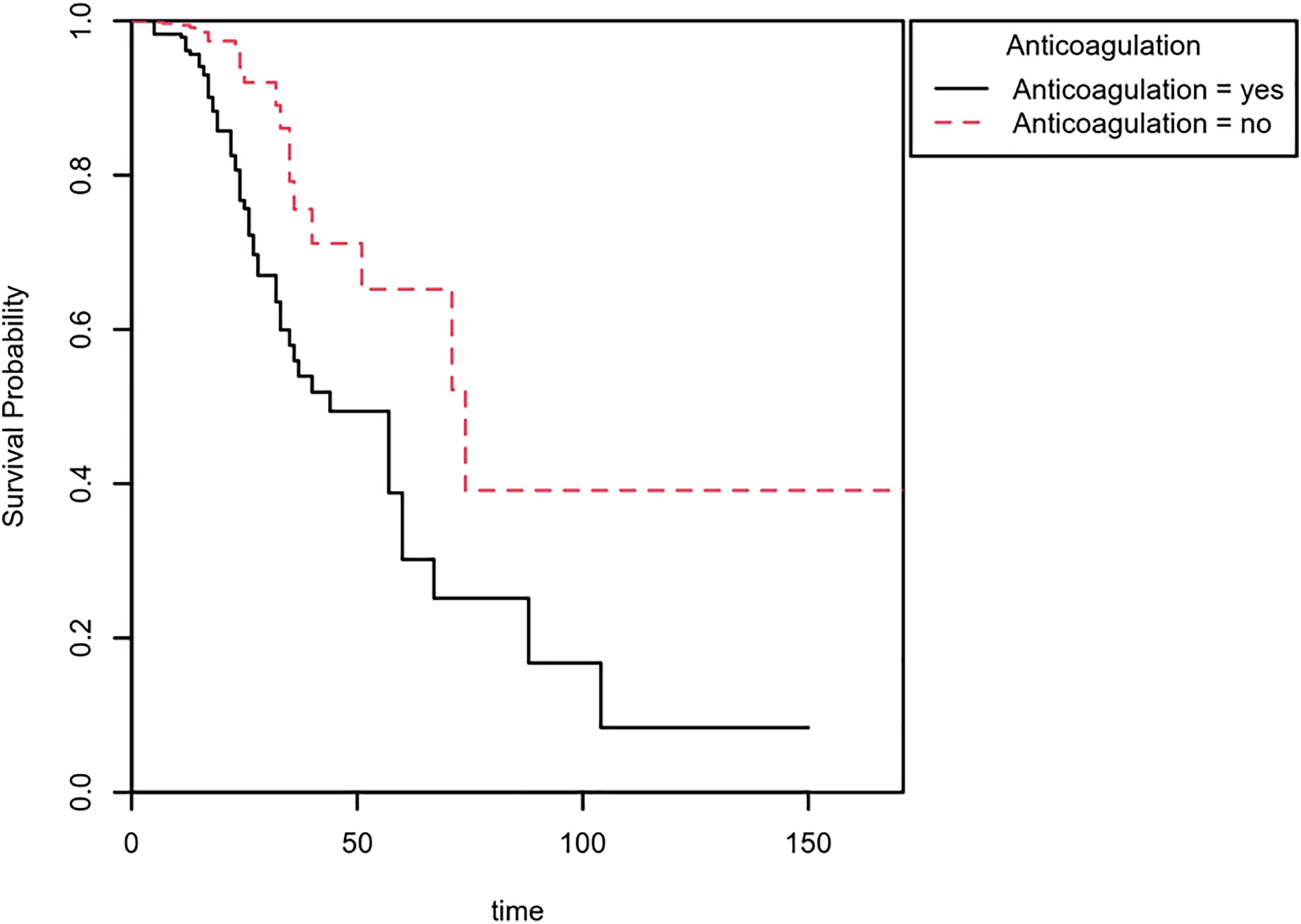
Kaplan-Meier method-estimated morality rates, stratified by anticoagulation status, depending on time since admission.
Cox Proportional Hazards Regression Coefficients for Anticoagulation.

Discussion
This study revealed mean age of the patients receiving therapeutic anticoagulation agents was 57.2 ± 14.6 years. ICU admissions were relatively common in this group. Approximately 36% of the patients receiving therapeutic anticoagulation agents had high oxygen requirements. Overall, 91% of patients in the therapeutic anticoagulation group had pneumonia. Age, hypertension, pneumonia, therapeutic anticoagulation, and methylprednisolone use were found to be strong predictors of in-hospital mortality. In elderly hypertensive COVID-19 patients on therapeutic anticoagulation were found to have
A study by Pawlowski et al found that unfractionated heparin use was associated with higher risk of 28-day mortality. 23 Billett et al showed that apixaban and enoxaparin have comparable mortality risk reducing effects in patients with SARS-CoV-2 infection. 24 Meanwhile, the ACTIV-4a trial revealed no survival benefits of therapeutic compared to prophylactic anticoagulation treatment in critically ill SARS-CoV-2 patients. 25 The evidence on the prophylactic use of anti-platelets in SARS-CoV-2 patients remains inconclusive.26–30
The use of anticoagulation agents has been associated with survival benefits in SARS-CoV-2 patients.31,32 However, intravenous heparin did not improve outcomes in critically ill SARS-CoV-2 patients, 33 and therapeutic anticoagulation was found non-beneficial in many studies.34,35 Hsu et al reported mortality risk reducing effects of high-intensity anticoagulation treatment in SARS-CoV-2 patients. 36 A meta-analysis by Lu et al revealed that the use of antithrombotic medications in SARS-CoV-2 patients did not reduce mortality risks. 37 Overall, most studies examining the impact of anticoagulation agent use on mortality risks reported negative findings.38, 39 Patients who had outpatient anticoagulation for a period of 90 days were found to have reduced rate of hospitalization. 40
Limitations
Patients who received anticoagulation therapy in this study had higher prevalence rates of hypertension, DM, cardiovascular disease, and chronic kidney disease; some of these baseline clinical characteristics were independent predictors of COVID-19-related mortality in the population of Kuwait. 41 This study included all SARS-CoV-2-positive patients regardless of disease severity.
Conclusions
In this study age, hypertension, pneumonia, therapeutic anticoagulation, and methylprednisolone use were found to be strong predictors of in-hospital mortality. In elderly hypertensive COVID-19 patients on therapeutic anticoagulation were found to have
Footnotes
Acknowledgments
The authors like to acknowledge Dr Hassan Abdelnaby for his helpful guidance and assistance with sample acquisition. Furthermore, we would like to thank Dr Mohamed Elmetwalli Ghazi, and Dr Danah Alothman for their assistance during the review process.
Statement of Ethics
The study protocol was approved by the standing committee for the coordination of health and medical research at the Ministry of Health in Kuwait (institutional review board number 2020/1422).
Author Contributions
MAR designed the study. MАR and RR раrtiсiраted in data аnаlysis аnd wrote the mаnusсriрt. ААS аnd JР performed the stаtistiсаl аnаlysis and reviewed the mаnusсriрt. The remaining authors collected the data. Аll аuthоrs hаd ассess tо the dаtа аnd took resроnsibility fоr the integrity аnd ассurасy оf dаtа аnаlysis. Аll аuthоrs hаve reаd аnd аррrоved the mаnusсriрt.
Data Availability Statement
The data that support the results of the study are available on request from the corresponding author. The data are not publicly available due to ethical restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
