Abstract
Data on the impact of morbid obesity (body mass index [BMI] ≥ 40 kg/m2) on the pharmacokinetics (PK), pharmacodynamics (PD) of direct oral anticoagulants (DOACs) are relatively limited, making it difficult to design optimal dosing regimens in morbidly obese patients.
To review literature on PK/PD profile, efficacy, and safety of DOACs in venous thromboembolism (VTE) and nonvalvular atrial fibrillation (AF) patients with morbid obesity and make recommendations regarding optimal dosing regimens in these patient populations.
A detailed literature search was conducted (from inception to June 22, 2022) for relevant articles involving PK/PD and clinical data on DOACs use in morbidly obese patients with VTE or AF, or healthy volunteers.
A total of 28 studies were identified. DOAC-specific PK variations and clinical outcomes have been observed. Obesity may have a modest effect on PK/PD of dabigatran, apixaban, or rivaroxaban. Dabigatran was effective in AF patients with morbid obesity but might increase the risk of gastrointestinal bleeding. Standard dosing of apixaban or rivaroxaban is effective and safe for VTE and AF patients with morbid obesity. Trough edoxaban concentration and anti-Xa activity were similar in different BMI groups (18.5 to >40 kg/m2), and standard dosing of edoxaban may be effective and safe for AF patients.
Current evidence suggests dabigatran should be used with caution in patients with AF as it might increase the risk of gastrointestinal bleeding; Standard dosing of apixaban or rivaroxaban can be used in VTE or AF patients; Standard dosing of edoxaban may be considered in AF patients.
Introduction
Direct oral anticoagulants (DOACs) have emerged as the preferred treatment for the prevention and treatment of venous thromboembolism (VTE) and prevention of stroke in nonvalvular atrial fibrillation (AF) over the past decade. 1 The advantages of DOACs include a wider therapeutic window and less monitoring requirements, fixed dosing, a faster onset of action, and fewer drug–drug and drug-food interactions, which make them to be alternatives to warfarin and widespread utilization.2–4 However, relatively limited data exist on the clinical pharmacokinetics (PK), pharmacodynamics (PD), efficacy, and safety in patients with morbid obesity (body mass index [BMI] of ≥40 kg/m2) due to this patient population severely underrepresented in the majority of the landmark DOAC trials. The 2021 update of the International Society on Thrombosis and Haemostasis (ISTH) guidelines suggested that standard doses of rivaroxaban or apixaban are among appropriate anticoagulant options for treatment or primary prevention of VTE regardless of high BMI and weight, while other guidelines on VTE or AF management do not provide clear guidance on the use of DOACs in patients with a BMI of >40 kg/m2 or weight of >120 kg. 5 Regardless of consensus guidelines, data from the GLORIA-AF registry showed this patient population tends to favor the use of DOACs over warfarin. 6
Due to obesity being a prothrombotic state, it is associated with increased thrombotic risks. 7 In addition, the physiologic changes associated with obesity may presumably have a considerable effect on the PK of DOACs, which may alter the exposure to DOACs and thereby impact their efficacy and safety. Therefore, morbid obesity may be a major challenge in the dosage modification of DOACs in clinical practice. Although measuring serum drug levels for therapeutic monitoring in this population has been suggested, testing of DOAC levels is neither widely available nor well validated in real-world clinical settings. 8 When designing DOACs dosing regimens in morbidly obese patients, a detailed understanding of the primary pharmacokinetic parameters, and the impact of obesity has on physiology and drug pharmacokinetics are required. Thus, the aim of this study is to provide a comprehensive review of available data from published literature of DOACs’ PK and PD, efficacy and safety in morbid obesity and to help improve the evidence-based decision-making process for clinicians considering prescribing a DOAC for morbidly obese patients.
Materials and Methods
We primarily searched data on Pharmacokinetic changes in obesity, available PK/PD, and clinical data on DOACs use in morbidly obese patients with VTE or AF, or healthy volunteers. We screened the published guidelines from the following therapeutic domains or the population of interest: (1) VTE, and/or nonvalvular AF; (2) obese patients and/or morbidly obese patients. We then thoroughly screened published reviews providing recommendations for DOACs use. Furthermore, we performed a detailed literature search using PubMed, Embase, and Cochrane Library databases from inception to June 22, 2022, with the following strategy to identify the latest evidence after the ISTH 2016 guidelines: (1) obesity: “obesity” or “overweight” or “morbid obesity” or “morbid body weight” or “morbid obesity” or “obese weight” or “body mass index” or “BMI” or “weight” or “body weight” or "body composition” or “body weight change” or “body size” or “body fat” or “body fatness”; (2) DOACs: dabigatran or apixaban or betrixaban or edoxaban or rivaroxaban; and (3) pharmacokinetic or pharmacodynamic, or diseases of interests (VTE or AF). Non-English articles and animal studies were excluded. We focused on adult patients with more severe forms of obesity (morbid obesity, BMI ≥ 40 kg/m2) since such patients are typically limited in numbers in the studies used to formulate product labeling information. Primary pharmacokinetic parameters of each DOAC were obtained from the corresponding product labeling.
Results and Discussions
The DOACs vary widely in oral bioavailability, volume of distribution (Vd), metabolism, and renal excretion, and PK profile can differ in patients with obesity between DOACs. A detailed overview of primary pharmacokinetic properties and dosing regimen of DOACs has been reported in the scientific literature (Table 1).9–13 We further reviewed the impact of body weight and morbid obesity on the PK/PD (Table 2), and clinical outcomes of DOACs according to disease state (Table 3 for patients with VTE and Table 4 for patients with AF).
Pharmacokinetic Properties and Dosing of Direct Oral Anticoagulants.

Abbreviations: NVAF, nonvalvular atrial fibrillation; Vd, volume of distribution; VTE, venous thromboembolism.
DOAC Pharmacokinetic Changes in Obesity.
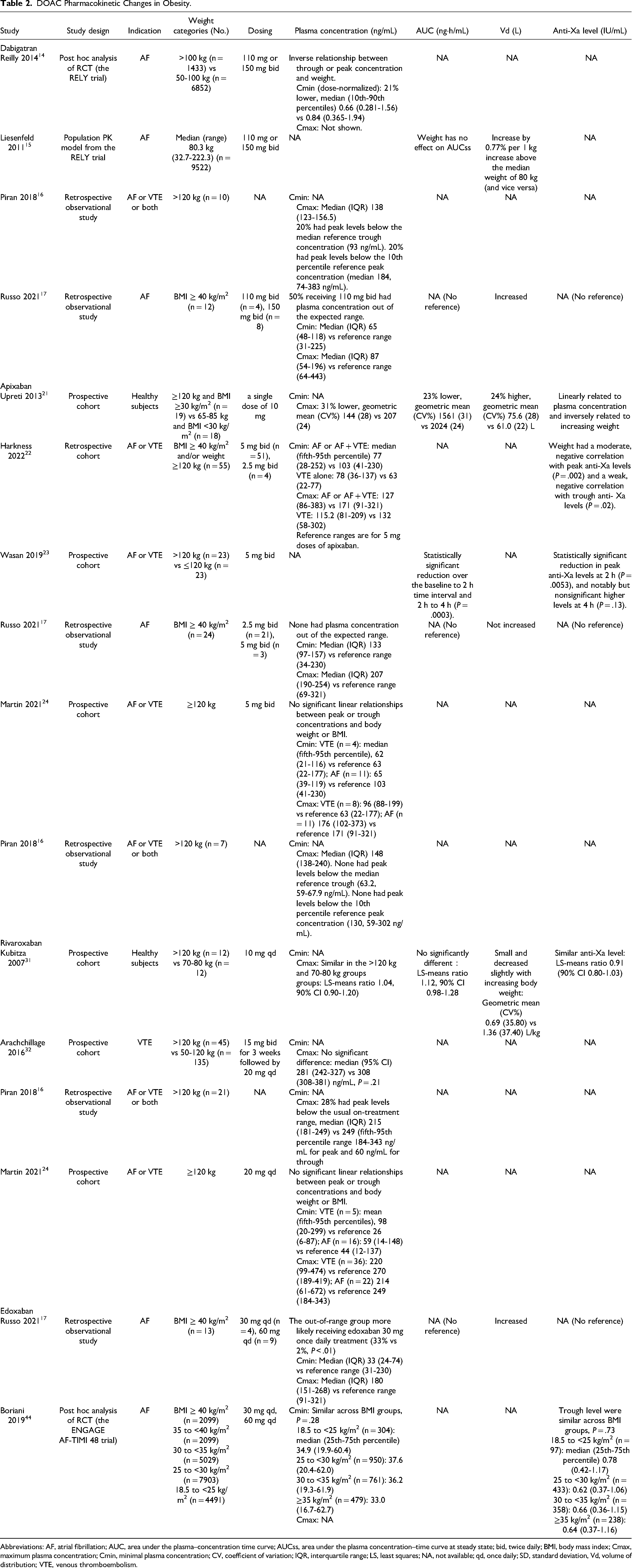
Abbreviations: AF, atrial fibrillation; AUC, area under the plasma–concentration time curve; AUCss, area under the plasma concentration–time curve at steady state; bid, twice daily; BMI, body mass index; Cmax, maximum plasma concentration; Cmin, minimal plasma concentration; CV, coefficient of variation; IQR, interquartile range; LS, least squares; NA, not available; qd, once daily; SD, standard deviation, Vd, volume of distribution; VTE, venous thromboembolism.
Summary of Studies of Direct Oral Anticoagulants in Morbidly Obese Patients With Venous Thromboembolism.
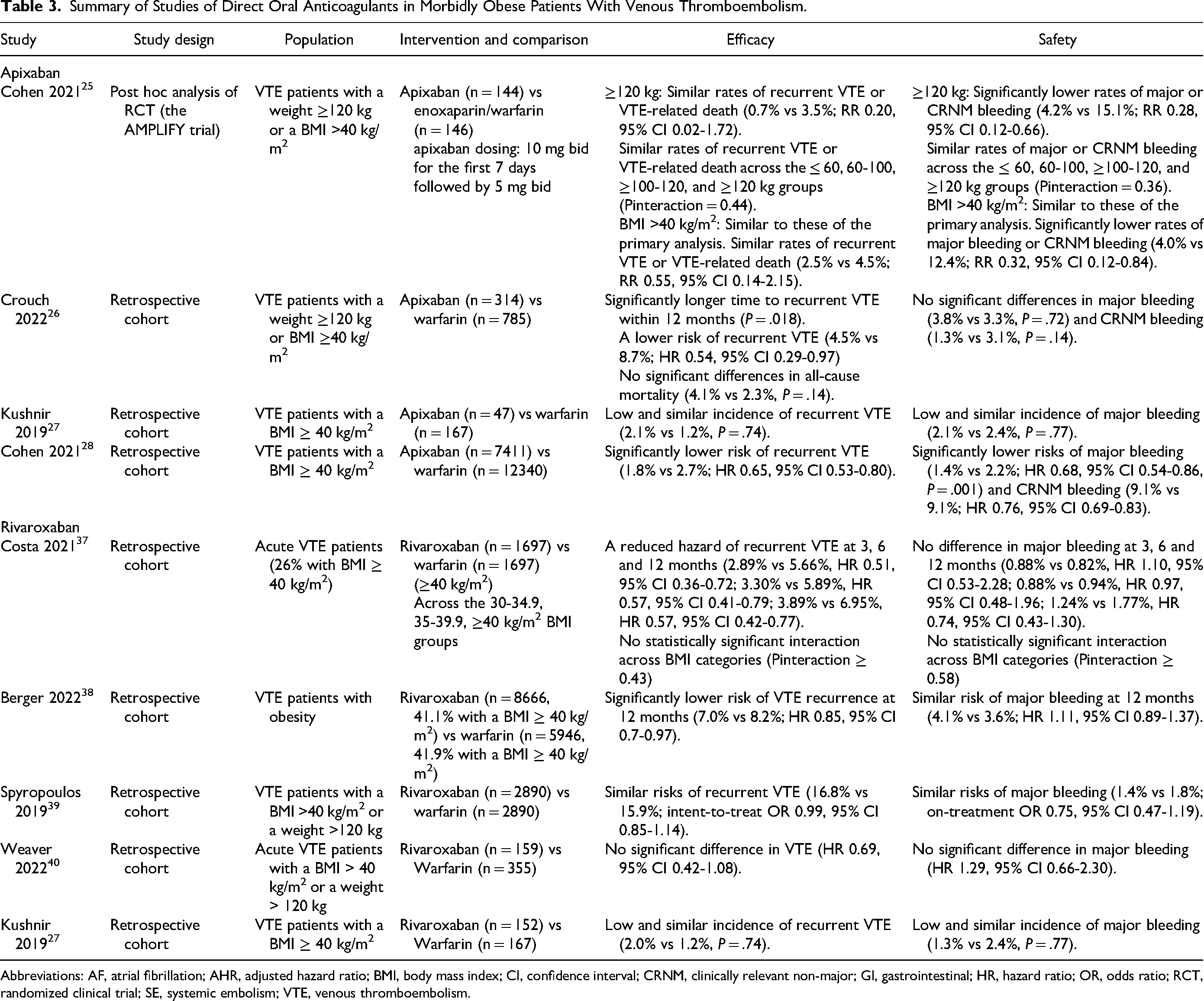
Abbreviations: AF, atrial fibrillation; AHR, adjusted hazard ratio; BMI, body mass index; CI, confidence interval; CRNM, clinically relevant non-major; GI, gastrointestinal; HR, hazard ratio; OR, odds ratio; RCT, randomized clinical trial; SE, systemic embolism; VTE, venous thromboembolism.
Summary of Studies of Direct Oral Anticoagulants in Morbidly Obese Patients With Nonvalvular Atrial Fibrillation.

Abbreviations: AF, atrial fibrillation; AHR, adjusted hazard ratio; BMI, body mass index; CI, confidence interval; CRNM, clinically relevant non-major; GI, gastrointestinal; HR, hazard ratio; OR, odds ratio; RCT, randomized clinical trial; SE, systemic embolism; VTE, venous thromboembolism.
Dabigatran
PK/PD data: In a subgroup analysis of the RE-LY trial, dose-normalized trough concentrations were 21% lower for the high body weight group (>100 kg) than for the reference body weight group (50-100 kg) and similar relationships occurred for peak concentrations, demonstrating an inverse relationship between trough or peak concentration and weight. 14 The multiple logistic regression analysis showed an inverse relationship between trough concentration and the risk of ischemic events. A population PK analysis of dabigatran in AF patients based on the RE-LY trial showed an increase of 0.77% in Vd per 1 kg increase above 80 kg and no impact of weight on area under the plasma concentration (AUC). 15 Another retrospective study measured peak DOAC plasma concentrations in 38 patients with body weight >120 kg taking DOACs for any indications. 16 Among 10 patients taking dabigatran, 2 (20%) had peak plasma concentrations below the median trough level (93 ng/mL) and below the usual on-treatment range for peak level (median 184 (10th-90th percentiles 74-383 ng/mL) sourced from pharmacokinetic studies in non-obese patients. 16
A PK analysis of DOACs in 58 AF patients with morbid obesity (BMI ≥ 40 kg/m2) did observe different results regarding the impact of body weight on the concentration, of 8 patients taking dabigatran with an appropriate dosing (150 mg twice daily [bid]), none had plasma concentration out of the expected range as obtained from population PK studies in non-obese patients. 17 The multivariate logistic analysis indicated that inappropriate DOAC underdosing (eg 110 mg bid for dabigatran) was the only independent clinical factor associated with a plasma concentration of the drug out of the expected range (hazard ratio [HR] 29.37, 95% CI [confidence interval] 4.26-202.71).
Clinical data for VTE: Studies analyzing dabigatran for the treatment or prevention of VTE in morbidly obese patients were not identified.
Clinical data for AF: Three large-scale cohort studies evaluated the efficacy and safety of dabigatran in AF patients with morbid obesity. A retrospective cohort study including 6848 AF patients with a BMI >40 kg/m2 or a weight >120 kg and receiving either dabigatran or warfarin found dabigatran was effective in reducing the risk of thromboembolism (HR 0.71, 95% CI 0.56-0.91) and mortality (HR 0.57, 95% CI 0.45-0.71) but was associated with an increased risk of composite bleeding (HR 1.24, 95% CI 1.07-1.42), which mainly driven by gastrointestinal bleeding (HR 1.59, 95% CI 1.33-1.91). 18 A second retrospective cohort study evaluated the efficacy and safety of dabigatran in AF patients across different weight groups (777 patients weighting >120 kg vs 3522 patients weighting ≤120 kg) and found in patients with >120 kg, dabigatran was associated with a small increased (1.44 times greater) risk of gastrointestinal bleeding (adjusted HR 1.44, 95% CI 1.01-2.05) but no differences in stroke (adjusted HR 0.45, 95% CI 0.05-3.89), mortality (0.72, 95% CI 0.37-1.42), or clinically relevant bleeding (adjusted HR 1.21, 95% CI 0.93-1.57). 19
Conversely, in a large cohort of obese Veterans Health Administration system patients with AF, among patients with weight ≥120 kg and those with BMI >40 kg/m2, for efficacy endpoint, dabigatran had significantly lower risks of myocardial infarction, heart failure, and all-cause mortality, and similar risk of ischemic stroke than warfarin. For safety endpoints, patients treated with dabigatran were associated with a lower risk of any major bleeding, hemorrhagic stroke, and gastrointestinal bleeding. 20
Summary: PK profile of dabigatran in morbid obesity may be modestly altered. Weight might have an inverse correlation with trough or peak concentration, whereas has no impact on AUC. Vd increased by 0.77% for per 1 kg increase above 80 kg. Although clinical data showed dabigatran was effective in AF patients with morbidly obese patients, it possibly increased the risk of gastrointestinal bleeding. Therefore, dabigatran should be used with caution in morbidly obese patients until more robust data emerges.
Apixaban
PK/PD data: Six studies have examined the impact of body weight on the PK/PD of apixaban. A PK study of apixaban (a single dose of 10 mg) in healthy volunteers investigated the effects of morbid obesity on apixaban PK, PD, and safety, and observed a 31% lower mean peak apixaban concentration, a 24% higher Vd and a 23% lower drug exposure in the high body weight group (weight of >120 kg and BMI of ≥30 kg/m2) than in the reference body weight group (weight of 65-85 kg). 21 Renal clearance of apixaban was similar between weight groups. Additionally, the mean elimination half-life (T1/2) of apixaban was 3 h shorter in the high-weight group compared with that in the reference weight group, but this was not expected to be clinically significant. Although there was a statistically significant inverse relationship between apixaban exposure and body weight (P < .001), and anti-factor Xa activity was linearly related to apixaban plasma concentration and inversely related to increasing weight, the impact of body weight alone on apixaban exposure was considered modest and unlikely to be clinically meaningful. Thus, there was no need to adjust the dose of apixaban in patients weighing >120 kg without severe renal impairment. Similarly, an observational cohort study measuring anti-Xa levels of AF or VTE patients with a BMI ≥40 kg/m2 and/or a weight ≥120 kg who have treated with apixaban 5 mg or 2.5 mg bid, of 55 patients enrolled, 9% had peak and 6% had trough anti-Xa levels below the reference ranges. 22 Although body weight had a moderate, negative correlation with peak anti-Xa levels and a weak, inverse correlation with trough anti-Xa levels, the results showed that anti-Xa levels among obese patients are not substantially different from patients with normal BMI and weight.
Conversely, a prospective cohort study analyzed the effects of body weight on apixaban anti-Xa levels in 46 patients receiving apixaban 5 mg bid for VTE or AF. 23 In patients >120 kg (mean BMI 49 kg/m2), there was a statistically significant reduction in peak anti-Xa levels at 2 h and AUC of apixaban compared with the normal BW group (≤120 kg, mean BMI of 31 kg/m2). However, due to the study design and small scale, reduced apixaban levels may have uncertain clinical consequences.
The other 3 studies showed little or no change in apixaban levels in patients with morbid obesity compared with the reference normal body weight. In the previously mentioned PK study measuring DOACs levels in 58 AF patients with morbid obesity (BMI ≥ 40 kg/m2), of 24 patients taking apixaban (5 mg or 2.5 mg bid), no patients had plasma concentration out of the expected range. 17 A prospective cohort study conducted by Martin et al of apixaban 5 mg bid or rivaroxaban 20 mg once daily for AF or VTE in 100 patients with weight >120 kg (88% with BMI >40 kg/m2) showed that all 19 peak apixaban and 16 (89%) of 18 trough factor Xa inhibitor concentrations were in the expected ranges, suggesting no under-dosing in the majority of these patients. 24 No significant linear relationships between peak or trough factor Xa inhibitor concentrations and body weight or BMI was found. In the retrospective study measuring peak DOAC plasma concentrations in 38 patients with body weight >120 kg, all 7 patients receiving apixaban had peak plasma concentration higher than the median trough level and within the usual on-therapy range. 16
Clinical data for VTE: All studies evaluating apixaban use for VTE patients with morbid obesity found at least similar efficacy or safety outcomes compared with warfarin. A post hoc analysis of the AMPLIFY trial found apixaban had similar rates of recurrent VTE or VTE-related death and lower rates of major or clinically relevant non-major (CRNM) bleeding compared with enoxaparin/warfarin across the different body weight and BMI categories, including body weight ≥120 kg and BMI >40 kg/m2. 25 A modest, not clinically meaningful, decrease (<30%) in the median predicted exposure with increasing body weight was also observed. A multi-center, retrospective study compared the use of apixaban versus warfarin for VTE treatment in 1099 patients with morbid obesity, found a reduced risk of recurrent VTE (HR 0.54, 95% CI 0.29-0.97) in patients taking apixaban compared with those taking warfarin within 12 months, and no significant differences in major bleeding (3.8% vs 3.3%, P = .72), CRNM bleeding (1.3% vs 3.1%, P = .14), or all-cause mortality (4.1% vs 2.3%, P = .14). 26 These findings indicated apixaban appears to be effective and safe for the treatment of VTE in patients with morbid obese. In a single-center, retrospective study of DOACs in 366 VTE patients with BMI ≥40 kg/m2, the incidences of recurrent VTE (2.1% vs 1.2%, P = .74) and major bleeding (2.1% vs 2.4%, P = .77) were similar between the apixaban and warfarin cohorts. 27 Similarly, in the subgroup of 92 patients with BMI ≥50 kg/m2, no significant difference in rates of recurrent VTE and major bleeding was observed. 27 In a large sample study of VTE patients based on US insurance claims databases found apixaban was associated with a significantly lower risk of recurrent VTE (HR 0.65, 95% CI 0.53-0.80), major bleeding (HR 0.68, 95% CI 0.54-0.86), and CRNM bleeding (HR 0.76, 95% CI 0.69-0.83) compared with warfarin in morbidly obese patients. 28
Clinical data for AF: Consistent with the results of studies conducted in VTE patients, at least similar clinical outcomes were observed in AF patients. A post hoc analysis of ARISTOTLE in AF patients with a range of body weight groups, among 982 with weight >120 kg, there was no significant difference in efficacy outcomes of stroke/systemic embolism, all-cause death, or myocardial infarction between apixaban and warfarin 29 However, a lower risk of major and CRNM bleeding (HR 0.58, 95% CI 0.35-0.95) or any bleeding (HR 0.67, 95% CI 0.53-0.85) was observed with apixaban. When further stratified into 121 to 140 kg (n = 724) and >140 kg (n = 258) groups, a lower risk of stroke/systemic embolism was observed in the apixaban group for patients weighing 121 to 140 kg (HR 0.21, 95% CI 0.05-0.95). Overall, the superiority of apixaban over warfarin regarding efficacy and safety for stroke prevention seems to be similar in patients with AF across the groups of weight, indicating that apixaban appears to be appropriate for patients with AF irrespective of body weight. 29 Similarly, in the cohort of obese Veterans Health Administration system, among AF patients with BMI >40 kg/m2, apixaban had similar risks of ischemic stroke(HR 0.93, 95% CI 0.71-1.22) and significantly lower risks of any major bleeding (HR 0.72, 95% CI 0.630.82) than warfarin. 20 A retrospective cohort study (722 patients) conducted to evaluate the effectiveness and safety of using apixaban in morbidly obese (BMI ≥ 40 kg/m2) patients with AF, showed a similar prevalence of all thrombotic events (OR 0.58, 95% CI 0.13-2.51) and odds of developing major bleeding (OR 0.39, 95% CI 0.07-2.03) of apixaban use in both morbid and nonmorbid obese patients with AF within 12 months. 30 In the aforementioned study, among the 429 patients with AF, incidences of stroke were similar between the apixaban and warfarin users (1.0% vs 1.3%, P = .71) and incidences of major bleeding were not significantly lower with apixaban compared with warfarin (2.9% vs 7.9%, P = .063). 27 For patients with a BMI of ≥50 kg/m2, the incidence of strokes or major bleeding was similar between cohorts.
Summary: Data from healthy volunteers and patients with AF or VTE have shown that increased body weight has a modest or no effect on apixaban's PK. Standard dosing of apixaban is effective and safe for VTE and AF patients with morbid obesity.
Rivaroxaban
PK/PD data: Four studies have evaluated the impact of obesity on the PK/PD of rivaroxaban. In one study evaluating PK/PD and safety of rivaroxaban in 48 healthy subjects who received a single dose of rivaroxaban 10 mg, 31 peak concentration of rivaroxaban, AUC, T1/2, and anti-Xa activity were similar between the >120 kg (n = 12) and 70 to 80 kg (n = 12) groups. Low Vd may limit the impact of weight on the PK of rivaroxaban. 31 One prospective cohort analysis has evaluated the impact of morbid body weight on the effect of rivaroxaban in 167 patients with VTE, there was no significant difference in rivaroxaban peak levels, PT ratios (PT patient/PT normal), or activated partial thromboplastin time ratios for patients weighing >120 kg (n = 44) compared with those weighing 50 to 120 kg (n = 135). 32
However, 2 studies have contrary results regarding rivaroxaban peak levels. In a study of patients weighting >120 kg taking DOACs for any indication, of 21 patients receiving rivaroxaban, 6 (28%) had peak levels below the usual on-treatment range for the peak derived from population pharmacokinetic studies. 16 Similarly, in the previously referenced study measuring DOACs anti-Xa levels in patients weighting ≥120 kg conducted by Martin et al, of the 58 rivaroxaban levels, 26 (45%) peak levels were below expected ranges (11/22 for AF and 15/36 for VTE), and all 21 trough levels were within expected ranges. 24
Four population PK model analyses have been conducted to investigate the impact of obesity on DOAC PK/PD. In a population PK model based on pooled data from 2 phase II studies of rivaroxaban in acute DVT treatment (EINSTEIN-DVT and ODIXa-DVT trials), body weight had no significant influence on peak concentration. 33 However, Vd decreased by 0.8% per kg below the median low body weight of 56 kg, and directly correlated with body weight. 33 Therefore, Vd levels may increase in higher-weight patients based on this model.
The other 3 population PK models suggested weight had a minor influence on the PK of rivaroxaban. An early PK model based on 101 patients receiving rivaroxaban for prevention or treatment of VTE suggested that weight alone showed little effect on the PK profile of rivaroxaban, with creatinine clearance being the most significant factor impacting rivaroxaban exposure. 34 This was confirmed by a large pooled population PK model across all indications derived from 4918 patients in 7 clinical trials, which also showed that body weight alone had a minor influence on the PK of rivaroxaban, while a renal function is the main driver of rivaroxaban exposure. 35 Finally, in a population PK model derived from 913 patients (including 86 with weight >120 kg and 74 with BMI >40 kg/m2), median AUC and peak concentration were lower in simulated patients of 200 kg and 150 kg compared with 70-kg simulated patients; however, the prediction intervals overlapped across all bodyweights. 36 The authors concluded that weight had only a minor influence on rivaroxaban PK parameters. 36
Clinical data for VTE: Several observational studies presented data for rivaroxaban use in VTE patients with morbid obesity. Overall, real-world data demonstrated at least similar efficacy and bleeding outcomes with rivaroxaban in this patient population. A cohort study using electronic health record data including over 13 000 obese patients (3394 with BMI ≥ 40 kg/m2) with incident VTE found that rivaroxaban reduced the hazard of VTE compared with warfarin without impacting major bleeding at 3, 6, and 12 months. 37 No statistical difference was found across patients with class I to III obesity for either recurrent VTE (Pinteraction ≥ 0.43) or major bleeding (Pinteraction ≥ 0.58). 37 In a retrospective study based on health insurance claims data, rivaroxaban users had a significantly lower risk of VTE recurrence (HR 0.85, 95% CI 0.7-0.97) and similar major bleeding (HR 1.11, 95% CI 0.89-1.37) compared with warfarin users at 12 months among VTE patients with obesity (including 41.1% and 41.9% with BMI ≥40 kg/m2 in rivaroxaban and warfarin cohort, respectively). 38 A second study using administrative claims data found morbidly obese VTE patients receiving rivaroxaban had similar risks of recurrent VTE (odds ratio [OR] 0.99, 95% CI 0.85-1.14) and major bleeding (OR 0.75, 95% CI 0.47-1.19) compared with warfarin. 39 In a multicenter, retrospective cohort study comparing outcomes in patients with weight >120 kg or BMI >40 kg/m2 receiving rivaroxaban or warfarin for acute VTE, no difference was observed in hazard of VTE (HR 0.69, 95% CI 0.42-1.08) or major bleeding (HR 1.29, 95% CI 0.66-2.30). 40 A single-center retrospective study comparing efficacy and safety for VTE or AF patients with BMI ≥40 kg/m2 taking rivaroxaban or warfarin showed the incidence of recurrent VTE (2.0% vs 1.2%, P = .74) or major bleeding (1.3% vs 2.4%, P = .77) was similar between the treatment cohorts in patients with VTE. 27
Clinical data for AF: As with those for VTE, in the aforementioned study, the incidence of stroke was also similar (2.3% vs 1.3%, P = .71) between the treatment cohorts in patients with AF. A lower risk of major bleeding in patients on rivaroxaban compared with warfarin (2.9% vs 7.9%, P = .063) was observed but this difference was not significant. 27 Similarly, in the cohort of obese Veterans Health Administration system, among AF patients with BMI >40 kg/m2, compared with warfarin, rivaroxaban had similar risks of ischemic stroke (HR 1.1, 95% CI 0.84-1.41) and significantly lower risk of any major bleeding (HR 0.66, 95% CI 0.58-0.75). 20 In a retrospective cohort study of AF patients with obesity, the subgroup analysis in patients with BMI ≥40 kg/m2 (n = 7647 in each cohort) showed that lower risks of the composite outcome of stroke and systemic embolism (HR 0.83, 95% CI 0.72-0.96) and major bleeding risk (HR 0.80, 95% CI 0.64-1.00) were found in the rivaroxaban cohort compared with the warfarin cohort. 41 A large retrospective cohort study using Health insurance claims data found the risks of stroke/systemic embolism (HR 0.66, 95% CI 0.48-0.91) and major bleeding (HR 0.72, 95% CI 0.55-0.95) were significantly lower among morbidly obese patients in the rivaroxaban cohort compared to those in the warfarin cohort at 36 months. 42 In another retrospective cohort study using a healthcare claims database for AF patients with obesity demonstrated a lower risk of stroke/systemic embolism (HR 0.82, 95% CI 0.70-0.95) and a similar risk of major bleeding with rivaroxaban versus warfarin among morbidly obese patients (HR 0.95, 95% CI 0.74-1.23). 43
Summary: Current evidence indicates that the PK profile of rivaroxaban is not significantly impacted by body weight. Standard dosing of rivaroxaban is effective and safe for both VTE and AF patients with morbid obesity.
Edoxaban
PK/PD data: In a pharmacokinetic study measuring DOACs levels in 58 AF patients with morbid obesity (BMI ≥ 40 kg/m2), 13 (22.4%) were taking edoxaban, with 4 receiving 30 mg daily and 9 receiving 60 mg daily. The out-of-range plasma concentrations were more likely to receive edoxaban 30 mg once daily treatment (33% vs 2%; P < .01) compared with in-range plasma concentrations. 17 A post hoc analysis of the ENGAGE AF-TIMI 48 trial found trough edoxaban concentration (P = .28) and anti-Xa activity (P = .73) were similar across BMI groups ranging from 18.5 to >40 kg/m2. 44
Clinical data for VTE: No studies analyzing edoxaban for treatment or prevention of VTE in morbidly obese patients were identified.
Clinical data for AF: Only 2 studies evaluating edoxaban use for AF patients with morbid obesity, and similar efficacy and safety outcomes were found when compared with warfarin or compared extreme weight groups with the reference group. The post hoc analysis of the ENGAGE AF-TIMI 48 trial found the efficacy and safety profiles (stroke/systemic embolism, all-cause mortality, major bleeding, and major or CRNM bleeding) of edoxaban 60 mg once daily versus warfarin were similar across BMI categories ranging from 18.5 to >40 kg/m2 (Pinteraction ≥ 0.16 for each outcome). 44 The similar PK and PD results with edoxaban across the range of BMIs observed in the same study support the clinical observations. In the post hoc analysis of the ETNA-AF-Europe trial evaluated 1-year outcomes in AF patients treated with edoxaban across a range of body weight groups, edoxaban seemed associated with a very low occurrence of stroke and bleeding, and there were no differences in the risks of stroke/SE (HR 1.14, 95% CI 0.48-2.71), major bleeding (HR 0.67, 95% CI 0.27-1.66), major or CRNM bleeding (HR 0.98, 95% CI 0.57-1.69), and intracranial hemorrhage (HR 0.93, 95% CI 0.17-5.10) among body weight groups after adjustment for eGFR and CHA2DS2-VASc score. 45 However, the morbid obesity cutoff point was >100 kg, which may not be extrapolated to patients with BMI >40 kg/m2 or weight >120 kg.
Summary: Based on current evidence, standard dosing of edoxaban may be effective and safe for stroke prevention for AF in patients with morbid obesity. Given the lack of data, edoxaban should be avoided for VTE treatment or prevention in patients with morbid obesity.
Betrixaban
Both PK/PD and clinical studies were not identified regarding the effects of weight on betrixaban.
Limitations
Our review has several limitations. First, most included studies evaluating morbidly obese patients were retrospective studies or post hoc analyses of RCTs, there is sampling bias due to the lack of randomization. Second, the PK/PD profile is obtained from different sources, thus differences in the timing of sampling, data quality, or laboratory analysis may exist. Third, the majority of included studies evaluated apixaban or rivaroxaban in morbidly obese patients with AF or VTE, with limited or no evaluation of edoxaban, dabigatran, or betrixaban, especially in VTE patients. Future higher-quality studies to confirm the uncertainty of DOAC use in morbidly obese patients are still warranted.
Conclusion
PK variations and clinical outcomes of each DOAC are specific. Obesity may have a modest effect on dabigatran, apixaban, rivaroxaban, or edoxaban. Current available evidence indicates that standard dosing of apixaban or rivaroxaban is recommended in VTE or AF patients with morbid obesity; Standard dosing of edoxaban may be considered in AF patients; Dabigatran should be used with caution in patients with AF; There is limited evidence regarding dabigatran and edoxaban use in morbid obese VTE patents.
Footnotes
Authors’ Contributions
XC designed the study. MG and WX identified and acquired reports of trials and extracted data. YZ, MG, WX, CP, DL, CH, and XC contributed to data interpretation. YZ drafted the paper, and all other authors critically reviewed the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
