Abstract
We aimed to investigate the efficacy and safety of rivaroxaban for acute and long-term management of cerebral venous sinus thrombosis (CVST). This study reviewed CVST-diagnosed patients admitted to the First Affiliated Hospital of Guangxi Medical University from January 2015 to December 2020. The primary outcome was a composite of recurrent thrombosis or major bleeding events. The secondary efficacy outcomes included a disease recovery time (DRT) presenting the time from admission to the endpoint as recovery (the modified Rankin scale [mRS] score [0-1]) within 30 and 90 days, and length of hospital stay (LHS). Patients treated with rivaroxaban (38) and warfarin (45) were enrolled in the final analysis. The primary outcome had no significant difference (5.3% vs 11.1%, P = .576) between the 2 groups. The secondary efficacy outcome regarding the median 30-d DRT was 17 days (95% confidence interval [CI], 14.6-19.4) in the rivaroxaban group, compared with 26.0 days (95% CI, 16.8-35.2) in the warfarin group (hazard ratio, 1.806; 95% CI, 1.051-3.103; log-rank P = .026). Two groups have a significant difference in LHS (P = .041). Patients with cerebral edema, intracerebral hemorrhage, and mild/moderate disability (admission mRS score [2-3]) treated with rivaroxaban recovered faster than those with warfarin (log-rank P < .05). Patients with cerebral edema, intracerebral hemorrhage, and mild/moderate disability treated with rivaroxaban had a shorter recovery time than those treated with warfarin within 1 month from admission, indicating that rivaroxaban a promising convenient therapy for CVST, helping them speedily restore social functions.
Introduction
Cerebral venous sinus thrombosis (CVST) is a rare neurovascular disorder that involves the formation of blood clots in the dural venous sinuses or cerebral veins; CVST has highly variable causes and clinical presentations1,2. The incidence of CVST is approximately 1.3 to 1.6 cases per 100,000 persons in high-income countries, and this incidence is even higher in low/middle-income countries 3 . Advances in neuroimaging techniques have improved the diagnoses of CVST 4 . When the standardized management procedures of CVST in the acute stage are followed, approximately 75% of CVST patients achieve a full recovery 5 . However, previous studies have found that patients with a CVST history are at an increased risk of recurrent venous thrombosis5–7. The current guidelines recommend the use of an oral anticoagulant (OAC), such as warfarin, at a standardized intensity (target international normalized ratio [INR] between 2.0 and 3.0) for 3 to 12 months, as long-term anticoagulation therapy5,8. This is to prevent recurrent venous thrombotic events (VTEs) after initial anti-coagulation therapy with low-molecular-weight heparin (LMWH)5,8. Recently, given that routine INR monitoring is no longer required, the use of direct oral anticoagulants (DOACs) has become more favorable for the management of venous thromboembolism and atrial fibrillation, than vitamin K antagonists (VKAs)9,10. Additionally, DOACs have fewer drug interactions and lower rates of intracranial hemorrhage than warfarin and are an effective, safe, and convenient alternative to VKAs9,10.
However, owing to the lack of supporting evidence, the current guidelines do not recommend the use of DOACs, such as dabigatran and rivaroxaban, for patients with CVST5,8. A recent multicenter, prospective, randomized clinical trial, RE-SPECT CVST, which included 120 participants with CVST that were randomly assigned to either dabigatran or warfarin treatment for 24 weeks, found that both dose-adjusted warfarin and dabigatran are safe and effective in preventing recurrent VTEs in patients with CVST 11 . Nevertheless, randomized controlled trials (RCTs) that investigate the efficacy of rivaroxaban therapy for CVST, such as CeREbral Venous Thrombosis (SECRET), are still ongoing 12 , and the study did not include the Chinese population.
Therefore, in the present study, we aimed to investigate and compare the clinical outcomes of patients with CVST who were treated with either rivaroxaban or warfarin, to evaluate the efficacy and safety of rivaroxaban for the treatment of CVST.
Patients and Methods
Study Population
In this single-center, retrospective study, we reviewed the electronic medical records of patients who were admitted to the First Affiliated Hospital of Guangxi Medical University, China from January 2015 to December 2020, who had a confirmed clinical diagnosis of CVST 13 , confirmed by neuroimaging, and who received LMWH as the initial anticoagulation therapy. The diagnosis of CVST was based on typical CVST clinical signs and symptoms, and was confirmed by strict neuroimaging techniques, such as magnetic resonance imaging venography, computed tomography venography, or digital subtraction angiography 13 . Only those patients who achieved clinical stability at discharge, after receiving acute CVST anticoagulant treatment, were included in our study. The following exclusion criteria were applied: aged ≤ 18 years old; combined diagnosis of CVST with head, face, and central nervous system infection or craniocerebral trauma; CVST with life-threatening complications; did not receive OAC therapy during hospitalization or received OAC therapy for less than 3 months; underwent endovascular recanalization; no recorded follow-up visits.
This study was approved by the ethics committee of the First Affiliated Hospital of Guangxi Medical University and was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from the patients or from their legally authorized guardians.
Anticoagulant Therapy and Patient Outcomes
LMWH was chosen for acute anticoagulant treatment; LMWH was bridged to long-term OAC therapy, which was comprised of either rivaroxaban or warfarin. Discharged patients were required to visit the hospital or the local clinics regularly. The initial dose of rivaroxaban was varied between 15 mg twice a day and 20 mg daily 14 . The traditional dose of rivaroxaban (15 mg twice daily for 3 weeks followed by 20 mg once daily) was cited from the Oral Rivaroxaban for Symptomatic Venous Thromboembolism by The EINSTEIN Investigators, which offers a simple, single-drug approach to the short-term and continued treatment of venous thrombosis that may improve the benefit-to-risk profile of anticoagulation 15 . Standard-dose rivaroxaban (20 mg once daily) is for routine use in China, for high risk of recurrence of DVT/PE and bleeding, such as these patients with complex complications 14 . Dose determination and dosing were performed by outpatient neuroscience specialists. Similarly, the dosage of warfarin was adjusted to maintain an INR of between 2.0 and 3.0. Based on previous studies, the duration of OAC therapy for CVST was determined based on underlying risk factors for recurrence and bleeding, and lasted for at least 3 months16,17, except for patients who discontinued OAC therapy because of symptom relief.
Patients recruited into the study were divided into 2 groups, according to the chosen OAC: rivaroxaban or warfarin, which mainly depended on the treating physicians’ decision. We collected the relevant clinical data and recorded the modified Rankin scale (mRS) score at the time of patient admission, discharge, and follow-ups to assess the efficacy and safety of these 2 OACs. The follow-up information was obtained from clinical visits and/or telephone interviews, and neuroimaging data was available until May 2021.
Baseline characteristics included gender (female), age, smoking history, thrombophilia, history of stroke/transient ischemic attack (TIA), infection, surgical history, D dimer level at admission, symptoms (headache, epilepsy, paralysis, aphasia, visual impairment), brain lesion (edema, hemorrhage, venous infarction), involved vessels, duration of LMWH use, start time of OAC, mRS score at admission.
The primary outcome was a composite of recurrent thrombosis events or major bleeding while on anti-coagulation 11 . Recurrent thrombosis events, including both venous and arterial events, were defined as any of the following: recurrence of CVST, deep venous thrombosis of any limb, pulmonary embolism, splanchnic vein thrombosis, myocardial infarction, and ischemic stroke 18 . Major bleeding referred to fatal bleeding, leading to a decrease in hemoglobin level of at least 20 g/L, the need for a blood transfusion of at least 2 units, and/or substantial bleeding in a vital organ or critical area (including an intracranial, pericardial, retroperitoneal, intraocular, intraspinal, intraarticular, or intramuscular area) 19 .
The secondary efficacy outcomes were 30-d disease recovery time (DRT) and 90-d DRT, measured using the mRS score, length of hospital stay (LHS), duration of OAC use, and cerebral venous and sinus recanalization, as measured by the change in the score of occluded cerebral veins and sinuses 20 .
MRS score was to measure the ability of patients for living independently, including physical function, mobility, and participation in daily life 21 , which was measured by specialized neurologists determined at admission, at discharge, and at follow-up visits within 30d and 90d of admission. MRS score of 0 to 1 indicates complete recovery or the absence of obvious disability, meaning that patients were able to complete daily activities despite having mild symptoms; the score of 2 to 3 represents patients had mildly or moderately disabled; the score of 4 to 5 indicates patients with severe disability and the score of 6 represents death. The DRT was defined as the time from admission to the occurrence of a good outcome, which was in turn defined as an mRS score of 0 to 1 14 .
DRT referred to the time to the endpoint of recovery, while 30-d and 90-d DRT referred to the time to recovery within 30 and 90 days, respectively. The cerebral venous and sinus recanalization was measured by the change in the number of occluded cerebral veins and sinuses after up to 30 days from admission, was presented as no change or as worsened (if there was at least 1 new vein or sinus occluded) or improved (if there was at least 1 vein or sinus recanalized better than before) 11 . This was based on evidence obtained by 2 neurologists from the last follow-up computed tomography/magnetic resonance (CT/MR) angiography.
The secondary safety outcomes included minor bleeding events (such as gingival bleeding, epistaxis, skin ecchymosis, and increased menstruation), adverse effects post-CVST, venous thrombosis-associated death, and all-cause death. Adverse effects post-CVST referred to paroxysmal headaches and memory loss, usually caused by fatigue and weather changes. The clinical signs and symptoms assessed during this study included headaches, seizures, pareses, aphasias, and visual symptoms. The risk factors for thrombophilia included the presence of factor V Leiden and factor II G20210A mutations, protein C/S or antithrombin III deficiency, hyperhomocysteinemia, hematological system diseases, antiphospholipid antibody syndrome, malignancy, long-term oral contraceptive use, and pregnancy/puerperium.
Statistical Analyses
To compare the differences between the groups, Student's t-test was used to analyze fibrinogen level on admission, and to analyze age, body mass index (BMI), platelet count, and activated partial thromboplastin time (APTT) at the time of discharge.
The Mann–Whitney U-test was used to compare the hemoglobin counts, mean platelet volumes (MPV), prothrombin times (PT), INRs, and thrombin times (TT) at the time of patient admission and discharge; mRS and APTT at the time of admission; fibrinogen level at the time of discharge; and D-dimer level, serum creatinine (SCr) level, duration of LMWH and OAC treatment, start time of OAC, time to last magnetic resonance imaging (MRI) at the time of admission, and LHS between the 2 treatment groups. The Chi-square test was applied to the categorical variables to compare the differences in frequency and percentage between the 2 groups.
A Cox model was used to estimate the hazard ratio (HR) for comparison of the speed of recovery between the groups. Kaplan–Meier curves were created to estimate the cumulative incidence of 30-d and 90-d recovery. Based on these analyses, we calculated the range of the median survival value and the 2-sided 95% confidence interval (CI).
We applied the log-rank test and calculated the log-rank P-value to compare DRTs between the 2 groups. To compare the between-group DRT and estimate the effect of this variable, P values and HRs were calculated using Cox regression. We also used the multivariable Cox proportional-hazards model to assess the independent effects of baseline characteristics on recovery time.
All tests were 2-tailed and P < .05 was considered statistically significant. The statistical analyses were conducted using SPSS v26.0 (IBM Inc., Chicago, IL, USA).
Results
Demographical Characteristics and Clinical Data of the Patients
A total of 83 patients meeting the inclusion criteria were finally enrolled in our study (Table 1), wherein 38 and 45 patients were treated with rivaroxaban and warfarin, respectively. Of the 38 rivaroxaban-treated patients, 6 patients received the traditional dose of rivaroxaban (15 mg twice daily for 3 weeks followed by 20 mg once daily) compared with 32 directly treated with standard-dose rivaroxaban (20 mg once daily) (15.7% vs 84.2%).
Demographical Characteristics and Clinical Data of the Patients.
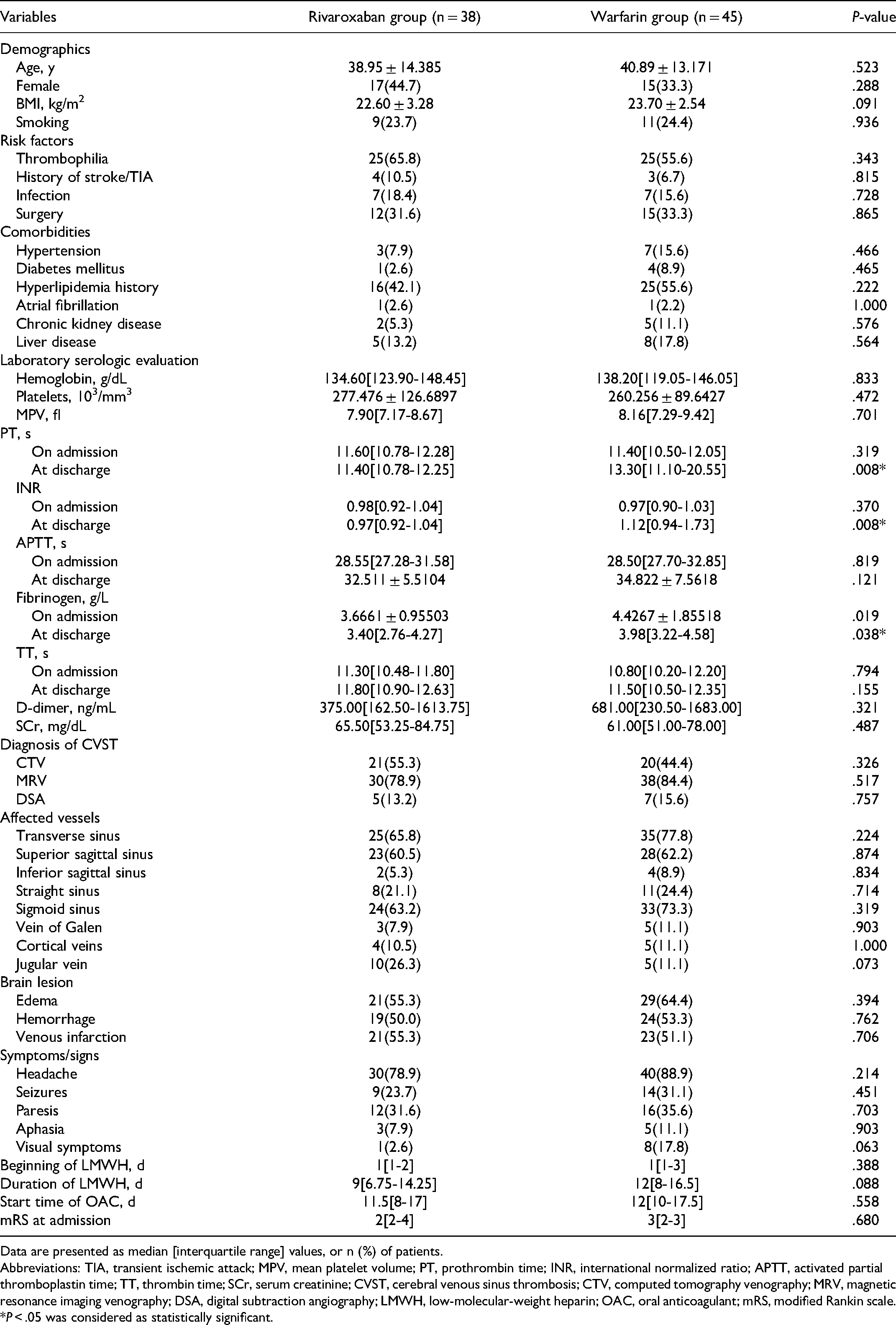
Data are presented as median [interquartile range] values, or n (%) of patients.
Abbreviations: TIA, transient ischemic attack; MPV, mean platelet volume; PT, prothrombin time; INR, international normalized ratio; APTT, activated partial thromboplastin time; TT, thrombin time; SCr, serum creatinine; CVST, cerebral venous sinus thrombosis; CTV, computed tomography venography; MRV, magnetic resonance imaging venography; DSA, digital subtraction angiography; LMWH, low-molecular-weight heparin; OAC, oral anticoagulant; mRS, modified Rankin scale.
*P < .05 was considered as statistically significant.
These 2 groups were well matched with the population characteristics and clinical data (Table 1), and no significant differences were observed in age, sex, BMI, cigarette smoking, and potential thrombosis risk factors such as thrombophilia and history of recent surgery, infection, and stroke/TIA (P > .05) between the 2 groups. No significant differences between the 2 groups were detected in terms of the prior medical history of hypertension, diabetes, hyperlipidemia, or comorbidities including atrial fibrillation, chronic kidney disease, and liver disease. The laboratory serologic levels were similar in both groups, except for PT, INR, and fibrinogen levels at discharge; We supposed PT and INR levels were higher in patients who received warfarin, as a result of the need to reach the target INR between 2.0 and 3.0 through warfarin dose adjustment (P = .008; P = .008; and P = .038, respectively; Table 1). Although fibrinogen levels at discharge were significantly higher in the warfarin group, they were still within the normal ranges in both groups of patients. Our study mainly compared the differences in characteristics of predrug clinical presentation in terms of sub-acute, including headache, seizures, paresis, aphasia, and visual symptoms, and showed no significant difference between the 2 groups. As for the diagnosis strategy for CVST, symptoms, affected vessels, brain lesion, the beginning of LMWH, duration of LMWH, and the start time of OAC use, the 2 groups showed similar characteristics, indicating a good match. Besides, the mRS on admission showed no significant difference between the 2 groups.
Primary Outcome and Secondary Efficacy and Safety Assessment
Primary and Secondary Efficacy Outcomes
The primary outcome was a composite of recurrent thrombosis or major bleeding events. In the rivaroxaban group, one patient had major gastrointestinal bleeding and one had a recurrent stroke, without recurrent VTEs. In the warfarin group, one patient had a recurrent stroke, and 4 had recurrent VTEs, including 3 recurrent CVST and one recurrent lower extremity deep venous thrombosis. Among the patients with recurrent CVST, one patient had an episode of intracranial hemorrhage (bleeding area <5 mL) after 3 months of therapy, and the INR detected at the time of bleeding was 3.64. The INR detected during the period of recurrent CVST during the second month of therapy was 1.04-2.45. None of the major bleeding events resulted in death. Within the cohort, one patient of the rivaroxaban group died of breast cancer and 2 in the warfarin group died of pulmonary infection and acute heart failure, respectively. Overall, the rivaroxaban group tended to have decreased incidence of recurrence of a thrombus or a major bleeding event, but the difference in incidence was not significant when compared with that of the warfarin group (5.3% vs 11.1%, P = .576; Table 2).
Primary and Secondary Outcomes.

Abbreviations
In our study, we found that the mRS scores measured at the admission of all patients were ≥2. By comparing the time from admission to the occurrence of a good outcome, which is defined as an mRS score of 0 to 1, between patients treated with rivaroxaban and those treated with warfarin, we could observe the secondary efficacy outcome of the 2 drugs and the factors affecting CVST prognosis. The median 30-d DRT was 17 days (95% CI, 14.6-19.4) in the rivaroxaban group and 26 days in the warfarin group (95% CI, 16.8-35.2), with a significant difference (HR, 1.806; 95% CI, 1.051-3.103; log-rank P = .026; Figure 1). One month after admission, patients treated with rivaroxaban had a shorter recovery time than those treated with warfarin, and there was a statistically significant difference in the LHS (P = .041; Table 2). The multivariate analysis showed that cerebral edema, intracerebral hemorrhage, mRS score at admission, and OAC starting time were significant factors affecting 30-d DRT (P = .010; P = .014; P = .000; P = .001, respectively; Table 3) and rivaroxaban was revealed to be a favorable factor for 30-day discovery (HR: 1.871, 95% CI, 1.048-3.339, P = .034; Table 3). However, the 90-d DRT did not differ significantly between the 2 groups (HR, 1.523; 95% CI, 0.935-2.483; log-rank P = .082; Figure 2). Besides, there were no significant differences in the OAC use duration (P = .341; Table 2). Cerebral venous recanalization, assessed as a change in the score of occluded cerebral veins and sinuses between baseline and the last neuroimaging, showed improvements in the 23 patients (60.5%) in the rivaroxaban group and 26 (57.8%) in the warfarin group, but revealed no significant differences in these 2 groups (P = .800; Table 2). The time from the first imaging to the last in the 2 groups, which ranged from 1.5 to 5.475 months in the rivaroxaban group and 1 to 6 months in the warfarin group, also revealed no significant difference. In subgroup analysis, patients with cerebral edema, intracerebral hemorrhage, and mild/moderate disability (admission mRS score [2-3]) treated with rivaroxaban recovered faster than those with warfarin, the difference was statistically significant (log-rank P < .05; Figure 3).

Kaplan–Meier curves estimating the cumulative incidence of recovery within 30 days. The median 30-d DRT was 17 days (95% CI, 14.6-19.4) in the rivaroxaban group and 26 days in the warfarin group (95% CI, 16.8-35.2), revealing a significant difference (log-rank P = .026). The recovery was considered as a good outcome which referred to an mRS score of 0 to 1. The hazard ratio (HR) was used to compare speeds of recovery by a univariable Cox model with recovery as an end point, was 1.806 (95% CI, 1.051-3.103) in favor of the rivaroxaban group.

Kaplan–Meier curves estimating the cumulative incidence of recovery within 90 days. The 90-d DRT did not differ significantly between the 2 groups (HR, 1.523; 95% CI, 0.935-2.483; log-rank P = .082).

Kaplan–Meier curves estimating the cumulative incidence of recovery within 30 days for each subgroup.
Multivariate Cox Proportional Hazards Model of 30-d Disease Recovery Time.

Secondary Safety Outcomes
Differences were observed among all secondary safety outcomes, including minor bleeding events (7.9% vs 8.9%), adverse effects, post-CVST (13.2% vs 24.4%), VTE-associated death (0% vs 0%), and all-cause death (2.6% vs 4.4%); however, these differences were not statistically significant (P > .05; Table 2).
Discussion
As a new OAC that could inhibit factor Xa directly, rivaroxaban has been approved for the acute/long-term treatment of deep vein thrombosis and as a precautionary measure for systemic embolism or stroke in patients who have suffered from atrial fibrillation since December 2011, given its non-inferior clinical efficiency to warfarin and the less frequent intracranial and fatal bleeding events22–24. Recent studies have shown that rivaroxaban has advantages in terms of efficacy and safety, compared with warfarin. In a meta-analysis of previous real-world studies, rivaroxaban was shown to be more effective in preventing stroke and thromboembolism in patients with atrial fibrillation and was associated with a decreased risk of intracranial hemorrhage, than warfarin 25 . Another study found that morbidly obese patients with atrial fibrillation who were treated with rivaroxaban had lower healthcare resource utilization and costs than those treated with warfarin 26 .
Thus far, evidence supporting the use of oral factor Xa inhibitors for CVST treatment has mostly been based on small-sample case reports and retrospective studies. In a study by Geisbüsch et al 14 , a similar clinical benefit of rivaroxaban to that of warfarin was found for the first time in 7 patients receiving CVST treatment. Other studies comparing DOACs with warfarin had similar results, that is, an oral factor Xa inhibitor tended to be a reasonable option for CVST-patient therapy, and were non-inferior to VKAs in terms of efficacy and safety27,28. Based on these previous studies, we conducted a study with a relatively larger sample size and offered a more specific comparison between rivaroxaban and warfarin. We compared the efficacy and safety of rivaroxaban and warfarin and investigated the recovery time of CVST between these 2 groups. This is the first study to reveal an advantage of rivaroxaban in recovery efficiency.
In the RE-SPECT CVST trial, the primary composite outcome of recurrent VTE and major bleeding revealed no difference when comparing dabigatran with warfarin 11 , which provided promising evidence for dabigatran use in the treatment of CVST. Our study was based on this primary endpoint, and observed the recurrent thrombosis events, including both venous and arterial events or a major bleeding event while on anti-coagulation, where no differences in the use of rivaroxaban or warfarin were found as well. We found the risk of recurrent thrombosis events or a major bleeding event in patients with CVST who received regular and continuous anticoagulant therapy, with either rivaroxaban or warfarin, for at least 3 months was low. Although not statistically significant for the low number of cases, the primary outcome occurred with half the frequency in the rivaroxaban group than in the warfarin group, mainly due to a higher thrombotic recurrence or major bleeding event in the latter. The decreased effectiveness of warfarin therapy is likely due to inadequate control of INR. These results are in line with evidence of previous studies that rivaroxaban is at least non-inferior in efficacy compared with warfarin in indications other than CVST and presents a tendency to have fewer recurrent arterial/venous thrombotic events.
Among the secondary efficacy outcomes, we mainly found an advantage in recovery time with rivaroxaban. Patients treated with rivaroxaban had a shorter recovery time than those treated with warfarin within 1 month from admission (HR, 1.806; 95% CI, 1.051-3.103; log-rank P = .026; Figure 1); moreover, rivaroxaban group patients had a significantly shorter LHS than warfarin group (P = .041; Table 2). Another study showed that rivaroxaban therapy resulted in a significantly shorter LHS along with lower healthcare resource utilization and costs than warfarin treatment for atrial fibrillation in morbidly obese or overweight patients 26 , which is consistent with our results. These results are also in line with the findings of a comparative study on the use of rivaroxaban and warfarin in non-valvular atrial fibrillation and venous thromboembolism patients27,29,30. The ongoing SECRET trial is an open-label RCT trial that randomizing individuals with CVST 1:1 to rivaroxaban 20 mg daily with warfarin or LMWH therapy 12 . However, this study did not investigate the recovery time of CVST and did not include any Chinese population. The results drawn from our study were consistent with the studies mentioned above, which assessed the use of rivaroxaban for CVST treatment. These significant differences in the time of recovery may be related to its characteristic of rapid onset, persistent efficacy, no need for routine monitoring and routine dose adjustment, as a kind of new OAC that can directly inhibit Xa factor15,31,32. On the other hand, our study results reflected that patients with warfarin took longer time to achieve a satisfying therapeutic purpose, which may be due to the need for time to reach the target INR between 2.0 and 3.0 through warfarin dose adjustment.
Subgroup analysis was performed in order to better exclude the factors affecting the recovery time. Patients with cerebral edema, intracerebral hemorrhage, and mild/moderate disability (admission mRS score [2-3]) treated with rivaroxaban recovered faster than those with warfarin, the difference was statistically significant (log-rank P < .05; Figure 3). Shorter recovery time is found in patients with cerebral edema and intracerebral hemorrhage; this may be related to fewer incidents of intracranial hemorrhage caused by rivaroxaban compared with warfarin 25 .
There was no significant difference between the 2 groups in patients with severe disability, which may indicate that the 2 drugs could not achieve a good prognosis early in patients with severe neurological impairment and that there was no significant difference in the effect of the 2 groups on early recovery time.
In addition, in this study, we excluded patients receiving intravascular recanalization and found there was no difference in the risk of recurrent thrombotic events and long-term recovery time within 90 days of admission, which had similar results in a previous study 18 , it shows that in the majority of CVST patients who do not undergo endovascular recanalization, there may be no significant difference in thrombotic recurrence after long-term anticoagulant therapy.
Concerning the recanalization of cerebral venous sinus, there was no significant difference between the 2 groups in our study. A recent meta-analysis of CVST demonstrated that DOACs have similar efficacy and safety to those of VKAs, but a better recanalization rate. One possible explanation for this difference may be that the studies included in this meta-analysis had inconsistencies in the type of DOACs used, as well as included other DOACs other than rivaroxaban 33 .
In the multivariate analysis, we found that cerebral edema, intracerebral hemorrhage, mRS score at admission, and OAC starting time could be significant factors for the 30-d DRT (P = .010; P = .014; P = .000; and P = .001, respectively; Table 3). Severe brain edema is known to be a poor prognostic indicator of CVST, which is probably linked to the edematogenic and inflammatory responses in the brain tissue after CVST 34 . Dhadke et al also found that cerebral hemorrhage and cerebral edema were the most common adverse prognostic factors and causes of death in CVST patients 35 . International ISCVT studies with large samples have found that CVST combined with intracranial hemorrhage was a significant prognostic risk factor for dependency or death 5 . Multiple studies of CVST in the same period of follow-up visits ranging from 6 months to 96 months found that intracranial hemorrhage was the important risk factor for poor prognosis in CVST patients36–38. The results of our study are consistent with those of previous studies, and it is found that CVST patients with cerebral edema and intracranial hemorrhage may take longer time to reach the recovery event at an early stage.
Although both groups presented similar efficacy and safety, rivaroxaban therapy was significantly associated with lower hospitalization days and recovery time than warfarin therapy, thus helping patients restore their social functions faster.
This study had several limitations. First, despite the 2 groups being matched well before the statistical analyses, the retrospective nature inevitably caused several defects. Patients who received rivaroxaban/warfarin mainly depended on the treating physicians’ decision. Besides, we were unable to compare the follow-up imaging at the same time between groups. We also failed to continuously monitor the INR of patients; thus, we could not determine the time in the therapeutic range, indicating the ratio of days with INR between 2.0 and 3.0 to the total days of use, which was used as a measure of warfarin therapy quality 39 . Second, the sample size of our study was relatively small. Third, the 90-d mRS score was based on the follow-up visits obtained from clinical visits and/or telephone interviews, which could produce a certain recall bias. Therefore, more investigations should be carried out to examine our results.
Conclusion
To the best of our knowledge, this is the first study to compare the recovery time of CVST and the risk factors associated with CVST recovery in both the rivaroxaban and warfarin groups. Patients treated with rivaroxaban had a shorter recovery time than those treated with warfarin in our study within 1 month from admission. Cerebral edema, intracerebral hemorrhage, mRS score at admission, and OAC starting time were found to be risk factors for CVST recovery. Patients with cerebral edema, intracerebral hemorrhage, and mild/moderate disability (admission mRS score [2-3]) treated with rivaroxaban may recover faster than those with warfarin, indicating that rivaroxaban a promising convenient therapy for CVST, helping them speedily restore their social functions. Future studies with larger sample sizes are warranted to explore the use of rivaroxaban or other DOACs for CVST therapy.
Footnotes
Acknowledgments
This work was supported by the Natural Science Foundation of Guangxi Province (2013GXNSFBA019131, 2015GXNSFAA139171, 2020GXNSFAA259053) and the Guangxi Scholarship Fund of Guangxi Education Department. In addition, we thank Dr LWD from Guangzhou for valuable advice on data analysis. And, JYY wants to thank the persistent care and support from her good friends CY and QY.
Availability of Data and Materials
The datasets supporting the conclusions of this article are included within this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Guangxi Province (grant numbers 2013GXNSFBA019131, 2015GXNSFAA139171, 2020GXNSFAA259053).
Ethics Approval and Consent to Participate
This study was conducted with approval from the Ethics Committee of First Affiliated Hospital of Guangxi Medical University and the approval number is 2021(KY-E-120). This study was conducted in accordance with the declaration of Helsinki. Written informed consent was obtained from all participants.
