Abstract
To identify risk factors and develop a risk-prediction nomogram model for 1-year readmission due to major adverse cardiovascular events (MACEs) in patients with acute ST-segment elevation myocardial infarction (STEMI) after primary percutaneous coronary intervention (PCI). This was a single-center, retrospective cohort study. A total of 526 eligible participants were enrolled, which included 456 non-readmitted and 70 readmitted patients. Multivariate logistical regressions were performed to identify the independent risk factors for readmission, and a prediction nomogram model was developed based on the results of the regression analysis. The receiver operating characteristic curve, Hosmer-Lemeshow test, calibration plot, and decision curve analysis (DCA) were used to evaluate the performance of the nomogram. Female (OR = 2.426; 95% CI: 1.395–4.218), hypertension (OR = 1.898; 95% CI: 1.100–3.275), 3-vessel disease (OR = 2.632; 95% CI: 1.332–5.201), in-hospital Ventricular arrhythmias (VA) (OR = 3.143; 95% CI: 1.305–7.574), peak cTnI (OR = 1.003; 95% CI: 1.001–1.004) and baseline NT-proBNP (OR = 1.001; 95% CI: 1.000–1.002) were independent risk factors for readmission (all P < 0.05). The nomogram exhibited good discrimination with the area under the curve (AUC) of 0.723, calibration (Hosmer-Lemeshow test; χ2 = 15.396, P = 0.052), and clinical usefulness. Female gender, hypertension, in-hospital VA, 3-vessel disease, baseline NT-proBNP, and peak cTnI were independent risk factors for readmission. The nomogram helped clinicians to identify the patients at risk of readmission before their hospital discharge, which may help reduce readmission rates.
Introduction
ST-segment elevation myocardial infarction (STEMI) is an extremely aggressive type of cardiovascular disease that often leads to various cardiac events and sometimes even death. Primary percutaneous coronary intervention (PCI) is the preferred treatment for patients diagnosed with STEMI within 12 h of symptom onset. 1 Since the beginning of the twenty-first century, China has experienced a rapid increase in the prevalence and hospitalization rates of patients with STEMI, with hospital admissions increasing from 3.5 per 100,000 in 2001 to 15.4 in 2011. 2 Advances in treatment and the availability of PCI have reduced in-hospital mortality; nonetheless patients who survive STEMI remain at high risk for major adverse cardiovascular events (MACEs), and readmission following discharge. In the United States, PCI survivors have a high rate of readmission, from 6% to 17% across hospitals in different regions. 3 The 2010 to 2014 US Nationwide Readmissions Database showed that the 30-day readmission rate for all STEMI hospitalizations was 12.3%, with 41.6% readmitted for noncardiac reasons. 4 Readmissions result in a huge waste of medical resources and also cause a substantial increase in economic burden.3,4 Therefore, identification of potential and preventable risk factors for readmissions will enable healthcare facilities to focus on already limited resources and prevent unnecessary readmissions.
Many previous studies have focused on short-term or all-cause readmissions; however, long-term readmissions in STEMI patients have not been studied well, and all-cause readmissions do not reflect the true cardiovascular burden to provide sufficient and valuable information to the cardiologists. In this study, we investigated the factors influencing readmission for MACEs within 1 year in survivors of STEMI undergoing PCI and attempted to build a predictive nomogram model.
Methods
Study Population
This is a single-center, retrospective cohort study. In 2018, we reviewed the records of consecutive STEMI patients admitted to the First Hospital of Jilin University from January to December 2016 and conducted telephone callbacks to patients with STEMI who survived to discharge after PCI or their families to understand their physical condition, cardiac discomfort, readmission, and medication use within 1 year after discharge. Once the patients were diagnosed and without significant contraindications, they immediately underwent primary PCI in the first 12 h after admission. The exclusion criteria included (1) in-hospital death, (2) more than 10% data missing, and (3) readmission to the hospital for non-MACEs.
Data Collection
We included a set of variables based on literature review and their availability in our database. The primary source of data was the electronic medical records of the hospital, which included baseline demographics (age and gender), clinical data (admission information, alcohol intake, smoking habits, disease history, etc), angiographic and laboratory parameters (mainly serum biochemical indexes), and out-of-hospital medications. The study endpoint was the first readmission to any hospital for MACEs in patients with STEMI within 1 year after PCI, and information on readmissions was mainly collected through telephone callbacks.
Definitions
STEMI was diagnosed using standardized criteria following the 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction, 5 which stated that primary PCI should be performed in patients with STEMI and ischemic symptoms of less than 12-h duration. MACEs were defined as a composite of cardiac death, myocardial infarction, hospitalization for chest pain or congestive heart failure, late revascularization, or arrhythmia. 6 Number of years of cigarette smoking, to indicate cumulative smoking exposure (CSE), were calculated by the average number of cigarettes smoked per day multiplied by the duration of smoking in years. 7 Alcoholism was confirmed by assessing the amount of ethanol consumed, 8 considering the ethanol content of different alcoholic beverages. 9 Ventricular arrhythmias (VA) included ventricular fibrillation, ventricular tachycardia, non-sustained ventricular tachycardia (NSVT), and Lown's grade ≥2 premature ventricular complexes. 10
Statistical Analyses
The distributions of all quantitative variables were analyzed using the Shapiro-Wilk test. Normally distributed continuous data were presented as the mean ± standard deviation and were compared between the two groups using the independent samples t-test. Non-normally distributed continuous data were presented as median (interquartile range) and compared using the Mann-Whitney test. Categorical variables were presented as counts and percentages, and differences between two groups were compared by the chi-squared test or the Fisher exact test. Considering the results of univariate analysis results, clinical relevance, and the number of events available, we constructed two adjusted models with baseline covariates, including information on gender, age, hypertension, and diabetes mellitus, to correct for confounding factors and adjust the number of potential variables, before multivariate regression analysis. Multivariate logistical regressions were employed to identify the independent risk factors for rehospitalization. A nomogram was constructed based on the results of multivariate analysis. The performance of the nomogram was evaluated using the receiver operating characteristic (ROC) curve, Hosmer-Lemeshow test, calibration plot, and the decision curve analysis (DCA). All data analyses were performed using SPSS 26.0 (SPSS Inc., Chicago, IL, US) and R software (version 4.2.0, R Foundation for Statistical Computing, Vienna, Austria). A p-value < 0.05 (two-tailed) or the 95% confidence interval (CI) of the odds ratio (OR) excluding the value one was considered statistically significant.
Results
Baseline Characteristics
A total of 526 consecutive eligible patients were enrolled in this study; among these, 456 were non-readmitted patients and 70 were readmitted patients. The mean age of all participants was 59 years (standard deviation of 11 years), and 71.9% were male. Complete baseline and clinical characteristics of the study population are presented in Table 1. Compared with non-readmitted patients, those undergoing readmission were older (58.5 vs 61.4 years), more likely to be female and non-smokers, propensity to have hypertension and diabetes mellitus, a longer Symptom-Onset-to-Balloon time, the 3-vessel disease, a lower left ventricular ejection fraction (LVEF), and a greater prevalence of in-hospital VA. In terms of laboratory parameters, the readmitted group had higher levels of fasting plasma glucose (FPG), baseline NT-proBNP, peak cardiac troponin I (cTnI), and fibrinogen, and lower levels of albumin–globulin ratio (AGR) and hemoglobin (all P <0.05). There were no other statistically significant differences between the two groups in other outcomes (all P ≥0.05).
Baseline and Clinical Characteristics of Patients.
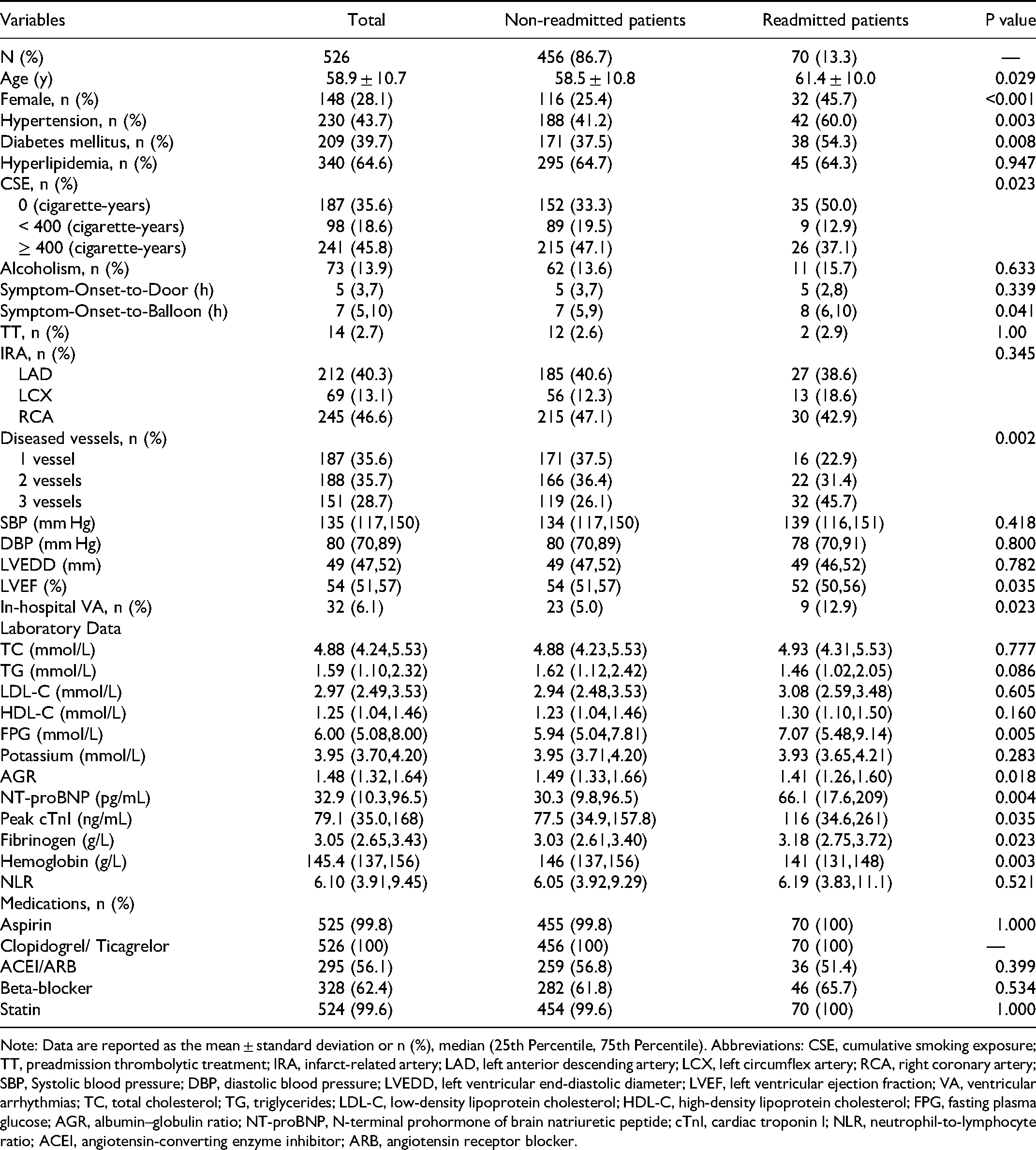
Note: Data are reported as the mean ± standard deviation or n (%), median (25th Percentile, 75th Percentile). Abbreviations: CSE, cumulative smoking exposure; TT, preadmission thrombolytic treatment; IRA, infarct-related artery; LAD, left anterior descending artery; LCX, left circumflex artery; RCA, right coronary artery; SBP, Systolic blood pressure; DBP, diastolic blood pressure; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; VA, ventricular arrhythmias; TC, total cholesterol; TG, triglycerides; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; FPG, fasting plasma glucose; AGR, albumin–globulin ratio; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; cTnI, cardiac troponin I; NLR, neutrophil-to-lymphocyte ratio; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker.
Univariate Analysis and variable Selection
For univariate analysis, each of the listed factors in Table 1 were used as the independent variable and readmission as the dependent variable. The following variables listed in Table 2 had a significant association with readmission: gender, age, hypertension, diabetes mellitus, CES, number of diseased vessels, in-hospital VA, peak cTnI, LVEF, AGR, NT-proBNP, hemoglobin, and fibrinogen (all P < 0.05). We performed preliminary confounding factors correction for these covariates by constructing two adjusted models with baseline covariates, taking into account clinical relevance, univariate analysis results, and the number of events available. Model 1 incorporated gender as a moderating variable, and model 2 additionally incorporated age, hypertension, and diabetes mellitus.
Univariate Analysis and Confounding Factors Correction.

Abbreviations: OR, odds ratio; CI, confidence intervals; Ref, reference; CSE, cumulative smoking exposure; VA, ventricular arrhythmias; cTnI, cardiac troponin I; LVEF, left ventricular ejection fraction; AGR, albumin–globulin ratio; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; *, P > 0.05.
Model 1: Unadjusted covariates + Gender.
Model 2: Model 1 + Age + Hypertension + Diabetes mellitus.
Univariate analysis results showed that smokers with <400 and with ≥400 cigarette-years had a lower risk of readmission compared with non-smokers (Crude OR = 0.439 and 0.525 respectively; both P < 0.05). After adjustment for gender in model 1, there was no significant difference in the risk of rehospitalization for either smokers with <400 (OR = 0.510; 95% CI: 0.232–1.124) or ≥400 cigarette-years (OR = 0.639; 95% CI: 0.361–1.130) compared with non-smokers. After adjustment for additional confounding factors in model 2, the difference in readmission between smokers and non-smokers remained statistically insignificant, and therefore smoking (cigarette-years) was excluded in the final multivariate model. Similarly, after adjustment in models 1 and 2, LVEF, AGR, hemoglobin and fibrinogen were excluded in the final multivariable regression model.
Multivariate Logistic Regression Analysis for Risk Factors
Finally, gender, age, hypertension, diabetes mellitus, the number of diseased vessels, in-hospital VA, baseline NT-proBNP, and peak cTnI were included in a multivariable logistic regression model in a forward stepwise (likelihood ratio) fashion and retained if the resultant p-value was <0.05 (two-tailed). As presented in Table 3, multivariate analysis demonstrated that female (OR = 2.426; 95% CI: 1.395–4.218), hypertension (OR = 1.898; 95% CI: 1.100–3.275), 3-vessel disease (OR = 2.632; 95% CI: 1.332–5.201), in-hospital VA (OR = 3.143; 95% CI: 1.305–7.574), peak cTnI (OR = 1.003; 95% CI: 1.001–1.004), and baseline NT-proBNP (OR = 1.001; 95% CI: 1.000–1.002) were independent risk factors for readmission (all P < 0.05).
Multivariate Logistic Regression Analysis of Readmission.

Abbreviations: SE, Standard Error; OR, odds ratio; CI, confidence intervals; Ref, reference; VA, ventricular arrhythmias; cTnI, cardiac troponin I; NT-proBNP, N-terminal prohormone of brain natriuretic peptide.
Development and Validation of a Readmission-Predicting Nomogram
A simple nomogram was formulated on the results of multivariate logistic regression analysis to facilitate the calculation of risk scores and thus easily evaluate the probability of individual readmission. According to Figure 1, the nomogram contained six predictors, including gender, hypertension, in-hospital VA, diseased vessels, NT-proBNP, and peak cTnI. The maximum readmission prediction probability of the obtained nomogram model was 90%. The sum of the points assigned to each prognostic factor corresponded to the probability of readmission due to MACEs. For instance, one female (21 points) STEMI patient with hypertension (15 points), in-hospital VA (27.5 points), 3-vessel disease (23 points), a NT-proBNP level of 200 pg/mL (5 points), and a peak cTnI level of 200 ng/mL (12.5 points) had a total score of 104 points, which corresponded to a 70% risk for readmission in one year.

The nomogram for predicting the risk probability of 1-year readmission for MACEs in STEMI patients after PCI. VA, ventricular arrhythmias; NT-proBNP, N-terminal prohormone of brain natriuretic peptide; cTnI, cardiac troponin I; MACEs, major adverse cardiovascular events; STEMI, acute ST-segment elevation myocardial infarction; PCI, percutaneous coronary intervention.
We evaluated the performance of the nomogram in the internal validation. The discriminative power of the nomogram in predicting readmission was acceptable, and the corresponding area under the curve (AUC) was 0.723 (95% CI: 0.665–0.780) (Figure 2). The calibration plot of the nomogram exhibited a good consistency between predicted values and observed values (Figure 3A). Hosmer-Lemeshow test also indicated well-calibrated data in the model (χ2 = 15.396; P = 0.052). Furthermore, according to the DCA result (Figure 3B), the model exhibited a good clinical utility and delivered a high net benefit to patients.

ROC curve of the nomogram. ROC, receiver operating characteristic; AUC, area under the curve.
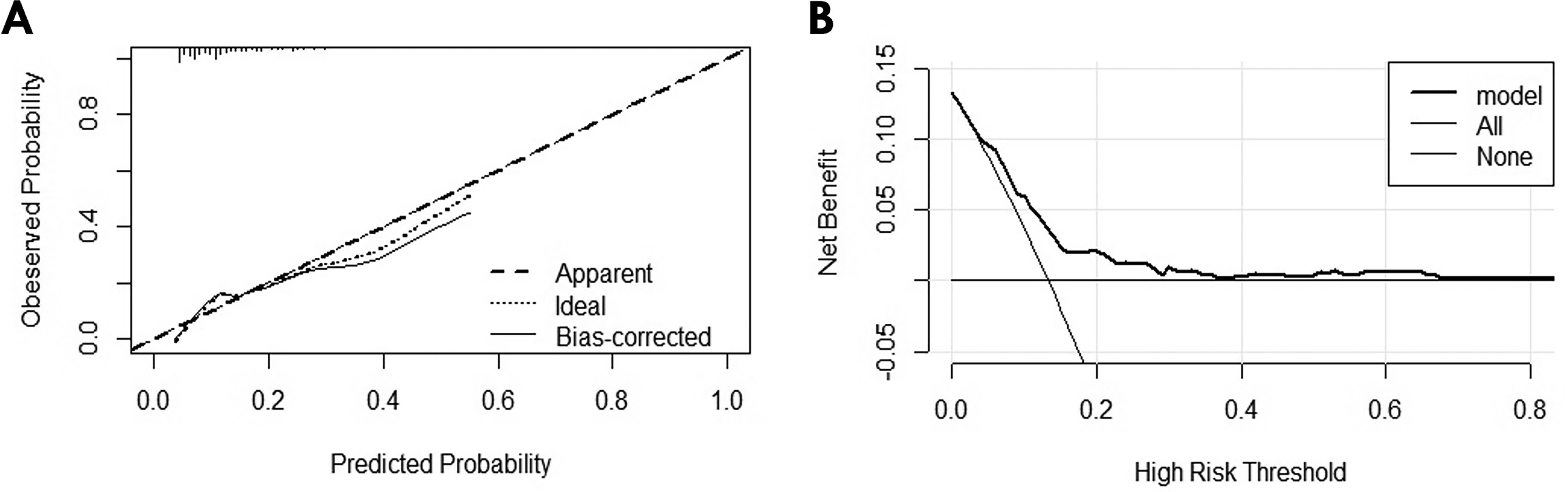
A: Calibration plot of the nomogram; B: DCA of the nomogram. DCA, Decision Curve Analysis.
Timing and Causes of Readmission
A total of 70 patients (13.3%) were readmitted for MACEs after discharge in the first year. Of all 70 readmissions, 23 (32.9%) were within the first month and 39 (55.7%) were within the first 3 months (Figure 4A). Chest pain was the leading cause of readmission, accounting for 72.9%, followed by congestive heart failure at 20% of all MACEs (Figure 4B).
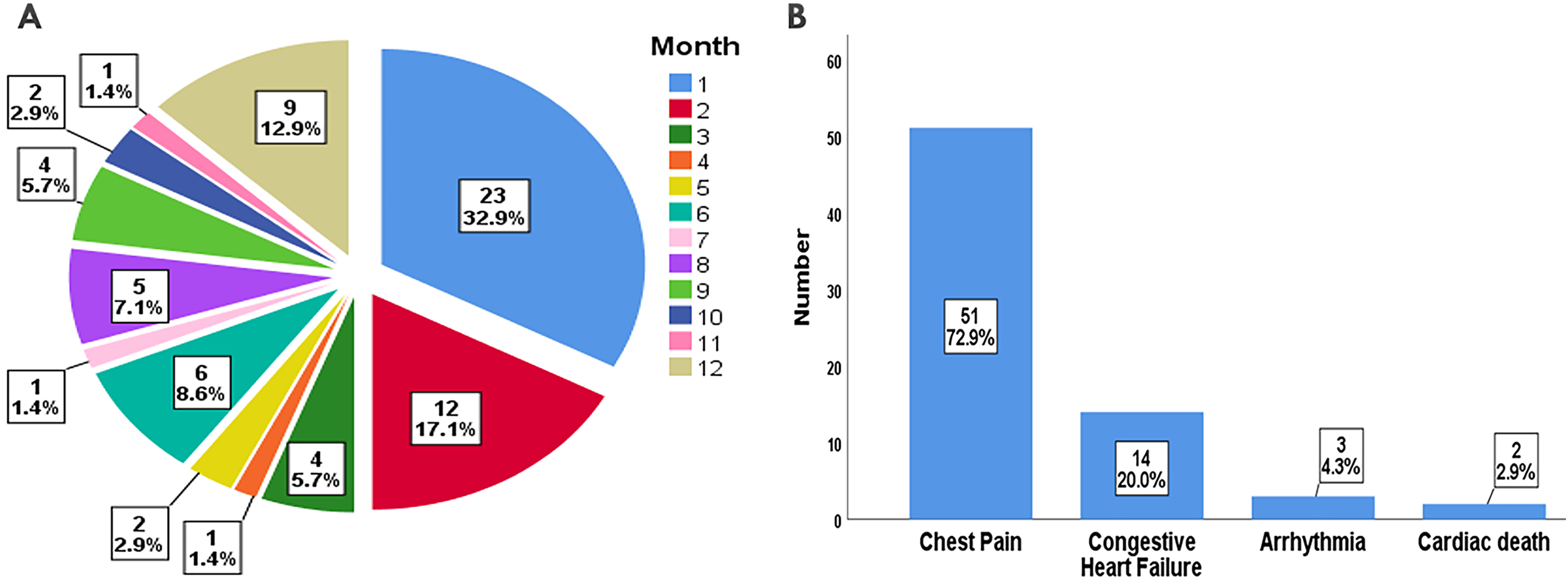
Timing (A) and causes (B) of readmission.
Discussion
Several earlier studies have focused on short-term or all-cause readmissions in patients with STEMI.11,12 However, recovery from myocardial infarction is a long-term process, and STEMI patients are usually elderly and suffer from diseases with high admission rates, such as hypertension, chronic obstructive pulmonary disease, and kidney disease. Therefore, short-term or all-cause readmission rates do not reflect the true readmission burden caused by cardiovascular events. According to our study, patients with STEMI had a high readmission rate for MACEs in the first month (32.9%) as well as in the first three months (55.7%) after discharge.
Smoking has been reported worldwide as the second most important risk factor for myocardial infarction 13 ; it is an important risk factor for incident heart failure hospitalization. 14 However, our study showed that smokers had a lower risk of readmission compared with non-smokers. This phenomenon is known as the “smoker's paradox”.15,16 Considering that unbalanced baseline covariates might be a potential confounder between smoking and readmission, we calculated the proportion of smoking men and women (71.7% vs 45.9%, P < 0.001). According to Spearman's correlation analysis, there was a significant correlation (r = −0.234, P < 0.001) between gender (male as reference) and cigarette-years. After correcting for gender (Model 1) and additionally for age, hypertension, and diabetes mellitus (Model 2), a causal relationship between smoking level and readmission was excluded (P >0.05). A comparison between the changes in OR and 95% CI values of the two models revealed gender as the most important confounder for smoking.
The proportion of women was much lower than that of men who were hospitalized for STEMI (28.1 vs 71.9%, P < 0.001), consistent with what had been reported in the literature. 17 However, once STEMI occurs, women usually have a higher risk of readmission than men, regardless of whether PCI was performed. 11 Our study also confirmed sex-based differences, with 2.4 times higher risk of readmission of female survivors than male survivors (OR = 2.426; 95% CI: 1.395–4.218; P = 0.002). This may be because women more often had comorbidities, such as hypertension and diabetes. In addition, women were more sensitive to pain. 18 Consistent with previous studies,12,19 our study showed chest pain was the predominant event for readmission, and the majority of chest pain events after post-PCI discharge were low risk and did not require intervention. 3 However, patients were not able to accurately identify such low-risk chest pain events, which in turn led to their readmissions. Some post-discharge interventions, such as motivational telephone consultation and clinical follow-up, were reported to help reduce readmissions for angina and other cardiac reasons post-PCI.20,21 Therefore, structured medical interventions and health education during hospitalization, together with a well-established follow-up system, psychological interventions, and professional medical guidance after discharge, might help reduce readmission rates, especially in the female patients.
After adjusting for confounding factors, the risk of readmission was nearly two times (OR = 1.898; 95% CI: 1.100–3.275; P = 0.021) higher in patients with hypertension than in those who did not have hypertension. Previous studies showed that hypertension significantly increased the risk of post-discharge MACEs in patients with acute myocardial infarction,22,23 and the same was observed in relatively healthy populations. 24 Few data are available on the risk of MACEs or readmission after intensified antihypertensive therapy. A study showed that in older patients with a baseline systolic blood pressure (SBP) of 160 mm Hg or greater, there was a significant reduction in cardiac events (fatal and non-fatal myocardial infarction and sudden cardiac death) if a target SBP of less than 150 mm Hg was achieved. 25 Interestingly, however, in another study on elderly hypertensive patients hospitalized for noncardiac conditions, intensification of antihypertensive regimens at hospital discharge increased the risk of all-cause readmission (HR = 1.23; 95% CI: 1.07–1.42) and serious adverse events (HR = 1.41; 95% CI: 1.06–1.88), and it was not associated with reduced cardiac events within 30 days and one-year. 26
Approximately 50% of STEMI patients had concurrent non-infarct-related artery (non-IRA) disease, defined as ≥50% stenosis in ≥1 non-IRA. 27 The 3-vessel disease (including the IRA and 2 non-IRA) usually predicted worse cardiac outcomes than single and double vessel disease 27 and was one of the primary causes of 30-day readmission of patients with STEMI. 12 This finding was further corroborated in our study, in which we found a 1.6-fold increase in risk of readmission in patients with the 3-vessel disease compared with those with single-vessel (IRA only) disease (OR = 2.632; 95% CI:1.332–5.201; P = 0.005). Although there was conflicting evidence for the safety and benefit of simultaneous revascularization of non-IRA lesions, recent studies have favored complete revascularization for a significantly better prognosis compared to IRA-only PCI28,29; current guidelines recommend routine complete revascularization of non-IRA lesions in STEMI patients with the multivessel disease before hospital discharge. 30 However, actual clinical application lags far behind the pace of the update of guidelines. To our knowledge, complete pre-discharge revascularization protocols have not yet been applied in most hospitals in China, probably due to safety and cost concerns. Most of the research data on complete revascularization are from the United States and Europe; therefore, this is a direction worth us exploring.
VA is a common cardiac complication, and two large studies have shown a 5% to 11% incidence of VA in patients with STEMI treated with primary PCI.31,32 In this study, the overall incidence of in-hospital VA was 6.1%, with a 12.9% incidence in the readmitted group and a 5.0% incidence in the non-readmitted group (P = 0.023). In the analyzed population, multivariate analysis identified in-hospital VA as the most important risk factor for readmission and the readmission rate in the presence of VA was 3.1 times higher than that in the absence of VA (OR = 3.143; 95% CI: 1.305 7.574; P = 0.011). Likewise, other investigators showed that in-hospital VAs were strong and independent predictors of worse out-of-hospital outcomes in the STEMI population after PCI.31,32
The peak level of cTnI rather than the baseline level was the biomarker of choice for predicting the size of acute myocardial infarction and was a convenient and reliable alternative to imaging techniques in terms of evaluating myocardial survival status and cardiac function. 33 Infarct size was one of the major determinants of adverse outcomes in patients with STEMI. 34 Peak cTnI could also independently predict short- and long-term adverse events in patients with STEMI after PCI, including low LVEF, death, and other cardiac events.33,35,36 Our study further confirmed that peak cTnI could independently predict readmissions for MACEs. Although we could not find a role of Symptom-Onset-to-Balloon time for readmission, early opening of IRA helped to reduce the size of myocardial infarction to thus possibly reduce the release of troponin and improve the prognosis of STEMI patients.
NT- proBNP is a sensitive biomarker for the diagnosis of heart failure. Elevated levels of NT-proBNP were observed to be correlated with MACEs. 37 High baseline levels of NT-proBNP often predicted a high incidence of adverse events during follow-up, including death, congestive heart failure, shock, and short-term mortality.38–40 Our study extended these earlier findings by demonstrating baseline NT-proBNP as an independent impact factor for readmission due to MACEs.
In the current study, we present for the first time, a well-performing and easy-to-use risk prediction nomogram on readmission, and the DCA indicated clinical utility. Finally, we emphasize that prevention is more important than treatment. There is a long way to go to improve screening for coronary heart disease in high-risk groups in both developing and developed countries.
Study Limitations
First, this is a single-center, retrospective cohort study, and prospective studies and large multi-center studies are needed to confirm these findings. Second, the present model did not undergo external validation. Third, we did not consider the effects of type of medical insurance, household income, and psychological factors on readmission rates. Despite these limitations, we believe the study results may help screen STEMI survivors at high risk for readmission and thus reduce readmission rates.
Conclusions
To conclude, female gender, hypertension, in-hospital VA, 3-vessel disease, baseline NT-proBNP, and peak cTnI were independent risk factors for readmission of STEMI patients after PCI. This nomogram exhibited a good performance and may help in risk stratification and clinical decision-making for readmission.
Footnotes
Author Contributions
Wensen Yao designed the study and performed the research. Jie Li and Wensen Yao analysed the data and were involved in writing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the Ethics Committee of the First Hospital of Jilin University (Changchun, China). In this study, no extra interventions were performed, thus, the institutional review board waived the informed consent of patients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
