Abstract
Development of cardiovascular diseases could originate in early childhood. However, reference values of hemostatic parameters and adipokines in preschool children remain to be explored. We measured blood levels of adipokines and parameters of the hemostatic/fibrinolytic systems in 167 healthy children aged 4 to 6 years at 9:00 to 10:30
Cardiovascular disease is the main cause of death worldwide. The development of cardiovascular diseases is multifactorial, including a possible association with the metabolic syndrome. 1 The metabolic syndrome is defined by a constellation of clinical features including visceral or central obesity, insulin resistance, high blood pressure, high triglycerides (TGs), and low high-density lipoprotein cholesterol (HDL-C). However, the correlation among the diagnostic criteria for metabolic syndrome in the pediatric age group and future cardiovascular disease developing in adulthood remains to be investigated, 2 despite the prevailing concept that lifestyle-related diseases sometimes originate in childhood. 3 The early stage of arteriosclerosis can be detected by increased carotid intima–media thickness, and this early marker of arteriosclerosis in childhood is associated with fluctuation of some hematological variables. 4 The prevalence of obesity in children has been increasing over the last 20 years in Japan as in all Western countries.5–8 The incidence of the metabolic syndrome in Japanese overweight children is comparable to that in US overweight children, and the critical period for the development of obesity is between 5 and 6 years of age.5,7 Establishing hematological reference values for metabolic syndrome in preschool children would therefore be useful in the overall assessment of the potential effects of intervention.
The hemostatic and fibrinolytic systems as well as adipokines have been implicated in the development of metabolic syndrome,9–12 although such variables are not included in the diagnostic criteria of the syndrome. While the developmental changes in the hemostatic/fibrinolytic systems during childhood have been studied,13–15 the available data are mostly for Western children; this is important because the metabolic syndrome-related parameters such as insulin resistance show race differences. 16 A full assessment of these variables and any association with the metabolic syndrome criteria remain to be investigated, partly due to the difficulty in morning blood sampling in young children. This study was designed to establish reference values for hemostatic/fibrinolytic variables and adipokines in Japanese preschool children after strict fasting and to determine any relationships with the components of metabolic syndrome.
Methods
Participants
The study comprised 167 preschool children, aged 5 to 6 years (females, 85), who attended kindergarten in Yokohama City or Kagoshima City. Children showing illness at examination including common cold or had history of significant disease were excluded from the study. The Ethics Committee of Human Research of Tsukuba University Hospital approved the study protocol in advance. In addition, parents of the participants attended an instruction lecture about the importance of prevention of metabolic syndrome from early childhood before the commencement of the study. Written informed consent was subsequently obtained from the parents.
Anthropometric and Biometric Assessment
Height and weight were measured using standard methods (TTM-HV; TSUTSUMI Co, Kyoto, and DC-320; TANITA Co, Tokyo, Japan), and the body mass index (BMI) was calculated as weight in kilograms divided by height in meter square. Blood pressure and heart rate were measured 3 times using an automated oscillatory system (TM-2571; A&D Co, Tokyo), between 9 and 10:30
Blood Sampling and Laboratory Analyses
Blood samples were collected from the antecubital vein in the morning (between 9 and 10:30
Measured Variables a
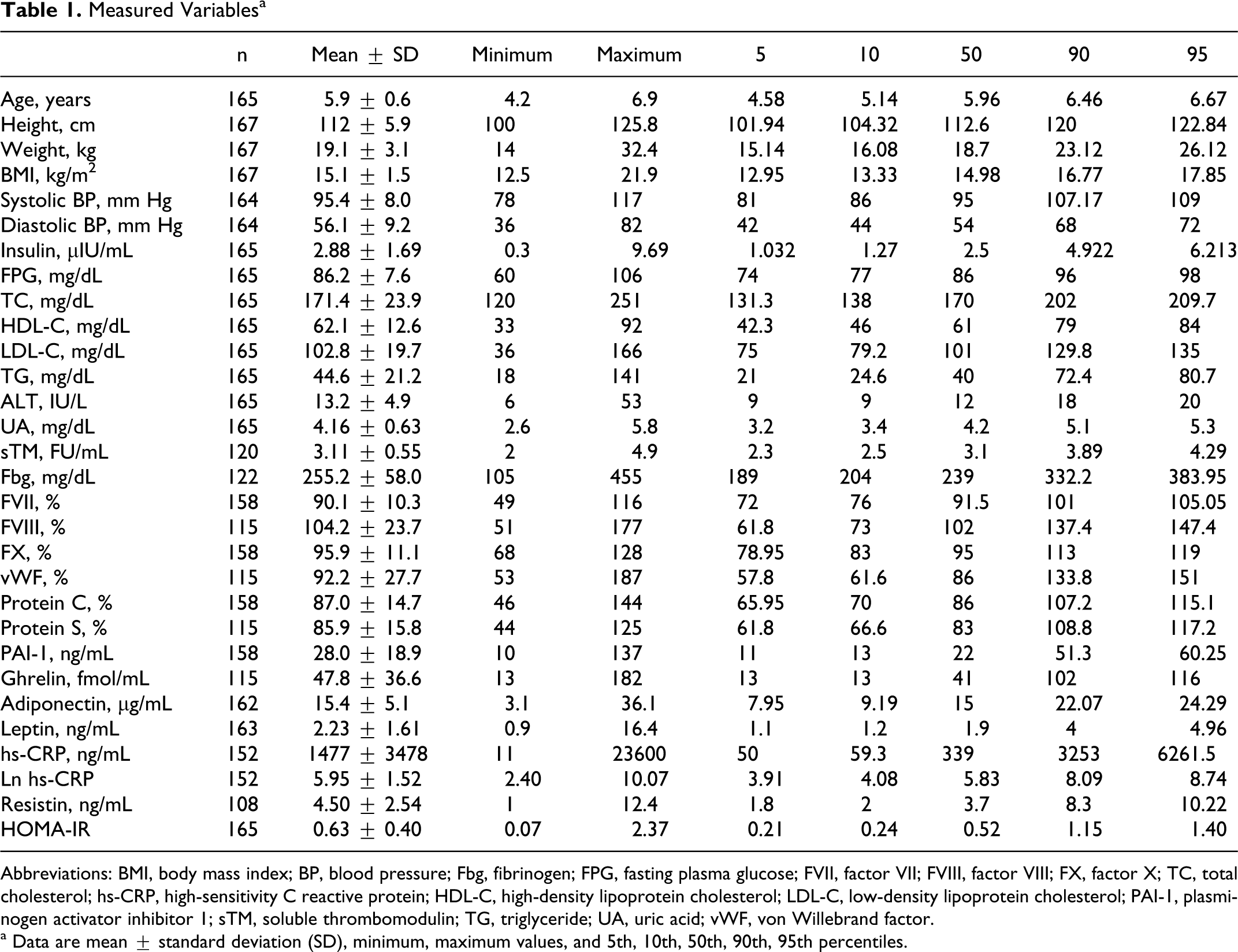
Abbreviations: BMI, body mass index; BP, blood pressure; Fbg, fibrinogen; FPG, fasting plasma glucose; FVII, factor VII; FVIII, factor VIII; FX, factor X; TC, total cholesterol; hs-CRP, high-sensitivity C reactive protein; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PAI-1, plasminogen activator inhibitor 1; sTM, soluble thrombomodulin; TG, triglyceride; UA, uric acid; vWF, von Willebrand factor.
a Data are mean ± standard deviation (SD), minimum, maximum values, and 5th, 10th, 50th, 90th, 95th percentiles.
Alanine aminotransferase (ALT), uric acid, TG, total cholesterol, HDL-C, low-density lipoprotein cholesterol (LDL-C), fibrinogen, insulin, high-sensitivity C reactive protein (hs-CRP), and fasting plasma glucose were measured by standard automated methods using the appropriate devices (JEOL, Sysmex, Mitsubishi Chemical Medience, Fujirebio, and Siemens Healthcare Diagnostics, Japan). The sTM was measured by enzyme immunoassay ([EIA]; Molecular Devices, Japan); leptin by radioimmunoassay ([RIA]; Aloka, Japan); desacyl-ghrelin, adiponectin, and resistin by enzyme-linked immunosorbant assay ([ELISA]; Mitsubishi Chemical Medience, Otsuka Pharmaceutical Co, and BioVender Laboratory Medicine, Japan). Coagulation factor (F) VII, FVIII, and FX were measured by clotting time methods, and von Willebrand factor (vWF) was assayed by the fixed platelet agglutination method (Siemens Healthcare Diagnostics). Protein C antigen, free protein S antigen, and plasminogen activator inhibitor 1 (PAI-1) in a complex with tissue plasminogen activator 1 (tPA-PAI-1 complex) were assayed by latex photometric immunoassay (Mitsubishi Chemical Medience and JEOL).
Data Analysis
All continuous variables were expressed as mean ± standard deviation (SD), with the 5th, 10th, 50th, 90th, and 95th percentile values calculated for each parameter. Logarithms of the values were also calculated for hs-CRP. The number of data was different among parameters (maximum 167 and minimum 108; see Table 1) because the specimen volume was insufficient to allow all measurements in some children, who needed to be resampled but refused.
The study cohort was divided into 2 groups: those with <90th percentile values of BMI and those with ≥90th percentile. Each continuous variable was compared between the 2 groups using the Student t test. Participants were then assigned to subgroups based on the number of the following cardiovascular risk factors: (1) BMI ≥ 90th percentile, (2) blood pressure (systolic or diastolic or both) ≥90th percentile, (3) plasma glucose level ≥ 90th percentile, (4) TG ≥ 90th percentile, and (5) HDL-C ≤ 10th percentile. The hematological parameters were then compared among the subgroups using analysis of variance (ANOVA) followed by a Tukey–Kramer-type multiple comparisons.
The relationships between the hemostatic/fibrinolytic parameters or adipokines and the components of the metabolic syndrome were tested by simple linear regression model, and significant variables were then subjected to stepwise linear regression analysis to identify independent predictors of the metabolic syndrome. A P value less than .05 was considered statistically significant.
Results
Table 1 details the anthropometric, biometric, and hematological data for all participants. Analysis of differences in various parameters between children with BMI < 90th percentile (n = 149) and those with BMI >/= 90th percentile (n = 18) showed that systolic blood pressure (92.9 ± 9.4 vs 103.8 ± 9.5 mm Hg, P < .001), diastolic blood pressure (55.2 ± 10.5 vs 61.7 ± 11.4 mm Hg, P = .017), heart rate (95.1 ± 14.5 vs 102 ± 9.5 bpm, P = .048), insulin (2.7 ± 1.4 vs 4.8 ± 2.6 μIU/mL, P = .004), FVII (89.6 ± 10.6 vs 94.1 ± 6.6, P = .021), FX (95 ± 10.8 vs 103.2 ± 11.6, P = .004), protein S (84.8 ± 15.1 vs 93.9 ± 19.8, P = .021), ghrelin (49.2 ± 37.7 vs 33.6 ± 19.3 fmol/mL, P = .018), leptin (2 ± 0.84 vs 4.5 ± 3.6 ng/mL, P = .01), and HOMA-IR (0.57 ± 0.32 vs 1.17 ± 0.64, P = .003) were significantly different. The other hematological parameters were not different between the 2 groups.
Among all participants, no, 1, and 2 cardiovascular risk factors (pertaining BMI, blood pressure, plasma glucose, TG, and HDL-C) were observed in 96 (58.5%), 44 (26.8%), and 24 (14.6%) cases, respectively. None had 3 or more risk factors. Blood levels of fibrinogen and leptin in participants with 2 or 1 risk factors were significantly higher than in those with no cardiovascular risk factors (Figure 1 ). The other parameters showed no significant association with the number of cardiovascular risk factors.
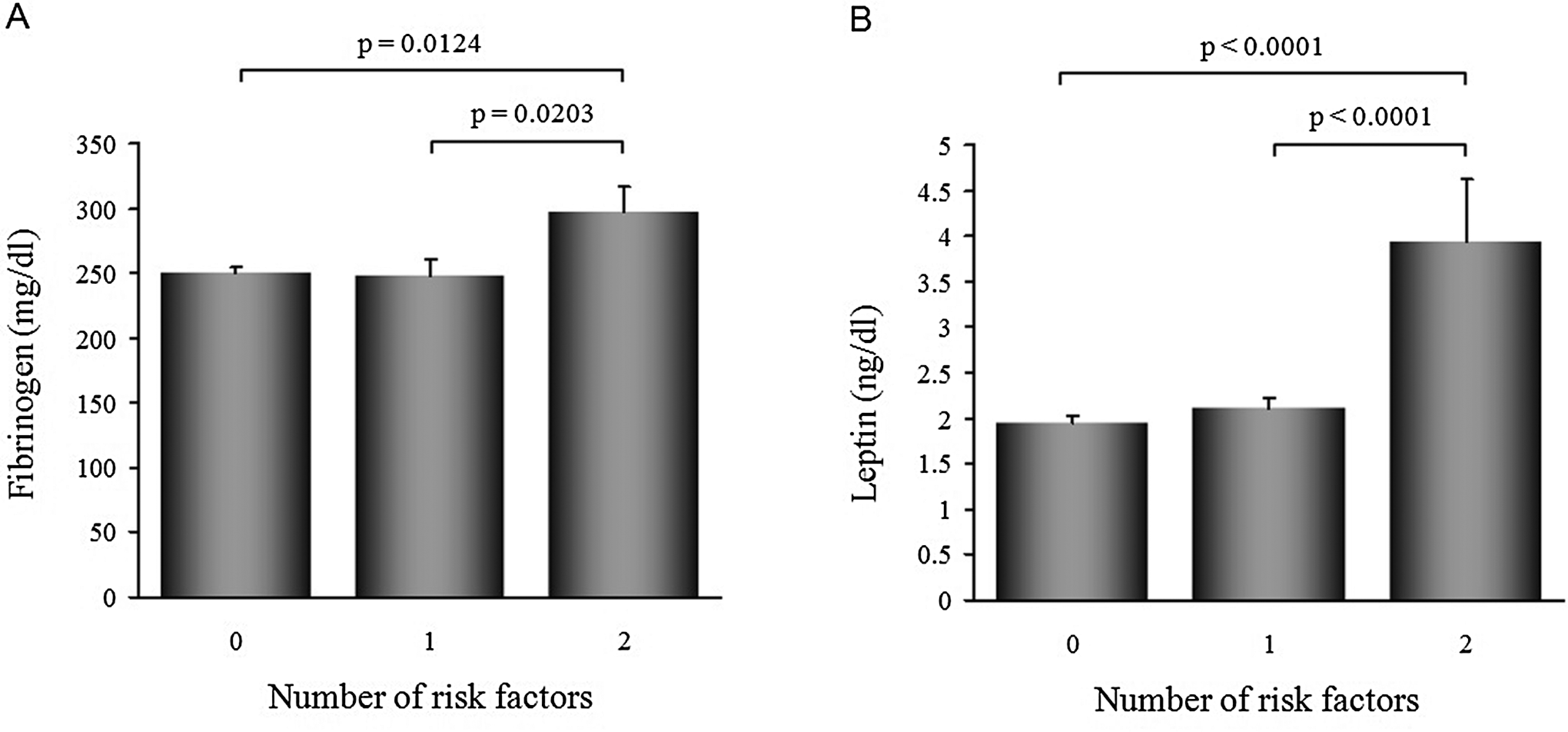
Comparison of various parameters among the 3 groups with no, 1, or 2 cardiovascular risk factors. Blood levels of fibrinogen (A) and leptin (B) were significantly higher in the group with 2 risk factors than in those with none or 1 risk factors. Data are expressed as mean ± standard error of the mean (SEM).
Stepwise regression analysis identified ALT, uric acid, fibrinogen, FVIII, FX, vWF, protein C, protein S, PAI-1, and leptin levels as significant independent risk factors for the metabolic syndrome (Table 2 ).
Variables Independently Associated With Components of the Metabolic Syndrome.
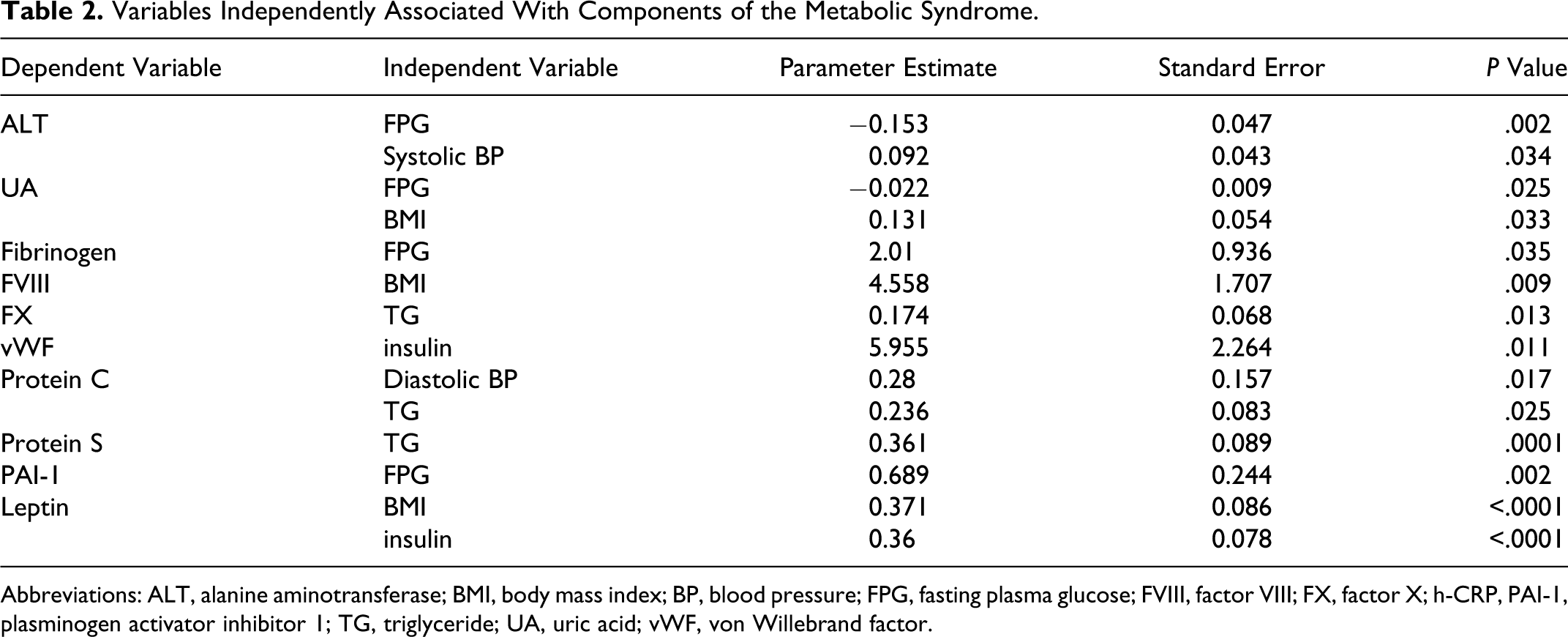
Abbreviations: ALT, alanine aminotransferase; BMI, body mass index; BP, blood pressure; FPG, fasting plasma glucose; FVIII, factor VIII; FX, factor X; h-CRP, PAI-1, plasminogen activator inhibitor 1; TG, triglyceride; UA, uric acid; vWF, von Willebrand factor.
Discussion
The present study demonstrated that many hemostatic/fibrinolytic parameters and adipokines, as well as ALT and uric acid, are associated with the components of the metabolic syndrome, even in healthy preschool children. The mean BMI in this study group was 15.1 ± 1.5 kg/m2 with a maximum value of 21.9. Based on the cutoff value of BMI for obesity in preschool children reported in our recent work, 7 only 10 children were judged overweight in this study, indicating that the study population could be considered a healthy one.7,8 Specifically, blood levels of FVII, FX, protein S, and leptin were higher in children with BMI ≥ 90th percentile, and that of ghrelin was lower, compared to the other group. Stepwise regression analysis identified blood levels of fibrinogen (Fbg), FVIII, FX, vWF, protein C, protein S, and PAI-1 were determined by 1 or 2 components of the metabolic syndrome, although the independent components related to each dependent variable varied. Furthermore, children who had one or more known cardiovascular risk factors had significantly higher values of fibrinogen and leptin than those with no risk factors. These results lend support to the notion that hemostatic/fibrinolytic parameters or substances secreted by adipose tissues are associated with the development of metabolic syndrome in early childhood.
Several parameters involved in blood coagulation and fibrinolysis systems are known predictors of cardiovascular diseases.9,10,17–20 These include fibrinogen, tissue factor (TF), FVII, FVIII, FX, vWF, protein C, sTM, PAI-1, and leptin. Among them, PAI-1 and leptin have recently attracted much interest because they are secreted by adipose tissue and their blood levels correlate with obesity and the amount of visceral fat mass. Both PAI-1 and leptin levels are also related to each other independent of the fat mass, 18 and both are recognized as cardiovascular risk factors.21–23 In the present study, PAI-1 was significantly associated with fasting plasma glucose, while leptin showed a strong relationship with BMI and fasting insulin levels, suggesting the association with insulin resistance. Increased PAI-1 levels is currently considered a true component of the metabolic syndrome, through which the risk of development of cardiovascular disease increases. 24 In the present study, PAI-1 and leptin levels were associated with features of the metabolic syndrome, indicating that the above-mentioned association is valid even in preschool children. Desacyl-ghrelin is another adipokine considered to lower cardiovascular risk through the activation of endothelial nitric oxide synthase. 25 Participants with high BMI in the present study had low ghrelin levels. Furthermore, serum concentrations of uric acid increase in proportion with leptin 26 and uric acid is also strongly associated with several components of the metabolic syndrome, 27 tendencies that were reproduced in the present study. Increased adipose tissue, and progression of the metabolic syndrome, have also been associated with increases in other indicators of prothrombotic activity such as increased plasma levels of fibrinogen, vWF, FVII, FVIII, and FX.19,24 These changes were also observed in the present study.
Fibrinogen is both a procoagulant factor and an activator of inflammation, thus a typical cardiovascular risk factor. 28 The present study indicated that even in children, fibrinogen levels increased with BMI and with the number of cardiovascular risk factors. Increased fibrinogen has been associated with impaired activation of protein C, 29 which is interesting because thrombin binds to both fibrinogen and TM through a common region. In addition, thrombin is a procoagulant when bound to fibrinogen but exerts potent anticoagulant activity through the activation of protein C when it binds to TM on the cell surface. It is therefore conceivable that fibrinogen levels influence serum levels of sTM and that increased sTM in the circulation is related to a decrease in cardiovascular complications. 30 The present study demonstrated a significant association between protein C/S levels and certain components of the metabolic syndrome (TG and blood pressure). Previous studies on obese adults also showed increased activated protein C levels and their decrease after weight loss. 31 Protein S also exerts anticoagulant activity usually as a cofactor of protein C but also through the stimulation of TF pathway inhibitor. 32 However, sTM did not show any significant correlations with such metabolic syndrome-related parameters in this study. This might be due to the small number of participants analyzed and/or obese children.
Our study also presented clinically significant reference values for metabolic syndrome- or overweight-related variables in healthy preschool children, including those involved in hemostasis and fibrinolysis systems. Some of these values such as developmental changes from infancy to adolescence were reported previously,13–15 although these studies did not specify the time of blood sampling or whether fasting was applied. Some parameters, especially those related to fibrinolysis, are significantly influenced by circadian oscillation; for instance, PAI-1 activity peaks in the morning. 33 Our results obtained in the morning after strict fasting are therefore important for future research and could be useful for the establishment of diagnostic criteria or prevention strategies for the metabolic syndrome in Japanese children.
Study Limitations
The numbers of participants analyzed and that of obese children were relatively small in this study. The results should be useful to provide reference values for hemostatic/fibrinolytic parameters and adipokines in Japanese preschool children. Further studies are needed to confirm how the abnormalities observed herein in those parameters could be implicated in later-life development of cardiovascular diseases.
Conclusions
The present study demonstrated that, even in preschool children, many hemostatic/fibrinolytic or adipose tissue-related variables show significant associations with the components of the metabolic syndrome, implicating a role for these systems in the insidious progression of cardiovascular diseases from early age.
Footnotes
Acknowledgments
The authors are grateful to the Directors and staff of the Himawari Kindergarten, Yokohama, and the Kinjoh Kindergarten, Kagoshima, for their contribution in the survey. We are also grateful to SRL Inc, Tokyo, Japan, for the help in measuring some of the hematological parameters in this study.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported in part by a grant-in-aid from the Japanese Ministry of Health, Labor and Welfare (Comprehensive Research on Cardiovascular and Life-style Related Disease, H18-049). The authors have no further financial or corporate involvement to disclose.
