Abstract
Aim
Non-vitamin K antagonist oral anticoagulants (NOACs) were developed as an alternative to warfarin to prevent thromboembolism in patients with atrial fibrillation (AF), prosthetic heart valves, venous thromboembolism (VTE), or other thrombotic disorders. The aim of this study is to explore the trends in prescribing OACs, including warfarin and NOACs, in Shanghai, China.
Methods
Prescription data of OACs were retrospectively collected from Rx Analysis System from 2010 to 2020 in Shanghai, China. Comparisons were made on the trends of each OACs according to different indications, age groups, and hospital grades. The costs and the contribution of individual OACs were also explored.
Results
Growing trends in overall prescriptions for OACs were observed. The prescriptions of NOACs were significantly increased since 2016, while the prescriptions of warfarin kept decreasing since 2017. A highly statistically significant increase in prescriptions of Rivaroxaban was observed from 2016 to 2020 (P < .001). Despite the price reduction of rivaroxaban in 2018, the total cost of rivaroxaban continued to rise (P < .001). Rivaroxaban emerged as a preferred NOAC in both indications of AF and VTE, and accounted for more than three-quarters of the total costs for OACs since 2019. Compared with rivaroxaban, the prescription numbers of dabigatran and apixaban were much smaller, and the growth of prescriptions were much slower. Differences in prescribing patterns in different indications, age groups, and grades of hospitals were also founded.
Conclusion
There has been a rapid increase in the use of OAC over the last 11 years in Shanghai, China. NOACs have been adopted rapidly, and have been gradually replacing warfarin. Warfarin remains the top choice for certain patients with valvular heart disease. Future studies are warranted considering changes in the OAC use in a larger scale, as well as the rationality and its influence factors on OAC use.
Introduction
Oral anticoagulants (OACs) are used to prevent thromboembolism in patients with atrial fibrillation (AF), prosthetic heart valves, venous thromboembolism (VTE), or other thrombotic disorders. Vitamin K antagonists, chiefly warfarin, have been the mainstay of oral anticoagulant therapy for nearly 70 years. 1 The last decade has witnessed the emergence and the application of non-vitamin K antagonist oral anticoagulants (NOACs), including thrombin inhibitor dabigatran, and Factor Xa inhibitors rivaroxaban, apixaban, edoxaban, and betrixaban. 2 NOACs now have been developed as alternatives to warfarin, which showed at least noninferior or even superior to warfarin in efficacy with lower risk of bleeding. Other advantages of NOACs include fewer requirements of blood monitoring, less frequent follow-up, more immediate effects of drug onset and offset, and fewer food and drug interactions, which can lead to better patient adherence. 3 Accordingly, current clinical guidelines strongly recommend the NOACs over warfarin in many treatment indications. European Society of Cardiology made a Class IA recommendation favoring NOACs over warfarin for stroke prevention in nonvalvular AF patients in 2020. 4 CHEST guideline of antithrombotic therapy for VTE disease also strongly recommended NOACs over warfarin in patients with VTE as treatment-phase anticoagulant therapy in 2021. 5
NOACs have been increasingly and widely used in countries and regions in recent years.6–8 In US, the prescriptions of NOACs increased from 14.1% of all OAC prescriptions in 2013 to 57.3% in 2018. Similarly, NOACs achieved a dominant (62%) patient share over warfarin in 2019 in primary care England. 9 It has been 14 years since the first NOAC rivaroxaban was approved for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. There are four NOACs (dabigatran, rivaroxaban, apixaban, and edoxaban) that have been approved in China Now. Nevertheless, there is little knowledge on how NOACs are accepted by clinicians and patients in China. Therefore, the current study aims to i) examine the trends in prescribing OACs, including warfarin and NOACs in Shanghai, China between 2010 to 2020; ii) make comparisons on the use of each OACs according to different indications, age groups, and hospital grades; and lastly iii) analyse the possible reasons of the prescription patterns.
Methods
Data Sources and Study Design
Prescription data of OACs in this study were retrospectively extracted from 2010 to 2020 from Rx Analysis System (RAS), which was founded in 2005 by China National Pharmaceutical Industry Information Center for supporting clinical pharmaceutical research. 10 RAS involves outpatient prescription data in more than 60 hospitals in Shanghai, covering three levels of hospitals, including tertiary hospitals, secondary hospitals, and community health service centers. The database randomly selected prescription data of outpatients from 6 consecutive days in each month.
In this study, outpatient prescriptions of OACs, including warfarin, rivaroxaban, dabigatran, apixaban, and endoxaban were collected from 2010 to 2020. The prescription data of betrixaban were not available, as it was not approved in China. The prescription volume of endoxaban was low, since it was the last OAC approved in China in December, 2018. Accordingly, the analyses in this study focused on warfarin, rivaroxaban, dabigatran, and apixaban. Patients were eligible for inclusion if they were prescribed OAC during the study period. Patients who were dispensed OAC without any indications of anticoagulation were excluded. A prescription item was defined as a single OAC prescribed by a doctor on one treatment visit. Information on patient's code, prescription date, department, sex, age, diagnosis, as well as the generic names, specifications, quantities, and prices of the drugs were extracted. Prescriptions with missing data in the above fields were excluded. Each patient's identity was encrypted, as patient's code were reorganized by the dataset. This study was approved by the ethics committee of Ren Ji Hospital (KY2021-274-B), and a waiver of informed consent was obtained.
Data Analysis
Prescriptions were analyzed on treatment visits and costs for each OAC by year to assess the trends in prescribing patterns. Costs in Chinese Yuan (CNY) calculated by adding up all the costs of OACs for each treatment visit were converted into US dollars ($) using exchange rate of each year. The data were stratified by diagnoses (AF and VTE), age groups (< 65, 65-75, and ≥ 75), departments (cardiology, internal medicine, orthopedics, and surgical department), and hospital grades (tertiary hospital, secondary hospital, and community health service center [CHSCs]). Comparisons on baseline characteristics across patients receiving different OACs were assessed using χ2 test or rank-sum test where appropriate. Linear regression analysis with year as the independent variable and number of treatment visits or costs as the dependent variable, using the data from 2016 to 2020. The annual percentage increase or decrease was calculated by dividing the regression coefficient by the baseline of number of treatment visits or costs from 2016. All the statistical analyses were conducted using IBM SPSS version 26. Statistical significance was set at P value <.05 for all tests.
Results
Characteristics of Included Prescriptions
A total of 180 105 prescriptions of four OACs, including warfarin (67.0%), rivaroxaban (24.1%), dabigatran (8.9%), and apixaban (0.1%) were extracted (Table 1). All OAC prescriptions were from 56 hospitals, involving 29 tertiary hospitals, 17 sary hospitals, and 20 CHSCs (Table S1). The proportion of male patients was similar to that of female patients. More than 60% patients receiving OACs were over 65. About half of the patients using rivaroxaban and dabigatran were over 75. The vast majority of OAC prescriptions were prescribed in tertiary hospitals (71.6%) and secondary hospitals (27.0%). Meanwhile, OACs were more frequently prescribed in the department of cardiology (69.9%) compared with other departments such as internal medicine (19.2%) and surgical departments (7.6%). The common comorbidities of patients receiving OACs were hypertension (33.4%), diabetes (23.1%), coronary artery disease (11.3%), heart failure (9.3%), chronic renal disease (9.2%), previous bleeding (8.0%), and previous stroke (5.3%) (Table S1). Minor differences in prevalence of comorbidities were observed among patients using different NOACs.
The Summary Data of Included OACs Prescriptions from 2010 to 2020.
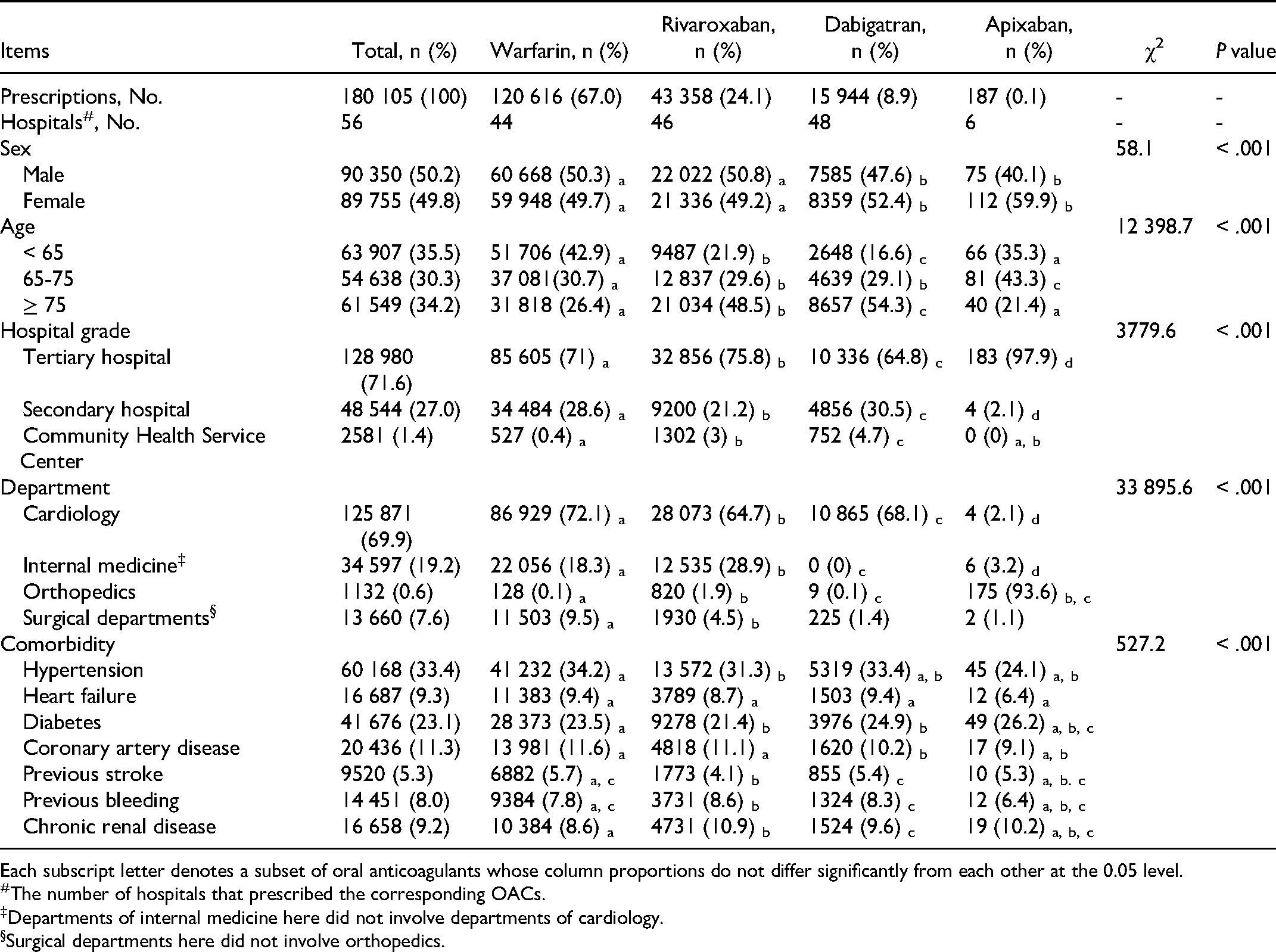
Each subscript letter denotes a subset of oral anticoagulants whose column proportions do not differ significantly from each other at the 0.05 level.
The number of hospitals that prescribed the corresponding OACs.
Departments of internal medicine here did not involve departments of cardiology.
Surgical departments here did not involve orthopedics.
Overall Trends of OAC Prescriptions
The overall trends of OAC prescriptions were presented with the number and the proportion of treatment visits, as well as the costs (Figure 1). The number of treatment visits for overall OACs increased significantly from 2010 to 2020 (β = 2742.0, 95% CI 2223.9 to 3069.2, P < .001), with an average increase of 60.6% (95% CI 49.1 to 72.0) a year, a total of 637.6% over the 11 years examined (Table S2). An obvious upward trend was also observed in total costs of overall OACs, increasing from 203 hundred US dollars in 2010 to 22 710 hundred US dollars in 2020 (Table S3). For warfarin, the number of treatment visits continued to increase from 2010 to 2015 (β = 2013.7, 95% CI 2010.4 to 2017.0, P < .001), and turned to decline steadily from 2016 to 2020 (β = −1540.9, 95% CI −1544.3 to −1537.6, P < .001) (Figure 2). The proportion of warfarin prescriptions began to decrease gradually since 2015 with the approvals and the growing use of NOACs. Similar tendency was found in the costs for warfarin (β = −59.1, 95% CI −60.1 to −58.2, P < .001), as the price of warfarin was relatively stable (Table S4). For rivaroxaban, a highly statistically significant increase (β = 5726.8, 95% CI 5719.0 to 5734.6, P < .001) in the number of treatment visits was observed from 2016 to 2020 (Figure 2), with an average increase of 786.6% (95% CI 785.6 to 787.7) per year, or 2565% overall (Table S2). Rivaroxaban accounted for 4.37% of all OAC visits in 2016 compared with 58.12% in 2020. Although significant reduction in the price of rivaroxaban has been achieved since 2018, the costs for rivaroxaban continued to rise rapidly (β = 4082.2, 95% CI 4066.1 to 4098.4, P < .001), with the total cost being 17 934 hundred US dollars in 2020 (Table S3). Compared to rivaroxaban, the growth of prescriptions for dabigatran was slower (β = 1091.1, 95% CI 1083.6 to 1098.7, P < .001), and the proportion fluctuated between 14% and 16% in 2018 to 2020. The costs also grew slowly from 1112 hundred US dollars in 2016 to 4293 hundred US dollars in 2020 (β = 736.0, 95% CI 730.6 to 741.3, P < .001), with an average increase of 66.1% (95% CI 65.7 to 66.7) per year. Although apixaban has been approved in 2013 in China, it was rarely prescribed with a very low proportion below 1%.

Trends in prescriptions and costs of OACs over 11 years (2010 to 2020). Linear regression analysis with year as the independent variable and number of treatment visits or costs as the dependent variable, using the data from 2010 to 2020.
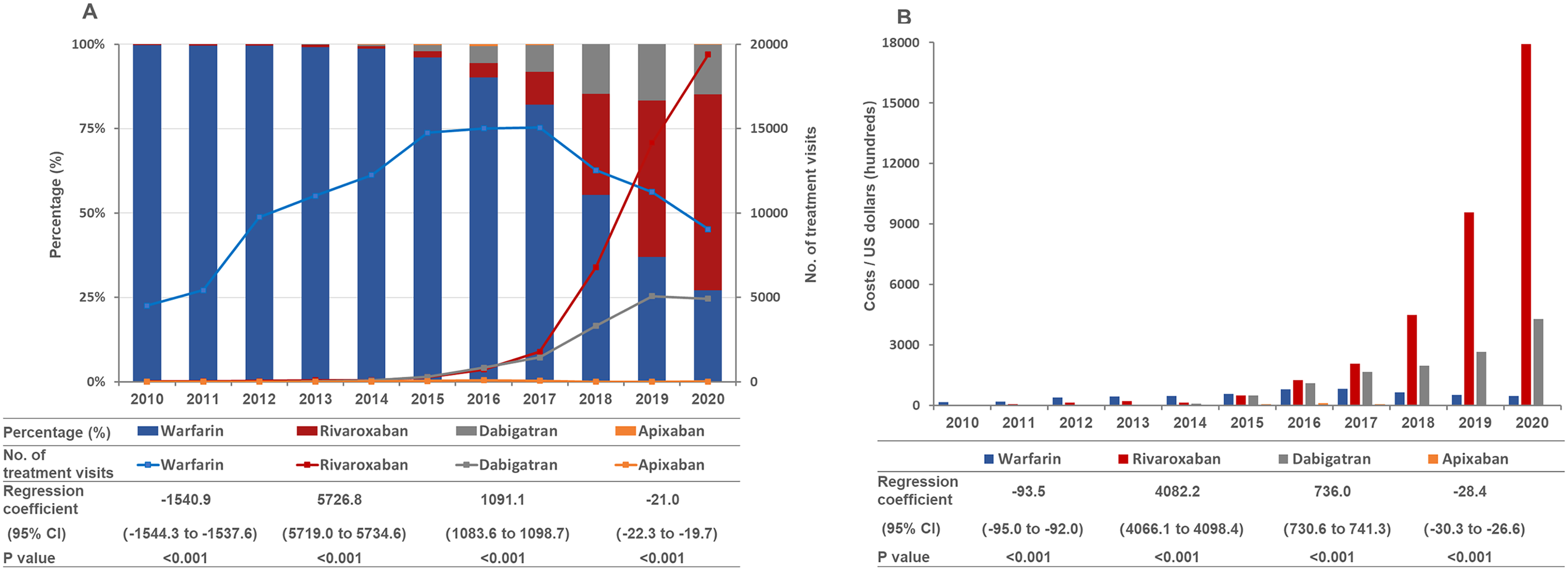
The number of treatment visits and the percentage (A) and the costs (B) for each OAC. Linear regression analysis with year as the independent variable and number of treatment visits or costs as the dependent variable, using the data from 2016 to 2020.
Trends by Indications
OACs are mostly used for stroke prevention in patients with nonvalvular AF and treatment of VTE, including deep vein thrombosis (DVT) and pulmonary embolism (PE). 11 The trends of OAC prescriptions were also described according to these two indications (Figure 3 and Figure S1). Generally, the prescription volume of OACs were much larger in AF patients than that in VTE patients (Figure S1). Dabigatran was first approved NOAC for stroke prevention in nonvalvular AF patients in 2013 in China (Table S5). Nevertheless, the extensive use of dabigatran in AF patients started from 2016 (β = 1080.8, 95% CI 1073.4 to 1088.2, P < .001), with the proportion increasing from 7% in 2016% to 20% in 2019 (Figure 3A and Table S6). A highly statistically significant upward trend (β = 4725.5, 95% CI 4715.8 to 4735.3, P < .001) was observed in prescriptions of rivaroxaban for AF patients from 2016 to 2020 after its approval in 2015. About 58% OAC prescriptions for patients with nonvalvular AF in 2020 were rivaroxaban, occupying the major market share (Table S6). In contrast, apixaban was rarely used for stroke prevention in AF patients, as it was not approved for the indication in China. In terms of prescriptions in prophylaxis and treatment of VTE, rivaroxaban also became the most popular OAC used since 2018, accounting for 51.56% in 2018% to 78% in 2020 (Figure 3B and Table S7). Dabigatran made up less than 10% of OAC prescriptions, and the proportion declined to only 3% in 2020. With the growing use of NOACs, the prescriptions of warfarin steadily declined for both AF patients (β = −812.7, 95% CI −816.2 to −809.3, P < .001) and VTE patients (β = −140.3, 95% CI −141.8 to −138.9, P < .001) since 2016, with only 23% and 19% share in 2020 for AF and VTE, respectively.

The number of treatment visits and the percentage for each OAC in AF (A) and VTE (B). Linear regression analysis with year as the independent variable and number of treatment visits as the dependent variable, using the data from 2016 to 2020.
For warfarin, rivaroxaban, and dabigatran, the majority of prescriptions were prescribed in AF (Figure 4), and the prescriptions in VTE treatment were much larger than those of VTE prophylaxis. There were still a minority of OAC prescriptions that were prescribed for other indications, such as atrial thrombosis and ventricular thrombosis, which were not presented. Besides the use in AF and VTE, warfarin can also be used in reduction of thrombotic risk in valvular heart disease (VHD) (Figure 4), including valvular AF, heart valve repair or replacement surgery. Whereas, the prescriptions of warfarin for VHD also started to slip since 2017.

The number of treatment visits for warfarin (A), rivaroxaban (B), and dabigatran (C) in different indications. Linear regression analysis with year as the independent variable and number of treatment visits as the dependent variable, using the data from 2016 to 2020.
Trends by Age Groups
Different tendencies were also found among patients in different age groups (Figure 5). For rivaroxaban, the fastest growth in prescriptions was observed in patients over 75 (β = 2857.8, 95% CI 2851.8 to 2863.8, P < .001) between the year of 2016 and 2020 (Figure S2). The prescriptions of dabigatran in patients over 75 (β = 599.7, 95% CI 593.5 to 605.9, P < .001) grew faster than those in patients between the age of 65 to 75 (β = 346.7, 95% CI 341.7 to 351.7, P < .001) and patients under the age of 65 (β = 123.2, 95% CI 121.7 to 124.7, P < .001). The prescriptions of warfarin in patients under the age of 65 began to decline since 2015 (Figure 5A), while the prescriptions in patients over 65 turned to decrease in 2017 (Figure 5B and Figure 5C). It is notable that about half of the users of rivaroxaban and dabigatran were over the age of 75, while only 26% patients using warfarin were older than 75 (Table S8).
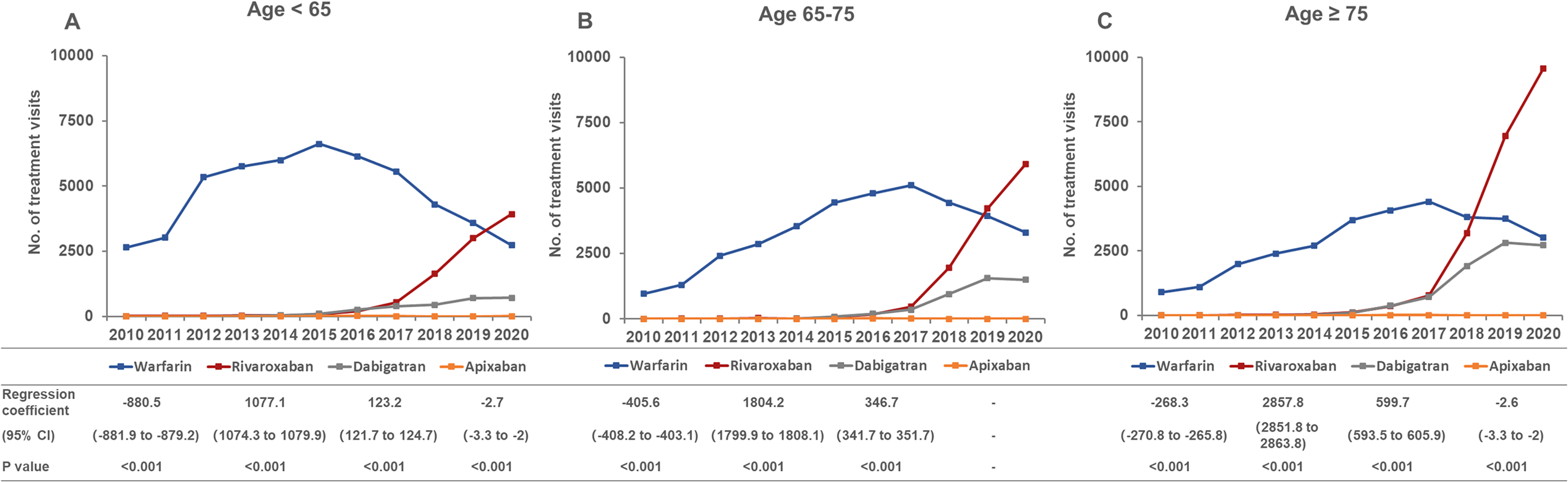
The number of treatment visits for each OAC in different age groups. Linear regression analysis with year as the independent variable and number of treatment visits as the dependent variable, using the data from 2016 to 2020.
Trends by Grades of Hospitals
The prescribing patterns of each OAC were also varied among different grades of hospitals (Figure 6). Most of OACs prescriptions were from tertiary hospitals (21 107 prescriptions in 2020), and only a small part of OACs were prescribed in CHSCs (1448 prescriptions in 2020). Apixaban was only available in tertiary hospitals, while warfarin, rivaroxaban, and dabigatran could be prescribed in all grades of hospitals. The prescriptions of warfarin started to decrease in tertiary hospitals since 2015 (Figure 6A), while the number in CHSCs kept increasing steadily since 2016 (β = 42.9, 95% CI 42.0 to 43.9, P < .001) (Figure 6B). Nevertheless, more than half of prescriptions for warfarin were still prescribed in tertiary hospitals (Figure S3).

The number of treatment visits for each OAC in different grades of hospitals. Linear regression analysis with year as the independent variable and number of treatment visits as the dependent variable, using the data from 2016 to 2020.
The prescriptions of rivaroxaban were growing rapidly since 2016 in all the three grades of hospitals, including tertiary hospitals (β = 3766.1, 95% CI 3759.5 to 3772.6, P < .001), secondary hospitals (β = 1756.2, 95% CI 1751.1 to 1761.3, P < .001), and CHSCs (β = 452.2, 95% CI 448.2 to 456.3, P < .001) (Figure 6). Rivaroxaban became the most popular OACs prescribed in 2020 regardless of grades of hospitals, accounting for 64.7%, 45.1% and 60.4% of OAC prescriptions in tertiary hospitals, secondary hospitals, and community health service centers, respectively. In 2020, 30% of rivaroxaban prescriptions were from secondary hospitals and CHSCs, indicating the improvements in availability of rivaroxaban in primary hospitals (Figure S3). The prescriptions of dabigatran were also in the upward trend in each grade of hospitals since 2016, with the highest increasing rate found in secondary hospitals (β = 625.5, 95% CI 622.6 to 628.4, P < .001), but the growth rates were much slower than those of rivaroxaban (Figure 6). Different from the prescribing pattern of rivaroxaban, over half (52%) of the prescriptions of dabigatran were prescribed in secondary hospitals and CHSCs in 2020 (Figure S3).
Discussions
This study describes the trends in prescribing OACs after the introduction of NOACs to the market in China. The current data indicate the rising trends in overall prescriptions for OACs. The prescriptions of NOACs were significantly increased in Shanghai since 2016, while the prescriptions of warfarin kept decreasing since 2017. Rivaroxaban emerged as a preferred NOAC in both indications of AF and VTE, and accounted for more than three-quarters of total costs for OACs since 2019. In contrast, dabigatran and apixaban were not so commonly prescribed. Another important observation was the difference in prescribing patterns in different indications, age groups, and grades of hospitals.
OACs were most prescribed in AF patients for stroke prevention, which was in accord with the situation in other countries and regions.12–14 and the increase of OAC prescriptions was considerable (Figure 1 and Figure S1). The population of AF patients are much larger than that of VTE patients. 15 It is reported that the prevalence of AF was around 2.3% in China, 16 while the annual incidence of VTE ranged from 14 to 57 per 100 000. 17 With the extended longevity of Chinese population, increased awareness of AF, and the convenient screening tools, the number of AF patients may keep growing. 16 In addition, lifelong anticoagulation is recommended to AF patients, while short-term anticoagulation, usually 3 to 6 months, is required for most VTE cases. 18 Therefore, it is understandable that the majority of OAC prescriptions were prescribed in AF patients. Moreover, different anticoagulation therapies need to be determined according to patients’ conditions. For patients with nonvalvular AF, NOACs are made as the Class IA recommendation by the current clinical guidelines. 19 Whereas, for patients with valvular AF, warfarin is preferred, and NOACs are discouraged, especially for mechanical prosthetic valve and moderate to severe mitral stenosis. 19 Warfarin is also preferred over NOACs in patients with VHD, antiphospholipid antibody syndrome, end-stage renal disease, and Child-Pugh Class C liver disease. 19 Recently, NOACs have been explored in patients with biological valves or after valve repair, and proven non-inferior to warfarin.19–21 This was in line with the fact observed in this study that the prescriptions of warfarin prescribed in VHD patients began to decrease since 2017 (Figure 4). In addition, the prescriptions of NOACs in older patients (over 75) grew faster than those in patients between the age of 65 to 75 and under the age of 65 (Figure S2). Older patients with AF are considered to have favorable outcomes (safer and more effective) on NOACs than on warfarin,22,23 even for patients over the age of 90. 24 The benefits of NOACs in those elderly patients mainly attribute to the lower risk of intracranial hemorrhage with no difference in risk of stroke compared with warfarin.24,25
Despite the favorable safety profile, non-inferior efficacy compared with warfarin, and ease of use, the high price of NOACs used to be prohibitive for many patients. With the involvement of National Reimbursement Drug List and the price reduction, NOACs are now considered to be more cost-effective options compared with warfarin for Chinese patients.26–28 Although there is no consensus in terms of most recommended NOAC, more preference to prescribing rivaroxaban was observed compared to dabigatran since 2017, which was consistent with studies in other countries.12,14 It is estimated that once-daily dosing in AF may contribute to the extensive use of rivaroxaban, as it is closely associated with the improvement of patients’ medication adherence. Moreover, active marketing strategy and promotion of rivaroxaban may be another important factor for its dominant market share. The prescription volume of apixaban in this study was very small and barely grew in the last decade. Although it is a talented NOAC for stroke prevention in AF patients with the lowest risk of major bleeding,25,29 apixaban was only approved for DVT prophylaxis in patients undergoing knee or hip replacement surgery. Edoxaban was the last approved NOAC in China in 2018, and was not involved in National Reimbursement Drug List until December 2020. Therefore, the use of edoxaban was very limited and was not presented in this study.
There were huge differences in the size of OAC prescription volume among each grade of hospitals. Although CHSCs are built with core responsibilities in preventing and managing chronic diseases in China, 30 a vast majority of health resources were concentrated at tertiary hospitals and secondary hospitals, and the share of outpatient visits at CHSCs was very small, which could explain the small volume and the slow growth of OAC prescriptions in CHSCs in this study. It is notable that the prescriptions of rivaroxaban grew faster than those of warfarin and dabigatran in CHSCs, indicating that rivaroxaban might be more available in CHSCs or be more familiar to clinicians in CHSCs than warfarin and dabigatran. The prescriptions of warfarin decreased rapidly in tertiary hospitals since 2017, and the decline was more smoothly in secondary hospitals. One possible explanation might be that clinicians in tertiary hospitals preferred to switch warfarin to NOACs for eligible patients because of the favorable outcomes of NOACs, while clinicians in secondary hospitals and CHSCs were accustomed to maintaining prescriptions for patients using warfarin. Therefore, with the increased awareness and understanding of NOACs among clinicians in secondary hospitals and CHSCs, NOACs may be more widely used in the future.
Studies have been conducted considering the trends of OAC use in other countries and regions.7,9,12,31 Similar trends of significantly increasing use of NOACs were observed in the last decade after their introduction to the market, indicating the wide acceptation and preference of clinicians in the use of NOACs all over the world. Different from the dominant share of rivaroxaban in this study, apixaban was the most prescribed NOAC in England and Norway, despite its delayed market entry.9,13 For elderly patients, NOACs were found preferred in this study, which was in accord with the trend in U.S. that the proportion of NOACs increased from 35.4% in 2020 to 57.8% in 2014 for patients over 65 years old. 32 On the contrary, patients using warfarin were on average older than those receiving NOACs in Norway and State of Texas.13,31 In addition, it is assumed that more and more patients would be initially prescribed NOACs instead of warfarin,33,34 and patients already used warfarin might switch to NOACs in the future, especially for patients with low therapeutic range.35,36 These might because current clinical guidelines strongly recommend the NOACs over warfarin in many treatment indications,4,5,19 and patients receiving NOACs were reported to have higher adherence and satisfaction than those received warfarin.37–39 Nevertheless, it is uncertain whether switching from warfarin to NOACs could benefit the patients. 40 One study found reduced risk of gastrointestinal bleeding, intracranial hemorrhage, and all-cause mortality in patients switching from warfarin to dabigatran compared with warfarin users. 41 Whereas, another study reported that patients switching to rivaroxaban had an increased risk of gastrointestinal bleeding. 42 Moreover, one meta-analysis found that use of dabigatran in prior warfarin users was associated with a slightly increased risk of arterial thromboembolism and gastrointestinal bleeding, but a decreased risk of intracranial hemorrhage. 43 Accordingly, despite the benefits of NOACs brought to patients, use of NOACs in warfarin-experience users should be with cautions.
This study must be interpreted considering several limitations. First, the results obtained in this study were based on sampling data, which could only present the prescribing trend of OACs and could not represent the exact number of patient visits and costs. Rising prescriptions or treatment visits could not fully indicate the rising number of OAC users, because longer-term anticoagulation and improved patient adherence could also increase the prescription number. Second, although the sampling data of OAC prescriptions were representative, it did not include prescriptions from online pharmacies and private hospitals, which might affect the generalizability of the results. Third, some information captured from the medical records was incomplete or have some errors or inconsistencies. Therefore, the rationality of OAC use and the influence factors could not be conducted. Moreover, RAS is a database randomly selecting prescription data of outpatients, and the consecutive information of a certain patient is not available. Accordingly, the duration of OAC treatment as well as the change of OAC regimen could not be analyzed in this study.
Conclusions
There has been a rapid increase in the use of OAC over the last 11 years in Shanghai, China. NOACs have been adopted rapidly, achieving more than 70% of all OAC use. Rivaroxaban has emerged as a preferred NOAC in both AF and VTE patients. Despite the growing use of NOACs, warfarin remains the top choice for patients with mechanical prosthetic valve and moderate to severe mitral stenosis. Future studies are warranted considering changes in the OAC use in a larger scale, as well as the rationality and its influence factors on OAC use.
Supplemental Material
sj-docx-1-cat-10.1177_10760296221132551 - Supplemental material for Prescribing Trends of Oral Anticoagulants from 2010 to 2020 in Shanghai, China: A Retrospective Study
Supplemental material, sj-docx-1-cat-10.1177_10760296221132551 for Prescribing Trends of Oral Anticoagulants from 2010 to 2020 in Shanghai, China: A Retrospective Study by Chi Zhang, Jia Wang, Ya Yang, Er-Li Ma, Hou-Wen Lin, Bing-Long Liu and Zhi-Chun Gu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Contributions
Z.G. and C.Z. designed this study. J.W. and Y.Y. collected and analyzed the data. E.M. AND B.L. supervised the finding of this study. C.Z. wrote the manuscript. Z.G. and H.L. revised the manuscript.
Data Availability
All data are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by the ethics committee of Ren Ji Hospital (KY2021-274-B), and a waiver of informed consent was obtained.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Clinical Research Innovation and Cultivation Fund of Ren Ji Hospital, Shanghai “Rising Stars of Medical Talent” Youth Development Program – Youth Medical Talents – Clinical Pharmacist Program, Research Project of Drug Clinical Comprehensive Evaluation and Drug Treatment Pathway, Three-Year Plan for Promoting Clinical Skills and Innovation in Municipal Hospitals of Shanghai Shenkang Hospital Development Center, Ren Ji Boost Project of National Natural Science Foundation of China, (grant number RJPY-LX-008, SHWRS (2020) 087, SHYXH-ZP-2021-001, SHDC2022CRS035, RJTJ-JX-001).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
