Abstract
Background
Advanced age and type 2 diabetes (T2D) are common in patients with nonvalvular atrial fibrillation (NVAF). We evaluated the impact of age on the effectiveness and safety of rivaroxaban versus warfarin in this population.
Methods
We analyzed electronic health record data from November 2010, to December 2019 including adults with NVAF and T2D, newly started on rivaroxaban or warfarin. Propensity score-overlap weighted hazard ratios (HRs) for stroke/systemic embolism (SSE), hospitalization for major or clinically relevant nonmajor bleeding (CRNMB), vascular death, major adverse limb events (MALE), major bleeding, and intracranial hemorrhage (ICH) were calculated for older (≥80 years) and younger (<80 years) cohorts.
Results
We included 32 078 rivaroxaban and 83 971 warfarin users (6606 rivaroxaban and 25,335 warfarin patients were aged ≥80 years). No significant interaction for rivaroxaban versus warfarin by age was observed for any outcome, including SSE (HR = 1.05 vs 0.95), hospitalization for major or CRNMB (HR = 1.06 vs 0.90), vascular death (HR = 0.92 vs 0.90), MALE (HR = 0.80 vs 0.76), major bleeding or ICH.
Conclusions
The effectiveness and safety of rivaroxaban versus warfarin remained consistent across patient age subgroups.
Introduction
Patients with diabetes have a 49% increased risk of developing atrial fibrillation (AF) compared with those without.1,2 These patients, specifically those with nonvalvular AF (NVAF), are at a fivefold increased risk of stroke and up to a twofold increased risk of mortality.3,4 Additionally, in patients aged 80 years and older, more than 1 in 5 strokes are attributable to AF, while a quarter of patients >60 years old have diabetes. 3 To reduce the risk of morbidity and mortality in these patients, oral anticoagulants (OACs) such as vitamin K antagonists (VKA) and direct-acting OACs (DOACs) are typically prescribed. These agents decrease thrombus formation and subsequently decrease mortality.
Data from randomized controlled trials, administrative claim database analyses, and electronic health record (EHR) studies have demonstrated that DOACs, such as rivaroxaban, are at least as effective and safe as warfarin in preventing stroke and systemic embolism (SSE) in patients with NVAF and type 2 diabetes (T2D).5–8 Data comparing outcomes associated with rivaroxaban and warfarin use in NVAF patients with T2D stratified by advanced age are scarce. These data are important given the increased risk of AF and T2D with age, the attributable risk of stroke due to AF when T2D in present, and older patients’ baseline risk of bleeding compared to a younger population.3,9,10
In this study, we aimed to assess the incidence rates of thrombosis, vascular death, and bleeding events in patients with NVAF and T2D who were prescribed rivaroxaban or warfarin and were stratified by age under versus over 80 years.
Methods
The rivaroxaban in diabetes and NVAF study (RIVA-DM, Trial Registration: NCT04509193) was a retrospective cohort study that utilized the US Optum® De-Identified EHR data from November 1, 2010, through December 31, 2019. The US Food and Drug Administration approval date of rivaroxaban for NVAF was in November 2011; therefore, utilization back to November 2010 was needed to have a 12-month pre-index information for all patients. The EHR data set utilized included longitudinal patient-level medical record data for more than 90 million patients seen at over 700 hospitals and 7000 clinics across the United States. 11 It contains data on insured and uninsured patients of all ages, which provide a representative sample of US patients. Records of prescriptions and over-the-counter medications (as prescribed or self-reported by patients), laboratory results, vital signs, anthropometrics, clinical diagnostic codes (International Classification of Diseases, Ninth Revision [ICD-9] and Tenth Revision [ICD-10]), and procedural codes (ICD-9, ICD-10, CPT-4, Healthcare Common Procedure Coding System, Revenue codes) are contained in the data set. The use of Optum® De-Identified EHR data has been determined by the New England Institutional Review Board to not constitute research involving human subjects and was therefore exempt from institutional review board oversight.
Patients were included if they were aged ≥18 years, diagnosed with NVAF and T2D, were OAC naive, newly initiated on rivaroxaban or warfarin after November 1, 2011 (defined as the index date), were active in the data set for at least 12 months prior to the index data, and had documented care in the EHR from at least one provider in the 12 months prior to the index date. Patients were excluded if there was presence of valvular heart disease, any prior OAC use, received rivaroxaban dose other than 15 mg or 20 mg once daily, had venous thromboembolism as an alternative indication for OAC use, underwent recent orthopedic surgery, or were pregnant. Both the specificity and sensitivity for billing codes to identify NVAF were high (≥98% and ≥80%, respectively). 12 The specificity for billing codes to identify T2D was high (>94%), but the sensitivity was low (≤59). 13 To address the lower sensitivity of T2D codes, we also included patients in this study if they had both hemoglobin A1c ≥6.5% and were receiving a noninsulin antihyperglycemic medication at baseline.
To adjust for potential confounding between the rivaroxaban and warfarin cohorts, propensity scores were calculated using a multivariable logistic regression model. 14 The propensity score model consisted of commonly used variables and risk factors for differential OAC exposure identified at baseline, including demographics, comorbidities, laboratory values, vital signs, and outpatient medication use. The complete list of covariates used in the propensity score can be seen in Table 1. Comorbid disease presence was determined via billing codes and/or supporting laboratory and clinical data. The absence of data suggesting a comorbidity exists was assumed to mean the absence of the disease. When dependence on billing codes was required to identify a disease, we utilized validated coding algorithms (ie, Centers for Medicare and Medicaid Services Chronic Conditions Data Warehouse, Elixhauser or Charlson Comorbidity Indices) whenever possible.15–18 For continuous variables with <25% missing data, we imputed values using a multiple imputation approach based on a fully conditional specification linear regression model with all other available covariates and outcomes of interest included. 19 Propensity scores were subsequently used to assign weights to individual patients in the analysis via an overlap weighting approach.20,21 Overlap weighting assigns weights that are proportional to the patients’ probability of belonging to the opposite treatment cohort. By design, overlap weighting results in the exact balance of all variables included in the propensity score model. This method of adjustment retains all patients in the data set and gives less weight to patients with extreme propensity scores.
Unweighted and Weighted Baseline Characteristics of Included Patients in Subgroup Analysis.

Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin-receptor blockers; BMI, body mass index; CABG, coronary artery bypass graft; CHA2DS2-VASc, Congestive heart failure, Hypertension, Age ≥75 years (2 points), Diabetes mellitus, Stroke or transient ischaemic attack (2 points), Vascular disease, Age 65–74, Sex category (female); CHADS2,Congestive heart failure, Hypertension, Age ≥75 years, Diabetes mellitus, Stroke or transient ischaemic attack (2 points); DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; GLP, glucagon-like peptide; H. pylori, Helicobacter pylori; HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly; INR, international normalized ratio; MI, myocardial infarction; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; SBP, systolic blood pressure; SD, standard deviation; SNRI, serotonin-norepinephrine reuptake inhibitors; SSRI: selective serotonin reuptake inhibitors.
Covariate not included in the propensity score model.
Baseline characteristics were analyzed and reported using descriptive statistics. Categorical variables were reported as percentages and continuous variables as means ± standard deviations (SDs). Propensity score-overlap weighted Cox proportional hazards regression models using a robust estimator were used to calculate hazard ratios (HR) and 95% confidence intervals (CI) for all outcomes. Patients were censored in the Cox models at the time of outcome occurrence, end-of-EHR activity, or end-of-data availability (December 31, 2019).
The study's primary outcomes included the incidence rates (%/year) of developing SSE (effectiveness outcome defined by a diagnosis code for stroke in the primary position associated with a hospitalization) and hospitalization due to major or clinically relevant nonmajor bleeding (CRNMB; safety outcome defined per the Cunningham algorithm). 22 Secondary outcomes included the composite of SSE/vascular death, any major bleeding (a Cunningham algorithm bleeding-related billing code in the primary coding position associated with an inpatient or outpatient encounter), and major adverse limb events (MALE; defined as revascularization or major amputation of the lower limbs and identified by ≥1 inpatient or outpatient diagnosis or procedural billing code in the primary or nonprimary position), along with individual components of composite outcomes.
All outcomes were compared between rivaroxaban and warfarin for a population aged <80 years and ≥80 years. Statistical comparison of age population results was made using the Benjamini and Yekutieli adjustment method for multiple hypothesis testing, with a P < .05 considered statistically significant in all cases. 23 Statistical analysis and database management was done using IBM SPSS version 27.0 (IBM Corp., Armonk, NY) and R Studio version 4.0.2.
Results
We identified 32,078 rivaroxaban patients (25,472 aged <80, 6606 aged ≥80) and 83,971 warfarin patients (58,636 aged <80, 25,335 aged ≥80) with NVAF and T2D. Baseline characteristics prior to propensity score-overlap weighting are depicted in Table 1. After propensity score-overlap weighting, rivaroxaban and warfarin cohorts had identical (standardized difference = 0 for all) comorbidity incidences and mean laboratory and clinical parameters as intended by methodology. In the age <80 population, the mean age was 67 ± 9, CHA2DS2VASc (Congestive heart failure, Hypertension, Age ≥75 years [2 points], Diabetes mellitus, Stroke or transient ischaemic attack [2 points], Vascular disease, Age 65-74, Sex category [female]) was 3.8 ± 1.3 and HAS-BLED (Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly, Drugs/alcohol concomitantly) was 1.4 ± 0.8. In the age ≥80 cohort, the mean age was 83 ± 2, CHA2DS2VASc was 4.41 ± 1.2, and HAS-BLED was 1.7 ± 0.7. In both age cohorts, approximately 38% of patients (31 961 patients aged <80 years and 12 361 patients aged ≥80 years) had an intermediate hospital frailty score, while 8177 (25.6%) of patients aged ≥80 years and 13 373 (15.9%) aged <80 years were determined to be highly frail (20 538 (64.3%) of patients aged ≥80 years had intermediate to high frailty scores). The mean follow-up time for rivaroxaban and warfarin patients was 2.9 ± 1.9 and 2.9 ± 2.0 years, respectively.
Propensity score-overlap weighted analyses found no significant interaction for the relative effectiveness or safety of rivaroxaban versus warfarin across the older or younger age groups for the outcomes of SSE (HR: 1.05, 95% CI: 0.92-1.19 vs HR: 0.95, 95% CI: 0.87-1.04) and hospitalization for major or CRNMB (HR: 1.06, 95% CI: 0.96-1.18 vs HR: 0.90; 95% CI: 0.84-0.96) (Table 2). No significant interaction for rivaroxaban versus warfarin across the older or younger age groups for the composite outcome of SSE or vascular death or MALE were observed (Table 3). Vascular death, MALE, as well as major bleeding and intracranial hemorrhage (ICH) were observed significantly less frequently with rivaroxaban compared with warfarin regardless of patient age.
Primary Outcomes.
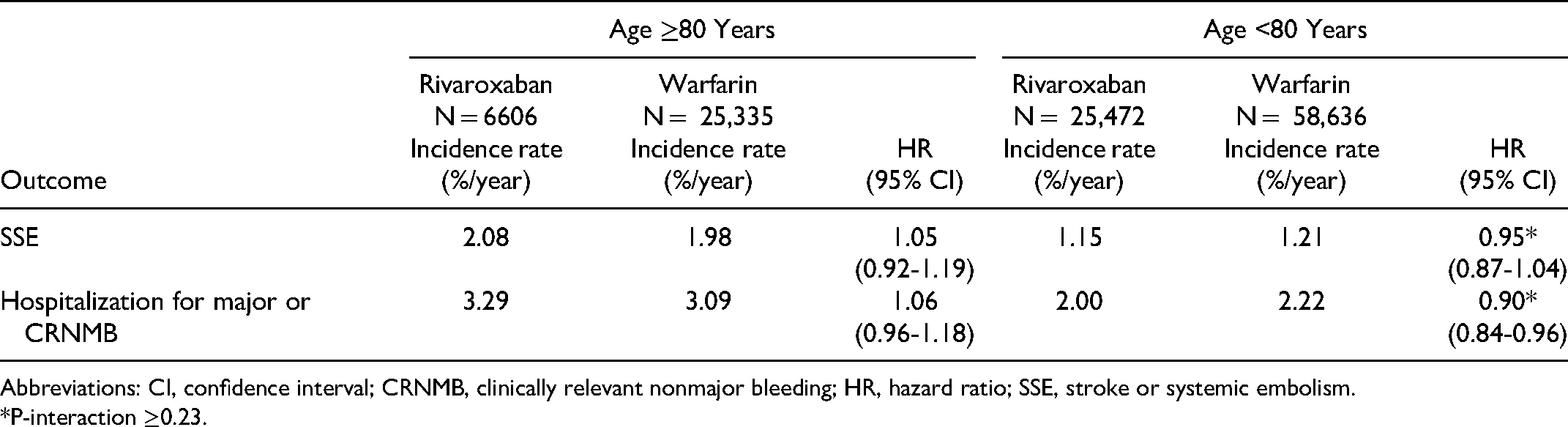
Abbreviations: CI, confidence interval; CRNMB, clinically relevant nonmajor bleeding; HR, hazard ratio; SSE, stroke or systemic embolism.
*P-interaction ≥0.23.
Secondary Outcomes.
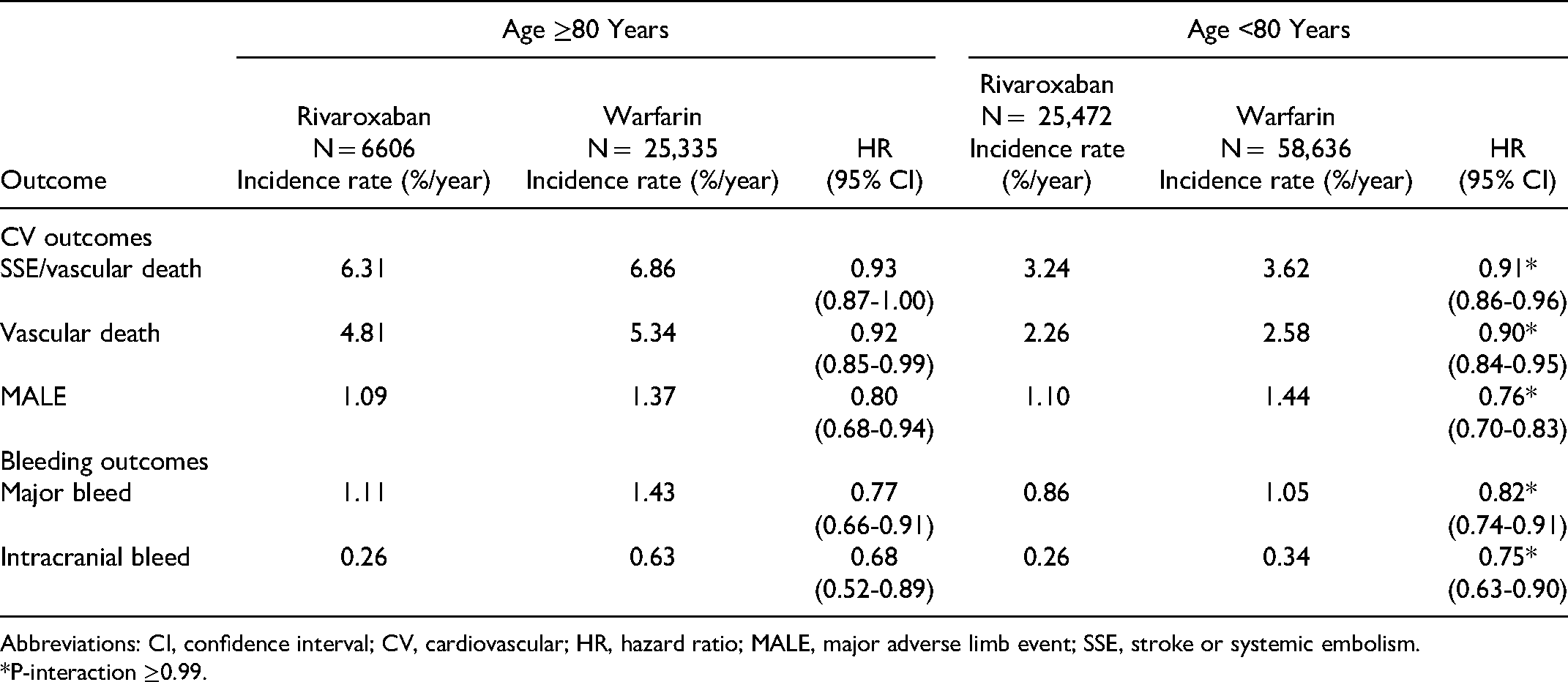
Abbreviations: CI, confidence interval; CV, cardiovascular; HR, hazard ratio; MALE, major adverse limb event; SSE, stroke or systemic embolism.
*P-interaction ≥0.99.
Discussion
In this study of more than 116,000 patients with NVAF and comorbid T2D, patients aged ≥80 years or older and aged <80 years received a consistent incidence rates of SSE and and hospitalization for major or CRNMB when receiving rivaroxaban compared with warfarin. Moreover, rivaroxaban use was associated with significantly lower rates of vascular death, MALE, any major bleeding, and ICH versus warfarin, regardless of patient age. These data support the use of rivaroxaban as an efficacious and safe alternative to warfarin in older patients with NVAF with concomitant T2D. Rivaroxaban has the advantages of easier dosing, no need for drug monitoring, and fewer drug-drug or drug-food interactions compared to warfarin.
Our study findings are very much in-line with current AF treatment guidelines. 24 The European Society of Cardiology (ESC) and European Association for the Study of Diabetes (EASD) collaborative guidelines on the management of diabetes, pre-diabetes, and cardiovascular diseases recommend DOACs (such as rivaroxaban) over a VKA in patients with diabetes aged >65 years with NVAF and a CHA2DS2VASc score ≥ 2, (class 1A recommendation). 24 Given the accumulating data on the safety of DOACs in patients of advanced age with T2D, the practice of preferentially using DOACs over a VKA in a patient with diabetes would seem warranted.
The present study reinforces findings from the phase 3 Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) and the bleeding risk in elderly Subjects Aged more than 80 years in atrial FIbrillation treated by Rivaroxaban (SAFIR) observational study.6,25,26 ROCKET AF randomized 14,264 patients with NVAF to receive either rivaroxaban or warfarin for the prevention of SSE. 6 In a subanalysis of ROCKET AF stratified by the presence or absence of diabetes, 40% of patients had diabetes at the time of treatment initiation. 5 The substudy demonstrated the relative effect of rivaroxaban and warfarin on SSE, major bleeding, major or CRNMB, and ICH appeared similar in patients with and without diabetes (P-interaction ≥.17). Of all patients randomized in ROCKET AF, 44% were of more advanced age (≥75 years) at treatment start. 6 Upon a separate analysis stratified by age, investigators demonstrated the incidence rates for patients receiving rivaroxaban compared with warfarin were similar between older and younger patients for SSE (P-interaction ≥.31). 25 While older patients in ROCKET AF experienced more major bleeding events overall, no difference in the treatment effect of rivaroxaban versus warfarin between older and younger subjects was noted (P-interaction ≥.34). More recently, the prospective, observational SAFIR study compared 1-year outcomes in newly initiated rivaroxaban (N = 995) and VKA patients (N = 908) with NVAF and aged ≥80 years. Similar to our present study, SAFIR found major bleeding risk was significantly lower in rivaroxaban-treated patients compared with VKA-treated patients in a propensity score–matched analysis (HR: 0.53, 95% CI: 0.33-0.85) and ICH occurred less frequently in rivaroxaban-treated patients than in VKA-treated patients in the propensity score–matched analysis (HR: 0.26, 95% CI: 0.09-0.80). No difference between anticoagulation cohorts was observed for the ischemic stroke (HR: 0.44, 95% CI: 0.15-1.30) outcome.
Our study has limitations worthy of discussion. First, because of the nonrandomized, retrospective nature of this EHR study, misclassification, sampling, and confounding bias may impact its internal validity. 27 We attempted to mitigate misclassification bias by using validated coding schema and leveraging objective laboratory and clinical observation data available in an EHR but not claims data sets.11,16–18,22 We used propensity score-overlap weighting to balance patients on numerous important demographics, clinical and medication covariates to reduce the risk of confounding bias.20,21 Although propensity score–based methods may serve to balance comparison groups, residual confounding due to variables not included or unavailable for inclusion (ie, time since diabetes diagnosis) into the propensity score model cannot be ruled out. 14 Second, the cause of death was also not available in the data set; therefore, we had to utilize an algorithm consisting of hospitalization due to vascular cause within 365 days of death to identify “vascular” mortality. This method has been shown in previous observational studies to provide vascular mortality rates similar to those reported in “like” randomized controlled trials. 8 Third, the EHR data set utilized for this study included only US patients. 11 Our results may be less generalizable to populations outside the United States. Finally, the EHR data set used for this study did not capture prescription medication claims but rather information on medications prescribed or self-reported. 11 The lack of formal prescription claims data (generated when a patient picks up their medication at a pharmacy) makes ascertainment of OAC exposure (persistence and adherence) difficult. Consequently, we did not perform an on-treatment analysis.
Conclusion
The effectiveness and safety of rivaroxaban relative to warfarin remained consistent across older and younger patient subgroups, supporting rivaroxaban as an alternative for older patients with NVAF with concomitant T2D. Vascular death, MALE, major bleeding, and ICH were observed less frequently with rivaroxaban versus warfarin regardless of patient age.
Footnotes
Acknowledgments
The authors thank Hillary Sotheran for providing editorial assistance in the preparation of the manuscript, with funding from Bayer AG.
Ethics Approval
The use of the Optum EHR data set was reviewed by the New England Institutional Review Board and was determined to be exempt from oversight, as this research project did not involve human subject research and the investigators were supplied only de-identified and HIPAA-compliant data.
Declaration of Conflicting Interests
CIC has received research funding and consulting fees from Bayer AG, Janssen Pharmaceuticals, Alexion Pharmaceuticals and presentation fees from Medscape. OSC has received presentation fees from and is an employee of Janssen Pharmaceuticals. CWB and NS have no conflicts of interests to disclose. BV, LH and KA are employees of Bayer AG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bayer AG.
Trial Registration
NCT04509193.
