Abstract
Podoplanin (PDPN) promotes platelet aggregation and activation by interacting with C-type lectin-like receptor 2(CLEC-2) on platelets. The interaction between the upregulated PDPN and platelet CLEC-2 stimulates venous thrombosis. PDPN was identified as a risk factor for coagulation and thrombosis in inflammatory processes. Hypercoagulability is defined as the tendency to develop thrombosis according to fibrinogen and/or D dimer levels. Nephrotic syndrome is also considered to be a hypercoagulable state. The aim of this study is to investigate the association of soluble PDPN/CLEC-2 with hypercoagulability in nephrotic syndrome. Thirty-five patients with nephrotic syndrome and twenty-seven healthy volunteers were enrolled. PDPN, CLEC-2 and GPVI concentrations were tested by enzyme-linked immunosorbent assay (ELISA). Patients with nephrotic syndrome showed higher serum levels of PDPN and GPVI in comparison to healthy controls (P < .001, P = .001). PDPN levels in patients with nephrotic syndrome were significantly correlated with GPVI (r = 0.311; P = .025), hypoalbuminemia (r = −0.735; P < .001), hypercholesterolemia (r = 0.665; P < .001), hypertriglyceridemia (r = 0.618; P < .001), fibrinogen (r = 0.606; P < .001) and D-dimer (r = 0.524; P < .001). Area under the curve (AUC) for the prediction of hypercoagulability in nephrotic syndrome using PDPN was 0.886 (95% CI 0.804-0.967, P < .001). Cut-off value for the risk probability was 5.88 ng/ml. The sensitivity of PDPN in predicting hypercoagulability was 0.806, and the specificity was 0.846. When serum PDPN was >5.88 ng/ml, the risk of hypercoagulability was significantly increased in nephrotic syndrome (OR = 22.79, 95% CI 5.92-87.69, P < .001). In conclusion, soluble PDPN levels were correlated with hypercoagulability in nephrotic syndrome. PDPN has the better predictive value of hypercoagulability in nephrotic syndrome as well as was a reliable indicator of hypercoagulable state.
Introduction
Podoplanin (PDPN) is a sialomucin-like single transmembrane glycoprotein that is primarily expressed on the surface of lymphatic endothelial cells, kidney podocytes, type-I alveolar cells, fibroblastic reticular cells in the lymph nodes, synovial cells in the joint, but not in vascular endothelial cells. 1 Studies have shown that PDPN plays a role in embryonic development of the lymphatic system and lungs, vascular integrity, cellular migration and platelet activation.1,2 Mice deficient for PDPN exhibit striking developmental defects, suggesting an important role of this glycoprotein in embryonic development.3,4 PDPN is also found to be upregulated on particular cells of the immune system such as subsets of inflammatory macrophages and Th17 cells, and may be important for regulating immune responses during inflammation.5,6 PDPN is implicated in chronic inflammatory diseases, such as psoriasis, multiple sclerosis, and rheumatoid arthritis.7,8 Furthermore, PDPN promotes platelet aggregation and activation by interacting with C-type lectin-like receptor 2(CLEC-2) on platelets.9,10 CLEC-2 is highly and almost specifically expressed in platelets and megakaryocytes, and is identified as a receptor for platelet activating snake venom, rhodocytin. Up to now, PDPN is the only known endogenous ligand for CLEC-2.9,10 The proposed prothrombotic mechanism and thrombotic events of PDPN is activation of platelets via CLEC-2.11–13 Evidence suggests that the interaction between the upregulated PDPN and platelet CLEC-2 stimulates venous thrombosis both in an inferior vena cava stenosis mouse model of deep venous thrombosis and in an in vivo mouse model of infection-driven thrombosis.13,14 The thrombus formation with CLEC-2-deficient platelets was severely impaired in vitro. 13 Patients with primary brain tumors develop venous thromboembolism, an event strongly associated with overexpression of PDPN. 15 PDPN was identified as a risk factor for coagulation and thrombosis in inflammatory processes and cancer because of its ability to induce and activate platelets. 16 After platelet activation, numerous procoagulant factors and other mediators from platelet granules are released that may induce hypercoagulability and thrombus formation. Hypercoagulability is defined as the tendency to develop thrombosis and implies that one individual has a heightened potential to develop thrombosis when compared to a normal individual. 17
Nephrotic syndrome is characterized by heavy proteinuria, edema, hypoalbuminemia, and hyperlipidemia. Patients with nephrotic syndrome have an increased risk of thromboembolism, which is associated with substantial morbidity and mortality. The incidence of deep venous thrombosis, pulmonary embolus and renal vein thrombosis has been reported to be approximately 25% of patients with nephrotic syndrome.18,19 Current studies have shown that the nephrotic syndrome is a thrombophilic or hypercoagulable state.18–20 It is presently thought that the mechanisms of hypercoagulability are mutifactorial in the nephritic syndrome. Abnormalities of balance between procoagulant and fibrinolytic proteins have promoted thrombosis. The increase in fibrinogen, in particular, is a hepatic synthetic response to the hypoalbuminemia of nephrotic syndrome. D-dimer is an indicator of coagulation activation and fibrinolysis, and a marker of hypercoagulability and thrombotic activity. 17 In addition to the imbalance between anticoagulation and procoagulant mechanisms, increased platelet activation and aggregability are closely related to prothrombotic state and hypercoagulability during the development of nephrotic syndrome.18,19 Platelet hyperaggregability, increased release of active substances, and elevated surface expression of activation-dependent platelet markers have been observed in nephrotic syndrome. Therefore, coagulation associated with platelet activation and aggregability is important processes that contribute to development of nephrotic syndrome. Recent studies have demonstrated a role for the PDPN/CLEC-2 in driving hypercoagulable state and thrombosis during platelet activation and deep vein thrombosis. 16 Inhibition of PDPN and CLEC-2 deficiency result in significantly reduced extension of thrombosis.12–14 However, the role of PDPN/CLEC-2 has not been examined in the nephrotic syndrome and the relationship between PDPN/CLEC-2 and hypercoagulable state is less determined. The aim of this study is to investigate the soluble PDPN/CLEC-2 levels and to assess their association with hypercoagulability in patients with nephrotic syndrome.
Materials and Method
Study Population
Thirty-five patients aged over 18 years in the Department of Nephrology, First Affiliated Hospital of Harbin Medical University, China, were enrolled in this study. All the patients were newly diagnosed with primary nephrotic syndrome based on clinical manifestations, laboratory tests and kidney biopsy. The exclusion criteria for the study were as follow: (1) patients with secondary nephrotic syndrome, (2) patients with autoimmune diseases, diabetes mellitus, hepatitis, liver cirrhosis or abnormal liver function, (3) patients at high risk of hypercoagulable state, such as the acute phase of infection, recent trauma, surgery, pregnancy and cancer, (4) patients receiving hormones, anticoagulants or diuretics. In addition, twenty-seven healthy age and sex matched individuals were selected as healthy controls. This study (2022IIT040) was approved by the Research Ethics Committee of the Hospital of Harbin Medical University and informed consent was obtained from all the participants.
Clinical Data Collection
Demographics, clinical, and laboratory data were obtained via direct interview and electronic medical record database, including age, gender, albumin, alanine aminotransferase, aspartate aminotransferase, total cholesterol, triglyceride, low-density lipoprotein cholesterol, highdensity lipoprotein cholesterol, blood glucose, uric acid, urea, creatinine, 24 h urine protein, leukocyte, erythrocyte, thrombocyte, fibrinogen, prothrombin time, activated partial thromboplastin time, and D-dimer. Hypercoagulability is defined as the tendency to develop thrombosis according to the fibrinogen and/or D-dimer. The hypercoagulable state was defined as fibrinogen levels >3.5 g/L and/or D dimer levels >0.55 mg/L FEU. Estimated glomerular filtration rate (eGFR) was calculated according to the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula (ml/minute/1.73 m2 body surface area). 21
Blood Collection and Measurement of Soluble PDPN, CLEC-2 and GPVI
Before clinical treatment peripheral blood were obtained in vacuum tubes (Becton Dickinson and Company, USA) in the morning before breakfast and centrifuged at 1500 g for 15 min. The supernatants were stored at 80°C until assayed. PDPN and GPVI concentrations were measured using commercially available enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer's instruction (Cloud-Clone Corp. Houston, USA). CLEC-2 concentrations were measured using commercially available ELISA kits according to the manufacturer's instruction (RayBiotech, Inc.Georgia, USA). All samples were analyzed in duplicate. The results were expressed as nanograms per milliliter. The low limits of detection for PDPN, GPVI and CLEC-2 were 0.156 ng/ml, 0.0156 ng/ml and 1.229 ng/ml respectively.
Statistical Analysis
Data were expressed as mean ± standard deviation (SD) for quantitative variables and as percentages for categorical variables. Normal distribution of the data was evaluated by the Kolmogorov–Smirnov test. Comparison of quantitative data between two groups was tested using the appropriate Student's t test. Chi-squared tests were used to compare categorical data. Non–normal distributed data were analyzed by means of nonparametric tests. Pearson correlation coefficient was used for the correlation analysis between measured indexes and laboratory parameters. Receiver operating characteristic curve (ROC) was performed to identify the predictive value of PDPN. The appropriate cut-off values were calculated to maximize the sensitivity and specificity. All P values were two-tailed and a value of ≤.05 was considered statistically significant. SPSS version 23.0 software was used to perform the analysis
Results
Baseline Characteristics of Patients with Nephrotic Syndrome and Healthy Controls
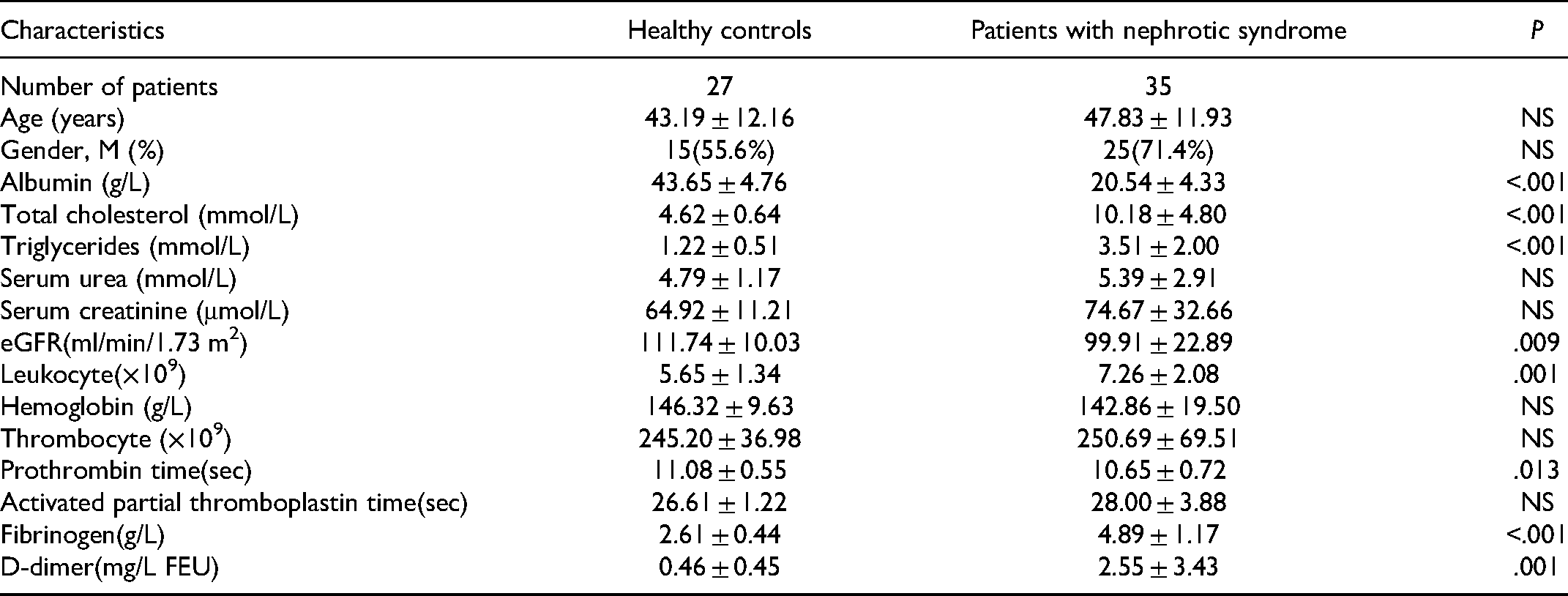
Thirty-five patients with nephrotic syndrome and twenty-seven healthy controls were included in the study. The baseline characteristics of all patients and healthy controls are shown in Table 1.
The Clinical Characteristics in all the Patients and Healthy Controls.
Soluble PDPN/CLEC-2 and GPVI
The concentrations of CLEC-2 in patients with nephrotic syndrome and healthy controls were (5.88 ± 0.23)ng/ml and (6.19 ± 0.60)ng/ml, respectively (P = .091, Figure 1A). The concentrations of PDPN in patients with nephrotic syndrome and healthy controls were (10.51 ± 4.48)ng/ml and (3.78 ± 1.03)ng/ml, respectively (P < .001, Figure 1B). Patients with nephrotic syndrome showed higher serum levels of PDPN in comparison to healthy controls. Serum levels of GPVI in nephrotic syndrome patients were significantly higher than those in healthy controls (38.60 ± 15.00 ng/ml versus 27.46 ± 6.96 ng/ml, P = .001, Figure 1C).

Soluble CLEC-2, PDPN and GPVI levels in the patients with nephrotic syndrome and healthy controls. A. Analysis of serum CLEC-2 levels shows no significant differences between patients with nephrotic syndrome and healthy individuals (P = .091). B and C. Patients with nephrotic syndrome showed higher serum levels of PDPN and GPVI in comparison to healthy controls (P < .001 and P = .001).
Correlation between serum Soluble PDPN and GPVI in Patients with Nephrotic Syndrome
To determine whether the elevated PDPN is related to the platelet activation of nephrotic syndrome, correlation between serum levels of PDPN and GPVI were analyzed. It was found that serum PDPN was found to be positively correlated with GPVI (r = 0.311; P = .025, Figure 2).

Correlation of soluble PDPN levels with GPVI. The levels of PDPN are positively correlated with GPVI (r = 0.311; P = .025).
Correlation of PDPN with Albumin, Total Cholesterol, Triglyceride, Fibrinogen and D-dimer in Patients with Nephrotic Syndrome
Next, spearman correlation analysis of soluble PDPN levels associated with hypoalbuminemia, hypercholesterolemia, hypertriglyceridemia, fibrinogen and D-dimer was performed, to identify correlations of PDPN with hypercoagulability. Results showed that soluble PDPN levels were significantly correlated with hypoalbuminemia (r = −0.735; P < .001), hypercholesterolemia (r = 0.665; P < .001), hypertriglyceridemia (r = 0.618; P < .001), fibrinogen (r = 0.606; P < .001) and D-dimer (r = 0.524; P < .001, Figure 3) in nephrotic syndrome.

Correlation of soluble PDPN with albumin, total cholesterol, triglyceride, fibrinogen and D-dimer. Serum PDPN were significantly correlated with hypoalbuminemia (A. r = −0.735; P < .001), hypercholesterolemia (B. r = 0.665; P < .001), hypertriglyceridemia (C. r = 0.618; P < .001), fibrinogen (D. r = 0.606; P < .001) and D-dimer (E. r = 0.524; P < .001) in patients with nephrotic syndrome.
Predictive Values of Soluble PDPN Levels for Hypercoagulability in Nephrotic Syndrome
Subsequently, Predictive values of soluble PDPN for predicting hypercoagulability was performed. The area under the curve (AUC) was analyzed using ROC analysis for PDPN. AUC for the prediction of hypercoagulability in nephrotic syndrome using PDPN was 0.886 (95% CI 0.804-0.967, P < .001, Figure 4), the Youden index was 0.652, and cut-off value for the risk probability was 5.88 ng/ml. The sensitivity of PDPN in predicting hypercoagulability was 0.806, and the specificity was 0.846. When serum PDPN was >5.88 ng/ml, the risk of hypercoagulability was significantly increased in nephrotic syndrome (OR = 22.79, 95% CI 5.92-87.69, P < .001).
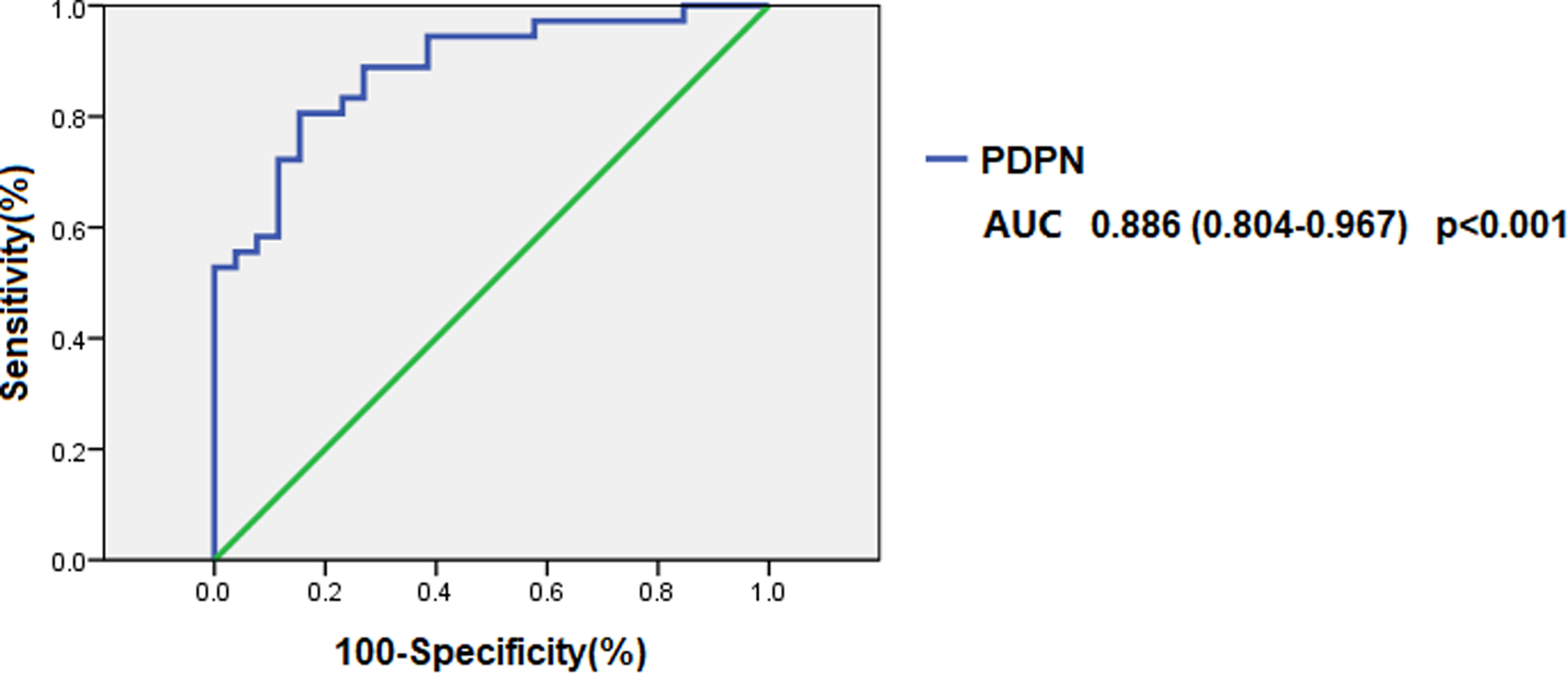
Receiver operating characteristic (ROC) curves of PDPN for predicting hypercoagulability in nephrotic syndrome. AUC indicates area under curve. The number in parentheses of legends indicates 95% confidence of intervals.
Discussion
The present study demonstrated elevated soluble PDPN and glycoprotein VI (GPVI) levels in nephrotic syndrome patients. Moreover, there is a significant correlation between PDPN and GPVI levels. Hypercoagulable state results in kidney injury and the progress of nephrotic syndrome and is important part of early diagnosis of thromboembolic disease. Platelet activation and hyperactivity are found to accompany the nephrotic syndrome and result in an increased risk of thromboembolic events. Increased levels of various active substances including β-thromboglobulin, Platelet-derived growth factor (PDGF), interleukin-7 and P-selectin, which play an important role in platelet activation, have been reported in nephrotic syndrome.18,19,22 Endothelial cells damage is the vital pathologic processes in the initial step of vascular damage that precedes and leads to platelet activation and adhesion. GPVI, the primary collagen receptor on platelets, has been shown to mediate both platelet activation and adhesion via collagen at sites of endothelial cells damage. Previous data have suggested that platelet activation induce platelet MMPs to cleave GPVI on the platelet surface generating soluble forms. 23 Cleavage of platelet receptors in response to activation may act as a physiologic mechanism for thrombosis. GPVI signaling also leads to propagate thrombus growth. 24 Therefore, GPVI is considered as a marker of platelet activation, and elevated GPVI may play critical roles in platelet activation and coagulation during nephrotic syndrome.
The present study is the first to examine serum levels of PDPN/CLEC-2 in nephrotic syndrome and to reveal an elevated soluble PDPN level. PDPN induces platelet aggregation and activation through direct binding of its extracellular domain to CLEC-2 expressed on the platelet surface, and increases the risk of venous thromboembolism.11–13 Current studies demonstrate that high expressions of PDPN in some tumor cells induce platelet aggregation, correlates with hypercoagulability, and is associated with increased risk of thromboembolism.15,25–27 Although PDPN is not expressed in the normal vessel wall, its presence has been reported in certain pathological states, in which PDPN was shown to be up-regulated in the subendothelial wall and to become exposed at sites of breach thereby triggering platelet activation and thrombosis. 28 Inhibition of PDPN also resulted in reduced accumulation of platelets at the inferior vena cava wall after stenosis.11,13 Furthermore, PDPN expression levels correlated with the extent of thrombosis, and the interaction between PDPN and CLEC-2 is responsible for thrombus stabilization. In mice with platelet specific CLEC-2 depletion, venous thrombosis was significantly reduced and transfusion of wildtype platelets restored thrombus formation.11,13 Recent study found the interaction of PDPN and CLEC-2 to be an important driver for thrombosis in inflammation, and upregulation of PDPN on inflammatory cells.5–7,29 These results strongly indicate that PDPN and CLEC-2 play a crucial role in the process of thrombus formation. Therefore, known as a promising predictive biomarker, elevation of PDPN expression might not only reflect physiological regulatory, but also be a sign of hypercoagulability and high risk factor for thromboembolism. In the present study elevated soluble PDPN levels are associated with hypercoagulable state and thromboembolism in nephrotic syndrome, and that the result of significant correlation between PDPN and GPVI confirms this point further.
Based on the previously reported role of PDPN in the process of hypercoagulable state and thromboembolism, we hypothesize that PDPN might be released into the circulation of nephrotic syndrome patients in a soluble form. Nephrotic syndrome is thought to be an immune inflammatory disease. Circulating immune inflammatory cells and subendothelial cells, on which upregulation of PDPN is detected in the process of immune inflammation,5,28 might be a source of soluble PDPN. After that, platelet and coagulation activation via PDPN/CLEC-2 during nephrotic syndrome might induce a hypercoagulable state, which leads to an increased propensity of thrombus formation at sites of slow blood flow and blood stagnation. In future studies, it will be of particular interest to investigate detailed pathophysiologic mechanisms of how PDPN expression induces thrombosis via PDPN/CLEC-2 in nephrotic syndrome.
CLEC-2 expressed on platelets is a physiological binding partner of PDPN and involved in platelet aggregation and thrombosis. Mice with general inducible deletion of CLEC-2 are associated with reduced platelet accumulation and protected against thrombosis. 13 However, no elevated CLEC-2 is detected, and no correlation is confirmed between CLEC-2 and PDPN levels in nephrotic syndrome in the present study. The reason for this is that CLEC-2 may be down-regulated in a negative feedback by forming complexes with soluble PDPN that can function as a negative regulator. In addition, another hypothesis is that CLEC-2, in contrast to GPVI, is only expressed on the surface of platelets and does not appear in circulation in soluble form in nephrotic syndrome.
Subsequently, when analyzing correlation of PDPN with albumin, cholesterol and triglycerides in patients with nephrotic syndrome, the results demonstrated that PDPN levels are significantly associated with hypoalbuminemia, hypercholesterolemia and hypertriglyceridemia. Hypoalbuminemia is generally accepted as the strongest association with venous thromboembolism risk. Hypoalbuminemia increases bioavailability of arachidonic acid released by platelets and thromboxane A2 synthesis, enhances the recruitment of other platelets to the platelet-rich thrombus, which also promotes platelet adhesiveness and aggregation. Besides, hypercholesterolemia and hypertriglyceridemia also promote platelet adhesiveness and hyperactivity, and are associated with hypercoagulable state of nephrotic syndrome. Therefore, in the present study the significant correlation of PDPN with hypoalbuminemia, hypercholesterolemia and hypertriglyceridemia supports its role in thromboembolic events in nephrotic syndrome.
Another crucial result of the present study is that PDPN expression was also significantly associated with levels of fibrinogen and D-dimer. Fibrinogen is the most abundant coagulation protein and marker for thrombin activity. 17 D-dimer levels are established markers in clinical practice for a thrombophilic or hypercoagulable state. Results from previous studies have indicated that D-dimer is not only a sign of hypercoagulability, but its continuous increase is closely related to the complications of thromboembolism. 17 The significant correlation between PDPN, fibrinogen and D-dimer further confirms that high expression of PDPN may be able to relate to hypercoagulable state and more prone to develop thrombosis in nephrotic syndrome. The present study shows that when serum PDPN levels in nephrotic syndrome was >5.88 ng/ml, the risk of developing hypercoagulability was significantly increased. Therefore, we speculate that PDPN has the better predictive value of hypercoagulability in nephrotic syndrome as well as is a reliable indicator of hypercoagulable state.
Some limitations of this study need to be discussed. First of all, a relatively small cohort of patients from a single medical center is enrolled. A single sample at a certain point in time may fail to reflect the natural course of disease. Multicenter and prospective studies are needed to make dynamic analysis of PDPN and to confirm the results of this study in the further. In addition, there is no experimental in vivo evidence for a mechanistic relationship between PDPN expression and development of hypercoagulable state and thrombosis. However, results observed in this study are in concordance with previously published coagulation mechanism related to PDPN/CLEC-2, and indicate that this study population is representative for patients with nephrotic syndrome. Finally, the correlation between PDPN levels, antiphospholipid antibodies and thrombin levels is not analyzed. These factors may contribute to the hypercoagulable state in nephrotic syndrome. Future studies may further investigate the role of specific molecular alterations associated with PDPN/CLEC-2 expression and coagulation.
Conclusion
In conclusion, soluble PDPN may be play critical roles in the progress of the platelets activation, aggregation, hypercoagulable state and thrombosis, and have the better predictive value of hypercoagulable state in nephrotic syndrome. This study highlights the possibility that PDPN/CLEC-2 is involved in the complications of thromboembolism in nephrotic syndrome. PDPN/CLEC-2 pathway may be a promising therapeutic target for anticogulation. Future studies will concentrate on regulatory mechanisms of PDPN/CLEC-2 pathway for prophylactic anticoagulation and thromboembolism treatment.
In addition, as the COVID-19 pandemic spread worldwide, recent studies have shown its association with the tendency of thrombotic complications. 30 And since the nephrotic syndrome is also a hypercoagulable state, its co-occurrence with the COVID-19 infection is likely to be associated with an even higher risk of thrombosis. 31 It would be interesting to study PDPN in this setting in future studies.
Footnotes
Acknowledgements
The authors appreciate the support provided by all colleagues involved in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Project of Heilongjiang Provincial Health Commission (grant number 2019-013, 2019-017).
